ABSTRACT
Objectives: The aim of our longitudinal study was to explore changes in HRQOL over a 6-month period and to identify factors associated with the HRQOL of HSCT recipients.
Method: Our study comprised 191 HSCT patients; their data were collected before transplantation and at 30, 90, and 180 days posttransplantation. The Functional Assessment of Cancer Therapy-Bone Marrow Transplant (FACT-BMT) questionnaire was used to assess HRQOL.We also evaluated the patients’ demographic characteristics and clinical histories to determine the relative contributions of these factors to HRQOL outcomes.
Results: Before HSCT, the patients reported a mean overall HRQOL of 110.31 (SD, 14.99); this reached a minimum of 105.07 (SD, 18.85) at day 30 after HSCT and increased steadily over time to 106.71 (SD, 18.34) at day 90 and 108.16 (SD, 18.34) at day 180 after HSCT.Compared with baseline, overall HRQOL changed with the mean of −5.24 (SE 1.55; P = .001), and −3.60 (SE 1.55; P = .022), respectively, at 30 days and 90 days after HSCT. Overall HRQOL returned to near pretransplant levels at 180 days after HSCT (SE 1.47; P = .146).Generalized estimating equation (GEE) models showed that household income (β = 6.590; P < .001), transplant-related complications (β = −6.101; P < .001), and patient age (β = 0.243, P = .045) were associated with HRQOL.
Conclusion: The patients’ overall HRQOL was severely impaired in the early stages of posttransplantation, and patients experienced the worst HRQOL at 30 days. They had improved significantly by 180 days posttransplantation. We also found that household income, transplant-related complications, and age were independent predictors of early HRQOL.We therefore concluded that the HRQOL of HSCT patients in the early stages posttransplantation deserved more attention.
Introduction
Hematopoietic stem cell transplantation (HSCT) is the most effective way of treating hematologic malignancies [Citation1]. Before treatment, patients undergoing HSCT undergo high-dose chemotherapy and radiotherapy in order to eradicate tumour cells and abnormally proliferating cells. Thereafterdonor hematopoietic stem cells are injected and the patient's hematopoietic system is eventually rebuilt. More than 60,000 individuals worldwide undergo allogeneic or autologous HSCT each year,with a concomitant rise in the number of survivors [Citation2]. Advances in HSCT techniques and supportive care have led to dramatic improvements in relapse mortality in patients with high-risk hematologic malignancies. Nearly 60% of HSCT recipients exhibit long-term survival with no disease [Citation1,Citation3].
HSCT has greatly improved the survival of patients; however, this procedure has severe toxic effects, both physiological and psychological, which cannot be ignored [Citation4–9]. The advent of reduced-intensity conditioning regimens has improved patients’ tolerance, yet side effects still occur. These effects – caused by high-dose cytotoxic drugs, immunosuppressive drugs, and systemic irradiation – include nausea, emesis, mucositis, cystitis, and diarrhea [Citation10,Citation11]. They severely inhibit the immune system and increase the individual's susceptibility to infection. Posttransplantation complications include acute or chronic graft-versus-host disease (GVHD), severe infection, hemorrhagic cystitis, pneumonia, relapse, cataracts, and infertility [Citation12–16]. Acute or chronic GVHD is the major and most common complication, with an incidence of 50% to 70% and 30% to 50%, respectively [Citation17]. The physiological side effects result in decreased physical performance, thus negatively affecting patients’ HRQOL. In addition, patients undergoing HSCT often experience psychological effects – fear, anxiety, and uncertainty – as well as financial difficulties [Citation4,Citation18,Citation19]. All these have psychosocial consequences that influence life satisfaction and HRQOL.
HRQOL is generally conceptualized as a multidimensional construct referring to patients’ perceptions of the impact of a disease and its treatment on their physical, psychological, and social functioning and well-being [Citation20]. In recent years, HRQOL has gradually become an important part of the evaluation and management of HSCT recipients before and after transplantation [Citation4–6,Citation20–27]. However, studies addressing HRQOL in HSCT patients are subject to overreliance on cross-sectional surveys, diverse research designs, different types of patients studied, variations in methods of HRQOL assessment, and differences in the time points at which HSCT recipients were studied, resulting in great heterogeneity between studies. In addition, we found that authors paid more attention to the trajectory of HRQOL during the longer term in longitudinal studies and that few authors have focused on HRQOL at the early stages of transplantation. However, we regard this period as of great clinical significance, as patients’ bone marrow is affected, leading to increased susceptibility to infection and other complications. These developments are conducive to physiologic and psychological distress and likely to reduce HRQOL.
In view of the foregoing, we chose to focus our study more on HRQOL in the early stages of transplantation. We investigated 191 HSCT recipients’ HRQOL before transplantation and on days 30, 90, and 180 posttransplantation. Our aim was to identify the trajectory of HRQOL during the early stages of transplantation and to determine the most salient influencing factors so as to plan for intervention at the earliest time.
Methods
Participants
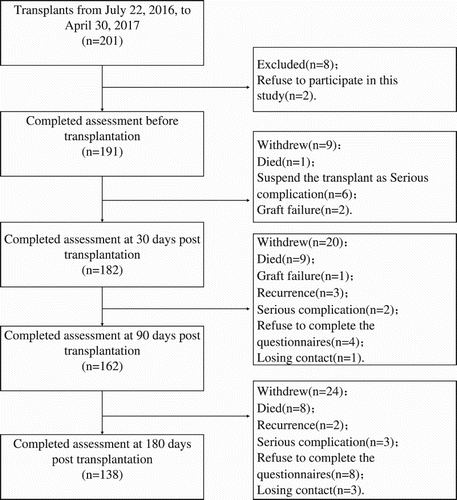
All the participants were undergoing an allogeneic/autologous bone marrow or peripheral blood stem cell transplant for a hematologic malignancy. Patients were included if they were 18 years old or older and were willing to participate in the study. Patients with mental illness or cognitive impairment were excluded, as were those who experienced graft failures, whose illness was recrudescent, who were too sick to participate, or who died. Participants were recruited from 22 July 2016, to 30 April 2017. Sample size calculations were based on the detection of medium effect sizes (power = 0.8) with an a priori estimation of 10–15 predictor variables: a sample size of 120 was required [Citation28]. Finally, A total of 191 participants were enrolled in the study and each completed the questionnaires before transplantation. The number of participants on days 30, 90, and 180 was 182, 162, and 138, corresponding to participation rates of 95.3%, 84.8%, and 72.3%, respectively (). This study was approved by the ethics committee of our hospital and all subjects provided informed consent in accordance with the Declaration of Helsinki.
Study measures
Health-related quality of life
HRQOL was measured by the Functional Assessment of Cancer Therapy – Bone Marrow Transplant (FACT-BMT, V4.0) questionnaire [Citation29], which includes 5 domains: physical well-being (PWB), social/family well-being (SWB), emotional well-being (EWB), functional well-being (FWB), and BMT-specific concerns (BMTS). Responses were ranked from 0 to 4 (0, not at all; 1, a little bit; 2, somewhat; 3, quite a bit; 4, very much). The 5 domain scores were summed to generate a total score. The total scores ranged from 0 to 148, and the higher the score, the better the HRQOL. The reliability coefficients range from 0.86 to 0.89 [Citation29].
Sociodemographic characteristics
Sociodemographic characteristics included age, sex, marital status, residence, education level, and household income [Citation19,Citation23,Citation26,Citation30]. For analysis, marital status included both unmarried and married; and there was no category for divorce or separation in this study. Area of residence was categorized as rural or nonrural. Education was determined according to the highest level attained, including junior high school or below, high school, baccalaureate degree, and above). Household income was divided into 4 categories, from low to high ().
Table 1. Characteristics and clinical data of patients in the study.
Clinical data collection
Clinical materials included disease diagnosis (acute leukemia/nonacute leukemia), stem cell source (allogeneic/autologous), preparative regimen (busulfan and cyclophosphamide or not), platelets (the level of platelet measured when patients got out of the purifying ward), and transplant-related complications (including infection, GVHD, urocystitis, etc.) [Citation4,Citation27,Citation31–33] (). We obtained this information from the HSCT follow-up group, which was responsible for the follow-up of HSCT patients in the department of hematology.
Statistical analysis
Data analysis was carried out using IBM SPSS Statistics 19. Data were presented as mean ± SD, for continuous variables and count (percentage) for categorical variables. Repeated measures ANOVA was used to assess the changes between different time points and the LSD method for comparison between different measurement points. The GEE method was used to identify the risk factors for HRQOL. All statistical assessments were evaluated with a 2-sided alpha level of 0.05.
Results
Sample baseline characteristics
A total of 138 patients provided serial HRQOL data. The mean age of participants was (33.31 ± 11.40) years (range, 18–62). Of the 138 patients, 82 (59.4%) were male and 56 (40.6%) were female. Only 10 (7.2%) of the 138 patients underwent autologous transplantation; the others 128 (92.8%) received allogeneic transplantation. describes the characteristics of the study participants at different points in the treatment process. The dropout sample size was 53 (27.7%). Of these 53 patients, 18 died of serious GVHD, 6 stopped the transplant temporarily owing to serious complications while undergoing pretreatment, 3 experienced graft failures, 5 relapsed, and 5 were too sick to complete the questionnaires. Twelve refused to complete the questionnaires, as most thought that filling out the questionnaires would not help them in any way, and 4 could not be contacted (). We compared the baseline characteristics of the missing sample (n = 53) and the rest of the samples (n = 138), which came from patients who completed all the measurements at the 4 time points and found that there was no statistical difference in characteristics between the 2 groups (). Therefore the missing values were ignored in our data analysis.
Table 2. Baseline characteristics comparison of the missing sample with the rest of the sample.
Changes in HRQOL scores over time
Overall HRQOL
Before HSCT, patients reported a mean overall HRQOL of 110.31 (SD, 14.99); it reached a minimum of 105.07 (SD, 18.85) at day 30 after HSCT and increased steadily over time to 106.71 (SD, 18.34) at day 90 and 108.16 (SD, 18.34) at day 180. Compared with baseline, overall HRQOL changed with the mean of −5.24 (SE 1.55; P = .001), and −3.60 (SE 1.55; P = .022), respectively, at 30 days and 90 days after HSCT. Overall HRQOL had returned to near pretransplant levels 180 days after HSCT (SE 1.47; P = .146) ().
Table 3. Functional assessment of cancer therapy–bone marrow transplantation (FACT-BMT) and its dimensions and mean scores at 4 different time points and tests of difference for patients before HSC, and at 30, 90, and 180 days post transplantation.
PWB HRQOL
Patients reported a mean PWB HRQOL of 22.14 (SD, 4.05) at baseline, 20.38 (SD, 5.47) at day 30, and 21.58 (SD, 4.46), 21.92 (SD, 4.12) at days 90 and 180 after HSCT. Patients experienced the worst physical function at 30 days after transplantation. The deterioration from baseline had no statistical significance at 90 (SE, 0.44; P = .208) and 180 days (SE, 0.42; P = .603) ().
SWB HRQOL
SWB HRQOL showed a continuous decline with means 23.63 (SD, 4.16), 22.24 (SD, 4.88), 21.90 (SD, 4.83) and 21.29 (SD, 5.45) at baseline and at 30, 90, and 180 days, respectively. Within SWB HRQOL, the decrease from baseline was significant at 30 (SE, 0.38; P < .001), 90 (SE, 0.41; P < .001), and 180 (SE, 0.45; P < .001) days after HSCT ().
EWB HRQOL
On the other hand, EWB HRQOL showed a rising trend, with means 18.85 (SD, 3.77), 19.57 (SD, 3.70), 19.76 (SD, 3.45) and 20.04 (SD, 3.28) at baseline and at 30, 90, and 180 days, respectively. The improvement at day 30 was not significant (SE, 0.38; P = .061). However, it was significant at days 90 (SE, 0.40; P = .024) and 180 (SE, 0.36; P = .001) after HSCT ().
FWB HRQOL
The score of FWB HRQOL also changed dynamically at different time points (F = 3.794; P = 0.013). It reached its highest level before HSCT, with a mean of 14.93 (SD, 6.53) and then descended to its lowest level, to 13.43, at 30 days after HSCT (SD,6.58). These changes were significant (SE, 0.59; P = .012). Improvement began at 90 days, with a mean of 13.89 (SD, 5.72) after HSCT, representing a change of −1.04 (SE, 0.57; P = .068). It then and returned nearly to baseline level (SE, 0.57; P = .929) at 180 days after HSCT ().
BMTS HRQOL
Last, BMTS HRQOL was highest before HSCT, with a mean of 30.76 (SD, 4.59). Patients experienced the most serious damage at 30 days after HSCT, with a mean of 29.45 (SD, 4.39); this decline was statistically significant (SE, 0.43; P = .003). And there was a mean of 29.58 (SD, 4.78) at 90 days after HSCT, still lower than pretransplant levels (SE = 0.49; P = .017). But patients began to recover at 180 days after HSCT, with a mean change of −0.74 (SE = 0.43; P = .086) from baseline ().
Factors associated with HRQOL 180 days after HSCT
summarizes the GEE model to find risk factors for HRQOL. Household income (β = 6.590; P < .001), transplant-related complications (β = −6.101; P < .001), and patient age (β = 0.243, P = .045) were associated with HRQOL.
Table 4. Factors predicting health-related quality of life.
Conclusion
This prospective study has evaluated changes in the HRQOL of patients with hematologic and lymphoid malignancies treated with hematopoietic stem cell transplantation with a follow-up of 6 months and has also recognized factors associated with HRQOL in the early stages following transplantation. We found that patients’ overall HRQOL was severely impaired early – that is, they experienced the worst HRQOL at 30 days posttransplantation – but they experienced a significant improvement by 180 days posttransplantation. We also have confirmed that better HRQOL was associated with higher household income, absence of transplant-related complications, and older age.
We concluded that the HRQOL of patients in the early stages of transplantation should receive more attention. The deterioration in patients’ HRQOL in the early stages of transplantation has many causes. The main one is that the immune and hematopoietic systems are not yet completely rebuilt in the early stages of recovery. This reduces the body's power of resistance and can lead to infections and other complications. Distressing symptoms of reduced physical performance and functioning and increased high levels of fatigue affect patients’ HRQOL negatively [Citation34]. Furthermore, the long period of hospitalization can produce social isolation and a lack of social support, resulting in anxiety and depression [Citation6,Citation35]. With the recovery of hematopoietic function, the HRQOL gradually begins to increase, reaching a statistically higher level by 180 days posttransplantation. However, the recovery process is very long and difficult. Study [Citation36] has confirmed that the HRQOL in HSCT patients after an average of 20 years posttransplantation is still below that of the general population.
Another fact that should arouse attention is the trajectory of social/family well-being. There was a continuous deterioration in social/family well-being during the study period. After transplantation, a deterioration in social function is a common problem [Citation37–39]. Research [Citation37] has found that 48%, 23%, and 16% of patients had reduced social function at 6 months, 1 year, and 2 years after transplantation, respectively. In addition, reemployment after transplantation is an important part of the restoration of social function. Gruber et al. [Citation40] haveconfirmed that reemployed patients experience less anxiety and depression and have fewer sleep problems than unemployed patients.
Measures should be taken to help patients return to normal life as soon as possible after transplantation. The results of our multivariate analyses show that transplant-related complications, household income, and age were the chief factors affecting patients’ HRQOL. The effects of medical complications on HRQOL have been confirmed [Citation31,Citation41]. Although complications are more difficult to manage via nursing, we still have to do our best to prevent their occurrence. When complications arise, nurses should provide not only the best available care but also use their scientific knowledge to educate patients, informing them of the causes of complications as well as the prognosis and available treatments so as to eliminate patients’ worries and anxiety and improve their compliance and the overall efficacy of treatment.
We also found that higher household income is associated with greater HRQOL. Economic factors affect HRQOL in many ways, since they involve treatment costs, nutritional options, health conditions, and compliance with treatment [Citation19]. HSCT imposes a heavy economic burden to patients and their families even after discharge from the hospital. A survey [Citation42] from China found that economic factors had the greatest impact on the HRQOL of patients; only 15% believed that their economic conditions could meet the requirements of treatment. Similarly, the results of this study suggest that older patients had a better HRQOL. Older patients usually have more stable social networks, better interpersonal skills, more supportive friendships, and more satisfactory marriages [Citation26]; they often have a better state of mind and are more capable of coping with major life changes.
In conclusion, our study sheds light on changes in HRQOL before and after HSCT, suggesting that HRQOL in the initial period of treatment should receive more attention. We also demonstrated that transplant-related complications, household income, and age were risk factors associated with HRQOL. Nevertheless, weaknesses in our study must be acknowledged. One limitation is its relatively small sample size compared with multicenter studies. A second limitation might be that only FACT-BMT was used to assess the patients’ QoL without considering other measurements. Further, we did not discuss the influence of factors including anxiety, depression, caregivers’ roles, rehabilitation treatment, and so on, which may also have an effect on HRQOL. However, the results of this study still have practical value clinically. In the future we will focus more on these other factors and discuss their interactions so as to make the results more useful to the scientific community.
Acknowledgments
The authors gratefully acknowledge the time and energy contributed by the participants. We also would like to thank LetPub (www.letpub.com) for providing linguistic assistance during the preparation of this manuscript.
Disclosure statement
No potential conflict of interest was reported by the authors.
References
- Huang X, Wu D, Liu D. Practice of hematopoietic stem cell transplantation. Beijing: People’s Medical Publishing House; 2014.
- Wingard JR, Majhail NS, Brazauskas R, et al. Long-term survival and late deaths after allogeneic hematopoietic cell transplantation. J Clin Oncol. 2011;29(16):2230–2239. doi: https://doi.org/10.1200/JCO.2010.33.7212
- Majhail NS, Douglas Rizzo J. Surviving the cure: long term followup of hematopoietic cell transplant recipients. Bone Marrow Transpl. 2013;48(9):1145–1151. doi: https://doi.org/10.1038/bmt.2012.258
- Hjermstad MJ, Knobel H, Brinch L, et al. A prospective study of health-related quality of life, fatigue, anxiety and depression 3–5 years after stem cell transplantation. Bone Marrow Transpl. 2004;34(3):257–266. doi: https://doi.org/10.1038/sj.bmt.1704561
- Pillay B, Lee SJ, Katona L, et al. A prospective study of the relationship between sense of coherence, depression, anxiety, and quality of life of haematopoietic stem cell transplant patients over time. Psychooncology. 2015;24(2):220–227. doi: https://doi.org/10.1002/pon.3633
- Sherman AC, Simonton S, Latif U, et al. Changes in quality-of-life and psychosocial adjustment among multiple myeloma patients treated with high-dose melphalan and autologous stem cell transplantation. Biol Blood Marrow Tr. 2009;15(1):12–20. doi: https://doi.org/10.1016/j.bbmt.2008.09.023
- Wingard JR, Huang IC, Sobocinski KA, et al. Factors associated with self-reported physical and mental health after hematopoietic cell transplantation. Biol Blood Marrow Tr. 2010;16(12):1682–1692. doi: https://doi.org/10.1016/j.bbmt.2010.05.017
- Bhatia M, Kolva E, Cimini L, et al. Health-related quality of life after allogeneic hematopoietic stem cell transplantation for sickle cell disease. Biol Blood Marrow Tr. 2015;21(4):666–672. doi: https://doi.org/10.1016/j.bbmt.2014.12.007
- Andrykowski MA, Bishop MM, Hahn EA, et al. Long-term health-related quality of life, growth, and spiritual well-being after hematopoietic stem-cell transplantation. J Clin Oncol. 2005;23(3):599–608. doi: https://doi.org/10.1200/JCO.2005.03.189
- Pallera AM, Schwartzberg LS. Managing the toxicity of hematopoietic stem cell transplant. J Support Oncol. 2004;2(3):223–237.
- Arfons LM, Lazarus HM. Total parenteral nutrition and hematopoietic stem cell transplantation: an expensive placebo? Bone Marrow Transpl. 2005;36(4):281–288. doi: https://doi.org/10.1038/sj.bmt.1705039
- Al-Anazi KA, Al-Jasser AM. Infections caused by stenotrophomonas maltophilia in recipients of hematopoietic stem cell transplantation. Front Oncol. 2014;4:1–11.
- Liu QF, Luo XD, Ning J, et al. Association between acute graft versus host disease and lung injury after allogeneic haematopoietic stem cell transplantation. Hematology. 2009;14(2):63–72. doi: https://doi.org/10.1179/102453309X385142
- Shakiba E, Yaghobi R, Ramzi M. Prevalence of viral infections and hemorrhagic cystitis in hematopoietic stem cell transplant recipients. Exp Clin Transpl. 2011;9(6):405–412.
- Li Z, Mewawalla P, Stratton P, et al. Sexual health in hematopoietic stem cell transplant recipients. Cancer Am Cancer Soc. 2016;121(23):4124–4131.
- Karrasch M, Schmidt V, Hammer A, et al. Chronic persistent parvovirus B19 bone marrow infection resulting in transfusion-dependent pure red cell aplasia in multiple myeloma after allogeneic haematopoietic stem cell transplantation and severe graft versus host disease. Hematology. 2017;22(2):93–98. doi: https://doi.org/10.1080/10245332.2016.1183288
- Elad S, Zeevi I, Or R, et al. Validation of the national institutes of health (NIH) scale for oral chronic graft-versus-host disease (cGVHD). Biol Blood Marrow Tr. 2010;16(1):62–69. doi: https://doi.org/10.1016/j.bbmt.2009.08.018
- Hamada N, Hasegawa R, Okamoto A, et al. Factors influencing health-related quality of life for patients after hematopoietic stem cell transplantation: a single institution analysis. J Hematopoietic Cell Transpl. 2017;6(1):36–44. doi: https://doi.org/10.7889/hct.6.36
- Hamilton JG, Wu LM, Austin JE, et al. Economic survivorship stress is associated with poor health-related quality of life among distressed survivors of hematopoietic stem cell transplantation. Psychooncology. 2013;22(4):911–921. doi: https://doi.org/10.1002/pon.3091
- Andersson I, Ahlberg K, Stockelberg D, et al. Patients’ perception of health-related quality of life during the first year after autologous and allogeneic stem cell transplantation. Eur J Cancer Care. 2011;20(3):368–379. doi: https://doi.org/10.1111/j.1365-2354.2009.01174.x
- Sirilla J, Overcash J. Quality of life (QOL), supportive care, and spirituality in hematopoietic stem cell transplant (HSCT) patients. Support Care Cancer. 2013;21(4):1137–1144. doi: https://doi.org/10.1007/s00520-012-1637-y
- Mcquellon RP, Russell GB, Rambo TD, et al. Quality of life and psychological distress of bone marrow transplant recipients: the ‘time trajectory’ to recovery over the first year. Bone Marrow Transpl. 1998;21(5):477–486. doi: https://doi.org/10.1038/sj.bmt.1701115
- Hamilton BK, Rybicki L, Dabney J, et al. Quality of life and outcomes in patients ≥ 60 years of age afterallogeneic hematopoietic cell transplantation. Bone Marrow Transpl. 2014;49(11):1426–1431. doi: https://doi.org/10.1038/bmt.2014.166
- Janicsák H, Masszi T, Reményi P, et al. Quality of life and its socio-demographic and psychological determinants after bone marrow transplantation. Eur J Haematol. 2013;91(2):135–140. doi: https://doi.org/10.1111/ejh.12126
- Pidala J, Anasetti C, Jim H. Quality of life after allogeneic hematopoietic cell transplantation. Blood. 2009;114(1):7–19. doi: https://doi.org/10.1182/blood-2008-10-182592
- Niederbacher S, Them C, Pinna A, et al. Patients’ quality of life after allogeneic haematopoietic stem cell transplantation: mixed-methods study. Eur J Cancer Care. 2012;21(4):548–559. doi: https://doi.org/10.1111/j.1365-2354.2012.01345.x
- Wong F, Francisco L, Togawa K, et al. Long-term recovery after hematopoietic cell transplantation: predictors of quality-of-life concerns. Blood. 2010;115(12):2508–2519. doi: https://doi.org/10.1182/blood-2009-06-225631
- Green SB. How many subjects does it take to do a regression analysis. Multivar Behav Res. 1991;26:499–510. doi: https://doi.org/10.1207/s15327906mbr2603_7
- Kopp M, Schweigkofler H, Holzner B, et al. EORTC QLQ-C30 and FACT-BMT for the measurement of quality of life in bone marrow transplant recipients: a comparison. Eur J Haematol. 2000;65(2):97–103. doi: https://doi.org/10.1034/j.1600-0609.2000.90143.x
- Socié G, Mary JY, Esperou H, et al. Health and functional status of adult recipients 1 year after allogeneic haematopoietic stem cell transplantation. Brit J Haematol. 2001;113(1):194–201. doi: https://doi.org/10.1046/j.1365-2141.2001.02678.x
- Lynch KD, Lyon DE, Ameringer SA, et al. Symptoms, cytokines, and quality of life in patients diagnosed with chronic graft-versus-host disease following allogeneic hematopoietic stem cell transplantation. Oncol Nurs Forum. 2015;42(3):265–275. doi: https://doi.org/10.1188/15.ONF.265-275
- Lee SJ, Fairclough D, Parsons SK, et al. Recovery after stem-cell transplantation for hematologic diseases. J Clin Oncol. 2001;19(1):242–252. doi: https://doi.org/10.1200/JCO.2001.19.1.242
- Danaher EH, Ferrans C, Verlen E, et al. Fatigue and physical activity in patients undergoing hematopoietic stem cell transplant. Oncol Nurs Forum. 2006;33(3):614–624. doi: https://doi.org/10.1188/06.ONF.614-624
- Larsen J, Nordstrom G, Ljungman P, et al. Symptom occurrence, symptom intensity, and symptom distress in patients undergoing high-dose chemotherapy with stem-cell transplantation. Cancer Nurs. 2004;27(1):55–64. doi: https://doi.org/10.1097/00002820-200401000-00007
- Andrykowski MA. Psychiatric and psychosocial aspects of bone marrow transplantation. Psychosomatics. 1994;35(1):13–24. doi: https://doi.org/10.1016/S0033-3182(94)71803-X
- La Nasa G, Caocci G, Efficace F, et al. Long-term health-related quality of life evaluated more than 20 years after hematopoietic stem cell transplantation for thalassemia. Blood. 2013;122(13):2262–2270. doi: https://doi.org/10.1182/blood-2013-05-502658
- Lee SJ, Loberiza FR, Antin JH, et al. Routine screening for psychosocial distress following hematopoietic stem cell transplantation. Bone Marrow Transpl. 2005;35(1):77–83. doi: https://doi.org/10.1038/sj.bmt.1704709
- Morrison EJ, Ehlers SL, Bronars CA, et al. Employment status as an indicator of recovery and function one year after hematopoietic stem cell transplantation. Biol Blood Marrow Tr. 2016;22(9):1690–1695. doi: https://doi.org/10.1016/j.bbmt.2016.05.013
- Bush NE, Donaldson GW, Haberman MH, et al. Conditional and unconditional estimation of multidimensional quality of life after hematopoietic stem cell transplantation: a longitudinal follow-up of 415 patients. Biol Blood Marrow Tr. 2000;6(5A):576–591. doi: https://doi.org/10.1016/S1083-8791(00)70067-X
- Gruber U, Fegg M, Buchmann M, et al. The long-term psychosocial effects of haematopoetic stem cell transplantation. Eur J Cancer Care. 2003;12(3):249–256.
- Cohen MZ, Rozmus CL, Mendoza TR, et al. Symptoms and quality of life in diverse patients undergoing hematopoietic stem cell transplantation. J Pain Symptom Manag. 2012;44(2):168–180. doi: https://doi.org/10.1016/j.jpainsymman.2011.08.011
- Aifei F, Shuping W, Li D, et al. Analysis of postoperative life quality and influence factors of patients with hematopoietic stem cell transplantation. Int J Blood Transfus Hematol. 2015;38(5):377–382. Chinese