Abstract
Cardiovascular diseases remain the principal contributor to global mortality and morbidity. Chronic stress has emerged as a strong prognostic factor for cardiovascular diseases. We aimed to measure hair cortisol concentration (HCC) in patients with angiographically confirmed coronary atherosclerosis and find the association between HCC and classic cardiovascular risk factors in a case-control study. The study included 500 angiographically confirmed coronary atherosclerosis patients and 500 age and sex-matched controls having no coronary stenosis. Hair cortisol concentration was assessed from a single sample of scalp hair by ELISA. Perceived stress scale 10 was used to evaluate the stress level of the participants. Spearman rank correlation was applied to find the association between hair cortisol level and classic cardiovascular risk factors. Multivariate regression was executed to assess the independent contribution of hair cortisol concentration as a coronary atherosclerosis risk factor. Median hair cortisol concentration was significantly high in patients with angiographically documented coronary atherosclerosis compared to controls (158(17.6–1331.3) pg/mg versus 73 (13.4–889) pg/mg. Hair cortisol concentration showed a significant positive correlation with BMI r = 525 and Hb1Ac r = 665 (both, p = ≤0.0001). In multivariate logistic regression analysis after adjusting for all the confounding variables, HCC, matched odds ratio (MOR), 10.73;95%CI (1.60,42.09) p-value = 0.001 remained the most significant predictor of coronary atherosclerosis.
Hair cortisol concentrations (HCC) were significantly high in patients with angiographically confirmed coronary atherosclerosis compared to controls. HCC showed a significant positive correlation with diabetes and obesity and remained a major predictor of coronary atherosclerosis in the final analysis.
LAY SUMMARY
1. Introduction
Cardiovascular diseases (CVDs) remain the leading contributor to global morbidity and mortality (Nafisa et al., Citation2018). The burden of CVDs is receding in the West but it is escalating in several lower middle income and developing countries (Roth et al., Citation2018). South Asians are at high CVDs risk compared to other Asian populations (Volgman et al., Citation2018). CVDs are the biggest non-communicable killer in Pakistan accounting for 29% of the overall mortality. Coronary heart disease (CHD) and stroke are the leading manifestation of CVDs in Pakistan. CHD alone contributes 20.28% to proportional mortality (WHO, Citation2018)
Despite the number of well-documented coronary atherosclerosis risk factors in literature and clinical practice, recently there has been a promising focus on the role of psychosocial stress in the genesis of cardiovascular diseases. Chronic stress negatively impacts general health status (McEwen, Citation2012) and fosters disease (Miller & Blackwell, Citation2006). Psychosocial stressors, including workplace stress, financial and marital stress, have enhanced vulnerability for developing CVDs including acute myocardial infarction(AMI) (Dupre et al., Citation2015; Xu et al., Citation2015). The higher incidence of CHD, stroke and other types of CVDs, such as atrial fibrillation is linked with stress (Kivimäki & Steptoe, Citation2018). Analyses of prospective follow-up data further support the associations between stress and cardiovascular disease (Kivimäki & Steptoe, Citation2018). Psychosocial stress factors promote systemic inflammation, cellular adhesion, endothelial dysfunction, and a pro-thrombotic state (von Känel, Citation2012).
Dysregulation of the hypothalamus-pituitary-adrenal axis (HPA) is a possible biological phenomenon linking psychosocial stress with CVDs. In humans, the major part of the stress response is incorporated by HPA-axis. Stress, physical or emotional alters the activity of the HPA axis and the autonomic nervous system (ANS). The HPA-axis stimulates the synthesis of glucocorticoid cortisol, which is known for its mediating effects on lipids and sugar metabolism (Brotman et al., Citation2007). Excessive cortisol secretions are linked with elevated cardiovascular risk (Arnaldi et al., Citation2003; Izawa et al., Citation2019; Manenschijn et al., Citation2013; Pereg et al., Citation2011).
Under non-stressed basal conditions, cortisol secretion follows circadian rhythm and aids in adjusting to the day and night cycles. Cortisol level measured in serum, saliva, or urine is affected by daily physiological fluctuations. It does not reflect the cumulative exposure to stress over long periods, as it gives the only snapshot of cortisol at a single point. The study of hair cortisol concentration (HCC) is a rapidly emerging field of research that provides a novel method for investigating the physiological implications of prolonged chronic stress exposure.
A high level of perceived stress has been linked with cellular aging (Mathur et al., Citation2016) and several pathophysiological alterations relating to CVDs (Nabi et al., Citation2013; Sumner et al., Citation2016). Keeping in view, the current study was conducted, firstly, to explore the association between hair cortisol concentration and classic CVDs risk factors. Secondly, to find the prognostic significance of hair cortisol concentration, a biological marker of stress, as a risk factor for coronary atherosclerosis.
2. Subjects and methods
2.1. Subjects
We carried out a hospital-based case-control study at Rawalpindi Institute of Cardiology (RIC) and Pir Mehar Ali Shah Arid Agriculture University to find the association of hair cortisol content with angiographically documented coronary atherosclerosis in the Pakistani population. RIC is a nonprofit tertiary level hospital in Rawalpindi, where all patients with diagnosed or suspected cardiovascular problems from Punjab, Khyber Pakhtoon Khaw, Azad Kashmir, and Gilgit Baltistan are referred for diagnosis and treatment. The research ethics committee at Pir Mehr Ali Shah Arid Agriculture University Rawalpindi approved the study. The study personnel herself daily visited the pre-cath ward of RIC for two years (April 2015 and May 2017) to enroll the patients. The recommendation to perform coronary angiography in clinically indicated subjects was based on the American Heart Association (AHA)/American College of Cardiology (ACC) guidelines for coronary angiography (1999).
2.2. Questionnaire-based information collection
Patients listed for their first coronary angiography were selected and relevant clinical information for all participants was obtained through documentary sources including patient’s files hospital records and structured interviews. A special questionnaire comprised of previously validated components from earlier studies was compiled to collect information regarding demographics, lifestyle (diet, smoking status), and family history of the study population. The questionnaire was translated and administered in the national language Urdu. Self-administered questionnaires were not used due to the study participants’ low literacy level and lack of skill to handle such questionnaires.
2.3. Operational definitions of classic risk factors
All traditional coronary atherosclerosis risk factors were operationally defined as following, hypertension: according to JNC VII guideline, diabetes mellitus: glycated hemoglobin (HB1Ac) ≥6.5% or as the history of treatment with either insulin or oral hypoglycemic medication, dyslipidemia: triglycerides (TG) ≥ 150 mg/dl; low-density lipoprotein (LDL-C) ≥130 mg/dl, total cholesterol (TC) ≥ 200 mg/dl, and density lipoprotein (HDL-C) ≥40 mg/dl, body mass index (BMI) was defined in the light of WHO recommendation for South Asians. The smoking status of the participants was categorized by self-report. Nonsmokers, if they had never smoked, current smokers if they had smoked in the last year and former smokers if they had abandon smoking more than one year ago. Positive family history of heart disease was defined as a diagnosis of CVDs in a first-degree male relative <55 years of age or a first-degree female relative <65 years of age. The information regarding stress and physical activity was collected through the perceived stress scale (PSS) (Cohen et al., Citation1983) and a short version of the international physical activity questionnaire (Craig et al., Citation2003). Subjects with a PSS- stress score of 27–40 were classified as highly stressed.
A face-to-face interview lasting for about 20–25 min was conducted by the study personnel to collect baseline information regarding age, ethnicity schooling, lifestyle factors (current and past smoking, physical activity), and family history of CVDs. Inclusion criteria for enrollment as case and control were based on an angiographic diagnosis. Two experienced cardiologists performed coronary angiograms. Coronary atherosclerosis was defined as more than 50% stenosis identified in a minimum of two views in case of single-vessel involvement (Rosenthal, Citation2015). A normal coronary angiogram was defined as having no luminal stenosis in any vessel in a minimum of two views. All participants gave informed written consent.
Cases
Patients with abnormal coronary angiograms aged between 25 and 75 years and who had never been on lipid-lowering therapies were included in the study. Patients who do not fall in the age bracket of 25–75, do not have enough hair at the back of the head, suffering from any documented disease including cardiomyopathy, valvular heart disease, congenital heart disease, and having coronary artery stenosis less than 50% were not enrolled the study.
Controls
For each case, one age (in 5- year bands) and sex-matched individual with normal coronary angiogram was enrolled as control. Individuals who do not have enough hair at the back of the head, are on lipid-lowering therapies, suffering from any documented disease, having minimal coronary artery stenosis were not enrolled in the study.
2.4. Estimation of biochemical parameters
Biochemical parameters including TC, TG, HDL, fasting plasma glucose, LDL-C, and Hb1Ac were measured with Beckman Coulter Au 480 automatic chemistry analyzer.
2.5. Cortisol immunosorbent assay
2.5.1. Collection of the hair sample
A hair sample equal to the half diameter of a lead pencil was collected non-invasively from the back of each individual’s head by cutting close to the scalp with clean surgical scissors by the study personnel (Cooper et al., Citation2012). The scalp end of the hair sample was aligned, tied with thread, and affixed with tape to collection form. The collection forms were placed in zip bags and kept at room temperature until analysis.
2.5.2. Quantification of hair cortisol
The most proximal 3 cm part was snipped from the hair segment and 10 mg of hair was weighed precisely to 0.01 mg using an analytical balance. Analysis of the most proximal 3 cm segment of hair gives information about 3 months of systemic cortisol exposure. Hair samples were placed in a glass vial, washed twice by immersing in 2.5mlof isopropanol for two minutes at room temperature, and left to dry overnight (Kirschbaum et al., 2009). Dried hair specimens were then minced into small granules with surgical scissors. The granular hair mass was transferred to a 2 ml cryovial and 1 ml of (HPLC grade) methanol was added. The vials were sealed with tape to minimize evaporation and incubated for 16 h in a water bath at 52 °C with constant shaking(Van Uum et al., Citation2008). Next, the sample vials were spun at 10,000 rpm for 2 min, and approximately 0.8 ml of the supernatant was transferred into a sterile 2 ml cryovial. The methanol was evaporated at 50 °C until the sample is fully dried (takes almost 20 min). The vaporization left a white residue that was reconstituted by adding 250 µl of phosphate-buffered saline solution (PBS) at pH 8.0 (Stalder et al., Citation2010) and stored at −80 °C till further analysis. Cortisol concentration was measured with ELISA salivary cortisol kit (Alpco Diagnostics, Salem, NH), according to the manufacturer’s instructions. Internal quality control was prepared by pooling different hair extracts and run to ensure the reproducibility of results. 20 µl PBS reconstituted hair cortisol extracts and quality control standard was loaded in the wells. Absorption was read at 450 nm on a Vmax plate reader. The ELISA kit’s limit of detection is 1.14 ng/ml. Average optical densities of standards, controls, and samples were calculated.
2.6. Statistical analysis
Study participants were stratified by case and control for analysis. Scale (continuous) variables were presented as mean ± Standard Deviation (SD) and median(minimum-maximum). Categorical variables were described as frequency (percentages). Baseline data of cases and controls were compared using Chi-square test, Student’s t-test, and Wilcoxon rank-sum test, as appropriate for skewed or normal distribution (Fay & Proschan, Citation2010). The comparison among different vascular anatomies was carried out using the Kruskal-Wallis test with Dunn’s multiple comparisons test. Association between HCC and continuous variables (classic risk factors) was assessed by Spearman rank correlation. Subjects on antidiabetic drugs (Four percent of cases and seven percent of controls) were excluded from the correlation analysis. After the log10 transformation of HCC, univariate and multivariate conditional logistic regression analyses were executed to determine the independent association of various risk factors with the manifestation of coronary atherosclerosis (Pearce, Citation2016). Receiver operator curve (ROC) analysis was done to estimate the discriminative ability of the hair cortisol concentration to predict the incidence of coronary atherosclerosis (Hanley & McNeil,1982). Statistical Package for Social Sciences (SPSS) version 25 and Graph Pad Prism version 7 were used to analyze the data. For all analyses, two-tailed p-values were calculated.
3. Results
The mean age of the study population (n = 1000) was 51.69 ± 9.51 years. The major age group (38.0%) affected by angiographically documented coronary atherosclerosis was 46–-55 years old. Among cases, 25.8% unstable angina, 17.8% had non-ST segment elevation myocardial infarction, and 56.4% ST-elevation myocardial infarction. A significant difference in the prevalence of obesity, diabetes, hypertension, positive family history of CVDs, physical inactivity, and smoking status was observed between the case and control group. The higher percentage of cases belonged to low socioeconomic status and were stressed as compared to the control group. Mean levels of triglycerides, LDL-C, and HCC were significantly raised in cases compared to controls ().
Table 1. Clinical characteristics of the subjects at the time of enrollment in the study.
Table 2. Correlation of Hair Cortisol levels with classic risk factors for coronary atherosclerosis.
Hair cortisol concentration
Mean HCC were significantly high in patients with coronary atheroscrelosis (226.82 ± 212.1 pg/mg) compared to controls (85.38 ± 67.83 pg/mg mean), median [158.86(17.65–1331.33)] vs [72.77(13.41–889.07)] pg/mg; . Median hair cortisol concentrations were significantly high in hypertensive subjects compared to normotensive 128.8(21.84,1331) vs 80.94 (13.41,1043) . Mean HCC in different vascular anatomies were as follow, SVD (mean = 191.3 pg/mg; SE = 13.39), DVD (mean = 209.5 pg/mg; SE = 14.99) and TVD (mean = 292.1 pg/mg; SE = 20.69). Comparisons of HCC among different vascular anatomies showed a significant difference for SVD vs TVD (p-value = 0.0029) and DVD vs TVD (p-value = 0.0189) as well as highly significant differences were observed for normal vs SVD, normal vs DVD, normal vs (for all p-value ≤ 0.0001) .
Figure 1. (A) Wilcoxon test for comparison of mean hair cortisol concentration in cases and controls. (B) Mann Whitney U test for comparison of hair cortisol concentration in hypertensive and normotensive subjects. (C) Kruskal Wallis test for comparison of hair cortisol in different vascular anatomies. (D) Kruskal Wallis test for comparison of hair cortisol concentration in different BMI group. (E) Receiver operator curve analysis to determine the cut off value of hair cortisol concentration to predict the risk of coronary atheroscrelosis.
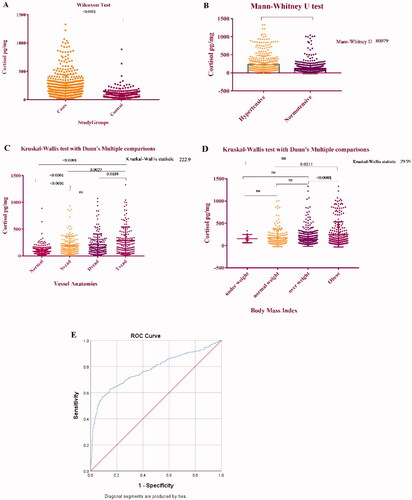
Additionally, median HCC was higher in obese subjects 126.6(19.35,1331) vs 104.34 (16.13,1005.88) compared to normal-weight individuals .
Correlation of Hair Cortisol Concentration with classic risk factors for coronary atherosclerosis ()
In multivariate regression analysis including all classic CVDs risk factors, HCC matched odd ratio (MOR), 10.73 95%CI; p-value = 0.001 and family history MOR 5.48; 95% CI (1.76–17.05) p-value= 0.003) remained the strong predictors of coronary atheroscrelosis whereas BMI(MOR), 1.23 95%CI; p-value = 0.028 and LDL-C; MOR,1.02; 95% CI; p-value= 0.006, showed a moderate association with coronary atheroscrelosis ()
Table 3. Univariate and Multivariate Conditional logistic regression analysis (Enter Method).
ROC curve analysis for hair cortisol concentration and coronary atherosclerosis
ROC analyses were conducted to explore the diagnostic potential of HCC . Cut off the value of HCC ≥104.07 pg/mg (AUC 0.775; 95% CI, 0.745–0.802; p < 0.001) showed the optimum balance of sensitivity (70.2%) and specificity (72.0%).
4. Discussion
A significantly high mean HCC was observed in patients with angiographically confirmed coronary atherosclerosis compared to controls. These findings are in line with other recently conducted case-control studies where higher HCC is found to be linked with different forms of CVD. Izawa et al. (Citation2019) found higher HCC in patients with acute coronary syndrome (Izawa et al., Citation2019). Similarly, an association between higher HCC and CHD (Bossé et al., Citation2018), aneurysmal subarachnoid hemorrhage (Colledge et al., Citation2017) recent event of AMI (Pereg et al., Citation2011), compared to controls was reported by other research groups. Pereg et al. (Citation2011) assessed HCC in AMI patients at presentation to hospital and compared it with the HCC level of 56 patients hospitalized for other diseases. Hair cortisol was observed to be the significant predictor of AMI after adjusting for other CVDs risk factors (Pereg et al., Citation2011). In another study, elevated HCC is linked with the severity of heart failure (Pereg et al., Citation2013). In a recent Swedish study significantly higher median HCC was observed in AMI patients compared with controls (Faresjö et al., Citation2020). Additionally, in a study conducted on elder citizens, hair cortisol concentration (fourth HCC quartile) was found to be linked with a 2.7 times increased risk for cardiac ailments including peripheral artery disease, CHD, and stroke (Manenschijn et al., Citation2013). Contrarily, Abell et al. (Citation2016) found no association raised HCC and CHD or stroke (Abell et al., Citation2016).
Cortisol’s role in the development of certain CVDs risk factors such as diabetes, obesity, and hypertension, cannot be ignored due to its key involvement in glucose and lipid metabolism, blood pressure, and immune responses regulation (Girod and Brotman, Citation2004). Elevated HCC showed a positive association with the number of CVDs risk factors that is BMI and waist circumference (Jackson et al., Citation2017) in adults, triglycerides (Kuehl et al., Citation2015), cholesterol (Mazgelytė et al., Citation2019; Stalder et al., Citation2013) hypertension (Bautista et al., Citation2019) and glycated hemoglobin (Lehrer et al., Citation2016). These chronic stress-induced ailments may further contribute to elevated CVD risk.
Elevated HCC showed a significant correlation with BMI was in the current study. Chronic stress-induced cortisol exposure over a longer period is related to obesity, metabolic syndrome, and CVDs. The findings of this study collude with previous data which stated a significant positive association between HCC, BMI, and abdominal obesity (Jackson et al., Citation2017; Stalder et al., Citation2013) concurring the hypothesis that a minor rise in longstanding cortisol exposure is related to increased adiposity. Metabolic derangements induced by high HCC may foster atherogenesis culminating in the adverse cardiac event.
Our findings of high HCC in hypertensive participants compared with normotensive individuals are in line with those of (Bautista et al., Citation2019; Manenschijn et al., Citation2013; Mazgelytė et al., Citation2019) who reported a significant association between hypertension and elevated HCC levels while differing from others who could not establish a link between systolic blood pressure and HCC (Saleem et al., Citation2013; Younge et al., Citation2015) These varying outcomes could be attributed to the differences in characteristics of the study samples. Some patients may be using antihypertensive medicines.
Our findings of a significant positive correlation between elevated HCC and Hb1Ac are in line with recent data which support a statistically positive relationship between raised HCC and diabetes (Lehrer et al., Citation2016; Stalder et al., Citation2013). Elevated HCC was positively linked with diabetes in 654 middle to old aged participants from the German cohort, (Feller et al., Citation2014). In another study conducted on 3507 adults, positive associations between raised HCC and diabetes were reported (Abell et al., Citation2016). Likewise, a positive relationship between higher HCC and diabetes in 760 adults was reported by (Staufenbiel et al., Citation2015).
Elevated HCC has also been linked to adverse lipid biomarkers (Kuehl et al., Citation2015; Lehrer et al., Citation2016; Mazgelytė et al., Citation2019) however these associations were not significant in some studies (Wester et al., Citation2017). We like others (Kuehl et al., Citation2015; Wester et al., Citation2017) observed no significant relationship between increased HCC and raised cholesterol levels contrary to the findings of Stalder et al. (Citation2013) who reported a positive association between elevated HCC and cholesterol. We found no correlation between HCC and HDL-C levels (Stalder et al., Citation2013). These controversial results may arise due to the inclusion of participants with a wider age bracket and disease status.
In the current study, no significant association between HCC and current smoking status was observed which colludes with (Dettenborn et al., Citation2012) but is inconsistent with others who reported a significantly high HCC in current smokers compared with nonsmokers (Feller et al., Citation2014). These unclear results may arise from the fact that the association between smoking and HCC may be dependent on the period as well as the number of units smoked per day which is not evaluated yet.
Previous data indicate that chronic stress elevates CVDs risk ∼ by 40–60% (Steptoe & Kivimäki, Citation2012). Since the response to the perceived psychosocial stress leads to the activation of the HPA axis and elevated concentrations of cortisol which is frequently used as a biological marker of stress. It is hypothesized that cortisol acts as the mediator between stress and CVDs. Cortisol regulates lipid metabolism by promoting lipolysis in the peripheral fat depots and increasing lipogenesis in visceral fat tissue (Staufenbiel et al., Citation2013). It acts on renal mineralocorticoid receptors to retain salt and water, resulting in bigger plasma volume and modulation of blood pressure (Staufenbiel et al., Citation2013). Furthermore, persistently activated HPA axis and cortisol exposure leads to glucocorticoid resistance and a diminished ability of cortisol to suppress proinflammatory cytokine synthesis and induction of anti-inflammatory cytokine secretion (Tian et al., Citation2014). Excessive glucocorticoids secretions not only induce CVD risk factors but also speed up progression and the incidence of atherosclerotic cardiovascular disease (Walker, Citation2007).
We observed a significant association between HCC and perceived stress after univariate analysis but these results turned insignificant when adjusted for classic risk factors. Likewise, diabetes and hypertension could not attain statistical significance in the multivariate analysis. This may be attributed to medical treatment in these patients.
Clinical implications
This study has several clinical implications. Analysis of HCC may provide a reliable biological marker for the quantification of retrospective chronic stress exposure in neuropshycobiology studies. Assessment of HCC may help to identify the subjects at high risk of coronary atherosclerosis.
In conclusion, our findings suggest that elevated hair cortisol concentration, a biological marker of chronic stress, is a strong predictor of angiographically confirmed coronary atherosclerosis. HCC is significantly correlated with the classic CVD risk factors diabetes and obesity. Chronic stress-induced diabetes and obesity may have contributed to the elevated concentrations of hair cortisol and increased the risk for the development of coronary atherosclerosis.
Our study has some limitations.
Due to the case-control study design, any cause-effect relationships could not be determined. Secondly, a significant number of patients could not be enrolled in the study due to thinning of hair or baldness. Thirdly, due to the low literacy level of the participants, questionnaire-based information was collected through the interview which may have led to the reporting bias in some individuals.
Author contributions
AN carried out experiments, analyzed the data, and wrote the manuscript.
FH planned and supervised the study. MQ and MG provide technical assistance and helped in data analysis and manuscript writing.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Correction Statement
This article has been corrected with minor changes. These changes do not impact the academic content of the article.
Additional information
Funding
References
- Abell, J. G., Stalder, T., Ferrie, J. E., Shipley, M. J., Kirschbaum, C., Kivimäki, M., & Kumari, M. (2016). Assessing cortisol from hair samples in a large observational cohort: The Whitehall II study. Psychoneuroendocrinology, 73, 148–156. https://doi.org/https://doi.org/10.1016/j.psyneuen.2016.07.214
- Arnaldi, G., Angeli, A., Atkinson, A. B., Bertagna, X., Cavagnini, F., Chrousos, G. P., Fava, G. A., Findling, J. W., Gaillard, R. C., Grossman, A. B., Kola, B., Lacroix, A., Mancini, T., Mantero, F., Newell-Price, J., Nieman, L. K., Sonino, N., Vance, M. L., Giustina, A., & Boscaro, M. (2003). Diagnosis and complications of Cushing’s syndrome: A consensus statement. The Journal of Clinical Endocrinology & Metabolism, 88(12), 5593–5602. https://doi.org/https://doi.org/10.1210/jc.2003-030871
- Bautista, L. E., Bajwa, P., Shafer, M. M., Malecki, K., McWilliams, C. A., & Palloni, A. (2019). The relationship between chronic stress, hair cortisol and hypertension. International Journal of Cardiology. Hypertension, 2, 100012.
- Bossé, S., Stalder, T., & D’Antono, B. (2018). Childhood trauma, perceived stress, and hair cortisol in adults with and without cardiovascular disease. Psychosomatic Medicine, 80(4), 393–402. https://doi.org/https://doi.org/10.1097/PSY.0000000000000569
- Brotman, D. J., Golden, S. H., & Wittstein, I. S. (2007). The cardiovascular toll of stress. The Lancet, 370(9592), 1089–1100. https://doi.org/https://doi.org/10.1016/S0140-6736(07)61305-1
- Cohen, S., Kamarck, T., & Mermelstein, R. (1983). A global measure of perceived stress. Journal of Health and Social Behavior, 24(4), 385. https://doi.org/https://doi.org/10.2307/2136404
- Colledge, F., Brand, S., Zimmerer, S., Pühse, U., Holsboer-Trachsler, E., & Gerber, M. (2017). In individuals following aneurysmal subarachnoid haemorrhage, hair cortisol concentrations are higher and more strongly associated with psychological functioning and sleep complaints than in healthy controls. Neuropsychobiology, 75(1), 12–20. https://doi.org/https://doi.org/10.1159/000477966
- Cooper, G. A., Kronstrand, R., & Kintz, P. (2012). Society of Hair Testing guidelines for drug testing in hair. Forensic Science International, 218(1–3), 20–24.
- Craig, C. L., Marshall, A. L., Sjöström, M., Bauman, A. E., Booth, M. L., Ainsworth, B. E., Pratt, M., Ekelund, U., Yngve, A., Sallis, J. F., & Oja, P. (2003). International physical activity questionnaire: 12-country reliability and validity. Medicine and Science in Sports and Exercise, 35(8), 1381–1395. https://doi.org/https://doi.org/10.1249/01.MSS.0000078924.61453.FB 12900694
- Dettenborn, L., Tietze, A., Kirschbaum, C., & Stalder, T. (2012). The assessment of cortisol in human hair: Associations with sociodemographic variables and potential confounders. Stress (Amsterdam, Netherlands), 15(6), 578–588. https://doi.org/https://doi.org/10.3109/10253890.2012.654479
- Dupre, M. E., George, L. K., Liu, G., & Peterson, E. D. (2015). Association between divorce and risks for acute myocardial infarction. Circulation: Cardiovascular Quality and Outcomes, 8(3), 244–251.
- Faresjö, T., Strömberg, S., Jones, M., Stomby, A., Karlsson, J.-E., Östgren, C. J., Faresjö, Å., & Theodorsson, E. (2020). Elevated levels of cortisol in hair precede acute myocardial infarction. Scientific Reports, 10(1), 22456–22458.
- Fay, M. P., & Proschan, M. A. (2010). Wilcoxon-Mann-Whitney or t-test? On assumptions for hypothesis tests and multiple interpretations of decision rules. Statistics Surveys, 4, 1–39. https://doi.org/https://doi.org/10.1214/09-SS051
- Feller, S., Vigl, M., Bergmann, M. M., Boeing, H., Kirschbaum, C., & Stalder, T. (2014). Predictors of hair cortisol concentrations in older adults. Psychoneuroendocrinology, 39, 132–140.
- Girod, J. P., & Brotman, D. J. (2004). Does altered glucocorticoid homeostasis increase cardiovascular risk?. Cardiovascular Research, 64(2), 217–226. https://doi.org/https://doi.org/10.1016/j.cardiores.2004.07.006 15485680
- Izawa, S., Miki, K., Tsuchiya, M., Yamada, H., & Nagayama, M. (2019). Hair and fingernail cortisol and the onset of acute coronary syndrome in the middle-aged and elderly men. Psychoneuroendocrinology, 101, 240–245.
- Jackson, S. E., Kirschbaum, C., & Steptoe, A. (2017). Hair cortisol and adiposity in a population‐based sample of 2,527 men and women aged 54 to 87 years. Obesity, 25(3), 539–544. https://doi.org/https://doi.org/10.1002/oby.21733
- Kirschbaum, C., Tietze, A., Skoluda, N., & Dettenborn, L. (2009). Hair as a retrospective calendar of cortisol production—increased cortisol incorporation into hair in the third trimester of pregnancy. Psychoneuroendocrinology, 34(1), 32–37. https://doi.org/https://doi.org/10.1016/j.psyneuen.2008.08.024
- Kivimäki, M., & Steptoe, A. (2018). Effects of stress on the development and progression of cardiovascular disease. Nature Reviews. Cardiology, 15(4), 215–229.
- Kuehl, L. K., Hinkelmann, K., Muhtz, C., Dettenborn, L., Wingenfeld, K., Spitzer, C., Kirschbaum, C., Wiedemann, K., & Otte, C. (2015). Hair cortisol and cortisol awakening response are associated with criteria of the metabolic syndrome in opposite directions. Psychoneuroendocrinology, 51, 365–370. https://doi.org/https://doi.org/10.1016/j.psyneuen.2014.09.012
- Lehrer, H. M., Dubois, S. K., Maslowsky, J., Laudenslager, M. L., & Steinhardt, M. A. (2016). Hair cortisol concentration and glycated hemoglobin in African American adults. Psychoneuroendocrinology, 72, 212–218.
- Manenschijn, L., Schaap, L., van Schoor, N. M., van der Pas, S., Peeters, G. M. E. E., Lips, P., Koper, J. W., & van Rossum, E. F. C. (2013). High long-term cortisol levels, measured in scalp hair, are associated with a history of cardiovascular disease. The Journal of Clinical Endocrinology & Metabolism, 98(5), 2078–2083. https://doi.org/https://doi.org/10.1210/jc.2012-3663
- Mathur, M. B., Epel, E., Kind, S., Desai, M., Parks, C. G., Sandler, D. P., & Khazeni, N. (2016). Perceived stress and telomere length: A systematic review, meta-analysis, and methodologic considerations for advancing the field. Brain, Behavior, and Immunity, 54, 158–169. https://doi.org/https://doi.org/10.1016/j.bbi.2016.02.002
- Mazgelytė, E., Karčiauskaitė, D., Linkevičiūtė, A., Mažeikienė, A., Burokienė, N., Matuzevičienė, R., Radzevičius, M., Janiulionienė, A., Jakaitienė, A., Dindienė, L., & Kučinskienė, Z. A. (2019). Association of hair cortisol concentration with prevalence of major cardiovascular risk factors and allostatic load. Medical Science Monitor : International Medical Journal of Experimental and Clinical Research, 25, 3573–3582. https://doi.org/https://doi.org/10.12659/MSM.913532
- McEwen, B. S. (2012). Brain on stress: how the social environment gets under the skin. Proceedings of the National Academy of Sciences of the United States of America, 109 Suppl 2(Supplement 2), 17180–17185. https://doi.org/https://doi.org/10.1073/pnas.1121254109
- Miller, G. E., & Blackwell, E. (2006). Turning up the heat: Inflammation as a mechanism linking chronic stress, depression, and heart disease. Current Directions in Psychological Science, 15(6), 269–272. https://doi.org/https://doi.org/10.1111/j.1467-8721.2006.00450.x
- Nabi, H., Kivimäki, M., Batty, G. D., Shipley, M. J., Britton, A., Brunner, E. J., Vahtera, J., Lemogne, C., Elbaz, A., & Singh-Manoux, A. (2013). Increased risk of coronary heart disease among individuals reporting adverse impact of stress on their health: The Whitehall II prospective cohort study. European Heart Journal, 34(34), 2697–2705. https://doi.org/https://doi.org/10.1093/eurheartj/eht216
- Nafisa, A., Gray, S. G., Cao, Y., Wang, T., Xu, S., Wattoo, F. H., Barras, M., Cohen, N., Kamato, D., & Little, P. J. (2018). Endothelial function and dysfunction: Impact of metformin. Pharmacology & Therapeutics, 192, 150–162. https://doi.org/https://doi.org/10.1016/j.pharmthera.2018.07.007
- Pearce, N. (2016). Analysis of matched case-control studies. BMJ, 352, i969. https://doi.org/https://doi.org/10.1136/bmj.i969
- Pereg, D., Chan, J., Russell, E., Berlin, T., Mosseri, M., Seabrook, J. A., Koren, G., & Van Uum, S. (2013). Cortisol and testosterone in hair as biological markers of systolic heart failure. Psychoneuroendocrinology, 38(12), 2875–2882. https://doi.org/https://doi.org/10.1016/j.psyneuen.2013.07.015
- Pereg, D., Gow, R., Mosseri, M., Lishner, M., Rieder, M., Van Uum, S., & Koren, G. (2011). Hair cortisol and the risk for acute myocardial infarction in adult men. Stress (Amsterdam, Netherlands), 14(1), 73–81. https://doi.org/https://doi.org/10.3109/10253890.2010.511352
- Rosenthal, R. L. (2015). The 50% coronary stenosis. The American Journal of Cardiology, 115(8), 1162–1165. https://doi.org/https://doi.org/10.1016/j.amjcard.2015.01.553
- Roth, G. A., Abate, D., Abate, K. H., Abay, S. M., Abbafati, C., Abbasi, N., Abbastabar, H., Abd-Allah, F., Abdela, J., Abdelalim, A., Abdollahpour, I., Abdulkader, R. S., Abebe, H. T., Abebe, M., Abebe, Z., Abejie, A. N., Abera, S. F., Abil, O. Z., Abraha, H. N., … Murray, C. J. L. (2018). Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. The Lancet, 392(10159), 1736–1788. https://doi.org/https://doi.org/10.1016/S0140-6736(18)32203-7
- Saleem, M., Herrmann, N., Swardfager, W., Oh, P. I., Shammi, P., Koren, G., Van Uum, S., Kiss, A., & Lanctôt, K. L. (2013). Higher cortisol predicts less improvement in verbal memory performance after cardiac rehabilitation in patients with coronary artery disease. Cardiovascular Psychiatry and Neurology, 2013, 340342. https://doi.org/https://doi.org/10.1155/2013/340342
- Stalder, T., Kirschbaum, C., Alexander, N., Bornstein, S. R., Gao, W., Miller, R., Stark, S., Bosch, J. A., & Fischer, J. E. (2013). Cortisol in hair and the metabolic syndrome. The Journal of Clinical Endocrinology and Metabolism, 98(6), 2573–2580. https://doi.org/https://doi.org/10.1210/jc.2013-1056
- Stalder, T., Kirschbaum, C., Heinze, K., Steudte, S., Foley, P., Tietze, A., & Dettenborn, L. (2010). Use of hair cortisol analysis to detect hypercortisolism during active drinking phases in alcohol-dependent individuals. Biological Psychology, 85(3), 357–360. https://doi.org/https://doi.org/10.1016/j.biopsycho.2010.08.005
- Staufenbiel, S. M., Penninx, B. W. J. H., De Rijke, Y. B., Van Den Akker, E. L. T., & Van Rossum, E. F. C. (2015). Determinants of hair cortisol and hair cortisone concentrations in adults. Psychoneuroendocrinology, 60, 182–194. https://doi.org/https://doi.org/10.1016/j.psyneuen.2015.06.011 26176863
- Staufenbiel, S. M., Penninx, B. W., Spijker, A. T., Elzinga, B. M., & van Rossum, E. F. (2013). Hair cortisol, stress exposure, and mental health in humans: A systematic review. Psychoneuroendocrinology, 38(8), 1220–1235.
- Steptoe, A., & Kivimäki, M. (2012). Stress and cardiovascular disease. Nature Reviews. Cardiology, 9(6), 360–370. https://doi.org/https://doi.org/10.1038/nrcardio.2012.45
- Sumner, J. A., Khodneva, Y., Muntner, P., Redmond, N., Lewis, M. W., Davidson, K. W., Edmondson, D., Richman, J., & Safford, M. M. (2016). Effects of concurrent depressive symptoms and perceived stress on cardiovascular risk in low‐and high‐income participants: Findings from the Reasons for Geographical and Racial Differences in Stroke (REGARDS) study. Journal of the American Heart Association, 5(10), e003930. https://doi.org/https://doi.org/10.1161/JAHA.116.003930
- Tian, R., Hou, G., Li, D., & Yuan, T.-F. (2014). A possible change process of inflammatory cytokines in the prolonged chronic stress and its ultimate implications for health. The Scientific World Journal, 2014, 1–8. https://doi.org/https://doi.org/10.1155/2014/780616
- Van Uum, S., Sauvé, B., Fraser, L., Morley-Forster, P., Paul, T., & Koren, G. (2008). Elevated content of cortisol in hair of patients with severe chronic pain: A novel biomarker for stress. Stress, 11(6), 483–488. https://doi.org/https://doi.org/10.1080/10253890801887388
- Volgman, A. S., Palaniappan, L. S., Aggarwal, N. T., Gupta, M., Khandelwal, A., Krishnan, A. V., Lichtman, J. H., Mehta, L. S., Patel, H. N., Shah, K. S., Shah, S. H., & Watson, K. E. (2018). Atherosclerotic cardiovascular disease in South Asians in the United States: Epidemiology, risk factors, and treatments: A scientific statement from the American Heart Association. Circulation, 138(1), e1–e34. https://doi.org/https://doi.org/10.1161/CIR.0000000000000580
- von Känel, R. (2012). Psychosocial stress and cardiovascular risk : current opinion. Swiss Medical Weekly, 142, w13502. https://doi.org/https://doi.org/10.4414/smw.2012.13502
- Walker, B. R. (2007). Glucocorticoids and cardiovascular disease. European Journal of Endocrinology, 157(5), 545–559. https://doi.org/https://doi.org/10.1530/EJE-07-0455
- Wester, V. L., Noppe, G., Savas, M., van den Akker, E. L., de Rijke, Y. B., & van Rossum, E. F. (2017). Hair analysis reveals subtle HPA axis suppression associated with use of local corticosteroids: The Lifelines cohort study. Psychoneuroendocrinology, 80, 1–6. https://doi.org/https://doi.org/10.1016/j.psyneuen.2017.02.024
- WHO. (2018). World Health Organization–Noncommunicable Diseases (NCD) country profiles. WHO.
- Xu, X., Bao, H., Strait, K., Spertus, J. A., Lichtman, J. H., D’Onofrio, G., Spatz, E., Bucholz, E. M., Geda, M., Lorenze, N. P., Bueno, H., Beltrame, J. F., & Krumholz, H. M. (2015). Sex differences in perceived stress and early recovery in young and middle-aged patients with acute myocardial infarction. Circulation, 131(7), 614–623. https://doi.org/https://doi.org/10.1161/CIRCULATIONAHA.114.012826
- Younge, J. O., Wester, V. L., van Rossum, E. F. C., Gotink, R. A., Wery, M. F., Utens, E. M. W. J., Hunink, M. G. M., & Roos-Hesselink, J. W. (2015). Cortisol levels in scalp hair of patients with structural heart disease. International Journal of Cardiology, 184, 71–78. https://doi.org/https://doi.org/10.1016/j.ijcard.2015.02.005
