?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.1. Introduction
Cartilage is a non-vascularized organ in which cells (i.e. chondrocytes) represent only 2% of the global volume. Thus, cartilage exhibits a very limited ability to repair. Cartilage diseases represent an important public health problem, whose prevalence increases with the population ageing. Main used treatments (e.g. arthroscopic lavage, synthetic implants, periosteal and perichondral transplantation, osteochondral allograft and autograft…) are generally non-lasting and may lead to inflammatory reactions. Furthermore, mechanical properties of the repaired cartilage are generally inferior to those of the native one. Therefore, there is a need in better understanding the main mechanisms governing the cartilage homeostasis, notably its growth process, and the corresponding major influencing parameters.
As a result, many mechanical or biological studies have been performed the last decades over the world. However, very few published data are based on the crossing of both mechanical and biological analysis on an in vitro model, excepted those performed on scaffold including seeded cells. This is specially the case for cartilage micropellet that is the standard in vitro model of cartilage growth from mesenchymal stem cells (MSC) to late hypertrophic chondrocytes (Barry et al. Citation2001). Indeed, this experimental model aims at representing the developmental stages of cartilage. To our knowledge, no study has investigated MSC-based micropellet.
Present work precisely aims at studying the generation of MSC-based cartilage micropellets over 5 weeks in culture, crossing biological and mechanical analysis, and at determining possible correlations between molecular, histological and mechanical markers of cartilage growth.
2. Methods
2.1. Cartilage micropellet culture
The in vitro cartilage micropellets were obtained from the differentiation of human MSC (isolated from bone marrow) into chondrocytes by 3D-culture in presence of the inducing factor TGFβ3. As control, micropellets were cultured without TGFβ3. All TGFβ3-based micropellets were then cultured up to 5 weeks and both biological analysis (i.e. RT-qPCR and immunocytochemistry) and mechanical characterisation (i.e. compression tests) were performed after respectively 7, 14, 21, 29 and 35 days of culture.
2.2. Molecular analysis
Molecular analysis of micropellets used RT-qPCR. Total RNA was extracted from micropellets by using the RNeasy kit (Qiagen). RNA (0.5 µg) was reverse transcribed using the M-MLV reverse transcriptase and PCR reactions performed using specific primers as described in (Maumus et al. Citation2017). Gene level is expressed as relative expression (2 − ΔCt) and normalized on the expression of RSP9 housekeeping gene.
2.3. Histologic analysis
Quality of cartilage matrix production was determined by immunohistochemical analysis. Micropellets were processed for routine histology and deparaffinized sections (3 µm) were stained with Safranin O/Fast for proteoglycan visualization. We used a 1/1000 dilution of anti-aggrecan antibody or 1/50 dilution of anti-type II collagen, and the Ultravision detection system anti-polyvalent HRP/DAB kit for immunohistochemistry.
2.4. Mechanical characterisation
Each micropellet was kept in 5 mL of phosphate buffered saline (PBS) at room temperature (22 °C ± 1 °C) until analysis. Micropellets were then placed between two platens for overall compression that consisted in one cycle of loading with an imposed displacement of 200 µm for a total duration of 1 s (0.5 s at constant speed for the compression and 0.5 s at constant speed to get back to the initial position). Both the force and the displacement were acquired simultaneously at 100 Hz. The force was measured with a miniature S Beam load cell.
Both dissipative and hyperelastic behaviors were then characterised. On the one hand, the dissipated energy for the complete cycle was computed by using the difference between the integrals of the force-displacement curves during loading (compression) and unloading (return to initial state). On the other hand, the hyperelastic density energy similar to the one proposed by Fung (Citation1967) coupled with a finite element (FE) model of a deformable sphere was implemented using lab-made LMGC90 software. The constitutive isotropic hyperelastic behavior of the micropellet was described by a Fung energy function:
Where E is the Young’s modulus representing the initial stiffness of the micropellet at small strains, I1 the first invariant of the Cauchy-Green right tensor, the Poisson’s ratio and b a parameter corresponding to the strain-hardening behavior of the sample. A quasi-incompressible behavior was implemented with a Poisson’s ratio of 0.499. A contact law without friction was used between the rigid compression platen and the deformable sphere. A non-linear minimization procedure was used to fit the force from the FE model to the experimental force, in order to identify the Young’s modulus E and the parameter b.
3. Results and discussion
Micropellets cultured with TGFβ3 exhibited a significantly higher expression of cartilage-specific genes (SOX9, type IIB collagen (COL2A1Δ2), aggrecan (ACAN)) than MSCs at day 0 or undifferentiated micropellets generated from MSCs cultured without growth factor. Interestingly, the corresponding initial Young’s moduli, estimated by using the FE model, increased by more than a 5.3 fold factor in the differentiated micropellets (160.4 ± 17.5 kPa) compared to the undifferentiated ones (32.8 ± 8.3 kPa, p < 0.001). In parallel, the dissipated energy of differentiated micropellets was 3.4 fold higher than the dissipated energy of undifferentiated micropellets at day 21 (307 ± 46 × 10 − 9 J vs 88 ± 24 × 10 − 9 J, p < 0.05).
Furthermore, molecular, histological and mechanical characterisation made on differentiated micropellets up to 5 culture weeks, show that all markers (i.e., cartilage-specific genes, accumulation of proteoglycans and type II collagen, Young’s modulus and dissipated energy) increased significantly over time. In particular, Young’s modulus of micropellets significantly increased from day 14 to day 29 (from 12.1 ± 2.3 kPa to 179.9 ± 18.8 kPa), and then stabilized at 168.9 ± 28.6 kPa by day 35. The non-linear behavior of micropellets, b, increased from day 14 to day 21, (respectively 3.8 ± 2.3 and 12.4 ± 1.1, p < 0.01), and then maintained until day 35 (16.1 ± 3.1, p > 0.05). Similarly, the dissipated energy increased with culture time until day 29 and then stabilized at day 35 (226 ± 39 × 10 − 9 J).
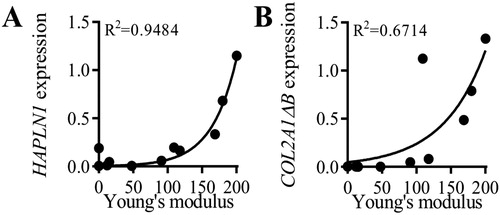
Finally and remarkably, we found a significant correlation between the expression of both link protein and type IIB collagen genes, and the Young’s modulus (). Similar correlations between the dissipative energy and these two chondrogenic markers were also found.
4. Conclusions
Present results show that MSC-based micropellets cultured with TGFβ3 exhibit a high correlation between chondrocyte marker expression and extracellular matrix mechanical stiffness. This highlights the potentiality of using cartilage micropellets as a relevant in vitro biomechanical and mechanobiological model of cartilage growth. Indeed, the cartilage micropellet model meets both biochemical and biomechanical characteristics required to investigate and recapitulate all stages of cartilage development. Current study used micropellet generated without any mechanical stimulation and future works will evaluate the impact of loading during micropellet culture. This will allow to discriminate the influence of mechanical loading versus biochemical treatments.
Acknowledgements
We thank S. Devic, G. Camp and P. Valorge from SERVEX (LMGC) for the design and the fabrication of the mechanical setup. M. Maumus and G. Dusfour contributed equally to this work.
Additional information
Funding
References
- Barry F, Boynton RE, Liu B, Murphy JM. 2001. Chondrogenic differentiation of mesenchymal stem cells from bone marrow: differentiation-dependent gene expression of matrix components. Exp. Cell Res. 268(2):189–200.
- Fung Y.C. 1967. Elasticity of soft tissues in simple elongation. Am J Physiol. 213(6):1532–1544.
- Maumus M, Manferdini C, Toupet K, Chuchana P, Casteilla L, Gachet M, Jorgensen C, Lisignoli G, Noël D. 2017. Thrombospondin-1 partly mediates the cartilage protective effect of adipose-derived mesenchymal stem cells in osteoarthritis. Front Immunol. 8:1638.