ABSTRACT
Electrical stimulation has been used to treat spasticity in children with cerebral palsy. Building on the benefits of electrical stimulation, a new assistive device, electro-suit Mollii® with imbedded electrodes has been tested. The aim of the study was to evaluate the possible effect of Mollii® on body function, activity and participation in self-selected activities. Six children, five to ten years of age, used the electro-suit for one hour, every other day for three months. The impact was evaluated after four weeks and after three months by measuring passive range of motion (ROM), muscle tone, pain, gross motor function and participation. All participants improved in the total score for Canadian Occupational Performance Measure (COPM), three of them showed significant clinical improvements. Pain was reduced for children who estimated pain when the study started. There were also small changes in Electro-suit Mollii® had a positive impact on activity and participation in self-selected activities among the children in this study. Further studies with more children over a longer time are necessary to evaluate the impact and usefulness over time.
Cerebral palsy (CP) is the most common neurological disability in children and youth, and in Sweden, its incidence is about 2.18 per 1000 births (Himmelmann & Uvebrant, Citation2014). CP is a clinical description of a chronic functional disability caused by a non-progressive lesion of the developing central nervous system on the human infant brain. The motor disorders are often accompanied by disturbances of sensation, cognition, perception, communication, secondary musculoskeletal impairments, and pain (P Rosenbaum et al., Citation2007). Spasticity is a common problem, and while the prevalence differs depending on the etiology, appears in about 80% of the persons with CP (Shevell et al., Citation2009). Spasticity is defined as a motor disorder characterized by a velocity-dependent increase in tonic stretch reflex and exaggerated tendon jerks (Lance, Citation1980). Furthermore, pain and spasticity often occur simultaneously in patients, and spasticity can be induced by sensory stimuli; thus, a direct link between spasticity and pain can be assumed (Voerman et al., Citation2010). The motor impairments are thought to be major contributors to movement dysfunction and impact on the child’s ability to participate in various life situations (Beckung & Hagberg, Citation2002).
There are various ways to treat spasticity, and the physiotherapeutic treatment can be training, stretching, mechanical loading and/or orthosis (Glickman et al., Citation2010), sometimes in combination with pharmacological treatment such as baclofen, botulinum neurotoxin (BoNT), or selective dorsal rhizotomy (Tedroff, Citation2013). Electrical stimulation has also been used for reducing spasticity (Bakheit, Citation2012, Moll et al., Citation2017) as well as increasing muscle function for patients with reduced volitional activity (Wright et al., Citation2012). According to P. Rosenbaum and Stewart (Citation2004), the components (activity, participation, body functions and structures, personal and environmental factors) are linked to one another. Therefore, when treating body structures, body functions would be affected, which in turn is expected to have a positive impact on activity and participation.
In a review by Wright et al., (Citation2012), neuromuscular electrical stimulation (NMES) for children with CP is cautiously advocated to minimize impairment and activity limitations during gait. Treatment with NMES on gluteus medius for children with CP showed an improved gait (Al-Abdulwahab & Al-Khatrawi, Citation2009). These results are in line with Van der Linden et al. (Citation2003), who also found that electrical stimulation for children with CP provided small, not significant changes in ROM and improved outcomes in walking (Van der Linden et al., Citation2003).
However, electric stimulation with simple electrodes, such as TENS and NMES, is difficult because of the difficulties in attaching the electrodes correctly and using it in training or activities in daily life. Today, an assistive device, electro-suit Mollii®, is available. This suit is a CE-marked device designed to reduce spasticity through electrical stimulation of the antagonistic muscles. Mollii® consists of a jacket, a pair of trousers, and a control unit. Fifty-eight electrodes are embedded in the suit for stimulation of selected muscles with a low-level electric stimulation (www.inerventions.se). The suit provides an opportunity to stimulate multiple muscle groups simultaneously. Therefore, the aim of this pilot study was to evaluate the suit’s possible effect on body function, activity, and participation in self-selected activities.
Materials and methods
The study had a purposive sampling, and the children were recruited from two habilitation centers in northern Sweden. To be included in the study, the children had to meet the following criteria: (1) be classified in Gross Motor Function Classification System (GMFCS) score I or II, (2) give their consent to refrain from treatment with botulinum toxin during the study, (3) be at least 104 cm tall, (4) have the ability to formulate goals, and (5) be able to express their experiences regarding the use of the suit. Exclusion criteria were injections of BoNT within three months of starting the study. Other criteria for exclusion were if the child had an implant such as intrathecal baclofen pump. Six children, one girl and five boys, aged between five and 11 years, fulfilled the inclusion criteria and were included in the study (). The study was approved by the Ethical Review Board in Stockholm, Sweden (D nr. 2014/1282-31/4). The parents received both oral and written information about the study. Full informed written consent was obtained from the parents of participating children.
Table 1. Participating children.
Measures
Body function
The passive range of motion (ROM) and spasticity assessment followed the assessment models used in the CPUP protocol (http://cpup.se/wp-content/uploads/2017/01/FT-manual-2017.pdf). Pain was measured with the Visual Analog Scale (VAS) (Alghadir et al., Citation2018).
Activity and participation
Gross motor function, standing and walking were measured with Gross Motor Function Measure (GMFM) dimensions D and E, a standardized observational instrument designed to measure change in gross motor function in children with cerebral palsy (Russel et al., Citation2000). Participation was measured by the Canadian Occupational Performance Measure (COPM), an instrument measuring participation in self-selected activities. COPM has good validity and reliability and the adapted COPM for children is a psychometrically robust tool (Cusick et al., Citation2007).
The intervention
The intervention was that the child had to wear the electro-suit Mollii® one hour every other day. The child and the parents were instructed to continue with the exercises and strains they already had without adding anything further. Examples of the continuing exercises were stretching, balance, and walking training. Assessment of joint mobility and muscle tone was performed in collaboration between the researchers and one person from the company. Increased muscle tone and/or decreased range of motion were decisive for the electrical stimulation. Therefore, the location of electrodes and intensity of stimulation was individual for each child. However, all children received stimulation from the electrodes for the trunk and buttocks, whether they experienced any symptoms or not ().
Table 2. Description of participants’ electrode locations and intensity of stimulation.
Procedure
At baseline, the first measurements took place, and the children set their goals individually or with some help from their parents. At four-week follow-up, the researchers also had a conversation with the child and parents about compliance, and some adjustments of the intensity of stimulation were performed. At three-month follow-up, the final measurement was performed and the goals were evaluated ().
Data analysis
The data were prepared and analyzed with descriptive statistics. Since the study was designed as a pilot study with six participants using the electro-suit Mollii®, no inferential statistics were calculated. The results are presented both for the group and individually.
Results
Data from the baseline measurements and the three-month follow-up are presented in the results section. The data from the four-week follow-up were essentially the same as at the three-month follow-up and, therefore, are not presented in the results.
The children pointed out different goals they wanted to achieve. After that, they estimated the performance of the task and how satisfied they were with the performance with COPM, illustrated in . All children perceived improvements, according to both performance and satisfaction. Three children estimated a clinically significant improvement in COPM after intervention.
Table 3. COPM baseline, 3 months, and clinical status.
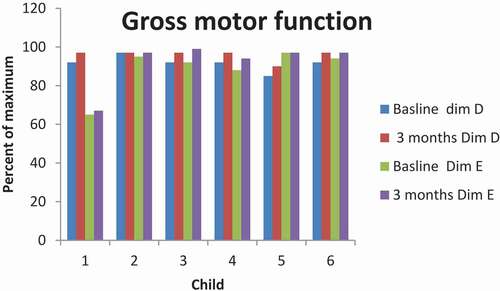
The gross motor function was evaluated regarding standing (dimension D) and walking, running, and jumping (dimension E) in GMFM. Dimension D: Five children had scores over 90% of maximum, and one child had 85% of maximum at baseline. Five children improved their results with 5% at three-month evaluation. One child had no improvement and had 97% of max at both measurement occasions. Dimension E: Five children had between 88% and 95% of maximum at baseline; one child had 65% of maximum. Five children improved their results between two and seven percent, while the result for the last child was unchanged ().
Figure 2. Illustration of gross motor function in standing (dimension D) and walking, running and jumping (dimension E).
The results related to pain showed that three children (3, 5, and 6, see ) experienced pain at baseline with an intensity of VAS 5, 5.5, and 6, respectively. At the follow-up after three months, the intensity was rated 0, 1, and 0, respectively.
Passive ROM in ankle dorsiflexion showed generally small changes (5°) with both flexed and extended knee. Two children (4 and 6) had larger changes (10°). The muscle tone was mostly unchanged ().
Table 4. Passive ROM ankle dorsiflexion with flexed respective extended knee and muscle tone in plantar flexors.
When measuring the passive ROM in hip abduction, no deterioration was seen in any of the hip joints. The abduction was unchanged in five hip joints and increased in the remaining seven joints. Regarding internal rotation, the ROM was mostly unchanged, but four joints showed increased rotation. The external rotation had increased in six hip joints, was unchanged in three joints, and decreased in two joints. However, all changes were small, at five or ten degrees ().
Table 5. ROM in hip joints at baseline and three-month follow-up.
Hip flexion increased in six joints, was unchanged in three joints, and ROM in the remaining three joints decreased. The muscle tone was unchanged in hamstrings in all joints except two, where the tone had increased ().
Table 6. ROM in hip joints at baseline and three months.
Discussion
The results of the study showed some improvements in activity and participation. All participants estimated an improvement in the total score for COPM, and the improvement was significant for three of them. There were small changes in ROM and muscle tone. The three participants who were bothered by pain when the study started experienced reduced pain or were pain free when the study ended. Reduced pain and better sleep when using electro-suit Mollii® was also reported in an interview study by (Nordstrom & Prellwitz, Citation2019). Theoretically, the pain relief effect could be explained by the electric stimulation from the suit, where both muscle fibers and fibers for sensation are stimulated with the frequency of 20 Hz, which is within the range of NMES (20–120 Hz) (CEFAR). It is well known that pain negatively affects participation for persons with CP (Ramstad et al., Citation2012) and a study by Vargus‐Adams and Martin (Citation2011) show that participation is regarded as important by the children, their parents, and medical professionals. However, children with CP participate less frequently in activities than children without disabilities (Michelsen et al., Citation2009) and children suffering from pain have even lower degrees of participation across many areas (Fauconnier et al., Citation2009). Reduced pain may therefore be one of the key factors in improving the children’s gross motor function and estimating higher scores in COPM.
All participants showed some improvements according to GMFM. The electrodes might give feedback when the child activates the stimulated muscles. All participants in this study got electrical stimulation for the trunk and buttocks, and the improvements in gross motor function are in line with studies on NMES showing insignificant but positive short-term results on muscle strength (Van der Linden et al., Citation2003; Wright et al., Citation2012) and improved gait (Al-Abdulwahab & Al-Khatrawi, Citation2009). P. L. Rosenbaum et al. (Citation2008) showed that participants who received treatment with botulinum toxin had improved results in GMFM. When the effect from the BoNT subsided, the participants remained on the same level of GMFM or continued improvement despite increasing spasticity. The results were explained as enhanced motor control in the motor cortex.
At baseline, all participants had reduced passive ROM and spasticity in dorsiflexion of the ankle, bilaterally or unilaterally. During the study, small changes were seen in ROM, and degree of muscle tone was mostly unchanged. The changes in passive ROM in the hip joint showed the most positive results. No deterioration was seen in any of the children’s hip joints in abduction; instead, it increased in seven joints and was unchanged in five joints. For five participants, the external rotation in the hip increased 5–10°, which is conforming to a study where electrical stimulation was placed over the gluteus maximus, and external rotation in the hip was increased, but the result was not significant (Van der Linden et al., Citation2003). The impacts of treatment with BoNT usually persist for about three or four months (FASS). The children had refrained from treatment with botulinum toxin during at least three months before baseline and for six to eight months at the end of the study. This study reveals unchanged or slightly increased ROM for some participants, which can be interpreted as a positive trend. In addition, when the study was finished, one child who usually received treatment by BoNT every third or fourth month wanted to wait for treatment because of the perceived impact of the suit. The interview study (Nordstrom & Prellwitz, Citation2019) with the children who participated in this study and their parents showed that both children and parents perceived some positive changes in activities that were not possible to measure in degrees of ROM and muscle tone.
There was no control group in this study, but the first author’s clinical experience was that if the participants abstained from treatment with BoNT, it would result in increased muscle tension and decreased ROM in affected muscles and joints. This is in line with Beckung et al. (Citation2002), Hägglund (Citation2013), and Lance (Citation1980), who stated that untreated spasticity can increase the risk of deformities in joints, which can cause pain and impaired physical exercise. Improved GMFM does not mean improved participation, although participation is not only related to gross motor function a better function can be a part of participation (Rosenbaum, Citation2008).
Goal setting, participation in self-selected activity and satisfaction
Those participants who had goals regarding sports or leisure activities had improved levels of activity and participation with friends. Participation is one of the core concepts in ICF-CF (Adolfsson et al., Citation2011), and in this study, participation was affected mostly positively. However, some goals were not achieved, which may be due to difficulties in goal setting. One of the participants had the goal to come out as quickly as his classmates during break at school. The child achieved the goal and was as quick as his classmates, but despite this, the estimated performance in COPM was at the same level as before, and the satisfaction was lower. The child stated that his participation had not improved. The low level of satisfaction may depend on the expectations of the outcome. The child had indirectly hoped that increased speed in walking and running would lead to greater participation in play. COPM is considered an instrument with good validity and reliability, even for children. Children who are under eight years old may have some difficulties with self-assessment and may need help from an adult (Cusick et al., Citation2007). In this case, the child was younger than eight years of age, and that may have contributed to difficulty in defining the actual goal. On the other side, some studies show that if the children have a self-chosen goal, they obtain better results from training (Cusick et al., Citation2007). Therefore, it is important for professionals and parents to support the child in choosing relevant goals.
There were few negative effects of the electro-suit. Two children experienced discomfort initially from the treatment when the control unit was activated. This resulted in an adjustment of the frequency of stimulation, and one of the children was satisfied with that and could use the suit as decided. The second child felt some discomfort even if the frequency was adjusted. Regarding compliance, it came up that the suit was difficult to put on and take off, which is confirmed in the interviews with the children and parents (Nordstrom & Prellwitz, Citation2019).
Method discussion
Some limitations with the study were the short timeframe of the study, the limited number of participating children (six), and that there was no control group. Another limitation was that the children had a low degree of spasticity, except for two children. Nevertheless, the results are valuable because the children were recruited by purposive sampling and have experience with other treatments to reduce spasticity. The participants were able to compare their experiences of the treatment with electro-suit Mollii® with treatment with BoNT. The children in the study were classified in GMCS level 1–2, and it was interesting to evaluate the impact of the suit on participants with more diverse GMFM levels and children with a higher degree of spasticity.
The children improved in activities and participation, but the measurable effects regarding passive ROM and degree of spasticity were small. The instruments used in the study are tested for reliability and validity, but there can still be a risk that the instruments are not sensitive enough for measuring small changes in ROM and muscle tone. For example, there can be small sources of errors that may influence the outcome a few degrees when passive ROM is measured by a goniometer. The five-grade Modified Ashworth Scale (MAS) may not be sensitive enough to register small changes in muscle tone. That can explain why the perceived impact in this study was greater than the measurable impact. When the goals were set, the children were able to receive help from their parents; how much help the children received is not known. The children were also allowed to receive help from their parents if they had difficulties in estimating by COPM. Children younger than eight years old might need help in estimating performance and satisfaction (Cusick et al., Citation2007). It seems that if the children themselves did not choose the goal, it was more difficult to estimate changes. Finally, the instructions to the participants not to add any new exercise were not followed up, which also can be a limitation in the study.
One strength of the study was that all participants completed the study and used the suit according to the instructions. The physiotherapist who performed the measurements had extensive experience working with children with cerebral palsy and with using the instruments. It was the same physiotherapist who performed all measures in the study. Another strength was that this study gives measurable effects on activity, participation and changes in body structures and body function and is a part of a study that includes individual interviews with the children and their parents (Nordstrom & Prellwitz, Citation2019).
Conclusion
It seems that electro-suit Mollii® affected the experience of pain, which in turn had a positive impact on gross motor function and activities for the children in the study. Changes in muscle tone and passive ROM were small and differed from unchanged, slightly improved to slightly decreased ROM and muscle tone.
For the future, it is important to make electro-suit Mollii® more user-friendly so the child can handle it independently. A greater number of participants would be an advantage to make comparisons between intervention – and control group. It should also be interesting to study children on higher GMFCS levels, to examine the effects of Mollii®. It is also important that professionals support children to set their own concrete and important goals.
Acknowledgments
The authors would like to thank the participants in this study.
Additional information
Funding
References
- Adolfsson, M., Malmqvist, J., Pless, M., & Granuld, M. (2011). Identifying child functioning from an ICF-CY perspective: Everyday life situations explored in measures of participation. Disability and Rehabilitation, 33(13–14), 1230–1244. https://doi.org/https://doi.org/10.3109/09638288.2010.526163
- Al-Abdulwahab, S. S., & Al-Khatrawi, W. M. (2009). Neuromuscular electrical stimulation of the gluteus medius improves the gait of children with cerebral palsy. NeuroRehabilitation, 24(3), 209–217. https://doi.org/https://doi.org/10.3233/NRE-2009-0470
- Alghadir, A. H., Anwer, S., Iqbal, A., & Iqbal, Z. A. (2018). Test-retest reliability, validity, and minimum detectable change of visual analog, numerical rating, and verbal rating scales for measurement of osteoarthritic knee pain. Journal of Pain Research, 11, 851–856. https://doi.org/https://doi.org/10.2147/JPR.S158847
- Bakheit, A. M. O. (2012). The pharmacological management of post-stroke muscle spasticity. Drugs & Aging, 29(12), 941–947. https://doi.org/https://doi.org/10.1007/s40266-012-0034-z
- Beckung, E., Brogren, E., & Rösblad, B. (2002). Sjukgymnastik för barn och ungdom: Teori och tillämpning. Studentlitteratur.
- Beckung, E., & Hagberg, G. (2002). Neuroimpairments, activity limitations, and participation restrictions in children with cerebral palsy. Developmental Medicine and Child Neurology, 44(5), 309–316. https://doi.org/https://doi.org/10.1111/j.1469-8749.2002.tb00816
- Bohannon, R. W., & Smith, M. B. (1987). Interrater reliability of a modified Ashworth scale of muscle spasticity. Physical Therapy, 67(2), 206–207. https://doi.org/https://doi.org/10.1093/ptj/67.7.1068
- CEFAR. http://www.cefar.se/treatments_j.asp?selLanguage=%7B16F02AFF-6B91-4E87-8DCF-0B286AAA0FB4%7D&selSection=%7B9AB1147F-A568-447D-872F-88336706F990%7D&id=&cat=NMES+%2F+Muskelstimulering
- Cusick, A., Lannin, N. A., & Lowe, K. (2007). Adapting the Canadian occupational performance measure for use in a paediatric clinical trial. Disability and Rehabilitation, 29(10), 761–766. https://doi.org/https://doi.org/10.1080/09638280600929201
- DePoy, E., & Gitlin, N. L. (1999). Forskning-En Introduktion. Studentlitteratur.
- Fass. www.fass.se/LIF/startpage
- Fauconnier, J., Dickinson, H. O., Beckung, E., Marcelli, M., McManus, V., Michelsen, S. I., & Colver, A. (2009). Participation in life situations of 8-12 year old children with cerebral palsy: Cross sectional European study. Bmj, 338. https://doi.org/https://doi.org/10.1136/bmj.b1458
- Glickman, L. B., Geigle, P. R., & Paleg, G. S. (2010). A systematic review of supported standing programs. Journal of Pediatric Rehabilitation Medicine, 3(3), 197–213. https://doi.org/https://doi.org/10.3233/PRM-2010-0129
- Graham, H. K., & Selber, P. (2003). Musculoskeletal aspects of cerebral palsy. Journal of Bone and Joint Surgery-British Volume, 85(2), 157–166. https://doi.org/https://doi.org/10.1302/0301-620X.85B2.14066
- Haas, B., Bergstrom, E., Jamous, A., & Bennie, A. (1996). The inter rater reliability of the original and of the modified Ashworth scale for the assessment of spasticity in patients with spinal cord injury. Spinal Cord, 34(9), 560–564. https://doi.org/https://doi.org/10.1038/sc.1996.100
- Hägglund, G. (2013). Stockholm. Läkartidningen, 15 (110), 765–766. Positiv utveckling med CPUP. ISSN: 0023-7205.
- Himmelmann, K., & Uvebrant, P. (2014). The panorama of cerebral palsy in Sweden. XI. Changing patterns in the birth‐year period 2003–2006. Acta Paediatrica, 103(6), 618–624. https://doi.org/https://doi.org/10.1111/apa.12614
- Lance, J. W. (1980). The control of muscle tone, reflexes, and movement: Robert Wartenberg lecture. Neurology, 30(12), 1303–1313. https://doi.org/https://doi.org/10.1212/WNL.30.12.1303
- Livanelioglu, A., & Gunel, M. K. (2007). Reliability of goniometric measurements in children with spastic cerebral palsy. Medical Science Monitor: International Medical Journal of Experimental and Clinical Research, 13(7), CR323–9. //487390[pii]
- Livingston, M. H., Rosenbaum, P. L., Russell, D. J., & Palisano, R. J. (2007). Quality of life among adolescents with cerebral palsy: What does the literature tell us? Developmental Medicine & Child Neurology, 49(3), 225–231. https://doi.org/https://doi.org/10.1111/j.1469-8749.2007.00225.x
- Lund, M. L., & Lexell, J. (2009). Participation and environmental factors in ICF. Possibilities and challenges when used in rehabilitation. [Delaktighet och omgivningsfaktorer i ICF. Mojligheter och utmaningar vid anvandning inom rehabilitering]. Lakartidningen, 106(13), 932–935. ISSN: 1652-7518.
- Michelsen, S. I., Flachs, E. M., Uldall, P., Eriksen, E. L., McManus, V., Parkes, J., Dickinson, H. O., Arnaud, C., Beckung, E., Dickinson, H. O., Fauconnier, J., Marcelli, M., Colver, A., & Parkinson, K. N. (2009). Frequency of participation of 8–12-year-old children with cerebral palsy: A multi-centre cross-sectional European study. European Journal of Paediatric Neurology, 13(2), 165–177. https://doi.org/https://doi.org/10.1016/j.ejpn.2008.03.005
- Moll, I., Vles, J. S., Soudant, D. L., Witlox, A. M., Staal, H. M., Speth, L. A., & Vermeulen, R. J. (2017). Functional electrical stimulation of the ankle dorsiflexors during walking in spastic cerebral palsy: A systematic review. Developmental Medicine & Child Neurology, 59(12), 1230–1236. https://doi.org/https://doi.org/10.1111/dmcn.13501
- Nordmark, E., Hagglund, G., & Lagergren, J. (2001). Cerebral palsy in southern Sweden I. prevalence and clinical features. Acta Paediatrica, 90(11), 1271–1276. https://doi.org/https://doi.org/10.1111/j.1651-2227.2001.tb01574.x
- Nordstrom, B., & Prellwitz, M. (2019). A pilot study of children and parents experiences of the use of a new assistive device, the electro suit Mollii. Assistive Technology, 1–8. https://doi.org/https://doi.org/10.1080/10400435.2019.1579267
- ØstensjØ, S., Øien, I., & Fallang, B. (2008). Goal-oriented rehabilitation of preschoolers with cerebral palsy—A multi-case study of combined use of the Canadian occupational performance measure (copm) and the goal attainment scaling (gas). Developmental Neurorehabilitation, 11(4), 252–259. https://doi.org/https://doi.org/10.1080/17518420802525500
- Palisano, R. J., Haley, S. M., & Brown, D. A. (1992). Goal attainment scaling as a measure of change in infants with motor delays. Physical Therapy, 72(6), 432–437. https://doi.org/https://doi.org/10.1093/ptj/72.6.432
- Pandyan, A. D., Johnson, G. R., Price, C. I., Curless, R. H., Barnes, M. P., & Rodgers, H. (1999). A review of the properties and limitations of the Ashworth and modified Ashworth scales as measures of spasticity. Clinical Rehabilitation, 13(5), 373–383. https://doi.org/https://doi.org/10.1191/026921599677595404
- Ramstad, K., Jahnsen, R., Skjeldal, O. H., & Diseth, T. H. (2012). Parent‐reported participation in children with cerebral palsy: The contribution of recurrent musculoskeletal pain and child mental health problems. Developmental Medicine & Child Neurology, 54(9), 829–835. https://doi.org/https://doi.org/10.1111/j.1469-8749.2012.04341.x
- Rosenbaum, P., Paneth, N., Leviton, A., Goldstein, M., Bax, M., Damiano, D., Dan, B. and Jacobsson, B. (2007). A report: The definition and classification of cerebral palsy April 2006. Developmental Medicine & Child Neurology Suppl, 109, 8–14. 2. PMID: 17370477.
- Rosenbaum, P., & Stewart, D. (2004). The World Health Organization international classification of functioning, disability, and health: A model to guide clinical thinking, practice and research in the field of cerebral palsy. Seminars in Pediatric Neurology, 11(1), 5–10. https://doi.org/https://doi.org/10.1016/j.spen.2004,01.002
- Rosenbaum, P. L. (2008). How do changes in body functions and structures, activity, and participation relate in children with cerebral palsy? Developmental Medicine & Child Neurology, 50(4), 283. https://doi.org/https://doi.org/10.1111/j.1469-8749.2008.02037.x
- Rosenbaum, P. L., Palisano, R. J., Bartlett, D. J., & Galuppi, B. E. (2008). Development of the gross motor function classification system for cerebral palsy. Developmental Medicine & Child Neurology, 50(4), 249. https://doi.org/https://doi.org/10.1111/j.1469-8749.2008.02045.x
- Russel, D. J., Avery, L. M., Rosenbaum, P. L., Raina, P. S., Walter, S. D., & Palisano, R. (2000). Improved scaling of the gross motor function measure for children with cerebral palsy: Evidence of reliability and validity. Physical Therapy, 80(9), 873–885. https://doi.org/https://doi.org/10.1093/ptj/80.9.873
- Shevell, M. I., Dagenais, L., & Hall, N. (2009). The relationship of cerebral palsy subtype and functional motor impairment: A population-based study. Developmental Medicine Child Neurology, 51(11), 872–877. https://doi.org/https://doi.org/10.1111/j.1469–8749.2009.03269.x
- Tedroff, K. (2013). Stockholm. Läkartidningen, 15 (110), 762–764. Behandling av spasticitet kan ge bättre vardagsliv.
- Turner-Stokes, L. (2009). Goal attainment scaling (GAS) in rehabilitation: A practical guide. Clinical Rehabilitation, 23(4), 362–370. https://doi.org/https://doi.org/10.1177/0269215508101742.
- Van der Linden, M., Hazlewood, M., Aitchison, A., Hillman, S., & Robb, J. (2003). Electrical stimulation of gluteus maximus in children with cerebral palsy: Effects on gait characteristics and muscle strength. Developmental Medicine & Child Neurology, 45(6), 385–390. https://doi.org/https://doi.org/10.1111/j.1469–8749.2003.tb00417.x
- Vårdhandboken. www.vardhandboken.se/texter/smartskattning-av-akut-och-postoperativ-smarta/smartskattningsinstrument.
- Vargus‐Adams, J. N., & Martin, L. K. (2011). Domains of importance for parents, medical professionals and youth with cerebral palsy considering treatment outcomes. Child: Care, Health and Development, 37(2), 276–281. https://doi.org/https://doi.org/10.1111/j.1365-2214.2010.01121.x
- Voerman, G. E., Erren-Wolters, C. V., Fleuren, J. F., Hermens, H. J., & Geurts, A. C. (2010). Perceived spasticity in chronic spinal cord injured patients: Associations with psychological factors. Disability and Rehabilitation, 32(9), 775–780. https://doi.org/https://doi.org/10.3109/09638280903304490
- Wright, P. A., Durham, S., Ewins, D. J., & Swain, I. D. (2012). Neuromuscular electrical stimulation for children with cerebral palsy: A review. Archives of Disease in Childhood, 97(4), 364–371. https://doi.org/https://doi.org/10.1136/archdischild-2011-300437

