Abstract
Aims: Peginterferon beta-1a 125 mcg administered subcutaneously every 2 weeks, a new disease-modifying therapy (DMT) for relapsing-remitting multiple sclerosis (RRMS), was approved in January 2015 by the Scottish Medicines Consortium. This study assesses long-term clinical and economic outcomes of peginterferon beta-1a compared with other self-injectable DMTs (interferon beta-1a [22 mcg, 30 mcg, and 44 mcg], interferon beta-1b, and glatiramer acetate 20 mg) in the treatment of RRMS, from the National Health Service and Personal Social Services perspective in Scotland.
Methods: A previously published, validated Markov cohort model was adapted for this analysis. The model estimates changes in patient disability, occurrence of relapses, and other adverse events, and translates them into quality-adjusted life years and costs. Natural history data came from the ADVANCE trial of peginterferon beta-1a, the London Ontario (Canada) database, and a large population-based MS survey in the UK. The comparative efficacy of each DMT vs placebo was obtained from a network meta-analysis. Costs (2015 British Pounds) were obtained from public databases and literature. Clinical and economic outcomes were projected over 30 years and discounted at 3.5% per year.
Results: Over 30 years, peginterferon beta-1a was dominant compared with interferon beta-1a (22, 30, and 44 mcg), and interferon beta-1b, and cost-effective compared with glatiramer acetate 20 mg. Results were most sensitive to variations in each DMT’s efficacy and acquisition costs. Deterministic and probabilistic sensitivity analyses confirmed the robustness of the results.
Limitations: The impact of improved adherence with peginterferon beta-1a on clinical and economic outcomes and the impact of subsequent DMTs after treatment discontinuation were not considered. Oral and infused DMTs were not included as comparators.
Conclusion Long-term treatment with peginterferon beta-1a improves clinical outcomes, while its cost profile makes it either dominant or cost-effective compared with other self-injectable DMTs for the treatment of RRMS in Scotland.
Introduction
Multiple sclerosis (MS) is a chronic and progressive disease of the central nervous system (CNS), characterized by localized areas of inflammation on nerve fibers, demyelination, and axonal degenerationCitation1,Citation2. MS is the leading cause of non-traumatic CNS morbidity and mortality in young and middle-aged adultsCitation3–5, and negatively impacts patients’ quality-of-life (QoL)Citation6. The estimated prevalence of MS in Scotland is ∼255 per 100,000, with an estimated annual incidence of 15.29 per 100,000Citation7. The mean annual total cost per patient in the UK, including direct medical and non-medical costs, was ∼£12,000 and £26,000 (in 2009 British Pounds) for patients with Expanded Disability Status Scale (EDSS) levels of 0–3 and 4–6.5, respectivelyCitation8. With the highest incidence and prevalence of MS among the 13 regions in the UKCitation7, Scotland bears a substantial economic MS burden.
Currently, there is no cure for MS, and disease-modifying therapies (DMTs) provide an alternative for disease control to patients with RRMSCitation9. Ten DMTs are approved by the Scottish Medicines Consortium (SMC) as of December 2015, including three types: (1) self-injectable DMTs (interferon beta-1a [22 mcg and 44 mcg] subcutaneous three times per week, interferon beta-1a 30 mcg intramuscular once per week, interferon beta-1b subcutaneous once every other day, glatiramer acetate subcutaneous [20 mg once daily and 40 mg three times per week], and peginterferon beta-1a subcutaneous every 2 weeks); (2) oral DMTs (dimethyl fumarate, fingolimod, and teriflunomide); and (3) intravenously infused DMTs (alemtuzumab and natalizumab)Citation9–11.
Self-injectable DMTs have been used to treat MS for ∼2 decadesCitation9. However, some patients may find the frequent injections burdensome, and adherence with these treatments, in terms of both compliance and persistence, has been poorCitation12. Poor adherence may limit the efficacy of these agents, leading to sub-optimal treatment outcomesCitation13 and a higher rate of healthcare resource utilizationCitation14,Citation15.
To prolong the half-life of interferon beta-1a, and thus decrease the required dose frequency, a PEGylated formulation of interferon beta-1a (peginterferon beta-1a) was developed. Peginterferon beta-1a builds upon the proven efficacy and safety profile of interferons and provides less frequent dosing, which may improve treatment adherenceCitation13,Citation16,Citation17. The ADVANCE trial, a global, 2-year, randomized, double-blind study, placebo-controlled during the first year, has shown the benefits of peginterferon beta-1a compared with placebo on relapses and disability progression, as well as favorable safety resultsCitation18, but its long-term clinical and economic consequences compared with other DMTs are still unknown.
Therefore, it was of interest to compare peginterferon beta-1a’s long-term costs and health benefits with those of other self-injectable DMTs in the treatment of patients with RRMS in Scotland, and to provide cost-effectiveness information for decision-makers considering peginterferon beta-1a for inclusion in healthcare formularies, or for patients in clinical practice.
Methods
Economic model
A Markov cohort model was developed for this analysis, based on a previously published and well-validated modelCitation19,Citation20 accepted by several health technology assessment (HTA) agencies, including the National Institute for Health and Care Excellence (NICE) in the UKCitation21–23.
To model the course of the disease, the model includes: (1) 10 health states defined by EDSS levels 0–9.5 during the relapsing-remitting (RR) phase of the disease; (2) 10 health states with the same definition during the secondary progressive (SP) phase; and (3) death ().
Figure 1. Model structure, Adapted from Gani et al.Citation20. Ovals represent health states. All health states may progress to death. Rectangles represent events that patients can experience at any time. Treatment-related AEs and treatment discontinuation can only occur for patients receiving treatment. AE, adverse event; EDSS, Expanded Disability Status Scale; RRMS, relapsing-remitting multiple sclerosis; SPMS, secondary-progressive multiple sclerosis; State N = current EDSS state.
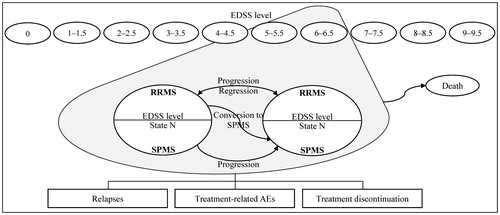
The cohort of patients, all with RRMS, starts the model with an initial EDSS distribution. Every year (model cycle length), patients with RRMS may: (1) remain at the same EDSS level with RRMS; (2) progress to a higher EDSS level (i.e. increased disability); (3) improve to a lesser EDSS level; (4) convert to SPMS; or (5) die. Patients converting to SPMS cannot improve to a lesser EDSS level or to RRMS; they can only stay at the same EDSS level, progress to a higher EDSS level, or die. Patients can experience relapses at any time. The annual risk of relapses depends on the disease phase and the patient’s EDSS level.
Treatment acts in two ways: slowing disability progression (i.e. delaying the transition to a higher EDSS level), and reducing the frequency of relapses. While on treatment, patients can experience treatment-related adverse events (AEs) at any time, and can discontinue as a result of three pre-defined events: conversion to the SP phase of the disease, progression to an EDSS level greater or equal to 7, or AEs and other reasons (i.e. dropouts). Patients who discontinue treatment follow the natural history of disease progression.
Patients accrue state- and event-specific costs and are assigned utilities to estimate quality-adjusted life years (QALYs) as they move between health states and experience relapses and treatment-related AEs. The model calculates and reports incremental health outcomes, costs, and incremental cost-effectiveness ratios (ICERs) for the different comparators.
Parameters and data sources
Perspective and time horizon
The analysis takes the perspective of the National Health Service and the Personal Social Services in Scotland. A time horizon of 30 years was selected to reflect the chronicity of RRMS, and that patients are expected to be on treatment until they die or discontinue treatment due to lack of efficacy (i.e. conversion to SPMS or progression to an EDSS level greater or equal to 7).
Discount rates allow for the adjustment of time preferences for a new treatment’s costs and benefits, by providing present valuesCitation24. Discount rates are not universal and vary according to the setting, location, and perspective of the analysisCitation24. As required by the SMC, “economic results should reflect the present value of the stream of costs and benefits accruing over the time horizon of the analysis”Citation25, using the applicable rates in the Treasury Green BookCitation26. Currently, the Treasury Green Book advises the use of an annual discount rate of 3.5% for both costs and benefitsCitation25.
Patient characteristics
The model population includes patients 18 years and older with RRMS. The population is 29.2% male with a mean age of 36.5 years, as observed in the ADVANCE trial. The initial EDSS distribution of the cohort was obtained from the baseline EDSS distribution of the intent-to-treat (ITT) population of the ADVANCE trial ().
Table 1. Initial EDSS distribution of the cohort, ARR natural history, utility, costs of disease management, and mortality by EDSS level.
Natural history
As reported in the study by Hernandez et al.Citation31, the probabilities of disability progression during RRMS were obtained by combining the data from the ITT population in the placebo arm of the ADVANCE trial with data from the longitudinal dataset of patients with MS from the London, Ontario (Canada) database, and transition probabilities from RRMS to SPMS and during SPMS were derived from the London, Ontario datasetCitation32,Citation33.
The annualized relapse rates (ARRs) by EDSS level are shown in . As reported in the study by Hernandez et al.Citation31, the ARRs for RRMS were derived from the ITT population in the placebo arm of the ADVANCE trial and the UK MS SurveyCitation27 and Patzold and PocklingtonCitation28 and the ARRs for SPMS were derived by extrapolating those derived for RRMS with ARRs from the UK MS SurveyCitation27 and Patzold and PocklingtonCitation28.
Treatment
The comparative efficacy for each DMT vs placebo (i.e. no DMT), as measured by ARRs and 3-month sustained EDSS progression, were obtained from a network meta-analysis of peginterferon beta-1a and self-injectable DMTs for RRMS ()Citation34. For all DMTs, it was assumed that there was no change in the treatment effect over time (i.e. no waning of treatment effect over time).
Table 2. Comparative efficacy, discontinuation risk, and AE-associated utility decrement.
Patients stopped treatment after progression to an EDSS level ≥7 or after conversion to SPMS. In addition, an annual probability of treatment discontinuation due to AEs and other reasons was applied to each DMT during the time horizon. The annual discontinuation risk was obtained from each DMT’s pivotal trials ().
The AEs included in the model are: abdominal pain, arthralgia, back pain, chest pain, cough, depression, diarrhea, fatigue, flu-like symptoms, flushing, gastroenteritis, headache, increased alanine aminotransferase (ALT), influenza, injection site reactions, leukopenia, nausea, pain in extremity, pruritus, rash, upper abdominal pain, and urinary tract infection.
The above-mentioned AEs are the most commonly reported (≥5% incidence in any treatment group) for peginterferon beta-1a in the ADVANCE trial, as well as AEs that occurred at an incidence of 3% or greater in the overall peginterferon beta-1a group compared to the placebo group (even if the overall incidence in the peginterferon beta-1a arms was <5%). As a conservative assumption, only AEs reported for peginterferon beta-1a were included for all the comparators. The annual incidence rates of AEs were obtained from the ADVANCE trial for peginterferon beta-1a and from published pivotal clinical trials for the comparatorsCitation17,Citation35–46.
The annual incidence rate of each AE for each treatment and study was calculated as the proportion of patients experiencing each AE over the study duration. The study duration was scaled to 1 year. When more than one study was used for a specific treatment, a weighted average of the annual incidence rate and the number of patients observed in each study was calculated to derive the annual incidence of each AE.
Serious AEs were considered for peginterferon beta-1a only: 0.03% of the peginterferon beta-1a patients had serious increased ALT, 5.88% had serious depression, 0.28% had serious injection site reactions, and 1.92% had serious urinary tract infections. As a conservative assumption, no serious AEs were considered for any of the other comparators.
Mortality
Age- and gender-specific all-cause mortality risks for the general population were obtained from national life tables (2013) for Scotland from the Office for National StatisticsCitation47. Mortality risks for patients with MS were calculated over imposing the relative risk of death by EDSS level presented by PokorskiCitation29 to the all-cause mortality risk ().
Utilities
Utilities are measured on a cardinal scale of 0–1, where 0 indicates death and 1 indicates perfect healthCitation48. Irrespective of the part of the scale being considered, one interval of change is the same as another (i.e. a change from 0.2 to 0.3 is equivalent to a change from 0.8 to 0.9)Citation48. States worse than death can also be accounted for, with such states taking a negative valueCitation48. To obtain QALYs, utilities are multiplied by the number of years in a particular health state.
Both patient and caregiver utilities were considered in this analysis (). Utility scores for patients by EDSS level were obtained by combining the data from the EuroQol five dimensions questionnaires (EQ-5D) data of the ITT population in the ADVANCE trial (EDSS levels 0–5.5) with the data from the UK MS Survey ()Citation27. For all EDSS levels, a disutility of 0.071 was subtracted to stratify by relapse vs no relapse. A disutility of 0.045 was subtracted to stratify by RRMS vs SPMSCitation27. Caregivers’ disutility values by EDSS were obtained from Gani et al.Citation20.
The disutility and duration associated with each AE were obtained from expert clinical opinion. For each AE included in the model, the utility decrement for a serious and for a non-serious event are first multiplied by their respective duration, and then weighted by the proportion of serious AEs. Finally, the annual incidence of the event is applied. The annual AE-associated utility decrements were applied only to patients receiving treatment ().
Costs
Direct medical costs include: (1) costs of disease management by EDSS level (excluding cost of DMTs); (2) cost of managing relapses; (3) costs of DMTs and DMT-related administration and monitoring; and (4) AE management costs. All costs are reported in 2015 British Pounds.
Disease management costs ()Citation8 and the cost of relapse (£2,145.28 per relapse)Citation49 were obtained from published literature and inflated to 2015. The inflation factor from 2009 to 2015 for the disease management costs was 1.1749Citation30. The inflation factor from 2005 to 2015 for the cost of relapse was 1.3218Citation30. Treatment acquisition (i.e. cost of DMTs), administration, monitoring costs, and their corresponding sources are shown in .
Table 3. Treatment-related costs.
Annual AE management costs were derived using a similar method to that applied for the annual AE-associated utility decrement. Management costs were calculated for each AE by first weighting the cost for a serious and for a non-serious event by the proportion of serious events. Next, the annual incidence of the event is applied. This weighted annual cost is calculated for all AEs, and then added up to calculate an annual AE management cost for each treatment (). Finally, these AE management costs were applied only to patients receiving treatment.
Model validation
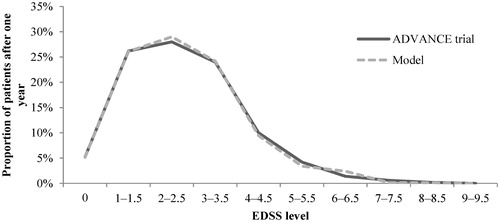
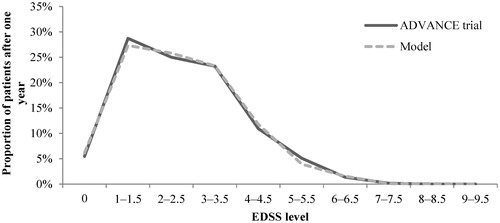
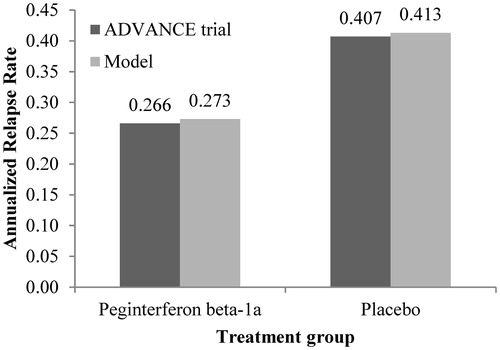
As in the study by Hernandez et al.Citation31, predictive validity was checked by comparing the model-reported ARR and the patients’ EDSS distribution after 1 year with the data from the ADVANCE clinical trial for peginterferon beta-1a and placebo. Overall, the ARR and EDSS distribution were comparable with the outcomes reported in the ADVANCE trials for peginterferon beta-1a and placebo (-).
Figure 2. ARR validation: Model outcomes vs ADVANCE trial after 1 year. ARR, annualized relapse rate.
Analyses
The life-table method for half-cycle correction was used to calculate the model outcomes for each treatmentCitation55. Deterministic sensitivity analyses were conducted to evaluate how the model outcomes varied in relation to changes in key model parameters, and to demonstrate whether the model results were robust across those variations. In order to account for statistical uncertainties of multiple key parameters, probabilistic sensitivity analyses were performed by simultaneously varying parameters using the following probability distributions: (1) log-normal, for rate ratios on ARRs and hazard ratios (HRs) on disability progression relative to placebo (based on their 95% confidence interval [CI]), incidence of AEs, and relative risks for MS mortality; (2) beta, for transition probabilities from RRMS to SPMS, annual treatment discontinuation risk, proportion of AEs reported as serious, disutility for serious and non-serious AEs, and patient and caregiver utility; (3) gamma, for the cost and duration of serious and non-serious AEs, cost of relapse, and disease management; and (4) Dirichlet, for transition probabilities between EDSS levels during RRMS and SPMS. Where a standard error or CI was not available for a selected parameter, 25% of the mean was assumed as standard error, a common approach in economic models.
Results
Base case analysis
Compared with all the DMTs included in this analysis, over 30 years, peginterferon beta-1a was associated with a slower rate of disability progression (i.e. accumulation of disability occurs at a slower pace) for patients with RRMS. It was also associated with a slower rate of conversion to SPMS and a longer time spent at EDSS levels under 7 (). Considering these benefits plus a lower total cost compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, and interferon beta-1b 250 mcg, over 30 years, peginterferon beta-1a was dominant (i.e. more effective and less costly). Compared with glatiramer acetate 20 mg, peginterferon beta-1a was associated with additional costs of £2,402 and additional QALYs of 0.42. Assuming a willingness-to-pay (WTP) threshold of £20,000 per QALY gained, peginterferon beta-1a was cost-effective compared with glatiramer acetate 20 mg.
Table 4. Base case outcomes over 30 years.
Deterministic sensitivity analyses
Deterministic sensitivity analyses were carried out to evaluate how sensitive the estimated incremental cost, incremental QALYs, and ICER were to changes in key model parameters, including costs of disease management by EDSS level (i.e. EDSS state costs), relapse costs, utility scores by EDSS level, natural history of relapse rates, treatment effect on disability progression and relapse rate, and risks of treatment discontinuation (or dropout). The results of the deterministic sensitivity analyses are presented as discounted, per patient, over 30 years, and are available in the Supplemental Online Material.
The incremental cost was most sensitive to variations in the risk of dropout (i.e. treatment discontinuation due to AEs and other reasons) across all the comparisons, except in the comparison with interferon beta-1a 22 mcg where incremental cost was most sensitive to the discount rate for costs. Note that, when the risk of treatment dropout is lower for a given treatment, patients accrue more treatment-related costs (i.e. drug acquisition and routine monitoring) over time. Conversely, a higher dropout rate decreases these costs over time. The incremental QALYs and the ICER were most sensitive to variations in the treatment effect of the DMTs on disability progression.
Compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, and interferon beta-1b, peginterferon beta-1a was dominant (i.e. more effective and less costly) or cost-effective (using the WTP threshold of £20,000 per QALY gained) in all the scenarios that were carried out, except for two scenarios:
The treatment effect of peginterferon beta-1a on disability progression (i.e. the hazard ratio of disability progression relative to placebo) was worsened by 20%. In this scenario, peginterferon beta-1a resulted in a lower total cost and a lower QALYs.
The treatment effect of the comparator on disability progression (i.e. the hazard ratio of disability progression relative to placebo) was improved by 20%. In this scenario, peginterferon beta-1a resulted in a lower total cost and a lower QALYs.
Compared with glatiramer acetate 20 mg, peginterferon beta-1a was cost-effective in all the scenarios (using the WTP threshold of £20,000 per QALY gained), except for the five scenarios:
The time horizon was reduced to 10 years. In this scenario, peginterferon beta-1a resulted in an ICER of £36,886 per QALY gained.
The treatment effect of glatiramer acetate 20 mg on disability progression (i.e. the hazard ratio relative to placebo) was improved by 20%. In this scenario, peginterferon beta-1a was dominated (i.e. less effective and more costly).
Treatment effect of peginterferon beta-1a on disability progression (i.e. the hazard ratio relative to placebo) was worsened by 20%. In this scenario, peginterferon beta-1a was dominated (i.e. less effective and more costly).
The annual risk of dropout of peginterferon beta-1a was increased by 20%. In this scenario, peginterferon beta-1a was dominant (i.e. more effective and less costly).
The annual dropout risk of glatiramer acetate 20 mg was reduced by 20%. In this scenario, peginterferon beta-1a was dominant (i.e. more effective and less costly).
Overall, the results of the one-way sensitivity analyses suggest that peginterferon beta-1a is cost-effective compared with all the DMTs considered in this analysis under a variety of scenarios, making the results robust.
Probabilistic sensitivity analysis
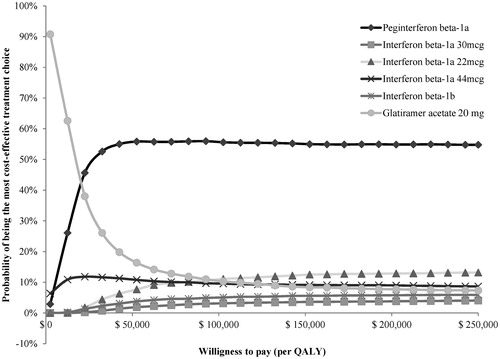
Assuming a WTP threshold of £20,000 per QALY gained, probabilistic sensitivity analyses indicated that peginterferon beta-1a remained cost-effective in 95%, 96%, 92%, 93%, and 75% of 5,000 replications compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, interferon beta-1b, and glatiramer acetate 20 mg, respectively. When all treatments were compared using multi-way cost-effectiveness acceptability curves, peginterferon beta-1a was the optimal treatment choice at any WTP threshold >£7,500 per QALY gained among all the treatments (Figure 5).
Discussion
The ADVANCE trial assessed the efficacy and safety of peginterferon beta-1a compared with placebo in patients with RRMSCitation18. The trial demonstrated the benefits of peginterferon beta-1a in terms of reducing the frequency of relapses and delaying disability progression compared to placebo over 1 yearCitation18. Peginterferon beta-1a was recently approved for use in patients with RRMS by the SMCCitation10, and findings from this economic analysis provide further information for clinicians and patients to make more informed choices when considering peginterferon beta-1a and for Health Boards to make funding decisions on peginterferon beta-1a. This analysis shows that long-term treatment with peginterferon beta-1a resulted in a number of clinical benefits, including a slower rate of disability progression during RRMS and of conversion to SPMS, and more QALYs gained compared with all the other self-injectable DMTs. These benefits are obtained at a reduced total cost compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, and interferon beta-1b; thus, peginterferon beta-1a dominates these treatments. Peginterferon beta-1a resulted in additional total costs compared with glatiramer acetate 20 mg. However, the resulting ICER falls below the assumed WTP of £20,000 per QALY gained, making peginterferon beta-1a cost-effective compared with glatiramer acetate 20 mg. Both the deterministic and probabilistic sensitivity analyses consistently support the findings of the base case analysis, indicating that peginterferon beta-1a is either a dominant or cost-effective treatment in the majority of the scenarios and replications.
To further assess the robustness of the results from this analysis, three issues concerning the quality of the model inputs need to be discussed. First, as noted in the Methods section, the transition probabilities of disability progression during RRMS were obtained from the ADVANCE trial and from the London, Ontario dataset, as reported in the study by Hernandez et al.Citation31. These sources had the following two limitations: (1) In actual practice, data from the ADVANCE trial may be considered unrepresentative of patients with RRMS due to the inclusion and exclusion criteria used in the trial and the short follow-up (1 year), which may be problematic for long-term extrapolation; (2) data from the London, Ontario database was collected about three decades ago and, thus, may not reflect the current natural history of disability progression. To address these issues, the recently published transition probabilities from the British Columbia (Canada) databaseCitation56 were used in one of the deterministic sensitivity analysis scenarios (detailed results of this scenario can be found in the Supplemental Online Material). Peginterferon beta-1a remained dominant compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, and interferon beta-1b. Additionally, peginterferon beta-1a was cost-effective compared with glatiramer acetate 20 mg.
Second, as noted in the Methods section, the ARRs were obtained from the ADVANCE trial and from the UK MS SurveyCitation27 and Patzold and PocklingtonCitation28, as reported in the study by Hernandez et al.Citation31. As with the transition probabilities of disability progression during RRMS, the ARRs from the ADVANCE trial may be considered unrepresentative of patients with RRMS. In addition, the UK MS SurveyCitation27 and Patzold and PocklingtonCitation28 collected data on ARRs well before the ADVANCE trial, several years before and about three decades before, respectively. The study by Inusah et al.Citation57 showed that the ARRs in patients with MS have fallen dramatically over the past years. Therefore, the ARRs from the ADVANCE trial and the UK MS SurveyCitation27 and Patzold and PocklingtonCitation28 used in the model combine data from several years ago from patients who experienced high ARRs and from recent patients with lower ARRs. Unfortunately, to our knowledge, there are no other publicly available population-based studies to supplement the ARRs observed in the ADVANCE trial. However, the natural history ARRs and the treatment effect of each DMT on the ARRs were varied in the deterministic sensitivity analysis scenarios (detailed results of these scenarios can be found in the Supplemental Online Material). These variations did not impact the model results. Peginterferon beta-1a remained dominant compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, and interferon beta-1b, and peginterferon beta-1a was cost-effective compared with glatiramer acetate 20 mg.
Third, network meta-analysis is a common and acceptable approach to assess the relative treatment effects of multiple treatments when these data are not available from head-to-head clinical trialsCitation58,Citation59. The treatment effect of peginterferon beta-1a used in this network meta-analysis was based on the 1-year, placebo-controlled ADVANCE study, which had a shorter follow-up time compared to 2–3 years for other studies of DMTs included in the network meta-analysisCitation34. Given that the results of the analysis are sensitive to treatment effects, the findings of the study should be interpreted carefully. The longer-term comparative efficacy for peginterferon beta-1a vs placebo or other DMTs is unavailable at this point. However, data from the 2-year ADVANCE study indicates that the observed treatment effect of peginterferon beta-1a vs placebo over the first year is very likely to be sustained for a longer termCitation60, which supports the findings from this analysis.
This is the first analysis assessing the cost-effectiveness of peginterferon beta-1a vs other self-injectable DMTs in Scotland, using a well-validated and accepted model structureCitation19–23. However, there are three limitations to consider. First, long-term, real-world data on adherence with peginterferon beta-1a relative to other self-injectable DMTs is not yet available; therefore, this analysis does not consider the impact of improved adherence on clinical and economic outcomes. However, as peginterferon beta-1a is expected to improve adherence due to its reduced dosing frequency, not considering the impact of improved adherence with peginterferon beta-1a is a conservative approach, likely to favor the other comparators and, thus, under-estimate the value of peginterferon beta-1a. Second, the impact of subsequent DMTs (i.e. second-line treatment after treatment discontinuation) is not considered in the analyses. Although including this feature would better reflect real life clinical practice, it could mask the true value of a specific DMT, as any benefits may result from other, subsequent treatments. Because of this, it was decided to exclude subsequent lines of DMT in the current analysis. Future analyses should examine the cost-effectiveness of different treatment sequences with and without peginterferon beta-1a to better reflect the treatment patterns of RRMS in real clinical practice. Third, oral DMTs (dimethyl fumarate, fingolimod, and teriflunomide) and intravenously infused DMTs (alemtuzumab and natalizumab) were not considered as comparators in this study. At the time our analyses were conducted, no published data were available on the efficacy of self-injectable DMTs compared with oral or intravenously infused DMTs. Future analyses should examine the cost-effectiveness of peginterferon beta-1a and the other self-injectable DMTs, compared with oral and intravenously infused DMTs to provide this additional information to payers and reimbursement agencies. A recent network meta-analysis published in September 2016 could be used as a reference for such analysesCitation61.
Conclusion
This analysis shows that peginterferon beta-1a is likely to have a greater positive impact on clinical outcomes over the long-term, while being less costly compared with interferon beta-1a 22 mcg, interferon beta-1a 30 mcg, interferon beta-1a 44 mcg, and interferon beta-1b. Compared with glatiramer acetate 20 mg, peginterferon beta-1a is a cost-effective treatment option, providing greater clinical benefits at a reasonable additional cost, resulting in an ICER under £20,000 per QALY gained. Based on these results and its recent approval by the SMC for patients with RRMS, peginterferon beta-1a would be a valuable addition to healthcare formularies in Scotland, and it should be reimbursed by Health Boards in Scotland.
Transparency
Declaration of funding
This study and resulting manuscript were funded by Biogen, the manufacturer of Peginterferon beta-1a (Plegridy). Biogen had no role in the design of the study, the collection, analysis, and interpretation of the data, or the writing the manuscript.
Declaration of financial/other interests
SC and SFSF are employees and shareholders of Biogen. LH, SG, and HTD are employed by Evidera, which provides consulting and other research services to pharmaceutical, medical device, and other organizations. In their salaried positions, they work with a variety of companies and organizations, and are precluded from receiving payment or honoraria directly from these organizations for services rendered. Evidera Inc. has a research consultancy agreement with Biogen. JME peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
| Abbreviations | ||
| AE | = | Adverse events |
| ALT | = | Alanine aminotransferase |
| ARR | = | Annualized relapse rate |
| CI | = | Confidence interval |
| CNS | = | Central nervous system |
| DMT | = | Disease-modifying therapy |
| EDSS | = | Expanded Disability Status Scale |
| EQ-5D | = | EuroQol five dimensions questionnaire |
| HTA | = | Health technology assessment |
| ICER | = | Incremental cost-effectiveness ratio |
| ITT | = | Intent-to-treat |
| MS | = | Multiple sclerosis |
| NICE | = | National Institute for Health and Care Excellence |
| QALY | = | Quality-adjusted life year |
| QoL | = | Quality-of-life |
| RR | = | Relapsing-remitting |
| RRMS | = | Relapsing-remitting multiple sclerosis |
| SMC | = | Scottish Medicines Consortium |
| SP | = | Secondary progressive |
| SPMS | = | Secondary progressive multiple sclerosis |
| UK | = | United Kingdom |
| WTP | = | Willingness to pay. |
Supplemental material
Download MS Word (47 KB)Acknowledgments
The authors would like to thank Clemence Bougeard, Market Access Analyst at Biogen, for her support obtaining the economic inputs for the model. They also thank Malinda O’Donnell, Director of Payer Communications at Evidera, for her review and editorial support on the manuscript, and Janet Dooley of Evidera’s Production Team for her assistance in the editing, formatting, and submission of the manuscript. Some of the results of this study have been previously presented at the International Society for Pharmacoeconomics and Outcomes Research 18th Annual European Congress (November 2015, Milan, Italy).
References
- Bainbridge JL, Corboy JR. Multiple sclerosis. In: Dipiro JT, Talbert RL, Yee GC, et al., editors. Pharmacotherapy: a pathophysiologic approach. 7th edn. New York, NY: McGraw-Hill Companies, Inc, 2008
- Compston A, Coles A. Multiple sclerosis. Lancet 2008;372:1502-17
- McDowell TY, Amr S, Langenberg P, et al. Time of birth, residential solar radiation and age at onset of multiple sclerosis. Neuroepidemiology 2010;34:238-44
- Wallin MT, Culpepper WJ, Coffman P, et al. The Gulf War era multiple sclerosis cohort: age and incidence rates by race, sex and service. Brain 2012;135:1778-85
- World Health Organization. Atlas: Multiple Sclerosis Resources in the World, 2008. Geneva: World Health Organization, 2008. http://www.who.int/mental_health/neurology/Atlas_MS_WEB.pdf. Accessed November 2, 2014
- Klevan G, Jacobsen CO, Aarseth JH, et al. Health related quality of life in patients recently diagnosed with multiple sclerosis. Acta Neurol Scand 2014;129:21-6
- Mackenzie IS, Morant SV, Bloomfield GA, et al. Incidence and prevalence of multiple sclerosis in the UK 1990–2010: a descriptive study in the General Practice Research Database. J Neurol Neurosurg Psychiatry 2014;85:76-84
- Karampampa K, Gustavsson A, Miltenburger C, et al. Treatment experience, burden and unmet needs (TRIBUNE) in MS study: results from the United Kingdom. Mult Scler 2012;18:41-5
- Evans D, Sykes J. Disease modifying drugs - a guide to treatments for relapsing MS. Letchworth Garden City, Hertfordshire, UK: Multiple Sclerosis Trust, 2015. https://support.mstrust.org.uk/shop?prodid=90. Accessed January 30, 2016
- Scottish Medicines Consortium. SMC - Briefing Note - No. 86, January 2015. Scottish Medicines Consortium advice to NHS-Scotland. Glasgow: Scottish Medicines Consortium, 2015. https://www.scottishmedicines.org.uk/files/briefing-notes/2014/SMC_BN_January_2015.pdf. Accessed April 15, 2015
- Scottish Medicines Consortium (SMC). Product Update: Glatriramer acetate 40mg/mL solution for injection prefilled syringes (Copaxone®). SMC No. (1108/15) Glasgow, Scotland: Scottish Medicines Consortium, 2015. https://www.scottishmedicines.org.uk/files/advice/glatiramer_acetate__Copaxane__Abbreviated_FINAL_Nov_2015_for_website.pdf. Accessed October 3, 2016
- Margolis JM, Fowler R, Johnson BH, et al. Disease-modifying drug initiation patterns in commercially insured multiple sclerosis patients: a retrospective cohort study. BMC Neurol 2011;11:122
- Menzin J, Caon C, Nichols C, et al. Narrative review of the literature on adherence to disease-modifying therapies among patients with multiple sclerosis. J Manag Care Pharm 2013;19:S24-S40
- Steinberg SC, Faris RJ, Chang CF, et al. Impact of adherence to interferons in the treatment of multiple sclerosis: a non-experimental, retrospective, cohort study. Clin Drug Investig 2010;30:89-100
- Tan H, Cai Q, Agarwal S, et al. Impact of adherence to disease-modifying therapies on clinical and economic outcomes among patients with multiple sclerosis. Adv Ther 2011;28:51-61
- Devonshire V, Lapierre Y, Macdonell R, et al. The Global Adherence Project (GAP): a multicenter observational study on adherence to disease-modifying therapies in patients with relapsing-remitting multiple sclerosis. Eur J Neurol 2011;18:69-77
- Fox RJ, Miller DH, Phillips JT, et al. Placebo-controlled phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N Engl J Med 2012;367:1087-97
- Calabresi PA, Kieseier BC, Arnold DL, et al. Pegylated interferon beta-1a for relapsing-remitting multiple sclerosis (ADVANCE): a randomised, phase 3, double-blind study. Lancet Neurol 2014;13:657-65
- Chilcott J, McCabe C, Tappenden P, et al. Modelling the cost effectiveness of interferon beta and glatiramer acetate in the management of multiple sclerosis. Commentary: evaluating disease modifying treatments in multiple sclerosis. BMJ 2003;326:522; discussion 522
- Gani R, Giovannoni G, Bates D, et al. Cost-effectiveness analyses of natalizumab (Tysabri) compared with other disease-modifying therapies for people with highly active relapsing-remitting multiple sclerosis in the UK. Pharmacoeconomics 2008;26:617-27
- Cooper K, Bryant J, Harris P, et al. Alemtuzumab for the treatment of relapsing-remitting multiple sclerosis: a single technology appraisal. Evidence Review Group Report commissioned by the NIHR HTA Programme on behalf of NICE. The Southampton Health Technology Assessments Centre (SHTAC), 2013. https://www.nice.org.uk/guidance/ta312/documents/multiple-sclerosis-relapsingremitting-alemtuzumab-evaluation-report3. Accessed November 2, 2014
- CRD and CHE Technology Assessment Group. Fingolimod for the treatment of relapsing remitting multiple sclerosis. University of York, Heslington, York: Centre for Reviews and Dissemination/Centre for Health Economics, 2011. http://www.nice.org.uk/guidance/ta254/documents/multiple-sclerosis-relapsingremitting-fingolimod-appraisal-consultation-evidence-review-group-report2. Accessed September 4, 2014
- Peninsula Technology Assessment Group (PenTAG). The effectiveness and cost-effectiveness of natalizumab for multiple sclerosis: an evidence review of the submission from Biogen. Evidence Review Group Report commissioned by NHS R&D HTA Programme on behalf of NICE. 2007. https://www.nice.org.uk/guidance/ta127/documents/multiple-sclerosis-natalizumab-evaluation-report-evidence-review-group-report2. Accessed Septembber 4, 2014
- Husereau D, Drummond M, Petrou S, et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS)–explanation and elaboration: a report of the ISPOR Health Economic Evaluation Publication Guidelines Good Reporting Practices Task Force. Value Health 2013;16:231-50
- Scottish Medicines Consortium (SMC). Guidance to manufacturers for completion of New Product Assessment Form (NPAF). Glasgow, Scotland: Scottish Medicines Consortium, 2016. http://www.scottishmedicines.org.uk/Submission_Process/Submission_guidance_and_forms/Templates-Guidance-for-Submission/Templates-Guidance-for-Submission. Accessed October 3, 2016
- HM Treasury. The Green Book: appraisal and evaluation in Central Government. London: HM Treasury, 2003, updated 2011. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/220541/green_book_complete.pdf. Accessed April 30, 2015
- Orme M, Kerrigan J, Tyas D, et al. The effect of disease, functional status, and relapses on the utility of people with multiple sclerosis in the UK. Value Health 2007;10:54-60
- Patzold U, Pocklington PR. Course of multiple sclerosis. First results of a prospective study carried out of 102 MS patients from 1976-1980. Acta Neurol Scand 1982;65:248-66
- Pokorski RJ. Long-term survival experience of patients with multiple sclerosis. J Insur Med 1997;29:101-6
- Eurostat. Harmonised Indices of Consumer Prices. HICP (2005 = 100)-monthly data (index). Luxembourg: European Commission, 2016. http://ec.europa.eu/eurostat/web/hicp/data/database. Accessed January 30, 2016
- Hernandez L, Guo S, Kinter E, et al. Cost-effectiveness analysis of peginterferon beta-1a compared with interferon beta-1a and glatiramer acetate in the treatment of relapsing-remitting multiple sclerosis in the United States. J Med Econ 2016;19:684-95
- Weinshenker BG, Bass B, Rice GP, et al. The natural history of multiple sclerosis: a geographically based study. Section 1. Clinical course and disability. Brain 1989;112:133-46
- Weinshenker BG, Bass B, Rice GP, et al. The natural history of multiple sclerosis: a geographically based study. Section 2. Predictive value of the early clinical course. Brain 1989;112:1419-28
- Tolley K, Hutchinson M, You X, et al. A network meta-analysis of efficacy and evaluation of safety of subcutaneous pegylated interferon Beta-1a versus other injectable therapies for the treatment of relapsing-remitting multiple sclerosis. PLoS One 2015;10:e0127960
- Vermersch P, Czlonkowska A, Grimaldi LM, et al. Teriflunomide versus subcutaneous interferon beta-1a in patients with relapsing multiple sclerosis: a randomised, controlled phase 3 trial. Mult Scler 2014;20:705-16
- Calabrese M, Bernardi V, Atzori M, et al. Effect of disease-modifying drugs on cortical lesions and atrophy in relapsing-remitting multiple sclerosis. Mult Scler 2012;18:418-24
- Bornstein MB, Miller A, Slagle S, et al. A pilot trial of Cop 1 in exacerbating-remitting multiple sclerosis. N Engl J Med 1987;317:408-14
- Cadavid D, Wolansky LJ, Skurnick J, et al. Efficacy of treatment of MS with IFNbeta-1b or glatiramer acetate by monthly brain MRI in the BECOME study. Neurology 2009;72:1976-83
- Comi G, Filippi M, Wolinsky JS. European/Canadian multicenter, double-blind, randomized, placebo-controlled study of the effects of glatiramer acetate on magnetic resonance imaging–measured disease activity and burden in patients with relapsing multiple sclerosis. European/Canadian Glatiramer Acetate Study Group. Ann Neurol 2001;49:290-7
- Ebers GC, Rice G, Lesaux, J, et al. Randomised double-blind placebo-controlled study of interferon (beta)-1a in relapsing/remitting multiple sclerosis. Lancet 1998;352:1498-504
- Etemadifar M, Janghorbani M, Shaygannejad V. Comparison of Betaferon, Avonex, and Rebif in treatment of relapsing-remitting multiple sclerosis. Acta Neurol Scand 2006;113:283-7
- Johnson KP, Brooks BR, Cohen JA, et al. Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis: results of a phase III multicenter, double-blind placebo-controlled trial. The Copolymer 1 Multiple Sclerosis Study Group. Neurology 1995;45:1268-76
- Lublin FD, Cofield SS, Cutter GR, et al. Randomized study combining interferon and glatiramer acetate in multiple sclerosis. Ann Neurol 2013;73:327-40
- Mikol DD, Barkhof F, Chang P, et al. Comparison of subcutaneous interferon beta-1a with glatiramer acetate in patients with relapsing multiple sclerosis (the REbif vs Glatiramer Acetate in Relapsing MS Disease [REGARD] study): a multicentre, randomised, parallel, open-label trial. Lancet Neurol 2008;7:903-14
- O'Connor P, Filippi M, Arnason B, et al. 250 microg or 500 microg interferon beta-1b versus 20 mg glatiramer acetate in relapsing-remitting multiple sclerosis: a prospective, randomised, multicentre study. Lancet Neurol 2009;8:889-97
- Schwid SR, Panitch HS. Full results of the Evidence of Interferon Dose-Response-European North American Comparative Efficacy (EVIDENCE) study: a multicenter, randomized, assessor-blinded comparison of low-dose weekly versus high-dose, high-frequency interferon beta-1a for relapsing multiple sclerosis. Clin Ther 2007;29:2031-48
- Human Mortality Database. National Life Tables 2013, Scotland. Berkeley, CA: University of California; Germany: Max Planck Institute for Demographic Research, 2015. http://www.mortality.org/cgi-bin/hmd/country.php?cntr=GBRTENW&level=1. Accessed April 30, 2015
- Whitehead SJ, Ali S. Health outcomes in economic evaluation: the QALY and utilities. Br Med Bull 2010;96:5-21
- Tyas D, Kerrigan J, Russell N, et al. The distribution of the cost of multiple sclerosis in the UK: how do costs vary by illness severity? Value Health 2007;10:386-9
- Curtis L, Burns A. Unit Costs of Health and Social Care 2015. Canterbury, UK: Personal Social Services Research Unit, The University of Kent, 2015. http://www.pssru.ac.uk/project-pages/unit-costs/2015/. Accessed February 12, 2016
- National Health Service (NHS). National Schedule of Reference Costs. Year 2014-15 - All NHS trusts and NHS foundation trusts - Outpatient Attendances Data. London: Department of Health, 2015. https://www.gov.uk/government/publications/nhs-reference-costs-2014-to-2015. Accessed January 30, 2016
- National Health Service (NHS). National Schedule of Reference Costs. Year 2014-15 - NHS trusts and NHS foundation trusts. Direct Access Pathology Services (DAPS): Haematology (DAPS05). London: Department of Health, 2015. https://www.gov.uk/government/publications/nhs-reference-costs-2014-to-2015. Accessed January 30, 2016
- National Health Service (NHS). National Schedule of Reference Costs. Year 2014-15 - NHS trusts and NHS foundation trusts. Direct Access Pathology Services (DAPS): Clinical Biochemistry (DAPS04). London: Department of Health, 2015. https://www.gov.uk/government/publications/nhs-reference-costs-2014-to-2015. Accessed January 30, 2016
- National Institute for Health and Clinical Excellence (NICE). Natalizumab Costing Template and Report. Technology Appraisal Guidance TA127: Natalizumab for the treatment of adults with highly active relapsing-remitting multiple sclerosis. London: National Institute for Health and Clinical Excellence, 2007. http://www.nice.org.uk/guidance/ta127/costing. Accessed September 4, 2014
- Barendregt JJ. The life table method of half cycle correction: getting it right. Med Decis Making 2014;34:283-5
- Palace J, Bregenzer T, Tremlett H, et al. UK multiple sclerosis risk-sharing scheme: a new natural history dataset and an improved Markov model. BMJ Open 2014;4:e004073
- Inusah S, Sormani MP, Cofield SS, et al. Assessing changes in relapse rates in multiple sclerosis. Mult Scler 2010;16:1414-21
- Jansen JP, Fleurence R, Devine B, et al. Interpreting indirect treatment comparisons and network meta-analysis for health-care decision making: report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: part 1. Value Health 2011;14:417-28
- Hoaglin DC, Hawkins N, Jansen JP, et al. Conducting indirect-treatment-comparison and network-meta-analysis studies: report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: part 2. Value Health 2011;14:429-37
- Kieseier BC, Arnold DL, Balcer LJ, et al. Peginterferon beta-1a in multiple sclerosis: 2-year results from ADVANCE. Mult Scler 2015;21:1025-35
- Fogarty E, Schmitz S, Tubridy N, et al. Comparative efficacy of disease-modifying therapies for patients with relapsing remitting multiple sclerosis: systematic review and network meta-analysis. Mult Scler Relat Disord 2016;9:23-30