Abstract
Petroleum ether (60–80°C) extract of Aerva lanata. (L.) Juss Ex. was prepared and partially purified by preparative thin-layer chromatography (TLC). The partially purified fraction (PPF) showed significant cytotoxicity against Daltons lymphoma ascites (DLA) tumor cell lines in vitro. and stimulated lymphocyte proliferation in in vitro. and in vivo. conditions. DLA-bearing animals when treated with PPF showed increase in life span compared to control animals. PPF was also found to be hepatoprotective as evidenced from the normal levels of liver marker enzymes compared to the elevated levels of these enzymes in DLA alone inoculated animals. The lipid, hemoglobin, and WBC levels were normal in PPF-treated animals, indicating a low proliferation of tumor cell in peritoneal cavity. Preliminary phytochemical analysis of PPF showed the presence of alkaloids. These results indicate that PPF contains nontoxic immunomodulatory compounds.
Introduction
Cancer continues to be one of the leading causes of mortality in the world and claims more than 6 million lives every year. An extremely promising strategy for cancer prevention today is chemoprevention, which is defined as the use of synthetic or natural agents (alone/combination) to block the development of cancer (Abdullaev et al., Citation2000). As a result, herbal medicine has become an integral part of standard health care, based on a combination of time-honored traditional usage and ongoing scientific research.
Currently, there is a growing trend to accept plant medicines because of their low toxicity and high medicinal effectiveness. Many researchers have reported the importance of various medicinal plants (Bhakuni et al., Citation1969; Mohanan & Devi, Citation1997). Burgeoning interest in medicinal herbs has increased scientific scrutiny of their therapeutic potential and safety. Some of the medicinal plants are believed to enhance the natural resistance of the body to infections (Girija & Kuttan, Citation1992a; Tiwari et al., Citation2004). Indian Ayurvedic agents are also capable of inducing differentiation of many tumor cell lines (Prasad et al., Citation1993).
The immune system plays an important role in physical and chemical carcinogenesis as well as pathophysiological mechanisms of many diseases (Mohammed, Citation1996; Nishiguri et al., Citation1996). The role of host immune function has become increasingly important in our understanding of the mechanisms that are involved in the body's ability to prevent cancer. In the establishment of lymphoma, some factor(s) are involved that directly or indirectly suppress bone marrow cytokine secretion that is essential for optimum immune response (Ghosal et al., Citation1990). It is known that several cancer chemopreventive agents can modulate immune functions (Sugiura et al., Citation1998). Immunomodulators are being used today in cancer therapy either in combination with chemotherapy or after chemotherapy and radiation therapy. They have a biphasic effect: some stimulate while some inhibit host defense system (Ramnath et al., Citation2001).
Aerva lanata. (L.) Juss. Ex. (Amaranthacae) is an important medicinal shrub growing in India along the wastelands (David, Citation1963). This is mainly used as a demulcent, diuretic, and in lithiasis (Khalidpad, Citation1973). The plant is very effective against cough, as vermifuge for children, and in the treatment of headache (Hassan, Citation1993). Different solvent fractions of the whole plant were found to have antimicrobial and cytotoxic activity against different cell lines (Chowdhury et al., Citation2002; Nevin & Vijayammal, Citation2003). The ethanol extract of A. lanata. was found to be effective against diabetes mellitus in alloxan-induced rats (Vetrichelvan & Jagadeesan, Citation2002).
Based on these facts, we focused our study on the effect of the partially purified fraction (PPF) to induce proliferation of lymphocytes in mice inoculated with Daltons lymphoma ascites (DLA) cell line, on life span, and on lipid levels of the inoculated animal. To justify this, an in vitro. lymphocyte proliferation study using different concentrations of the PPF was also done. In addition, a toxicological evaluation of the PPF was performed.
Materials and Methods
Chemicals and biochemicals
Petroleum ether (SD Chemicals, Mumbai, India), dimethyl sulfoxide (Ranbaxy, Mumbai, India), fetal calf serum, RPMI 1640 medium, histopaque, concanavalin A (Sigma Chemicals, St. Louis, MO, USA), and 3H-thymidine (Bharat Radio Isotope Technology [BRIT], Mumbai, India) were used.
Plant material
A. lanata. was collected from Thiruvananthapuram district, India, and was identified by our botanist, Dr. Remeshkumar, RA, Central Tuber Crops Research Institute (Thiruvananthapuram, India). The powdered shade-dried whole plant was subjected to Soxhlet extraction for 20 h using petroleum ether (60–80°C) and was partially purified by preparative silica gel G thin-layer chromatography (TLC) eluting with MeOH-acetone-triethanolamine (50:50:1.5). The partially purified fraction tested positive with Dragendroff's reagent, characteristic to alkaloids. Preparative TLC was done to obtain a large amount of this fraction and was used for in vivo. studies.
Assay of cytotoxicity
Cytotoxic effect of different concentration of PPF against DLA cell was conducted. Briefly, different concentrations of PPF in 10% DMSO (10–50 µg) was incubated with 1 × 106 cells/ml saline for 3 h at 37°C. After the incubation, viability of the cells was determined by the Trypan blue exclusion method. Percentage cytotoxicity was calculated after comparing with untreated control (Talwar, Citation1983).
Experimental tumorigenesis
Male Swiss bred albino mice from inbred stock having an average of 25 g body weight were used for the experiments. They were housed in standard environmental conditions and fed a rodent diet and water ad libitum.. The animals (6 mice/group) were grouped as follows: group I, control + 10% DMSO; group II, 1 × 106 DLA cells/0.1 ml saline; group III, PPF in 10% DMSO; group IV, 100 mg/kg body weight PPF administered 24 h after tumor cell inoculation; group V, 100 mg/kg body weight PPF administered 7 days after tumor cell inoculation. The experimental period was 16 days. After 16 days, blood was collected for estimating various hematological and pharmacological parameters.
Toxicological evaluation
The animals were grouped and treated as above. Clinical parameters, like serum oxaloactate transaminase (SGOT, E.C. 2.1.1) and glutamate pyruvate transaminase (SGPT, E.C. 2.6.1.2) were estimated by the procedure of Reitman and Frankel (Citation1957). Alkaline phosphatase (ALP, E.C. 3.1.3.1) was estimated using phenyl phosphate as substrate; 0.5 ml substrate was incubated at 37°C for 3 min followed by the addition of 0.1 ml serum and again incubated for 30 min at 37°C. Dinitrophenyl hydrazine (DNPH) (5 ml) was added to the mixture. The reaction was stopped by the addition of 5 ml 0.4 M NaOH. Optical density was measured at 512 nm (Posen, Citation1967). Lactate dehydrogenase (LDH, E.C. 1.1.1.27) was assayed colorimetrically using pyruvate as substrate in presence of NADH as coenzyme (Wootton, Citation1964).
Life span studies
Animals were grouped as follows: group I, 1 × 106 DLA cells/0.1 ml saline; group II, 100 mg PPF in 10% DMSO; group III, 100 mg/kg body weight PPF administered 24 h after tumor cell inoculation; group IV, 100 mg/kg body weight PPF administered 7 days after tumor cell inoculation. Life span studies were started 15 days after tumor inoculation and continued up to 65 days. The number of animals alive was measured every 10 days thereafter. Percentage increase in life span (%ILS) was calculated from the formula:
Lymphocyte proliferation assay
Mouse blood was collected in heparanized tubes by retrorbital bleeding. An equal amount of phosphate-buffered saline (PBS) was added to the tubes. Blood samples were layered over 1.5 ml histopaque and centrifuged at 1600 rpm for 20 min; lymphocytes formed at the interface were removed using a Pastuer pipette, counted, and adjusted to 2 × 105 cells in a total volume of 0.5 ml RPMI 1640 medium.
Lymphocytes were placed in a 5-ml screw-capped tube, 15 µg/tube of concanavalin were added and incubated in an atmosphere of 5% CO2 at 37°C for 18 h. 3H-Thymidine (2 µCi, sp. activity 18,500 µCi/mmol), concanavalin A (15 µg/tube), and PPF (50 and 100 µg) were added to the culture 18 h prior to harvesting the cells. Cells were washed with isotonic saline (containing 5% TCA) followed by methanol. After that, cells were suspended in 5 ml scintillation fluid and radioactivity measured in LKB-β liquid scintillation counter (Froebel et al., Citation1999; Ilback et al., Citation1991).
Hematological parameters
Blood was drawn from each mouse in the conventional way (Talwar, Citation1983). White blood cells (WBC) and hemoglobin (Hb) levels were determined. The differential count was carried out with Lieshmans stain (Wintrobe, Citation1961).
Serum cholesterol and triglycerides
Lipids were extracted from serum by the method of Folch et al. (Citation1957) using chloroform-methanol (2:1). Cholesterol was estimated by ferric chloride reagent as described by Zlatkis et al. (Citation1953). Triglycerides were measured according to Van Handel and Zilversmit (Citation1957).
Statistical analysis
The results were statistically analyzed by one-way ANOVA using SPSS 10 with Duncan's variance.
Results
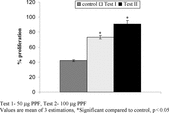
Cytotoxic effects of different concentrations of PPF on DLA cells in vitro. are shown in . PPF (50 µg) showed 100% death of cell lines. The total WBC and lymphocyte count was found to be significantly decreased in group II animals inoculated with DLA cells compared with normal animals. Group III animals (PPF alone treated) showed an increase in both WBC and lymphocyte count compared to normal and cancer control. In the differential count of WBC, the percentage of lymphocytes (30%) was found to be significantly decreased in DLA-inoculated animals compared to normal animals (65.9%). PPF treatment of DLA-bearing animals restored these parameters to the normal level (). PPF alone treated groups showed no significant difference from the normal animals. The in vitro. lymphocyte proliferation assay, using 50 and 100 µg of PPF, showed 53% and 73% increase in proliferation, respectively, compared to control (42%) ().
Table 1.. Cytotoxic effect of different concentrations of partially purified fraction (PPF) against Daltons lymphoma ascites (DLA) cells.
Table 2.. Effect of partially purified fraction (PPF) on hematological parameters in Daltons lymphoma ascites (DLA) control and treated animals.
The serum glutamate pyruvate transaminase (GPT), glutamate oxaloacetate transaminase (GOT), alkaline phosphatase (ALP), and lactate dehydrogenase (LDH) levels in DLA control animals were highly elevated but are regulated in PPF-treated groups. The enzyme levels in the PPF alone administered animals (group III) showed no significant change from the normal animals ().
Table 3.. Effect of partially purified fraction (PPF) on liver marker enzymes.
Life span of animals inoculated with DLA cells and PPF-treated is shown in . DLA alone treated animals have a very short life span, but with PPF-treated animals the life span was increased to 92% and 42%, respectively. The levels of serum cholesterol and triglyceride level of DLA control and treated animals are show in and . The cholesterol levels of DLA control animals were very high (281 mg/dl) compared to other groups. The triglyceride levels were much lower in the DLA control animals; in treated groups it was slightly higher compared with normal animals.
Table 4.. Life span of Daltons lymphoma ascites (DLA) cells inoculated and treated animals.
Discussion
The body's immunity has been shown to be suppressed in several diseases like AIDS and cancer, and the use of immunomodulatory agents can solve these problems to a greater extent. Immunomodulation is a procedure that enhances the immune function of the organism by interfering with its function. If drug treatment enhances immune reactions, it is termed an immunostimulative drug, which primarily implies stimulation of nonspecific system, that is, granulocytes, macrophages, complement, certain T lymphocytes, and different effector substances (Patwardhan, Citation1990). The greatest disadvantage of using synthetic immunomodulatory agents is their side effects, viz., neutropenia, anorexia, and proteinemia (Herberman, Citation1985). So plant-based immunomodulators can thus become a better choice for chemotherapy (Girija & Kuttan, Citation1992b; Leemol & Kuttan, Citation2000; Tanaka et al., Citation1999). Various plant extracts have immunostimulatory activity as evidenced by increased proliferation of lymphocytes and production of interleukin-2 (Rajagopal et al., Citation2003). Wagner (Citation1990) has reviewed various plant-derived natural products with immunostimulatory activity. They include alkaloids, quinones, terpenoids, phenolcarboxylic acids, and high-molecular-mass-compounds such as polysaccharides and glycoproteins.
Hematological evaluation also indicates that DLA-bearing mice experience significant immunosuppression that is evident from the low WBC and lymphocyte counts. In the case of PPF-administered DLA-bearing animals, the WBC and lymphocyte count were restored to normal levels.
In cancer chemotherapy, major problems are myelosuppression and anemia (Maseki et al., Citation1981; Price & Greenfield, Citation1958). The anemia encountered in tumor-bearing mice is mainly due to reduction in RBC/hemoglobin percentage, and this may occur either due to iron deficiency or due to hemolytic or myelopathic conditions (Fenninger & Mider, Citation1954). Treatment with PPF restored the hemoglobin content; RBC and WBC cell counts were near normal values. This indicates that the active component of PPF has protective action on the hematopoietic system. No autohemolysis was observed in any groups.
In vitro. lymphocyte proliferation studies using different concentrations of PPF showed an increase in the amount of lymphocytes, indicating that PPF is capable of causing the proliferation of the lymphocytes by enhancing DNA synthesis. The amount of radioactivity incorporated into DNA is proportional to the number of proliferating cells, which in turn is a function of the number of lymphocytes that are stimulated by a given antigen to enter the proliferate phase (Weinberg et al., Citation1998). The link between lymphocyte proliferation and antitumor activity is well established (Kyo et al., Citation1999, 1998).
The toxicity evaluation of PPF showed no toxic symptoms or any significant changes in the SGOT, SGPT, ALP, and LDH levels in normal mice. Meanwhile, highly elevated levels were observed in the DLA control animals. Aminotransferases are among a group of ubiquitous enzymes whose serum activity is elevated in many different disease states, including myocardial infarction, viral and toxic hepatitis, muscular dystrophy, and cancer. High LDH activity occurs in malignant diseases and is included among the tumor marker enzymes. In the DLA-inoculated mice, administration of PPF restored the elevated levels to the baseline values. These observations clearly proved that PPF has a significant hepatoprotective effect. These results agree with previous studies indicating that A. lanata. has a significant hepatoprotective effect (Majmudar & Shah, Citation1999).
Depletion of muscle proteins, glycogen, and lipids leads to an elevation of free cholesterol and a decrease in neutral fat. The loss of major nutrients, particularly excessive catabolism of lipids, leads to alterations in the activity of host tissue enzymes and changes in endocrine homeostasis and immunologic mechanisms, cachexic, chronic and acute cathetic conditions, finally leading to the death of animals (Theologides, Citation1979). Hypercholesterolemia has been reported in various disease conditions (Ellefson, Citation1976). Hypercholesterolemia associated with the DLA-inoculated animals may be due to the mobilization of fat from the stores to meet the increased demand of the tumor-bearing host. The lipemia subsides gradually as the fat stores are emptied to meet the caloric requirements of the host during acute cancer condition (Bloor, Citation1955). In the current study, serum triglyceride content decreased compared to normal animals. This is in agreement with the previous study in fibrosarcoma-bearing rats (Parimala, Citation1993).
Preliminary phytochemical analysis of PPF showed the presence of alkaloids. Previous photochemical analyses of A. lanata. have show the presence of alkaloids (β.-carboline and canthin-6-one) and flavonoid O.-acylglycosides (Zapesochnaya et al., Citation1991, 1992a,b). Alkaloids are an important class of natural compounds that possess various biological activities (Ganguly & Khar, Citation2002). Alkaloids from various plants have been shown to stimulate lymphocyte proliferation (Anis et al., Citation1999; Mikami et al., Citation1999; Wessel et al., Citation1987).
The alkaloid β-carbolines are highly lipophilic and are present in fruits and plants and found to act as antioxidants when absorbed and accumulated in the body (Herraiz, 2003). Previous studies have showed that the β-carboline harmine has cytotoxical activity. It induced crossing-over and mitotic gene conversion and DNA single- and/or double-strand breaks in yeast Saccharomyces cerevisiae. (Boeira, Citation2002). The alkaloid canthine, isolated from plants, has shown significant cytotoxicity against 12-O.-tetradecanoylphorbol-13-acetate (TPA) induced Epstein-Barr virus early antigen (EBV-EA) activation (Murakami et al., Citation2004). The lymphocyte proliferation activity and cytotoxic activity of PPF on DLA cells may be due to the presence of this alkaloid.
From these results, we conclude that PPF contains alkaloids having a nontoxic immunostimulatory effect. More work on the elucidation of the biochemical mechanisms associated with the effects of PPF on lymphoproliferation may be useful for maximizing the therapeutic effectiveness of the extract.
Acknowledgments
The authors are thankful to the Director, Regional Cancer Center, Thiruvananthapuram, India, for providing the DLA cell line.
References
- Abdullaev FI, Luna RR, Roitenburd BV, Espinosa AJ (2000): Pattern of childhood cancer mortility in Mexico. Arch Med Res 31: 526–531. [PUBMED], [INFOTRIEVE], [CSA]
- Anis KV, Kuttan G, Kuttan R (1999): Role of berberine as an adjuvant response modifier during tumor therapy in mice. Pharm Pharmacol Commun 5: 697–700. [CSA]
- Bhakuni DS, Dhar ML, Dhar MM, Dhawan BN, Hehrotra BN (1969): Screening of Indian medicinal plants for biological activity, Part II. Ind J Exp Biol 7: 250–262. [CSA]
- Bloor WR, Haven FL (1955): The weight and lipid content of the intestines in rats with Walker carcinoma 256. Cancer Res 15: 173–176. [PUBMED], [INFOTRIEVE], [CSA]
- Boeira JM, Viana AF, Picada JN, Henriques JA (2002): Genotoxic and recombinogenic activities of the two beta-carboline alkaloids harman and harmine in Saccharomyces cerevisiae.. Mutat Res 500: 39–48. [PUBMED], [INFOTRIEVE], [CSA]
- Chowdhury D, Sayeed A, Islam A, Shah Alam Bhuiyan M, Astaq Mohal Khan GR (2002): Antimicrobial activity and cytotoxicity of Aerva lanta.. Fitoterapia 73: 92–94. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- David P (1963): Bengal Plants, Vol 2. Calcutta, Botanical Survey of India, pp. 653.
- Ellefson RD, Caraway WT (1976): Lipids and lipoproteins. In: Tietz N, ed., Fundamentals in Clinical Chemistry. Philadelphia, WB Saunders, pp. 474–541.
- Fenninger LD, Mider GB (1954) In: Haddow A, Grenstein JP, eds., Advances in Cancer Research, Vol. 2. New York, Academic Press, p. 244.
- Folch J, Lees, M, Stanley GHS (1957): A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226: 497–509. [PUBMED], [INFOTRIEVE], [CSA]
- Froebel KS, Pakker NG, Aiuti F, Bofill M, Choremi-Papadopoulou H, Economidou J, Rabian C, Roos MTL, Ryder LP, Miedema F, Raab GM (1999): European concerted action on immunological and virological markers of HIV disease progression: Standardization and quality assurance of lymphocyte proliferation assays for use in assessment of immune function. J Immunol Methods 227: 85–97. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Ganguly T, Khar A (2002): Induction of apoptosis in human erythroleukemic cell line k562 by Trylophora alkaloids involves release of cytochrome c and activation of caspase 3. Phytomedicine 9: 288–295. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Ghosal D, Barua AK, Roy S, Bhattacharya HM (1990): Vaccine failure in malnourished animals: Use of a bio-immunomodulator to improve immunocompetence. Nutrition 6: 153–157. [CSA]
- Girija K, Kuttan R (1992a): Immunomodulatory acivity of a peptide isolated from viscum album. extract. Immun Inves 21: 285–290. [CSA]
- Girija K, Kuttan R (1992b): Immunological mechanism of action of the tumor reducing peptide from mistletoe extract (NSC 635089) cellular proliferation. Cancer Lett 30: 123–130. [CSA]
- Hassan D, Mazahanul A, Amadeer BS (1993): Botany. Dhaka, Dhaka University, p. 3.
- Herberman RB, Pinsky CM (1985): Polyribonucleotides for cancer therapy: Summary and recommendations for further research. J Biol Res 4: 680–683. [CSA]
- Herraiz T, Galisteo J (2003): Tetrahydro-beta-carboline alkaloids occur in fruits and fruit juices. Activity as antioxidants and radical scavengers. J Agric Food Chem 51: 7156–7161, [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Ilback NG (1991): Effects of methyl mercury exposure via placenta and milk impairs natural killer (NK) cell activity in the mouse. Toxicology 67: 117–124. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Khalidpad DB, Akkori SG (1973): Bharatia Banoushad, Vol. 5. Calcutta, India, Calcutta University Publishers, p. 576.
- Kyo E, Uda N, Kasuga S, Itakura Y(1998): Immunomodulatory effects of aged garlic extract. Conference on Recent Advances on the Nutritional Benefits Accompanying the Use of Garlic as a Supplement. Newport Beach, California, The Pennsylvania State University and the National Cancer Institute, November 15–17.
- Kyo E, Uda N, Kasuga S, Itakura Y, Sumiyoshi H (1999): Garlic as an Immunostimulant. Immunomodulatory Agents from Plants. Basel, Switzerland, Birkhäuser Verlag, pp. 273–288.
- Leemol D, Kuttan G (2000): Immunomodulatory activity of Withania somnifera.. J Ethnopharmacol 71: 193–200. [CSA], [CROSSREF]
- Majmudar FI, Shah MB (1999): Aerva lanata.: Its diuretic and hepatoprotective activity. Ind J Nat Prod 15: 9–12. [CSA]
- Maseki M, Nishiagaki I, Hagishara M, Tomoda Y, Yagi K (1981): Lipid peroxidation levels and lipid content of serum lipoprotein fractions of pregnant subjects with or with out preeclampsia. J Clin Chim Acta 41: 424–426. [CSA]
- Mikami M, Kitahara M, Kitano M, Ariki Y, Mimaki Y, Sashida Y, Yamazaki M, Yui S (1999): Suppressive activity of lycoricidinol (narciclasine) against cytotoxicity of neutrophil derived calprotectin and its suppressive effect on rat adjuvant arthritis model. Biol Pharm Bull 22: 674–678. [PUBMED], [INFOTRIEVE], [CSA]
- Mohammed Z (1996): Studies on the immunomodulatory effect of Ashwagandha.. J Ethnopaharmacol 50: 69–76. [CSA], [CROSSREF]
- Mohanan PV, Devi KS (1997). Effect of Sobatum. on tumor development and chemically induced carcinogenesis. Cancer Lett 112: 219–223, [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Murakami C, Fukamiya N, Tamura S, Okano M, Bastow KF, Tokuda H, Mukainaka T, Nishino H, Lee K-H (2004): Multidrug-resistant cancer cell susceptibility to cytotoxic quassinoids, and cancer chemopreventive effects of quassinoids and canthin alkaloids. Bioorg Med Chem 12: 4963–4968. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Nevin KG, Vijayammal PL (2003): Effect of Aerva lanata. on solid tumor induced by DLA cells in mice. Fitoterapia 74: 578–82. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Nishiguri C, Yorosh DB, Donawho C, Kripke ML (1996): The immune system in ultraviolet carcinogenesis. J Invest Dermatol Symp Proc 1: 143–146. [CSA]
- Parimala RK, Kumar, Sachidanandam P (1993): Effect of panchakavvyam in fibrosarcoma-bearing rats. Ancient Sci Life 13: 148–153. [CSA]
- Patwardhan B, Kalbag D, Patki PS, Nagsampagi BA (1990): Search of immunomodulatory agents: A review. Indian Drugs 28: 348–358, [CSA]
- Posen S (1967): Alkaline phosphatase. Ann Intern Med 67: 183–203. [PUBMED], [INFOTRIEVE], [CSA]
- Prasad KN, Parry P, Chang C (1993): Ayurvedic agents produce differential effects in murine and human melanoma cells in vitro.. Nutr Cancer 20: 79–86. [PUBMED], [INFOTRIEVE], [CSA]
- Price VE, Greenfield RE (1958): Anemia in cancer. In: Greenstein JP, Haddow A, eds., Advances in Cancer Research, Vol. 5. New York, Academic Press, pp. 199–200.
- Rajagopal S, Ajaya kumar R, Deevi D, Satyanarayana C, Rajagopalan R (2003): Andrographolide, a potential cancer therapeutic agent isolated from Andrographis paniculata.. J Exp Ther Oncol 3: 147–158. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Ramnath V, Girija K, Kuttan R (2001): Immunomodulatory effect of Abrin in normal mice. Amala Cancer Bull 21: 76–81. [CSA]
- Reitman S, Frankel S (1957): A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J Clin Pathol 28: 56. [PUBMED], [INFOTRIEVE], [CSA]
- Sugiura H, Inaba R, Iwata H, Nishida H, Tanaka T (1998): Modifying effects of Maharishi Amrit Kalash 4 and 5 on phagocytic and digestive functions of macrophages in male ICR mice. Environ Health Prev Med 3: 50–54. [CSA]
- Talwar GP (1983): A Hand Book of Practical Immunology. New Delhi, Vikas Publishing House pvt Ltd, p. 41.
- Tanaka T, Sugiura H, Inaba R, Nishikawara A, Murakami A, Koshimizu K, Ohigashi H (1999): Immunomodulatory action of citrus auraptene on macrophage functions and cytokine production of lymphocytes in female BALB/c mice. Carcinogenesis 20: 1471–1476. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Theologides A (1979): Cancer cachexia. Cancer 43: 2004–2012. [PUBMED], [INFOTRIEVE], [CSA]
- Tiwari U, Rastogi B, Singh P, Saraf DK, Vyas SP (2004): Immunomodulatory effects of aqueous extract of Tridax procumbens.in experimental animals. J Ethnopharmacol 92: 113–119. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Van Handel E, Zilversmit DB (1957): Micro method for direct determination of serum triglycerides. J Lab Clin Med 50: 152–157. [PUBMED], [INFOTRIEVE], [CSA]
- Vetrichelvan T, Jegadeesan M (2002): Anti-diabetic activity of alcoholic extract of Aerva lanata. (L.) Juss. Ex Schultes in rats. J Ethnopharmacol 80: 103–107. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Wagner H (1990): Search for plant derived natural products with immunostimulatory activity (recent advances). Pure Appl Chem 62: 1217–1222. [CSA]
- Weinberg A, Betensky RA, Zhang L, Ray G (1998): Effect of shipment, storage, anticoagulant and cell separation on lymphocyte proliferation assays for human immunodeficiency virus-infected patients. Clin Diag Lab Immunol 5: 804–807. [CSA]
- Wessel G, El-Menshawi B, Mahran G (1987): Toxic pyrrolizidine alkaloids of certain Boraginaceae. plants. Acta Pharm Suec 24: 199–204. [CSA]
- Wintrobe MM (1961): Clinical Haematology. Philadelphia, Lea and Febiger, pp. 391.
- Wootton IDP (1964): Micro analysis in Medical Biochemistry, 4th ed. London, J & A Churchill Ltd., pp. 117–118.
- Zapesochnaya G, Pervykh LN, Kurkin VA (1991): A study of herb Aerva lanata. III. Alkaloids. Chem. Nat Comp 27: 336. [CSA], [CROSSREF]
- Zapesochanaya G, Kurki V, Okhanov V, Mirshnikov A (1992a): Canthin-6-one and beta-carboline alkaloids from Aerva lanata.. Planta Med 58: 192–196. [CSA]
- Zapesochnaya GG, Kurkin VA, Okhanov VV, Pervykh LN, Miroshnikov AI (1992b): Stucture of alkaloids of Aerva lanata. Chem Nat Comp 27: 725. [CSA], [CROSSREF]
- Zlatkis A, Zak B, Boyle A (1953): A new method for the direct determination of cholesterol. J Lab Clin Med 41: 486. [PUBMED], [INFOTRIEVE], [CSA]