Abstract
Oral administration of a methanolic extract of Cressa cretica. Linn. (Rundati) (Convolvulaceae) (whole plant) at a dose level of 100 mg/kg/day for a period of 60 days led to a significant decrease in the weight of testis, epididymis, seminal vesicle, and ventral prostate. Cressa cretica. reduced the fertility of male rats by 100%. There was a marked reduction in the number of primary spermatocytes, secondary spermatocyte, and spermatids. Sertoli cell counts as well as the cross-sectional surface area were significantly decreased. Leydig cell nuclear area and the number of mature Leydig cells were also significantly decreased. The protein, sialic acid, glycogen, and cholesterol content of the testis, the fructose in the seminal vesicle, and protein and sialic acid in the epididymis were significantly decreased. Serum testosterone levels were also reduced after Cressa cretica. treatment. The RBC and WBC counts, hemoglobin, hematocrit, blood sugar, serum cholesterol, phospholipids, triglyceride, and HDL-cholesterol were within the normal range. The methanol extract of Cressa cretica. produced intrusion in testosterone production and affected spermatogenesis in male albino rats.
Introduction
Cressa cretica. Linn (Rundati) (Convolvulaceae) flowers exhibited cytotoxic and anti-inflammatory activity (Rizk & Heiba, Citation1990). Cressa cretica. is reported to be antibilous, antituberculosis, and expectorant (Rizk & El-Ghazaly, Citation1995). Shahat et al. (Citation2004) yielded five flavonoids (quercetin, quercetin-3-O.-glucoside, kaempferol-3-O.-rhamnoglucoside, and rutin) from the aerial parts of Cressa cretica.. Reports in the literature show that flavonoids produce antiandrogenic activity and affect male fertility in dogs (Bhargava, Citation1989) and rats (da Silveira et al., Citation2003). However, the possible effect of Cressa cretica. on the reproductive system of mammalian species has not been reported. The current study is an attempt to reveal the testicular and biochemical changes in male albino rats after the administration of a methanol extract from Cressa cretica..
Materials and Methods
Plant material and extraction
Cressa cretica. was collected from Chhaparwada dam, Dudu near Jaipur, and identified (voucher no. RUBL-19906) by the herbarium, Department of Botany, University of Rajasthan (Jaipur, India). The whole plants were shade-dried, powdered, and subjected to Soxhlet with methanol (100%) for 46–48 h. The filtrate obtained was concentrated under reduced pressure and washed with petroleum ether for the removal of fat portion. This fat portion was extracted out with benzene. A part of this final extract was employed to study the antifertility activity in male albino rats, and the remainder was used for the isolation of constituent compounds by means of chromatographic techniques over a Si-gel column. The following compounds were obtained: β.-sitosterol, n.-octacosanol, scopoletin umbelliferone, iso.-pimpinelline, isoflanone glycoside, coumaranochrome glycoside, and quercetin.
Animals
Twenty adult male Wistar rats weighing 150–160 g were used for the investigation. Animals were housed in standard rat cages and maintained under standard condition (12-h light/dark cycle: 25±3°C; 35–60 relative humidity), and water and food (commercial diet) were available ad libitum.. Drug and/or vehicle was administered to all animals by oral intubation. All the described experiments have been carried out under the approval of the departmental ethics committee.
Study protocol
Male rats of proven fertility were divided into two groups of 10 rats each. The daily dose of the plant extract was freshly dissolved in 0.5 ml of distilled water and administered to each treated animal every morning for 60 days.
Group 1: Control rats received 0.5 ml day−1 of the vehicle (i.e., distilled water).
Group 2: Rats treated with Cressa cretica. extract at 100 mg kg−1 day−1.
Mating studies
On day 56 toward the end of the treatment period, each male was housed individually with three proestrous females for 3–5 days. The females were scheduled for laparotomy 2 weeks later to determine the number and viability of fetuses. After mating, males were removed for autopsy.
Sperm motility and density
The motility of cauda epididymal spermatozoa was recorded. Percentage of motile sperm was calculated per unit area. Cauda epididymal and testicular sperm counts were assessed using a hemocytometer (Prasad et al., Citation1972).
Quantitative analysis
The evaluation of cell population dynamics was based on the counts of each cell type per cross tubular sections; various cell components were quantitatively analyzed using spherically appearing sections. Abercrombie's correcting factor was introduced (Berndtson, Citation1977) to correct for the better chance a large cell has to be counted. All types of interstitial cells, such as fibroblasts, mature and degenerating Leydig cells, were estimated applying a differential cell count. Diameters of seminiferous tubules were determined by tracing and measuring an average of 100 selected seminiferous tubules. Diameters of Leydig cell nuclei were measured at 800 × .
Hormonal assay
Serum testosterone levels were assayed from samples using a radioimmunoassay method (Belanger et al., Citation1980).
Tissue biochemistry
On day 61, testes, epididymides, seminal vesicles, and ventral prostate were removed, cleared of fat and connective tissue, weighed and kept at − 20°C until assayed for total protein (Lowry et al., Citation1951), sialic acid (Warren, Citation1959), and glycogen (Montgomery, Citation1957).
Blood and serum biochemistry
Blood was collected from heart and analyzed for RBC and WBC counts, hemoglobin, hematocrit, and blood sugar. Serum protein, cholesterol, triglycerides, phospholipids, and HDL-cholesterol were also estimated.
Statistical analysis
Data were analyzed by the Student's t.-test to determine the level of significance between control and treated groups.
Results
Body and organ weights
The oral administration of Cressa cretica. extract did not cause significant change in body weight, but the weight of testes, epididymides, seminal vesicle, and ventral prostate were significantly reduced (p < 0.01) ().
Table 1. Effect of crude extract of Cressa cretica. on the body weight, organ weight, sperm motility, density, and fertility in rats.
Sperm motility, density, and fertility
The Cressa cretica. extract treated rats showed significant reduction (p < 0.001) in the sperm concentration of testes and cauda epididymides. The sperm motility of the cauda epididymides was also reduced significantly (p < 0.001). The extract reduced the fertility of male rats by 100% ().
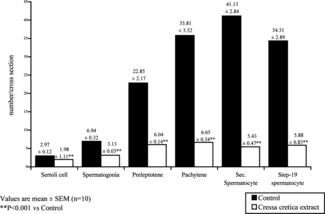
Cell population dynamics
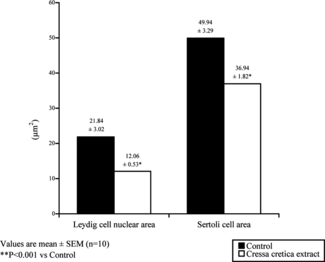
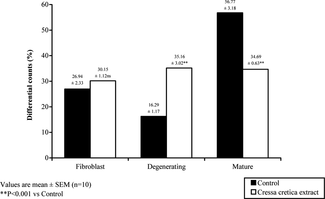
Cressa cretica. extract brought about a significant reduction in most of the germinal cell types. Testicular cell counts, that is, Sertoli cells, spermatogonia, preleptotene, pachytene, sec. spermatocyte and step-19 spermatids (), were shown to be reduced by 33.3%, 54.9%, 73.37%, 81.5%, 86.79%, and 83.1%, respectively. The number of mature Leydig cells were significantly reduced (). Sertoli cell area and the nuclear area of Leydig cells were also reduced ().
Hormonal assay
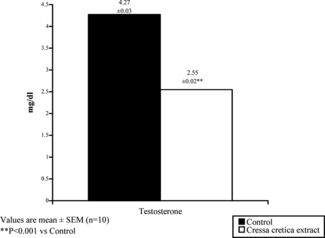
The level of serum testosterone was significantly (p < 0.001) decreased after treatment of Cressa cretica. extract in comparison to controls ().
Tissue biochemistry
Protein content of testes, epididymides, ventral prostate, and seminal vesicle were reduced after Cressa cretica. extract treatment as compared with controls. Sialic acid contents of the testes, epididymides, and auxiliary glands (seminal vesicle and ventral prostate) were depleted. Glycogen contents of testes were decreased significantly (p < 0.001), whereas testicular cholesterol was elevated significantly (p < 0.01). Fructose in seminal vesicle was decreased significantly (p < 0.001) ().
Table 2. Effect of Cressa cretica. L. extract treatment on biochemical parameters of male rats.
Blood and serum biochemistry
RBC and WBC counts, hemoglobin, hematocrit, blood sugar and serum protein, cholesterol, triglyceride, phospholipids, and HDL-cholesterol levels were within the normal range ().
Table 3. Effect of Cressa cretica. L. extract on some components of blood and serum in male rats.
Discussion
The plant Cressa cretica. contains flavonoids, active compounds that may be related to its different therapeutic properties according to folk medicine. Flavonoids present in the Cressa cretica. extract suggested a possible role of this plant as a potential agent in the field of male fertility regulation.
In the current study, the administration of Cressa cretica. extract decreased the weight of testis and accessory sex organs. The epididymis, seminal vesicle, and ventral prostate are all androgen-dependent organs, relying on testosterone for their growth and function (Klinefelter & Hess, Citation1998). A reduction in their weights may reflect a decreased bioavailability and production of androgens, which caused Leydig cell disintegration and reduced Leydig cell nuclei diameters, similar to results obtained with Curcuma longa. (Purohit & Bhagat, Citation2004). Reduced nuclear area evinced the impairment of Leydig cell function and lowered the number of mature Leydig cells. Androgen deprivation also suppresses spermatogenesis leading to low sperm concentration (Rao & Mathur, Citation1998; Rao & Shah, Citation1998), depletion in the number of spermatids; preleptotene and pachytene spermatocytes may affect the androgen-binding protein of Sertoli cells (Gupta et al., Citation2000).
Sperm count and motility, which were markedly reduced in the current study, show a statistically significant correlation with fertility (Bostofte et al., Citation1984). Treatment with the methanol extract possibly inhibits the activity of ATP in spermatozoa by uncoupling oxidative phosphorylation from the respiratory chain and preventing phosphorylation of ADP to ATP (Sharma & Jacob, Citation2002), thus rendering the spermatozoa immotile. Inadequate concentration and sluggishly motile or immotile sperm could not penetrate the cervical mucus and thus failed to fertilize the ova (Lohiya & Ansari, Citation1999; Sharma et al., Citation1999).
The glycogen content in the cell represents the energy storage. The Sertoli cells and spermatogonia contain glycogen and provide nourishment to the seminiferous tubular cells, and glycogen content is also found to be directly proportional to steroid hormone levels (Vijaykumar et al., Citation2004). A decrease in the glycogen content of the testis reduced the energy source for spermatogenic activity. Cressa cretica. treatment caused a significant decrease in total protein and sialic acid. Sialic acid is secreted by the epididymal epithelium and is coated on spermatozoa as they pass through the epididymis. They are concerned with changing the membrane surface of maturing spermatozoa, coating of spermatozoa with certain antigens, and in the development of their fertilizing capacity (Hinton & Palladine, Citation1995; Turner et al., Citation1995). It is likely that the extract might be interfering with the synthesis and/or their release from the principal cells of the epididymal epithelium. According to Lewis et al. (Citation1996), motile sperm consume fructose after ejaculation. Cressa cretica. extract administration caused reduction in fructose concentration, which shows the lack of an energy source in the female body during capacitation. Nontoxicity of the methanol extract of Cressa cretica. is further supported by the data obtained after examination of blood parameters, that is, RBC and WBC counts, hemoglobin, hematocrit, blood sugar and serum protein, cholesterol, triglyceride, phospholipids, and HDL-cholesterol, which remained unaltered. Oral administration of the 100% methanol extract of Cressa cretica. to male rats is responsible for the decline in testosterone production and also alters spermatogenic activity without adverse toxicity.
Acknowledgments
The authors are thankful to the Head, Department of Zoology and Chemistry; Prof. N.K. Lohiya, Coordinator, CAS, Department of Zoology, University of Rajasthan, Jaipur, India, for providing necessary facilities.
References
- Belanger A, Caron S, Picard V (1980): Simultaneous radioimmuno-assay of progestins, androgens and estrogens in rat testis. J Steroid Biochem 13: 185–192. [INFOTRIEVE], [CSA]
- Berndtson WE (1977): Methods for quantifying mammalian spermatogenesis: A review. J Am Sci 44: 818–833. [CSA]
- Bhargava SK (1989): Antiandrogenic effects of a flavonoids rich fraction of Vitex negundo. seeds: A histological and biochemical study in dogs. J Ethnopharmacol 27: 327–339. [INFOTRIEVE], [CSA], [CROSSREF]
- Bostofte E, Jorgen S, Rebbe H (1984): Interrelation among the characteristic of human semen and a new system for classification of male infertility. Fertile Sterile 41: 95–102. [CSA]
- da Silveirae Sa, R de C, Leite MN, Reporedo M de M, de Almeida RN (2003): Evaluation of long term exposure to Mikania glomerata. (Sprengel) extract on male Wistar rats' reproductive organs, sperm production and testosterone level. Contraception 67: 327–331. [CSA], [CROSSREF]
- Gupta RS, Kumar P, Dixit VP, Dhobal MP (2000): Antifertility studies of the root extract of the Barleria priorities. Linn in male albino rats with special reference to testicular cell population dynamics. J Ethnopharmacol 70: 111–117. [INFOTRIEVE], [CSA], [CROSSREF]
- Hinton BT, Palladine MA (1995): Epididymal epithelium: Its contribution to the formation of a luminal fluid microenvironment. Microsc Res Tech 30: 67–81. [INFOTRIEVE], [CSA], [CROSSREF]
- Klinefelter GA, Hess RA (1998): Toxicology of the male excurrent ducts and accessory sex glands. In: Korachks (ed.), Reproductive and Developmental Toxicology. New York, Marcel Dekker, pp. 553–592.
- Lewis DI, Aird IA, Biljan MM, Kingsland CR (1996): Effects of sperm activity on zinc and fructose concentration in seminal plasma. Hum Reprod 11: 2465–2467. [CSA]
- Lohiya NK, Ansari AS (1999): Male contraceptive agents. In: Joy KR, Krishna A, Halder C, eds., Comparative Endocrinology and Reproduction. New Delhi, Narosa Publishing House, pp. 260–277.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951): Protein measurement with Folin phenol reagent. J Biol Chem 193: 265–275. [INFOTRIEVE], [CSA]
- Montgomery R (1957): Determination of glycogen. Arch Biochem Biophys 67: 378–381. [INFOTRIEVE], [CSA], [CROSSREF]
- Prasad MRN, Chinoy NJ, Kadam KM (1972): Changes in succinate dehydrogenase levels in the rat epididymis under normal and altered physiologic conditions. Fertil Steril 23: 186–190. [INFOTRIEVE], [CSA]
- Purohit A, Bhagat M (2004): Contraceptive effects of Curcuma longa. (L.) in male albino rat. Asian J Androl 6: 71–74. [CSA]
- Rao MV, Mathur N (1998): Estrogen induced effects on mouse testis and epididymal spermatozoa. Expt Clin Endocrinol 92: 231–234. [CSA]
- Rao MV, Shah KD (1998): Endocrine approach to male fertility control by steroid hormone combination in rat: Rattus norvegicus. L. Indian J Exp Biol 36: 775–779. [INFOTRIEVE], [CSA]
- Rizk AM, El-Ghazaly GA (1995): Medicinal and Poisonous Plants of Qatar. University of Qatar, Scientific and Applied Research Centre, p. 101.
- Rizk AM, Heiba HI (1990): Antiinflammatory and cytotoxic activities of extracts of thirty indigenous species. Int J Crude Drug Res 28: 89. [CSA]
- Shahat AA, Abdel-Azim AA, Pieters L, Vlietinck AJ (2004): Flavonoids from Cressa cretica.. Pharm Biol 42: 349–352. [CSA]
- Sharma N, Jacob D (2002): Assessment of reversible contraceptive efficacy of methanol extract of Mentha arvensis. L. leaves in male albino mice. J Ethnopharmacol 80: 9–13. [INFOTRIEVE], [CSA], [CROSSREF]
- Sharma S, Kumar M, Goyal RB, Manivannan B, Lohiya NK (1999): Reversible antispermatogenic effect of gossypol in langur monkey (Presbytis entellus entellus.). Adv Contracep 15: 15–27. [CSA], [CROSSREF]
- Turner TT, Miller DW, Avery EA (1995): Protein synthesis and secretion by the rat caput epithelium in vivo influence of the luminal microenvironment. Biol Reprod 53: 1012–1019. [CSA], [CROSSREF]
- Vijaykumar B, Sangamma IA, Saraswati BP (2004): Antispermatogenic and hormonal effects of Crotalaria juncea. Linn. seed extracts in male mice. Asian J Androl 6: 67–70. [INFOTRIEVE], [CSA]
- Warren L (1959): A thiobarbituric acid assay of sialic acid. J Biol Chem 234: 1971–1975. [INFOTRIEVE], [CSA]