Abstract
The aim of this study was to investigate the antinociceptive and anti-inflammatory activities of the essential oil of Satureja thymbra. L. (Lamiaceae). Antinociceptive activity of the essential oil was measured by the formalin test in mice and by the light tail-flick and hot-plate methods in rats. The paw-edema test was performed in rats to evaluate the anti-inflammatory effect of the essential oil. Motor coordination was assessed with a rotarod apparatus. Although the essential oil produced an antinociceptive effect during both the early (50 and 100 mg kg−1) and late phases (25, 50, and 100 mg kg−1) of the formalin test, it did not exert any significant antinociceptive and anti-inflammatory effects in tail-flick and paw-edema tests, respectively. In addition, a significant antinociceptive effect was detected in the hot-plate test (100 and 200 mg kg−1). No significant change was detected in sensory motor performance. In conclusion, S. thymbra. L. essential oil does not exert any anti-inflammatory effect while it may have central analgesic activity in mice and rats.
Introduction
The genus Satureja. belongs to the family Lamiaceae, subfamily Nepetoideae, and the tribe Mentheae. One of the specific characteristics of the Nepetoideae subfamily is that its representatives contain more than 0.5% essential oil. Marked differences are reported in the essential oil composition between and within the subspecies of Satureja. (Abdollahi et al., Citation2003).
Satureja. species are widely used to treat various diseases. In particular, Satureja hortensis. L. (summer savory), which is one of the most important Satureja. species, has been used as a folk remedy to treat different symptoms like cramps, muscle pains, nausea, indigestion, diarrhea, and infectious diseases because of its antispasmodic, antidiarrheal, and antimicrobial properties (Uslu et al., Citation2003).
Satureja. genus is represented in Turkey with 15 different taxa, and 5 of them are endemic (Davis, Citation1982; Gören et al., 2003). Satureja thymbra. L. belongs to this genus and is widely distributed throughout the Western Anatolian Region with its gray color and pink flowers (Baytop, Citation1999).
Although antiviral (Abad et al., Citation1999), antinociceptive, anti-inflammatory (Hajhashemi et al., Citation2002), antibacterial, antifungal (Azaz et al., Citation2002; Sokovic et al., Citation2002), antispasmodic, antidiarrheic (Hajhashemi et al., Citation2000), and vasodilating (Sanchez de Rojas et al., Citation1996) effects have been reported for different Satureja. species growing in different parts of the world, no study has yet been performed regarding the pharmacological properties of S. thymbra.. The aim of the current study was to investigate the antinociceptive and anti-inflammatory activities of the essential oil of S. thymbra..
Materials and Methods
Plant material
Fresh plants from wild collections, gathered at the start of the flowering period in July 2002 from Davutlar National Park, Kusadasi, Aydin (a city located in the western part of Turkey) (N 37° 39′ 550″, E 27° 11′ 756″), were used. Mainly, the aerial parts of the plants that have a high proportion of buds and flowers were selected. The plant was identified by Professor Ozcan Secmen from Ege University, Faculty of Science, Department of Biology, Section of Botany. The voucher specimen of the plants used in the current study was kept for a record in the herbarium of Ege University, Faculty of Pharmacy, Department of Pharmaceutical Botany (http//www.izef.ege.edu.tr) (voucher no. 5667).
Preparation and analysis of essential oil
Plant materials were dried in shade at ambient temperature, cut into small pieces, placed in a Clevenger-type distillation apparatus with 2:1 of double-distilled water, and hydrodistilled for 3 h according to the European Pharmacopoeia. 1996. The essential oil was dried over anhydrous sodium sulfate and stored at 4°C before analysis. Essential oil was administered to mice and rats immediately after suspending in Tween 20 (Aldrich 27, 434-8 Hamburg, Germany).
The essential oil was analyzed by gas chromatography-mass spectroscopy (GC-MS). The analysis of the essential oil was also performed using a Hewlett-Packard (HP, Minnesota, USA) 6890 gas chromatograph, coupled with a HP 5973 mass selective detector and HP-5 capillary column (60 m × 0.25 mm i.d., film thickness 0.25 µm). For GC-MS detection, an electron ionization system with ionization energy of 70 eV was used. Helium was the carrier gas at a flow rate of 1 mL/min. The oven temperature was programmed from 50°C to 300°C at 10°C/min. Injector temperature was 150°C, and detector temperature was 250°C. Diluted samples (1:100 in ethyl acetate, v/v) of 1.0 µL were injected by autosampler in the splitless mode (Gulluce et al., Citation2003).
Identification of essential oil compounds was based on comparison of their relative retention time and mass spectra with those of commercial standards (for the main components) and retention indices (RIs) relative to C8–C32 n.-alkanes mixture. The results were also confirmed by computer matching of mass spectra with the Wiley 275 L mass spectra data library (Adams, Citation2001; Gulluce et al., Citation2003). The relative percentage of the essential oil constituents was calculated from the GC peak areas.
Animals
The protocol was approved by the Animal Ethical Committee of Ege University Faculty of Medicine (31.10.2002, no. 2002/37). Male albino mice (20–25 g) and Wistar rats (125–150 g) were used. Animals were housed in a room maintained at 22±1°C with an alternating 12-h light-dark cycle. Food and water were available ad libitum.. The animals were transported to a quiet laboratory at least 1 h before the experiment. All experiments assessing the analgesic activity were carried out between 0900 and 1200 h in normal room light and temperature (22±1°C).
Assessment of antinociceptive activity
The antinociceptive activity was measured in 15-h fasted male albino mice by the formalin test and in male Wistar rats by the light tail-flick and hot-plate methods.
All experiments conformed to ethical guidelines for investigation of experimental pain in conscious animals (Zimmermann, Citation1983). The number of animals and the intensity of noxious stimuli were the minimum possible with which to demonstrate reliable effects of the agents tested. Each animal was used once only and was humanely sacrificed immediately after completion of testing.
Formalin paw test
Formalin (2.5% v/v, 20 µL) was injected in the subplantar region of the right hind paw of mice and the duration of paw licking (an index of nociception) was monitored between 0 and 5 min (early phase) and 15 and 30 min (late phase) (Moore et al., Citation1991; Handy et al., Citation1995). The early phase response is believed to represent a direct irritant effect of formalin on sensory C fibers while the late phase response is most likely secondary to the development of an inflammatory response and the release of algesic mediators (Hunskaar & Hole, Citation1987). Essential oil (12.5, 25, 50, 100 mg kg−1) (n = 10 for each group) or vehicle (2% Tween 20) (n = 10) was administered 30 min before the formalin injection. Formalin test was repeated with 10 mg kg−1 morphine, 10 mg kg−1 morphine + 2 mg kg−1 naloxone, and 100 mg kg−1 essential oil + 2 mg kg−1 naloxone administration.
Light tail-flick test
The tail flick was evoked by a source of radiant heat, which was focused on the dorsal surface of the tail. Rats were examined for latency to withdraw their tails from a noxious thermal stimulus using a tail-flick meter (MAY-TF 0703, Ankara, Turkey) (n = 10 for each group). Each rat was tested twice before the administration of the essential oil, and the reaction times were averaged to obtain a baseline. The intensity of heat stimulus was adjusted to achieve a mean tail flick latency of 3–4 s in control animals. Each rat was then tested 30, 45, 60, 75, 90, 105, and 120 min after the i.p. administration of 50, 100, 200 mg kg−1 essential oil. Control rats received 2% Tween 20 instead of the essential oil. Light tail-flick test was repeated with 10 mg kg−1 morphine, 10 mg kg−1 morphine + 2 mg kg−1 naloxone, and 100 mg kg−1essential oil + 2 mg kg−1 naloxone administration. Treatments were terminated if the animals did not respond within 15 s in order to avoid tissue damage (D'Amour & Smith, Citation1941).
Hot-plate test
Rats were placed on an aluminum hot plate (MAY-AHP 0603, Ankara, Turkey) kept at a temperature of 55±0.5°C for a maximum time of 30 s (Franzotti et al., Citation2000). Reaction time was recorded (when the animals licked their fore and hind paws and jumped) before and 15, 30, 45, 60, and 90 min after i.p. administration of 50, 100, 200 mg kg−1 essential oil to different groups of 10 animals each. Morphine 10 mg kg−1 was used as the reference drug, and the test was repeated with 10 mg kg−1 morphine + 2 mg kg−1 naloxone, and 100 mg kg−1 essential oil + 2 mg kg−1 naloxone administration.
Assessment of anti-inflammatory activity
The anti-inflammatory activity was evaluated by the carrageenan-induced paw-edema test in the rat (Winter et al., Citation1962; Schapoval et al., Citation1998). Male Wistar rats were deprived of food overnight and treated by peroral route with 2% Tween 20 (as control group) and essential oil (50, 100, 200 mg kg−1), 30 min before 0.1 mL 1% carrageenan in isotonic saline was injected subplantarly into the left hind paw. The contralateral paw was injected with 0.1 mL saline and used as a control. Paw volume was measured by water plethysmometer (Lettica, LE 7500, Barcelona, Spain) before and 1, 2, 3, 4, 5, and 6 h after the injection of carrageenan into the plantar region of the left hind paw. The anti-inflammatory test was repeated with 10 mg kg−1 indomethacin administration.
Assessment of sensorimotor performance
Because sedation can affect the reaction to noxious stimuli, the integrity of motor coordination was assessed with a rotarod apparatus, at a rotating speed of 16 rpm. A preliminary selection of mice and rats was made on the day of experiment to exclude those that did not remain on the rotarod bar for two consecutive periods of 45 s each (Pieretti et al., Citation1999). Sensorimotor performance was assessed 30 min after the injection of 2% Tween 20 and 50, 100, 200 mg kg−1of essential oil (n = 10 for each group) in mice and rats separately. Results are expressed as percentage of animals that succeeded in remaining on the rod for 45 s, which was the cutoff time.
Drugs
The preparation of the essential oil was described above in detail. Formalin stocks (2.5% v/v) were freshly prepared with distilled water on the day of the experiment. Carrageenan, DMSO, morphine, and indomethacin were purchased from Sigma Chemical Co. (St. Louis, MO, USA), Tween 20 from Aldrich (Aldrich-27, 434-8 Germany). γ.-Terpinene (Fluka-86476), carvacrol (Fluka-22051), thymol (Fluka-89330), p.-cymene (Fluka-30039), β.-caryophyllene (Fluka-22075), and α.-thujene (Fluka-89231) were provided from Fluka-Seelze, Germany for GC analysis.
Statistical analysis
Results are shown as mean±SE mean. Statistical analysis was performed by ANOVA with Student-Newman-Keuls test, and p value of 0.05 or less was taken to indicate statistical significance.
Results
Chemical composition of the essential oil
The isolated pale yellow essential oil content was 2.4% (v/w) on a dried weight basis. The components of Satureja thymba. L. oil are listed in . Twenty-one components were identified representing 99.53% of essential oil. The main components of the essential oil were γ.-terpinene (40.99%), carvacrol (17.50%), thymol (13.19%), and P.-cymene (12.73%). Based on the percentage area, β.-caryophyllene (3.15%), α.-thujene (1.98%), and thymylmethylether (1.94%) were also determined as abundant components.
Table 1. Chemical composition of the essential oil of S. thymbra..
Assessment of antinociceptive activity
Formalin paw test
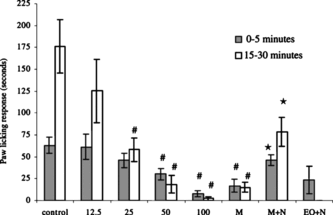
S. thymbra. extract caused a significant dose-related inhibition of licking and biting of the hind-paw responses during both the early (50 and 100 mg kg−1) and late phases (25, 50, and 100 mg kg−1) of formalin-induced hind-paw licking (p < 0.01). When morphine responses were compared with that of control, a significant inhibition of early and late phases was shown (p < 0.01). Naloxone reversed the morphine responses in both phases (p < 0.05); 100 mg kg−1 essential oil + naloxone administration was not found to be significantly different from 100 mg kg−1 essential oil administration alone (p > 0.05) ().
Figure 1 Antinociceptive effect of S. thymbra. extract (mg kg−1, i.p.) determined as the effect on early phase (striped columns) and late phase (dotted columns) formalin-induced hind-paw licking behavior in mice. Results indicate hind-paw licking time in seconds (s) and are mean±SEM, n = 10 for each group. Essential oil (12.5, 25, 50, 100 mg kg−1), vehicle (2% Tween 20) (control), morphine, morphine + naloxone, or essential oil + naloxone were administered before the formalin injection. EO, essential oil of S. thymbra., M, 10 mg kg−1 morphine; M + N, 10 mg kg−1 morphine + 2 mg kg−1 naloxone; EO + N, 100 mg kg−1 essential oil of S. thymbra. + 2 mg kg−1 naloxone administration. #p < 0.01 according to control animals. ⋆p < 0.05 according to morphine group.
Light tail-flick test
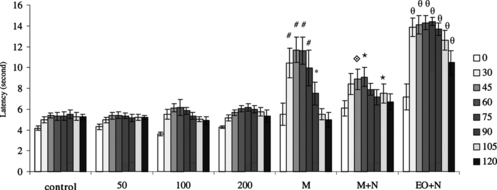
The essential oil administration did not increase the tail-flick latencies when compared with control rats (p > 0.05). When morphine responses were compared with that of control, it increased the latency significantly at 30, 45, 60, 75 (p < 0.01), and 90 min (p < 0.05). This increase in latency caused by morphine was reversed by naloxone administration at 45 (p < 0.01), 60, and 105 min (p < 0.05). When the effect of 100 mg kg−1 essential oil + naloxone administration was compared with 100 mg kg−1 essential oil alone, a significant increase in latency was observed at 30, 45, 60, 75, 90, 105, and 120 min (p < 0.01) ().
Figure 2 Latency to withdraw the tail from noxious thermal stimulation in rats after treatment with 2% Tween 20 (control) and 50, 100, and 200 mg kg−1 i.p. S. thymbra. essential oil injection. Data are expressed as mean and vertical lines show SEM (n = 10 for each group). EO, essential oil of S. thymbra.; M, 10 mg kg−1 morphine; M + N, 10 mg kg−1 morphine + 2 mg kg−1 naloxone; EO + N, 100 mg kg−1 essential oil of S. thymbra. + 2 mg kg−1 naloxone administration. *p < 0.05, #p < 0.01 according to control animals. ⋆p < 0.05, ·⋄p < 0.01 according to morphine group. θp < 0.01 according to 100 mg kg−1 EO.
Hot-plate test
Administration of 100 and 200 mg kg−1 essential oil of S. thymbra. increased the latency to the hot-plate test at 60 and 90 min (p < 0.05). Morphine administration increased the latency significantly at 15 and 45 min (p < 0.01) and at 30 and 60 min (p < 0.05). Naloxone reversed the morphine responses at 15, 30, and 45 min (p < 0.05). When the effect of 100 mg kg−1 essential oil + naloxone administration was compared with 100 mg kg−1 essential oil alone, a significant increase in latency was observed at 15 (p < 0.05), 30, and 45 min (p < 0.01) ().
Table 2. Effect of S. thymbra. essential oil on the latency time of rats submitted to the hot-plate test.
Assessment of anti-inflammatory activity
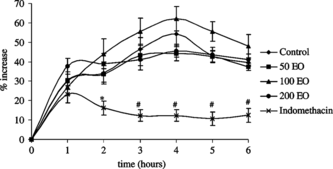
The intraplantar injection of carrageenan caused a time-dependent paw edema in the rat, although saline injection caused no swelling (data not shown). Peroral administration of S. thymbra. essential oil did not inhibit paw swelling, whereas a significant inhibition after 2 (p < 0.05), 3, 4, 5, and 6 h (p < 0.01) was obtained by indomethacin administration (). Percent increment in paw swelling was calculated by using the values before carrageenan injection.
Figure 3 Percent increase in carrageenan-induced paw edema in control rats treated with vehicle (2% Tween 20), in rats treated with 50, 100, and 200 mg kg−1 S. thymbra., and 10 mg kg−1indomethacin. Percent increment in paw swelling was calculated by using the values before carrageenan injection. Data are expressed as mean and vertical lines show SEM (n = 10 for each group). *p < 0.05, #p < 0.01 vs. control. EO, essential oil of S. thymbra..
Assessment of sensorimotor performance
The sensorimotor performance of control mice and rats were found as 92.6 ± 4.7 and 98.3 ± 2.6%, respectively. No significant change was observed with the administration of the essential oil in any of the doses tested (p > 0.05). In fact, in the treatment groups the percentage of animals with success in the rotarod test ranged between 85.8 ± 5.9% and 95.6 ± 3.9% for mice and 82.8 ± 5.7% and 97.6 ± 4.5% for rats (n = 10 for each group).
Discussion
In the current investigation, we have observed that while i.p. injection of S. thymbra. essential oil dose-dependently produced an antinociceptive effect in both phases of the mouse formalin paw test, the essential oil did not exert any significant antinociceptive and anti-inflammatory effect in the rat tail-flick and paw-edema tests, respectively. Besides, a significant antinociceptive effect was detected only with 100 and 200 mg kg−1 of essential oil in the hot-plate test performed on rats.
The formalin test is a valid and reliable model of nociception, and it is sensitive for various classes of analgesic drugs. The test produces a biphasic response: early phase is thought to result from direct chemical activation of nociceptive afferent fibers, and peripheral inflammatory processes seem to be responsible for the late phase (Tjolsen et al., Citation1992). Both phases of the test may be affected by different analgesics. Because of this reason, the formalin test has been used to clarify the possible mechanism of different analgesic drugs (Morteza-Semnani et al., Citation2002).
Centrally acting drugs such as opioids exert an inhibition in both phases (Shibata et al., Citation1989), as it is clearly consistent with our morphine results in formalin test, whereas peripherally acting drugs such as indomethacin, aspirin, and hydrocortisone only inhibit the late phase, which seems to be a result of an anti-inflammatory response (Hunskaar & Hole, Citation1987; Elisabetsky et al., Citation1995). The effect of S. thymbra. essential oil on the early and late phases of the formalin tests suggests that it may exert a central action.
On the other hand, the hot-plate test is sensitive to opioids and other centrally acting drugs like sedatives and muscle relaxants (Rosland et al., Citation1990). Our antinociceptive results with morphine also confirm this finding. Similarly, the analgesic activity obtained with 100 and 200 mg kg−1 of essential oil in the hot-plate test also supports the hypothesis that the essential oil may exert a central action.
Although the antinociceptive effects of the essential oil in formalin and hot-plate tests suggest that it may have a central analgesic effect, the lack of an antinociceptive effect in the light tail-flick test, which predominantly involves central mechanisms (Khanna & Bhaita, Citation2003), contradicts our results. However, when we compare these tests, the hot-plate and tail-flick tests measure the response to a brief, noxious stimulus, while the formalin test measures the response to a long-lasting nociceptive stimulus (Rosland et al., Citation1990). Because of this reason, the formalin test was believed to represent a more valid model for clinical pain (Hajhashemi et al., Citation2002). Therefore, according to our findings in the formalin and hot-plate tests, we suggest that S. thymbra. essential oil may exert a central analgesic effect.
The different results obtained among the different types of nociceptive tests might reflect the differential processing of noxious stimuli with different physical and temporal properties and which trigger different motor responses. According to our knowledge, it is not possible to discriminate the specific modulation of these responses by different molecules (Rojas-Corrales et al., Citation2003).
The observed antinociceptive effect of S. thymbra. essential oil may be due to one of its components, thymol, because the other main components were not shown to exert any analgesic activity. It was clearly seen that thymol—which is a naturally occurring phenol derivative that exerts an antinociceptive effect—exists as 13.19% in essential oil composition, and its analgesic activity may be mediated via blockade of voltage-operated sodium channels (Haeseler et al., Citation2002).
Morphine was used as a positive control for all three antinociceptive tests we have performed. In all of them, a significant antinociceptive effect was obtained with morphine and this effect was successfully reversed with the administration of naloxone, which is a specific opioid receptor antagonist. Similarly, the effect of 100 mg kg−1 essential oil + naloxone administration was compared with the effect of 100 mg kg−1 essential oil administered alone in all three tests in order to evaluate the possible opioid-like mechanism of the analgesic effect of the essential oil. While no significant difference was observed in the formalin test, a significant increase in latency was observed for different time points in the hot-plate and light tail-flick tests between 100 mg kg−1 essential oil + naloxone and 100 mg kg−1 essential oil administered groups. This increase in latency may be due to the possible partial opioid agonistic effect of S. thymbra. essential oil; it has clearly been shown that the analgesic effects of kappa partial agonists like nalbuphine, pentazocine, and butorphanol may be enhanced by low-dose naloxone administration (Gear et al., Citation2003). Therefore, the possible partial opioid agonistic effect of the components of the essential oil requires further investigation.
Moreover, an inhibition that was observed in the second phase of the formalin test made us think that it may be due to the peripheral action and anti-inflammatory effect of the essential oil. However, we have detected no anti-inflammatory effect in the carrageenan paw-edema test, which is a suitable test for the evaluation of anti-inflammatory drugs and has frequently been used to assess the antiedematous effects of natural products (Mascolo et al., Citation1987; Segura et al., Citation1998).
Species-related slight differences may also be considered while discussing these antinociceptive and anti-inflammatory results, while the essential oil may act differently in mice and rats.
Conclusions
S. thymbra. essential oil does not exert any anti-inflammatory effect, while it may have central analgesic activity in mice and rats. The mechanism of this analgesic effect needs to be elucidated by further investigations.
Acknowledgments
This study was designed and performed at Ege University, Center for Drug R&D and Pharmacokinetic Applications. The study was supported by Ege University Science Technology, Application and Research Center (project no. 02/ILAM/001).
References
- Abad MJ, Bermejo P, Gonzales E, Iglesias I, Irurzun A, Carrasco L (1999): Antiviral activity of Bolivian plant extracts. General Pharmacol 32: 499–503. [CSA]
- Abdollahi M, Salehnia A, Mortazavi SH, Ebrahimi M, Shafiee A, Fouladian F, Keshavarz K, Sorouri S, Khorasani R, Kazemi A (2003): Antioxidant, antidiabetic, antihyperlipidemic, reproduction stimulatory properties and safety of essential oil of Satureja khuzestanica. in rat in vivo.: A toxicopharmacological study. Med Sci Monitor 9: 331–335. [CSA]
- Adams RP (2001): Identification of Essential Oils Components by Gas Chromatography/Quadrupole Mass Spectroscopy. Carol Stream, Illinois, Allured Publishing Corp.
- Azaz D, Demirci F, Satil F, Kurkcuoglu M, Baser KH (2002): Antimicrobial activity of some Satureja. essential oils. Z Naturforschung C-A J Biosci 57: 817–821. [CSA]
- Baytop T (1999): Therapy with Medicinal Plants in Turkey (Past and Present), 2nd ed. Istanbul, Nobel Publications, p. 332.
- Davis PH (1982): Flora of Turkey and the East Aegean Islands, Vol 7. Edinburgh, Edinburgh University Press, p. 319.
- D'Amour FE, Smith DL (1941): A method determining loss of pain sensation. J Pharmacol Exp Ther 72: 74–79. [CSA]
- Elisabetsky E, Amador TA, Albuquerque RR, Nunes DS, Carvalho Ado C (1995): Analgesic activity of Psychotria colorata. Muell. alkaloids. J Ethnopharmacol 48: 77–83. [INFOTRIEVE], [CSA], [CROSSREF]
- Franzotti EM, Santos CV, Rodrigues HM, Mourao RH, Andrade MR, Antoniolli AR (2000): Anti-inflammatory, analgesic activity and acute toxicity of Sida cordifolia. L. (Malva-branca). J Ethnopharmacol 72: 273–278. [INFOTRIEVE], [CSA], [CROSSREF]
- Gear RW, Gordon NC, Miaskowski C, Paul SM, Heller PH, Levine JD (2003): Dose ratio is important in maximizing naloxone enhancement of nalbuphine analgesia in humans. Neuroscience Lett 351: 5–8. [CSA], [CROSSREF]
- Goren AC, Bilsel G, Altun M, Satil F, Dirmenci T (2003): Fatty acid composition of seeds of Satureja thymbra. and S. cuneifolia.. Naturforschung C-A J Biosci 58: 502–504. [CSA]
- Gulluce M, Sokmen M, Daferera D, Agar G, Ozkan H, Kartal N, Polissiou M, Sokmen A, Sahin F (2003): In vitro. antibacterial, antifungal and antioxidant activities of the essential oil and methanol extracts of herbal parts and callus cultures of Satureja hortensis. L. J Agric Food Chem 51: 3958–3965. [INFOTRIEVE], [CSA], [CROSSREF]
- Haeseler G, Maue D, Grosskreutz J, Bufler J, Nentwig B, Piepenbrock S, Dengler R, Leuwer M (2002): Voltage-dependent block of neuronal and skeletal muscle sodium channels by thymol and menthol. Eur J Anaesthesiol 19: 571–579. [INFOTRIEVE], [CSA], [CROSSREF]
- Hajhashemi V, Ghannadi A, Pezeshkian SK (2002): Antinociceptive and anti-inflammatory effects of Satureja hortensis. L. extracts and essential oil. J Ethnopharmacol 82: 83–87. [INFOTRIEVE], [CSA], [CROSSREF]
- Hajhashemi V, Sadraei H, Ghannadi AR, Mohseni M (2000): Antispasmodic and anti-diarrhoeal effect of Satureja hortensis. L. essential oil. J Ethnopharmacol 71: 187–192. [INFOTRIEVE], [CSA], [CROSSREF]
- Handy RLC, Wallace P, Gaffen ZA, Whitehead KJ, Moore PK (1995): The antinociceptive effect of 1-(2-trifluoromethylphenyl) imidazole (TRIM), a potent inhibitor of neuronal nitric oxide synthase in vitro., in the mouse. Br J Pharmacol 116: 2349–2350. [INFOTRIEVE], [CSA]
- Hunskaar S, Hole K (1987): The formalin test in mice: Dissociation between inflammatory and non-inflammatory pain. Pain 30: 103–114. [INFOTRIEVE], [CSA], [CROSSREF]
- Khanna N, Bhaita J (2003): Antinociceptive action of Ocimum sanctum. (Tulsi) in mice: Possible mechanisms involved. J Ethnopharmacol 88: 293–296. [INFOTRIEVE], [CSA], [CROSSREF]
- Mascolo N, Autore G, Capasso F (1987): Local anti-inflammatory activity of Tamus communis.. J Ethnopharmacol 19: 81–84. [INFOTRIEVE], [CSA], [CROSSREF]
- Moore PK, Oluyomi AO, Babbedge RC, Wallace P, Hart SL (1991): L-NG-nitro arginine methyl ester exhibits antinociceptive activity in the mouse. Br J Pharmacol 102: 198–202. [INFOTRIEVE], [CSA]
- Morteza-Semnani K, Saeedi M, Hamidian M, Vafamehr H, Dehpour AR (2002): Anti-inflammatory, analgesic activity and acute toxicity of Glaucium grandiflorum. extract. J Ethnopharmacol 80: 181–186. [INFOTRIEVE], [CSA], [CROSSREF]
- Pieretti S, Dal Piaz V, Matucci R, Giovannoni MP, Galli A (1999): Antinociceptive activity of a 3(2H)-pyridazinone derivative in mice. Life Sci 65: 1381–1394. [INFOTRIEVE], [CSA], [CROSSREF]
- Rojas-Corrales MO, Casas J, Moreno-Brea MR, Gibert-Rahola J, Mico JA (2003): Antinociceptive effects of tricyclic antidepressants and their noradrenergic metabolites. Eur Neuropsychopharmacol 13: 355–363. [INFOTRIEVE], [CSA], [CROSSREF]
- Rosland JH, Tjolsen A, Maehle B, Hole K (1990): The formalin test in mice, effect of formalin concentration. Pain 42: 235–242. [INFOTRIEVE], [CSA], [CROSSREF]
- Sanchez de Rojas VR, Somoza B, Ortega T, Villar AM (1996): Vasodilatory effect in rat aorta of eriodicytol obtained from Satureja obovata.. Planta Med 62: 272–274. [INFOTRIEVE], [CSA]
- Schapoval E, de Vargas M, Chaves C, Bridi R, Zuanazzi J, Henriques A (1998): Anti-inflammatory and antinociceptive activities of extracts and isolated compounds from Stachytarpheta cayennensis.. J Ethnopharmacol 60: 53–59. [INFOTRIEVE], [CSA], [CROSSREF]
- Segura L, Vila R, Gupta MP, Esposito-Avella M, Adzet T, Canigueral S (1998): Anti-inflammatory activity of Anthurium cerrocampanense. croat in rats and mice. J Ethnopharmacol 61: 243–248. [INFOTRIEVE], [CSA], [CROSSREF]
- Shibata M, Ohkubo T, Takahashi H, Inoki R (1989): Modified formalin test: Characteristic biphasic pain response. Pain 38: 347–352. [INFOTRIEVE], [CSA], [CROSSREF]
- Sokovic M, Tzakou O, Pitarokili D, Couladis M (2002): Antifungal activities of selected aromatic plants growing wild in Greece. Nahrung-Food 46: 317–320. [CSA], [CROSSREF]
- Tjolsen A, Berge OG, Hunskaar S, Rosland JH, Hole K (1992): The formalin test: An evaluation of the method. Pain 51: 5–17. [INFOTRIEVE], [CSA], [CROSSREF]
- Uslu C, Murat Karasen R, Sahin F, Taysi S, Akcay F (2003): Effects of aqueous extracts of Satureja hortensis. L. on rhinosinusitis treatment in rabbit. J Ethnopharmacol 88: 225–228. [INFOTRIEVE], [CSA], [CROSSREF]
- Winter CA, Risley EA, Nuss GW (1962): Carrageenan-induced edema in hind paw of rat as an assay for anti-inflammatory drugs. Prog Soc Biol Med 11: 544–547. [CSA]
- Zimmermann M (1983): Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16: 109–110. [INFOTRIEVE], [CSA], [CROSSREF]