ABSTRACT
Background: This study aimed to assess the impact of comorbidities (CMs) on costs, utilities and health-related quality of life (HRQOL) among HIV patients in Colombia.
Methods: A cross-sectional study was conducted among 138 HIV patients at an outpatient clinic in Bogotá to assess their costs, utilities (EQ5D-5L) and HRQOL (EQ-VAS). CMs and other covariates were gathered from patient records. Multiple regression models were conducted to assess the impact of CMs.
Results: Of all patients, 72% experienced CMs; of these, half experienced ≥2 CMs (N = 49). CMs had a statistically significant impact on utilities when the EQ5D-5L (P = 0.022) was applied among patient with ≥2 CMs. No statistical significance was found for the impact of CMs on costs.
Conclusion: This study suggests that having multiple CMs significantly influences utility, and CMs have some impact on HRQOL measured using the EQ-VAS but this effect is at the border of significance. No significant impact was observed on costs.
1. Introduction
The Human Immunodeficiency Virus (HIV) is one of the most well-known viruses and affects numerous individuals. A global report on the HIV epidemic showed that the incidence of HIV has decreased by 47%, from 3.4 million people in 2001 to 1.8 million new HIV infections in 2013 [Citation1,Citation2]. However, these results do not correspond to the situation in Colombia, where the incidence rate had only a small decrease of 0.44% from 2000 to 2013 [Citation1]. According to the World Health Organization, an estimated 140,000 people lived with HIV in Colombia in 2013, which is 0.3% of the total population [Citation3]. In most countries, HIV is no longer considered to be a mortal disease, but a chronic disease for which treatment requires lifelong access to adequate healthcare, with consequent increases in total healthcare costs [Citation4,Citation5].] In 2010, HIV was among the top four causes of burden of disease for Colombia [Citation6].]
Furthermore, HIV patients commonly experience additional diseases due to a dysfunctional immune system; these are further referred to as comorbidities (CMs). CMs that are found among HIV patients are cardiovascular diseases, hypertension, diabetes mellitus, and renal failure [Citation7–Citation10]. Having CMs plausibly enlarges the HIV burden, because CMs generally have an impact on the patient’s well-being and use of resources.
Costs, utilities, and health-related quality of life (HRQOL) are commonly used to describe the burden of a disease. Several studies have been performed to describe this burden among HIV patients [Citation11–Citation15], of which two recent studies [Citation16,Citation17] estimated the costs, utilities, and HRQOL among HIV patients in Bogotá. The outcomes showed an estimated mean cost per patient between 10,959 and 18,543 US dollars per year. Moreover, HIV-infected men had lower utilities than women (0.760 versus 0.88, P = 0.002), and in patients with severe HIV, men had lower utilities than women (0.83 versus 0.87, P = 0024). However, no statistical differences were found for HRQOL in HIV status and gender.
These two studies [Citation16,Citation17] did not consider the impact of CMs on costs, utilities or HRQOL, although the impact could be substantial when looking at HIV. To our knowledge, only two studies [Citation18,Citation19] examined this relationship. One study has shown that CMs have a significantly negative impact on utilities and HRQOL in HIV patients [Citation18] and the other study found a statistically significant difference (P < 0.001) for the impact of CMs on costs in HIV patients [Citation19]. Similarly, significant results for the impact of CMs on costs and utilities were found in other chronic disease, like diabetes and comorbid depression [Citation20,Citation21]. However, all these studies in HIV and diabetes patients were performed in Western countries and might not reflect the situation in developing countries like Colombia.
Therefore, further research is needed to study and verify the impact of CMs on costs and utilities among HIV patients in Colombia. Decision makers should be aware of the possible impact of CMs on the existing HIV burden. It plausibly increases the burden, which might require different policies. In addition to the importance of the impact of CMs on the HIV burden, it would be valuable for healthcare professionals to describe the prevalence and occurrence of CMs in Colombia.
The aim of this study was therefore to assess the impact of CMs on costs, utilities, and HRQOL among HIV patients in Colombia. A secondary aim was to assess which type and what number of CMs have the greatest impact on costs, utilities, and HRQOL. Additionally, the prevalence and occurrence of certain types of CMs among HIV patients were measured.
2. Methods
2.1. Population and setting
A quantitative cross-sectional study was conducted among HIV patients at the Asistencia Científica de Alta Complejidad (ACAC) that is located in Bogotá. ACAC is a private outpatient clinic that specializes in the treatment of HIV and several other diseases. This study used data from two previous studies about costs, utilities, and HRQOL that were performed at the outpatient clinic in 2014 [Citation16,Citation17]. The participants of this study were the same selected patients as the two previous performed studies, whereas the sample size for costs (N = 124) was smaller than the sample size for utilities and HRQOL (N = 138). In this article, the ‘costs’ sample is further referred to as CS and the ‘utility and HRQOL’ sample as UQS. Patients in these studies were recruited during a time frame of 3 weeks in April 2014, were 18 years and older, and received outpatient care. Exclusion criteria were literacy and limited cognitive abilities for filling in the questionnaires.
Additional data about CMs and other covariates of the selected patients were collected from the clinic’s database and patients’ records over the same period of time. No extra exclusion criteria were made in addition to those in the previously performed studies [Citation16,Citation17]. The ethics committee of ACAC approved this study and every participant signed the informed consent form before participating the previous studies.
2.2. Data collection and measurements
2.2.1. Total costs
Costs were collected according to the taxonomy of Drummond; these were the sum of ‘healthcare resource use’, ‘patient and family resource use’, and ‘resource use in other sectors’, also known as ‘healthcare costs’, ‘patient and family costs’, and ‘costs in other sectors’, respectively [Citation16,Citation22]. A bottom–up model was used to estimate these costs for every patient because it measures person-based costs and minimizes bias, in contrast to a top–down model [Citation23,Citation24]. Furthermore, a prevalence-based approach was used to assess the costs for a period of 3 months and was then extrapolated to 1 year, which is considered most suitable for assessing the total economic burden of a health issue [Citation23]. This was achieved by using patients’ records in the clinic’s database and a cost questionnaire. Two pilot studies were done to validate the cost questionnaire.
To determine ‘healthcare costs’ within the clinic, patient records were used. Healthcare costs include the use of all healthcare resources for the treatment of HIV, as well as the use of all healthcare resources when a patient suffered from CMs. With regard to prescription drugs, only costs of the antiretroviral drugs were available within the clinic’s pharmacy. For other drugs, the price list of the Universidad de Cordoba from 2013 was used [Citation25] and singular missing drug data were gathered from local pharmacies. Healthcare costs that were made outside the clinic were measured by using the cost questionnaire, such as other hospital visits and consulting physicians outside the clinic. All prices were expressed in Colombian Pesos (COP) and were inflated to the year 2014 if needed (in June 2015 $1 = COP1887) [Citation26].
The cost questionnaire was used to determine ‘patient and family costs’. Factors that were taken into account were traveling time to the hospital (ACAC and other healthcare organizations), waiting time, time of the actual consultation and out-of-pocket expenditures. Information was collected regarding paid help and also regarding unpaid help by friends or family. The productivity loss to absenteeism at work was included in the ‘costs in other sectors’ and was defined using the human capital approach [Citation27]. To value productivity loss, the gross domestic product per capita from 2012 was used [Citation28]. Furthermore, a sensitivity analysis excluding hyperlipidemia as a CM was conducted given the high prevalence of the disease and relatively low impact on costs.
2.2.2. Utility and HRQOL
The variables ‘utility’ and ‘HRQOL’ were measured with the EQ-5D-5L and the EQ-Visual Analogue Scale (EQ-VAS), respectively [Citation17]. The EQ-5D-5L allows the quality of life to be expressed in utilities and is a widely used instrument [Citation29]. It is a generic instrument and uses an indirect method for measuring utility, with five questions regarding mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. These questions were answered on a five-point categorical rating scale, varying from ‘no problems at all’ to ‘severe problems’ [Citation29]. The EQ-5D-5L is validated for Spanish-speaking countries and is composed following the Spanish value sets to determine the patient’s utility [Citation30,Citation31]. As the value set is not yet available for the new EQ-5D-5L instrument, an interim scoring algorithm was derived from the EQ-5D-3L value set, which should be acknowledged as a temporary solution. The utility is expressed in a value between 0 (death) to 1 (excellent health). The EQ-VAS uses a rating scale between the worst (0) and best imaginable (100) HRQOL of the patient to provide an individual score. A sensitivity analysis excluding hyperlipidemia as a CM was conducted given the high prevalence of the disease and relatively low impact on utility and HRQOL.
2.2.3. Comorbidities (CMS)
CMs were collected from patient registrations at ACAC over the same period; costs, utilities, and HRQOL were collected. The patient’s identification number was used to link the registered diagnoses with the selected patient. These diagnoses were registered as primary and secondary diagnoses. The primary diagnosis in this study was the HIV infection itself and secondary diagnoses were referred to as CMs. Only secondary diagnoses with a chronic character were included, assuming that chronic diseases have a plausible effect on costs, utilities, and HQOL. HIV-related complications, such as toxoplasmosis, were excluded since they have a temporary character and occur commonly among HIV-infected patients. These temporary CMs and other disabilities and disorders were most likely not related to utility and HRQOL and increased costs in comparison with chronic CMs. HIV medical specialists selected the chronic CMs which were included. Thereafter, the CMs were clustered according to the type of CM, such as metabolic disorders and cardiovascular diseases. These diseases were further classified into medical and mental CMs because the sample sizes of the former clusters were too small to assess the impact of the type of CM. Substance abuse disorders were excluded since only one subject was diagnosed with alcoholism.
2.2.4. Covariates
In line with previous studies, we included socioeconomic status (SES) [Citation32–Citation34], age [Citation8,Citation17,Citation25,Citation35,Citation36], gender [Citation17], and HIV status [Citation17,Citation18,Citation25,Citation36] as possible covariates. SES was defined according to the six social levels in Bogotá, expressed in value between SES1 and SES6, where SES1 is the poorest. SES2 represents the second poorest strata, SES3–SES4 represent the medium strata and SES5–SES6 represent the highest strata. According to the international federation of housing and planning, 68% of the population in Bogotá lives in SES2 or SES3 [Citation37]. Patients with SES1 and SES2 were collected through governmental websites through their personal identification number [Citation28,Citation38]. For ≥SES3, a social worker who is employed at the outpatient clinic categorized the patients based on social patients’ records. For those who did not have social records, the SES was determined by their address. People with SES6 were not treated at the outpatient clinic. Furthermore, as there were only 2 patients with a SES5 status and 8 patients with a SES4 status subjects were classified into three groups: SES1 (56 patients), SES2 (38 patients) and ≥SES3 (40 patients) to maintain comparable groups. Furthermore, HIV status was classified according to the Centers for Disease Control and Prevention (CDC). The CD4 cell count provides the HIV status classification; stage one is generally defined as ≥500 cells/μl, stage two as 200–499 cells/μl, stage 3 (also referred as AIDS) as <200 cells/μl, and the classification differs for patients who have had HIV for less than 6 years [Citation39]. HIV status, as well as the covariates age and gender, were collected from the patients’ database at the clinic. The CD4 count at the time of filling the questionnaire was used.
2.3. Data analyses
First of all, descriptive statistics were used to report data about the patients’ characteristics, costs, utilities, HRQOL, and CMs. The number and amount of participants were reported for sample size, gender, SES, and HIV status. The mean and standard deviation were reported for age, total costs, utility, and HRQOL. Furthermore, an overview table of all CMs according to the type of CM was made, including further classification into medical and mental CMs. Subsequently, (bar) graphs were made to present the average total costs, utility, and HRQOL according to the number of CMs.
Thereafter, multiple linear regression models were created to achieve the study aims. Three separate regression models were made for every dependent variable; total costs, utilities, and HRQOL. First, HIV status, age, and gender were tested for effect modification (P < 0.05). As the interaction terms were not significant, data were not stratified. Crude and adjusted models were estimated, in which all adjusted models were corrected for HIV status, age, gender, and SES. As the distribution of total costs was positively skewed, log transformed distribution was used during the regression analyses.
3. Results
3.1. Descriptive statistics
3.1.1. Patient characteristics
Patient characteristics are presented in . The same number of patients were included as in the two previous studies performed in Bogotá [Citation16,Citation17]; these were 124 participants to assess the impact of CMs on total costs (CS) and 138 participants to assess the impact on utility and HRQOL (UQS). More men than women were included in both study samples: 100 (81%) versus 24 (19%) in CS and 85 (62%) versus 53 (38%) in UQS.
Table 1. Characteristics of HIV patients at ACACa.
Three-quarters of the patients in both samples had CMs: 91 (73%) patients in CS, and 100 (72%) patients in UQS. Of the HIV patients with CMs, 44 (35%) patients had one CM and 47 (38%) patients had ≥2 CMs in CS, whereas in the UQS, 51 patients (37%) had one CM and 49 patients (36%) had ≥2 CMs ( and ). The most common types of CMs were (1) mental disorders: 57% in CS and 51% in UQS; and (2) metabolic disorders: 48% for CS and 46% for UQS ().
Table 2. Type of comorbidities.
Figure 1a. The mean total costs per year in Colombian pesos for having no comorbidities, 1 comorbidity and ≥ 2 comorbidities in HIV patients, reported with the 95 % confidence interval; . The mean total costs per year in Colombian pesos for having no comorbidities, 1 comorbidity and ≥ 2 comorbidities in HIV patients, reported with the 95 % confidence interval excluding hyperlipidemia.
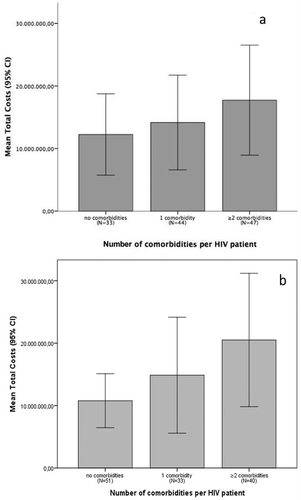
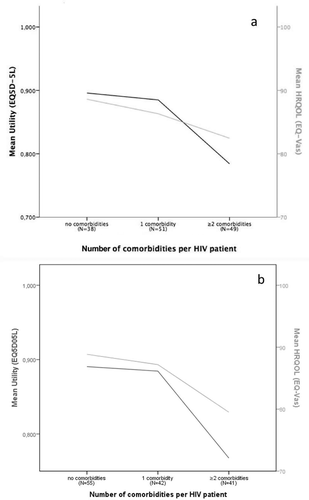
Figure 2a. The mean utility and mean Health-Related Quality of Life (HRQOL) according to the amount of comorbidities in HIV patients; : The mean utility and mean Health-Related Quality of Life (HRQOL) according to the amount of comorbidities in HIV patients excluding hyperlipidemia.
Furthermore, the majority of patients had a stage 3 HIV infection, also known as acquired immune deficiency syndrome (AIDS), and these patients were found to have more CMs than patients in an earlier stage of HIV. For example, in CS 62 (50%) patients with a stage 3 HIV infection suffered from CMs and 20 patients (16%) did not suffer from CMs, whereas 29 (23%) patients who had a stage 1 or 2 HIV infection also suffered from CMs and 13 (11%) patients with a stage 1 and 2 HIV infection did not suffer from CMs.
3.1.2. Costs, utility, and HRQOL
Mean healthcare costs were 12,640,195 Colombian Pesos (COP) per patient. Mean patient and family costs were 2,370,448 COP and mean total costs per patient were 15,001,538 COP (166,584,001 COP).
Subsequently, HIV patients with CMs spent more COP per year (16,002,151; SD 27,539) on average than did HIV patients without CMs (12,242,272; SD 18,340,257) (). The mean utility score for patients with CMs was lower than for patients without CMs (0.836; SD 0.224 versus 0.896, SD 0.187). A similar pattern was found for the HRQOL (84.4; SD 16.1 versus 88.6; SD 10.4) (). and show the mean total costs increasing according to the number of CMs and decreasing in utility and HRQOL according to the number of CMs. A large part of the patients suffered from hyperlipidemia (33.9%), which is generally asymptomatic and hence has a low impact on healthcare costs at early stages, particularly if untreated, this could cause difficulties in interpreting the overall results for >2 comorbidities as a predominant share of these occurrences are due to co-occurrence of hyperlipidemia. Hence, a sensitivity analysis was conducted, excluding hyperlipidemia as a CM. In this analysis, the total costs of patients with no comorbidities (N = 51) was 10,776,095 COP (SD: 15,367,612 COP); the total costs of patients with 1 comorbidity were 14,862,267 COP (SD: 26,188,870 COP); and the total costs of patients with ≥2 comorbidities were 20,503,877 COP (SD: 33,391,468 COP). and show the mean total costs increasing according to the number of CMs and decreasing in utility and HRQOL according to the number of CMs excluding hyperlipidemia.
3.2. Linear regression models
3.2.1. Costs
HIV patients with CMs had higher costs, but these expenditures did not differ significantly statistically from the expenditures of patients with no CMs, P = 0.356 (). There were no statistically significant differences for the type of CM in comparison with patients with no CMs after adjustment for confounding: P = 0.662 for patients with medical CMs, P = 0.724 for patients with mental CMs, and P = 0.104 for patients with both medical and mental CMs. Patients with one CM did not have statistically significant higher costs than patients without a CM (P = 0.790), nor did patients with two or more CMs (P = 0.164).
Table 3. Multiple linear regression models for the correlation between ‘having a comorbidity’, ‘type of comorbidity’ and ‘number of comorbidities’ with the ‘total costs’, ‘utility’ and HRQOL’ in HIV patients.
3.2.2. Utility and HRQOL
Lower utility and HRQOL scores were found for patients with CMs in all linear regression models, although only a few of them were found to be statistically significant (). Only patients with both medical and mental CMs showed a statistically significant impact for the type of CM on utility and HRQOL, P = 0.005 when using the EQ-5D-5L and P = 0.026 when using the EQ-VAS. HIV patients with two or more CMs showed a statistically significant impact on utility when using the EQ5DL, P = 0.022. However, similar results were not found when using the EQ-VAS (P = 0.069). In general, HIV patients who have only one CM did not experience a statistically significant lower utility or HRQOL score than HIV patients without CMs, after adjustment for confounding (EQ-5D-5L: P = 0.183, EQ-VAS: P = 0.162).
4. Discussion
In this study, we assessed the impact of CMs on costs, utilities, and HRQOL among HIV patients in Colombia. Statistically significant differences were found for utilities when HIV patients had two or more CMs and the number of CMs has some impact on the HRQOL of patients with HIV but is in the border of insignificance. Patients with both mental and medical CMs had a significant lower utility and HRQOL. However, no statistically significant impact of CMs on costs was found.
These findings were in contrast with the two previously performed studies that looked at the impact of CMs. According to Guaraldi et al. [Citation19], CMs were found to be statistically significant for total costs. The difference in statistical significance could be ascribed to the difference in controls. In our study, HIV-positive participants without CMs represented the control group and in Guaraldi et al. [Citation19] HIV-negative patients with the same CMs represented the control group. Moreover, both patient groups in our study were receiving HIV treatment care, such as antiretroviral therapy (ART). Guaraldi et al. [Citation19] found that ART is a significant associated factor with the total for direct healthcare costs in HIV patients (P < 0.001). This may also explain the insignificant differences in this study, because the costs for HIV care represent the total costs to a large extent. Furthermore, the majority of the study sample was known to have a stage 3 HIV infection (AIDS), which increases the use of ART and could further explain our result.
According to the findings of Anis et al. [Citation18], CMs were negatively associated with utilities and HRQOL. Anis et al. [Citation18] did not look at the type or amount of CMs but looked at CM as a dichotomous variable. In our study, CMs do affect the utility when HIV patients have two or more CMs, but no statistical significance was found when using CM as a dichotomous variable. Besides the fact that larger study samples were used, the definition of CMs could also explain the different results. In their study, Anis et al. included CMs with a temporary character, while we included only chronic CMs in our study. The chronic character and the patient’s ability to live with the chronic CM may be factors for an insignificant patient’s utility. This may also explain why having one CM does not affect the utility and having multiple CMs does affect the utility, because HIV patients with multiple CMs may be less able to live with the CMs. Furthermore, the statistically significant results for mental and medical CMs in comparison with having no CMs could be distorted by the number of CMs, because patients in this group have two or more CMs. Moreover, the insignificant impact on utility and HRQOL for different types of CMs may be due to all medical and mental CMs being taken together during the analyses because of the insufficient sample size of the clustered CMs. Certain CMs or clustered CMs might have an influence on the utility and HRQOL independently, if larger samples were used.
The additional findings for the prevalence and occurrence of different types of CMs were similar to the outcomes of other studies [Citation8,Citation9,Citation35]. Although not every study included mental CMs, this is a common type of CM based on our results. The prevalence of hyperlipidemia is high in our population (33.9%), yet literature has been scarce on reporting this prevalence. However, Green et al. [Citation40] have reported prevalence rates of dyslipidemia ranging from 33% to 82% in patients with HIV. Dyslipidemia, however, includes elevated total cholesterol, elevated LDL cholesterol, and elevated triglycerides (including severe hypertriglyceridemia). HIV medications have been shown to increase the risk of hyperlipidemia (e.g. HIV medicines in the protease inhibitor drug class) [Citation41].
The results of this study might be influenced by several limitations. A first limitation may be the small sample size for generalizing the results of the prevalence and occurrence of CMs. In particular, the prevalence of diabetes and hypertension seems low in our sample compared to previous studies. A second factor that may limit the generalizability is the cross-sectional data gathered at one specialized clinic. Care delivery at the ACAC is not standard in comparison with the rest of Bogotá or Colombia. Nevertheless, the data gathered about CMs and other covariates during this study is reliable because it was retrieved directly from patients’ records from a specialized HIV clinic. Third, this study only focused on chronic CMs and did not incorporate any temporary CMs. Especially in high income countries, however, temporary CMs can result in very expensive hospitalization episodes that can highly skew costs, Hence, neglecting temporary CMs may have led to an underestimation of the true costs. Fifth, the number of patients reporting substance abuse disorders was substantially low (i.e. one patient). Hence, this may be an indication of underreporting. For example, Campbell et al. have reported that in 2006 in the U.S. injection drug use was attributable to 24% of the total HIV infections [Citation42]. Finally, the definition of CMs may affect the outcomes; regardless there is no gold standard for defining CMs.
Notwithstanding these limitations, our study is one of the first to assess the impact of CMs on costs, utilities, and HRQOL in developing countries like Colombia. The results of this study could be useful for policymakers and healthcare professionals. Based on our findings, the overall costs are not much higher for HIV patients with CMs in comparison with HIV patients without CMs. However, utility decreases with the number of CMs, and this requires attention from healthcare professionals and policymakers. When patients experience several diseases, integrated care should be encouraged among healthcare professionals to optimize healthcare delivery, which would increase patient satisfaction and thus increase utility. Several studies of other chronic diseases have shown that integrated care increases utility and quality of life in patients [Citation43,Citation44]. Accordingly, stimulating integrated care for HIV patients with CMs would therefore be important. This would also be in line with the Primary Health Care strategy that has been introduced by the district government of Bogotá to decrease the high existing fragmentation and segmentation of providing services [Citation45]. Moreover, healthcare professionals should be aware of the existence of mental CMs as well as physical CMs and should redirect patients when necessary.
Further research is recommended to verify and specify the findings of this study. First, further studies should verify whether for less severe HIV (stages 1 and 2) patients with CMs, the costs of HIV care also represent most of the total costs. Second, the type of CMs could be more identified more specifically. Larger samples should be used to study the impact of specific CMs (e.g. hyperlipidemia) or clusters (e.g. metabolic disorders) on costs, utilities, and HRQOL.
5. Conclusion
A majority of HIV patients in Colombia have at least one comorbidity. Having multiple CMs significantly influences the utility of patients with HIV. Furthermore, patients with both mental and medical CMs had a significantly lower utility and HRQOL, although this may be distorted by the number of CMs. However, among HIV patients, having multiple CMs have some impact on HRQOL measured using the EQ-VAS only, but this effect is at the border of significance. In our study, HIV patients with CMs were not more costly than HIV patients without CMs, which is plausibly caused by the already relatively high costs of HIV care.
Key issues
HIV causes a high burden in costs, utility and health-related quality of life for patients worldwide, including in developing countries like Colombia.
In our study, about 72% of HIV patients, who participated in a study conducted at the Asistencia Cientifica de Alta Complejidad in Bogotá, experienced comorbidities (CMs); half of these patients had two or more CMs. The most prevalent comorbidities were mental disorders and metabolic disorders.
Having multiple comorbidities significantly influences utility; and has some impact on health-related quality of life (HRQOL) measured using the EQ-VAS, but this latter is in the border of insignificance. No significant impact on costs was observed in our study.
Given the great number of comorbidities in HIV patients and their potential influence on utility, integrated care should be encouraged among healthcare professionals to optimize healthcare delivery. This will increase the patient satisfaction, thus increasing utility.
HIV patients with comorbidities did not spend significantly more money for care than HIV patients without comorbidities, which is plausibly caused by the relatively high costs of HIV care.
Further research is needed in a larger population to study the impact of specific comorbidities instead of clustering in types of comorbidities.
Declaration of interest
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Additional information
Funding
References
- Murray CJ, Ortblad KF, Guinovart C, et al. Global, regional, and national incidence and mortality for HIV, tuberculosis, and malaria during 1990-2013: a systematic analysis for the global burden of disease study 2013. Lancet. 2014;384:1005–1070.
- UNAIDS. Global report: UNAIDS global report on the global AIDS epidemic 2013. WHO Library Cataloguing-in-Publication Data, 2013. Available from: http://wwwunaidsorg/sites/default/files/en/media/unaids/contentassets/documents/epidemiology/2013/gr2013/UNAIDS_Global_Report_2013_enpdf.
- WHO. Global Health Observatory Data Repository: Number of people (all ages) living with HIV estimates by country, 2012 [cited 2014]. Available from: http://appswhoint/gho/data/viewmain22100.
- Deeks SG, Lewin SR, Havlir DV. The end of AIDS: HIV infection as a chronic disease. Lancet. 2013;382:1525–1533.
- Thorpe KE, Ogden LL, Galactionova K. Chronic conditions account for rise in medicarespending from 1987 to 2006. Health Aff. 2010;29:718–724.
- Ortblad KF, Lozano R, Murray CJL. The burden of HIV: insights from the global burden of disease study 2010. Aids. 2013;27:2003–2017.
- Deeks SG, Phillips AN. HIV infection, antiretroviral treatment, ageing, and non-AIDS related morbidity. Bmj. 2009;338:3172.
- Vance DE, Mugavero M, Willig J, et al. Aging with HIV: a cross-sectional study of comorbidity prevalence and clinical characteristics across decades of life. J Assoc Nurses AIDS Care. 2011;22:17–25.
- Shah SS, McGowan JP, Smith C, et al. Comorbid conditions, treatment, and health maintenance in older persons with human immunodeficiency virus infection in New York city. Clin Infect Dis. 2002;35:1238–1243.
- Powderly WG. Non-AIDS-defining illnesses in the mature patient. Available from: http://hivinsite.ucsf.edu/InSite?page=kbr-07-04-18
- Clayson DJ, Wild DJ, Quarterman P, et al. A comparative review of health-related quality-of-life measures for use in HIV/AIDS clinical trials. PharmacoEconomics. 2006;24:751–765. Epub 2006/08/11.
- Schackman BR, Goldie SJ, Freedberg KA, et al. Comparison of health state utilities using community and patient preference weights derived from a survey of patients with HIV/AIDS. Med Decis Making. 2002;22:27–38.
- Cardona-Arias J, Pelaez-Vanegas L, Lopez-Saldarriaga J, et al. [health related quality of life in adults with HIV/AIDS in Colombia]. Biomedica. 2011;31:532–544.
- Valencia CP, Canaval GE, Marín D, et al. Quality of life in persons living with HIV AIDS in three healthcare institutions of Cali. Colomb Med (Cali). 2010;41:206–214.
- Bastardo YM, Kimberlin CL. Relationship between quality of life, social support and disease-related factors in HIV-infected persons in Venezuela. AIDS Care. 2000;12:673–684.
- Kuhlmann J, Keaei M, Conde R, et al. A cost of illness study of HIV/AIDS patients in Bogotá. Colombia; 2016. in submission.
- Keaei M, Kuhlmann J, Conde R, et al. Health utilities for patients with HIV/AIDS in Bogotá. 2016, Value in Health Regional Issues. [ Epub Ahead of Print].
- Anis AH, Nosyk B, Sun H, et al. Quality of life of patients with advanced HIV/AIDS: measuring the impact of both AIDS-defining events and non-AIDS serious adverse events. J Acquir Immune Defic Syndr. 2009;51:631–639.
- Guaraldi G, Zona S, Menozzi M, et al. Cost of noninfectious comorbidities in patients with HIV. Clinicoecon Outcomes Res. 2013;5:481–488.
- Egede LE, Zheng D, Simpson K. Comorbid depression is associated with increased health care use and expenditures in individuals with diabetes. Diabetes Care. 2002;25:464–470.
- Goldney RD, Phillips PJ, Fisher LJ, et al. Diabetes, depression, and quality of life: a population study. Diabetes Care. 2004;27:1066–1070.
- Drummond M. Methods for the economic evaluation of health care programmes. 3rd ed. New York: Oxford University Press; 2005.
- Larg A, Moss JR. Cost-of-illness studies: a guide to critical evaluation. PharmacoEconomics. 2011;29:653–671.
- Fox-Rushby J, Cairns J. Economic evaluation. 1st ed. Berkshire: Oxford University Press; 2005.
- Schouten J, Wit FW, Stolte IG, et al. Cross-sectional comparison of the prevalence of age-associated comorbidities and their risk factors between HIV-infected and uninfected individuals: the AGEhIV cohort study. Clin Infect Dis. 2014;59:1787–1797.
- The World Bank Group. Inflation, consumer prices (annual%). [cited 2014]. Available from: http://data.worldbank.org/indicator/FP.CPI.TOTL.ZG
- van den Hout WB. The value of productivity: human-capital versus friction-cost method. Ann Rheum Dis. 2010;69:89–91.
- Sistema de Indentificaión de Potenciales Beneficiarios de Programas Sociales (SISBEN). Consulta de Puntaje. 2015, Available from: https://www.sisben.gov.co/ConsultadePuntaje.aspx.
- EuroQol. EQ-5D-5L, Webpage. 2015. Available from: http://www.euroqol.org/eq-5d-products/eq-5d-5l.html.
- EuroQol. EQ-5D-5L User Guide. 2015. Available from: http://www.euroqol.org/about-eq-5d/publications/user-guide.html.
- EuroQol. EQ-5D-5L Value Sets. 2015. Available from: http://www.euroqol.org/about-eq-5d/valuation-of-eq-5d/eq-5d-5l-value-sets.html.
- Lopera MM, Einarson TR, Ivan Bula J. Out-of-pocket expenditures and coping strategies for people living with HIV: Bogota, Colombia, 2009. AIDS Care. 2011;23:1602–1608.
- Moussavi S, Chatterji S, Verdes E, et al. Depression, chronic diseases, and decrements in health: results from the world health surveys. Lancet. 2007;370:851–858.
- Everson SA, Maty SC, Lynch JW, et al. Epidemiologic evidence for the relation between socioeconomic status and depression, obesity, and diabetes. J Psychosom Res. 2002;53:891–895.
- Weiss JJ, Osorio G, Ryan E, et al. Prevalence and patient awareness of medical comorbidities in an urban AIDS clinic. AIDS Patient Care STDS. 2010;24:39–48.
- Goulet JL, Fultz SL, Rimland D, et al. Do patterns of comorbidity vary by HIV status, age, and HIV severity? Clin Infect Dis. 2007;45:1593–1601.
- International Federation for Housing and Planning. Colombia: Social Stratification by Law. 2012. Available from: http://www.ifhp.org/ifhp-blog/colombia-social-stratification-law
- Alcaldia Mayor de Bogotá D.C, Secretaría de Salud. Compraboder de Derechos. 2015. Available from: http://app.saludcapital.gov.co/comprobadordederechos/Consulta.aspxavailable
- CDC. Terms, definition, and calculations used in CDC HIV surveillance publications. 2015. Available from: http://www.cdc.gov/hiv/statistics/recommendations/terms.html.
- Green ML. Evaluation and management of dyslipidemia in patients with HIV infection. J Gen Intern Med. 2002;17:797–810.
- Gazzaruso C, Bruno R, Garzaniti A, et al. Hypertension among HIV patients: prevalence and relationships to insulin resistance and metabolic syndrome. J Hypertens. 2003;21:1377–1382.
- Campbell ANC, Tross S, Calsyn DA. Substance use disorders and HIV/AIDS prevention and treatment intervention: research and practice considerations. Soc Work Public Health. 2013;28:333–348.
- Bird S, Noronha M, Sinnott H. An integrated care facilitation model improves quality of life and reduces use of hospital resources by patients with chronic obstructive pulmonary disease and chronic heart failure. Aust J Prim Health. 2010;16:326–333.
- Koff PB, Jones RH, Cashman JM, et al. Proactive integrated care improves quality of life in patients with COPD. Eur Respir J. 2009;33:1031–1038.
- Méndez M, Andrea P Evaluation of a primary health care strategy implemented in a market-oriented health system: the case of Bogota, Colombia. Doctoral thesis 2014, Umeå University, Sweden.
