Abstract
Antibacterial Schiff bases derived from 1,2,4-triazoles as well as their metal complexes incorporating cobalt(II), nickel(II), copper(II) and zinc(II) have been synthesized and characterized. Physico-chemical studies suggest that an octahedral geometry for the cobalt(II), nickel(II) and zinc(II)and square-planer geometry for the copper(II) complexes. These complexes have been screened for antibacterial activity against three Gram-positive (Staphylococcus aureus, Staphylococcus epidermidis and Bacillus subtilis) and two Gram-negative (Salmonella typhi and Pseudomonas aeruginosa) bacterial strains, and results compared with the activity of the free ligands. The metal complexes were found to be more potent against one or more bacterial strains than the free ligands.
Introduction
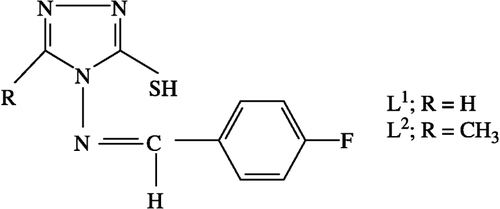
The discovery and development of effective antibacterial drugs with novel mechanisms of action has become an urgent task for infection diseases research programs [Citation1]. Vancomycin has been a drug of last report for the treatment of MDR. But the recent emergence of vancomycin-resistant enterococci (VRE) and vancomycin-intermediate resistant Staphylococcus aureus (VISA) is raising serious public health concern [Citation2]. Novel treatment and prophylaxis are urgently required to combat such a serious threat [Citation3]. At the same time most of the antibacterial, antifungal and/or antiviral drug resistance is becoming a major problem for medical purposes Citation4-6. The problem could overcome by the preparation of metal complexes, using a process of chelation via coordination of transition metal ions, and development of their novel antibiotics Citation7-8. Schiff bases possess a strong ability to form metal complexes [Citation9] and they deserve some attention because of their biological properties Citation10-13. The Co(II), Ni(II), Cu(II) and Zn(II) complexes of Schiff bases are also biologically active and they exhibit enhanced activities as compared to their parent ligands. Triazole derivatives Citation14-17 possesses a wide spectrum of biological activities and therefore Schiff bases containing a triazole moiety and their metal complexes are expected to be bioactive compounds. Continuing our investigation on the interaction of metal ions with antibiotics, we report here the synthesis and characterization of fluorobenzyl Schiff bases () and their metal complexes. These have been screened against various pathogenic bacterial strains, Staphylococcus aureus MTCC 3160, Staphylococcus epidermidis MTCC 2639, Bacillus subtilis MTCC 121, Salmonella typhi MTCC 733 and Pseudomonas aeruginosa MTCC 3541 by the minimum inhibitory concentration method [Citation18].
Material and Methods
All chemicals and solvents used were of AnalaR grade. All metal(II) were used as their acetate salts. Melting points were determined in open capillaries in an electrical melting point apparatus. IR Spectra were recorded on a Beckman IR-20 spectrophotometer in the region 4000–250 cm− 1. 1H NMR spectra were recorded in DMSO-d6 on a Brucker ACF 300 spectrometer at 300 MHz with reference to Me4Si (0.0 ppm). Electronic spectra of metal complexes were recorded in the region 1100–200 nm on a Hitachi U-2000 spectrophotometer. Conductance of the metal complexes was determined in dry DMF on a Systronics-306 conductivity meter. Magnetic measurements were carried out at the Institute Instrumentation Centre, IITR, Roorkee, on a vibrating sample magnetometer (model 155). The metal complexes were analyzed for their metal contents using standard methods [Citation19]. Thermal analyses of metal complexes were carried out in atmospheric air using a Perkin Elmer (Pyris Diamond) Instrument reference to Alumina Powder at IITR, Roorkee.
Synthesis of ligands
4-amino-5-mercapto-s-triazole (AMT) and 4-amino-5-mercapto-3-methyl-s-triazole (AMMT) were prepared by the reported procedure [Citation20]. Schiff bases namely, 4-(4-fluorobenzalideneamino)-5-mercapto-s-triazole (FBMT) and 4-(4-fluorobenzalideneamino)-5-mercapto-3-methyl-s-triazole (FBMMT), were prepared by refluxing a mixture of equimolar quantities of different s-triazoles (AMT and AMMT) with 4-fluorobenzaldehyde in ethanol. The product was filtered and recrystallized from the same solvent.
Synthesis of metal(II) chelates
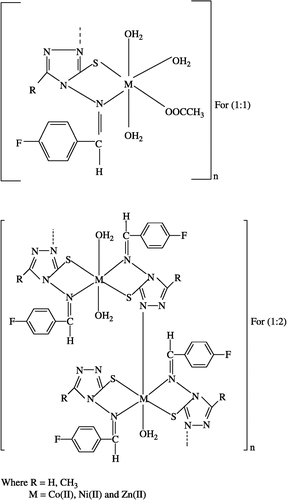
Aqueous ethanolic solution of metal acetates of Co(II), Ni(II), Cu(II) and Zn (II) were added to the hot ethanolic solution of the ligands in different molar ratios, which resulted in the precipitation of metal derivatives. The products so formed were filtered, washed with warm water, ethanol and finally with acetone and dried on a water bath.
In vitro antibacterial assay
All the synthesized ligands and their corresponding metal(II) chelates were screened in vitro for their antibacterial activity against the above mentioned bacteria using the minimum inhibitory concentration(MIC) method. MIC is the lowest concentration of the antimicrobial agents that prevents the development of visible growth after overnight incubation [Citation18]. The MIC of chemically synthesized compounds against test bacteria namely Staphylococcus aureus (MTCC 3160), Staphylococcus epidermidis (MTCC 2639), Bacillus subtilis (MTCC 121), Salmonella typhi (MTCC 733) and Pseudomonas aeruginosa (MTCC 3541) was determined by reported methods [Citation21]. All the test cultures were streaked on SCDA and incubated overnight at 37°C. Turbidity of all the bacterial cultures was adjusted to 0.5 McFarland standard [Citation22] by preparing bacterial suspension of 3–5 well-isolated colonies of same morphological type selected from a SCDA plate culture. The cultures were further diluted 10-fold to give an inoculum size of 1.2 × 107 CFU/ml. A stock solution of 4 mg/ml of each compound was prepared in DMSO and was appropriately diluted to give final concentrations of 64, 32, 16, 8, 4, 2, 1, 0.5, 0.25, 0.12, 0.06, 0.03 μg/ml. Standard antibiotics (linezolid and cefuroxime axetial) were also diluted in a same manner. 320 μl of each dilution was added to 20 ml molten and cooled MHA (separate flasks were taken for each dilution). After thorough mixing, the medium was poured into sterilized petri plates. The test bacterial cultures were spotted in a predefined pattern by aseptically transferring 5 μl of each bacterial culture on the surface of solidified agar–agar plates and the plate incubated at 35°C for 24 h.
Results and discussion
Chemistry
The ligands () were soluble in ethanol and methanol. All the metal(II) chelates were stable at room temperature, non-hygroscopic, insoluble in water and many common organic solvents, infusible at high temperature and were all considered to be polymeric in nature. The molar conductance values of 10− 3 M solutions of the metal complexes in dry DMF lie in the range 5–13Ω− 1 cm2 mol− 1, indicating their non-electrolytic nature. Structures of the Schiff bases were assigned with the help of IR, 1H NMR and analytical data ().
Table I. Spectral and analytical data for the Schiff bases.
IR spectra
IR spectra of the ligands reported in , show characteristic bands due to ν(N–H) and ν(S–H) at ∼3250 and ∼2700 cm− 1, respectively [Citation23]. Another band at ∼1100 cm− 1 is assigned to ν(C = S) [Citation23]. The deprotonation of thiol group is indicated by the absence of a band in the metal complexes () at ∼2700 cm− 1, which appears due to ν(S–H) in the spectra of ligands indicating the complexation of the metal atom to the sulphur. In the spectra of metal chelates a new band appears in the region 745–768 cm− 1 which is assigned to ν(C–S) and further confirms coordination through the sulphur atom. The band in the region 316–382 cm− 1 in the far IR-spectra further confirms metal-sulphur bond formation ().
Table II. Physical and analytical data for the Metal (II) complexes.
Table III. Spectral data for the metal(II) chelates.
The presence of coordinated water in the complexes [Citation23] is indicated by a broad trough band in the region 3600–2800 cm− 1 and two weaker bands in the region 832–840 cm− 1 and 700–722 cm− 1 due to the ν(–OH) rocking and wagging mode of vibrations, respectively [Citation24]. A strong band in the region 1715–1750 cm− 1 has been assigned to ν(OOCCH3) in the (1:1) (metal:ligand) complexes.
A strong band in the region 1593–1603 cm− 1 in the free ligands assigned to ν(–HC = N) exhibits shifting Citation25-29 to the higher side in the spectra of complexes indicating coordination through the azomethine nitrogen of Schiff bases and this can be explained by the donation of electrons from nitrogen to the empty d–orbitals of the metal atoms. Formation of a metal-nitrogen bond is further supported by the presence of a band in the region 490–545 cm− 1.
Thus the IR-spectra of ligands and metal complexes gives strong evidence for the complexation of the potentially multidentate ligands ().
Electronic spectra and Magnetic measurements
The cobalt complexes () generally exhibited two distinct bands in the region 10,940–11,000 and 20,000–21,000 cm− 1 which may be assigned to 4T1g(F) → 4T2g(F)(ν1) and 4T1g(F) → 4T1g(P)(ν3) transitions, respectively, and are suggestive of octahedral geometry around the cobalt ions Citation30-31. The electronic spectra of Ni(II) complexes showed d–d bands in the region 10,940–11,000, 15,000–17,000 and 22,000–26,000 cm− 1. These are assigned [Citation30] to the transitions 3A2g(F) → 3T2g(F)(ν1); 3A2g(F) → 3T1g(F)(ν2) and 3A2g(F) → 3T1g(P)(ν3), respectively, consistent with their well defined octahedral configuration. The electronic spectra of the Cu(II) complexes showed one broad band at ∼18,500 cm− 1, which is assigned to an 2Eg → 2T2g transition, a characteristic band of square-planer geometry Citation30-31 ().
The magnetic measurements for Co(II), Ni(II) and Cu(II) complexes showed three, two and one unpaired electrons and the magnetic moment values (4.3–5.2 BM), (2.8–3.5 BM) and (1.7–2.2 BM), respectively, for Co(II), Ni(II) and Cu(II) ion suggesting [Citation32] consistency with their octahedral and square-planer environment ().
NMR spectra
The NMR spectral data of the Schiff bases as well as their Zn(II) complexes were recorded in DMSO-d6 (Tables and ). The total number of protons present in the Schiff bases exhibited signals of the protons in their expected regions. In the PMR spectra of diamagnetic Zn(II) complexes a shift [Citation33] in electron density is observed from Schiff bases to its Zn(II) complexes. This downfield shift in the zinc complexes is due to the coordination of the azomethine nitrogen atom of the ligands, with the metal ion. The deprotonation of the thiol group in the spectra of Zn(II) complexes was also indicated by the disappearance of –SH protons.
Thermal studies
The thermal behaviour of all the complexes was almost the same. Therefore, only two complexes namely Co(C10H18N4SF)OAc·3H2O and Zn(C9H6N4SF)2·2H2O are discussed here in detail. In thermogravimetry (TG) the change in weight of a complex is recorded as a function of temperature during heating. The TG curves give information on the thermal stability and the products formed on heating. The TG curve is also supported by the derivative thermogravimetry (DTG) curves.
Thermal analyses of the complexes were carried out up to 900°C. The DTG curve of Co(C10H18N4SF)OAc·3H2O showed the loss of three water molecules up to 205°C with a mass loss of 4.07%(calc. 4.42%),8.43%(calc. 8.84%) and 13.12%(calc. 13.26%) on the TG curve Citation34-35. After 205°C, the decomposition of the organic part up to 391°C gave metal triazole with a mass loss of 57.15%(calc. 57.74%) on the TG curve and which was also supported by the DTA curve. All the triazole part decomposed at the temperature ranging from 391–748°C with a mass loss of 77.05%(calc. 77.64%) with the formation of CoS as the end product.
The sequence for thermal degradation of the complex Co(C10H18N4SF)OAc·3H2O are given below:
The thermoanalytical curve of Zn(C9H6N4SF)2·2H2O showed three clear steps in the ranges 70–220°, 220–520° and 520–730°C. The two water molecules were lost up to 220°C with a mass loss of 2.98%(calc. 3.31%) and 6.21%(calc. 6.63%) on the TG curve. The formation of Zn-s-triazole took place at 520°C with the decomposition of organic moiety, also indicated by the DTG curve with a mass loss of 50.93%(calc. 51.56%) on the TG curve Citation34-35. ZnS was left behind as the end product with a mass loss of 81.78%(calc.82.14%) due to decomposition of the s-triazole part.
The sequence for thermal degradation of the complex Zn(C9H6N4SF)2·2H2O are given below:
Biological results and discussion
Six chemically synthesized compounds were tested in vitro for their antibacterial activity against S. aureus, S. epidermidis, B. subtilis, S. typhi and P. aeruginosa (). The compounds were tested at different concentrations in DMSO solution using the MIC assay. The antibacterial results show that the activity of the ligands became more pronounced and significant when coordinated to the metal ions. This enhancement in activity may be due to an efficient diffusion of the metal complexes into the bacterial cell and/or interaction with the bacterial cell Citation36-39. Out of the six compounds tested, L2, 2, 6 and 9 possessed very good activity against Gram-positive bacteria namely S. epidermidis, S. aureus, B. subtilis. The minimum inhibitory concentration 0.03 μg/ml was shown by L2 and 9 against S. aureus, S. epidermidis, respectively. Compound 6 showed a MIC 0.06 μg/ml against S. aureus, S. epidermidis followed by compound 2, which showed a MIC 0.25 μg/ml against S. aureus. The minimum inhibitory concentration ranging from 2 to 16 μg/ml was shown by all the compounds (L1, L2, 2, 6, 9 and 13) of the series against all test bacteria, namely, S. aureus, S. epidermidis, B. subtilis, S. typhi and P. aeruginosa (). The antibacterial activity of these compounds was also compared with two commercial antibiotics, namely, linezolid and cefuroxime axetial.
Table IV. Minimum inhibitory concentration (MIC) (μg/ml) for six compounds against test microorganisms using the agar dilution assay technique.
| Abbreviations | ||
| B. subtilis: | = | Bacillus subtilis |
| CFU: | = | Colony Forming Unit |
| MHA: | = | Mueller Hilton Agar |
| MTCC: | = | Microbial Type Culture Collection and Gene Bank |
| MIC: | = | Minimum Inhibitory Concentration |
| P. aeruginosa: | = | Pseudomonas aeruginosa |
| S. typhi: | = | Salmonella typhi |
| SCDA: | = | Soyabean Casein Digest Agar |
| S. aureus: | = | Staphylococcus aureus |
| S. epidermidis: | = | Staphylococcus epidermidis |
Acknowledgements
This investigation received financial support from the University Grant Commission, New Delhi, No. F.12-47/2003(SR). UGC provided a project fellowship to one of the authors (M.S.B.).
Notes
*This paper was presented at the International Conference on Sustainable Development and Resource Utilization: Current Trends and Perspectives, P-51, 2005, Jaipur.
References
- Yang SW, Xu L, Mierzwa R, He L, Terracciano J, Patel M, Gullo V, Black T, Zhao W, Chan T, Chu M. Bioorg Med Chem 2004; 12: 3333–3338
- Sugie Y, Hirai H, Inagaki T, Ishiguro M, Kim YJ, Kojima Y, Sakakibara T, Sakemi S, Sugiura A, Suzuki Y, Brennan L, Duignan J, Huang LH, Sutcliffe J, Kojima N. J Antibiotic 2001; 54: 911–916
- Lara JG, Masalha M, Foster SJ. Drug Disc Today 2005; 10: 643–651
- Wright GD. Chem Biol 2000; 7: R127–R132
- Travis J, Potempa J. Biochim Biophys Acta 2000; 14: 35–50
- Smith HJ, Simons C. Proteinase and peptidase inhibition: Recent potential targets for drug development. Taylor and Francis, London 2001
- Rice SA, Givskov M, Steinberg P, Kjelleberg S. J Mol Microbiol Biotechnol 1999; 1: 23–31
- Scozzafava A, Supuran CT. J Med Chem 2000; 43: 3677
- Drabent K, Bialoska A, Ciunik Z. Inorg Chem Commun 2004; 7: 224–227
- Singh DP, Singh K, Singh PhA, Handa RN. J Inst Chemists (India) 2005; 77: 82
- Naik AD, Annigeri SM, Gangadharmath UB, Ravankar VK, Mahale VB, Reddy VK. Indian J Chem 2002; 41A: 2046–2053
- Sen AK, Singh G, Singh K, Handa RN, Dubey SN, Squattirito PJ. Proc Ind Acad Sci 1998; 110: 75–81
- Chohan ZH, Pervez H, Khan KM, Supuran CT. J Enz Inhib Med Chem 2005; 20(1)81–88
- Michael HP, Dines C. J Mol Struct 2004; 705: 177–187
- Singh K, Barwa MS, Tyagi P. Eur J Med Chem 2006; 41(1)147–153
- Chohan ZH, Pervez H, Rauf A, Scozzafava A, Supuran CT. J Enz Inhib Med Chem 2002; 17(2)117–122
- Chohan ZH, Scozzafava A, Supuran CT. J Enz Inhib Med Chem 2002; 17(4)261–266
- Greenwood D, Slack R, Peutherer J. Medical microbiology: A guide to microbial infections: Pathogensis, immunity, laboratory diagnosis and control15th ed. 1997
- Vogel AI. A text book of quantitative chemical analysis5th ed. Addition Wesly Longman, London 1999
- Bala S, Gupta RP, Sachdeva ML, Singh A, Pujari HK. Ind J Chem 1978; 16B: 481
- NCCLS. Method for dilution antimicrobial susceptibility test for bacteria that grow aerobically approved standards5th ed. Villanova, PA 2000
- McFarland J. J Am Med Assoc 1907; 14: 1176–1178
- Singh G, Singh PhA, Singh K, Singh DP, Handa RN, Dubey SN. Proc Nat Acad Sci (India) 2002; 72A: 87–94
- Shukla PR, Singh VK, Jaiswal AM, Narain J. J Ind Chem Soc 1983; 60: 321–324
- Gaur S, Sharma B. J Ind Chem Soc 2003; 8: 841–842
- Singh K, Singh RV, Tandon JP. Synth React Inorg Met-Org Chem 1987; 385–398
- Daniel TT, Natarajan K. Transition Met Chem 2000; 25: 311, (Dordrecht, Neth.)
- Sharma BD, Bailar JC. J Am Chem Soc 1955; 77: 5476–5480
- Nakamoto K. Infrared spectra of inorganic and coordination compounds5th ed. Wiley Interscience, New York 1997, Part B
- Lever ABP. Inorganic electronic spectroscopy. Elsevier, Amsterdam 1984
- Saied MEK. Ind J Chem 1994; 33A: 830–836
- Balhausen CJ. An introduction to ligands field. McGraw Hill, New York 1962
- Jhaumeer-Laulloo BS, Bhowon MG. Ind J Chem 2003; 42A: 2536–2540
- Dubey SN, Vaid BK. Ind J Chem 1992; 31A: 199–201
- Sharma PK, Sen AK, Dubey SN. Ind J Chem 1994; 33A: 1031–1033
- Chohan ZH, Supuran CT, Scozzafava A. J Enz Inhib Med Chem 2004; 19(1)79–84
- Chohan ZH, Praveen M. Synth React Inorg Met-Org Chem 2000; 30(1)175–182
- Chohan ZH, Praveen M. Appl Organomet Chem 2001; 15: 617–625
- Chohan ZH, Hassan MUI, Khan KM, Supuran CT. J Enz Inhib Med Chem 2005; 20(2)183–188