Abstract
Two novel structurally related phosphoramidate compounds, 1 and 2, with likely β-diketone system were synthesized and characterized by 1H, 13C, 31P NMR, IR spectroscopy and elemental analysis. Compound 2 exhibited a 31P NMR signal which was significantly shielded (8 ppm) relative to compound 1. Determination of human erythrocyte acetylcholinesterase (hAChE) inhibitory activity was carried out according to Ellman's modified kinetic method and the IC50 values of compounds 1 and 2 were 1.567 and 2.986 mM, respectively. The ki values of 1 and 2 were 1.39 to 2.65 min− 1 respectively. A comparison of the bimolecular rate constant (ki) and IC50 values for the irreversible inhibitors 1 and 2 revealed that the oxono analogue has greater affinity for hAChE than the thiono compound. Furthermore effects of two conventional oximes paralidoxime (A) and obidoxime (B) on reactivation of the inhibited hAChE were studied but low reactivity was shown by both the oximes.
Introduction
Acetylcholinesterase (AChE, EC 3.1.1.7) is a very important enzyme in biological systems because of its vital role in nerve impulse transmission [Citation1]. Many researchers have studied the pharmacological and toxicological properties of AChE inhibitors over the past fifty years [Citation2,Citation3]. Inhibitors such as the organophosphorus compounds with various potency towards AChE are being used increasingly as insecticides today [Citation4] and their synthesis and characterisation continues [Citation5]. The inhibitory effect of organophosphorus compounds especially those in the form of oxono ( ≡ P = O) and thiono ( ≡ P = S) is known Citation4Citation6-10 but, in accord with our literature search, this is first report on phosphoramidates containing the -(CO)NHP(X)- moiety (X = O,S).
Herein, two isosteric phosphoramidate 1 and 2, were synthesized and characterized by 1H, 13C, 31P NMR and IR spectroscopy and elemental analysis. In vitro inhibition of hAChE (pure and crude) by the phosphoramidates 1 and 2 was studied and the kinetic parameters, inhibitory potency (IC50) and bimolecular rate constant (ki), were determined. Classic studies have demonstrated that strong nucleophiles such as oximes are able to reactivate phosphoramidate-cholinesterase conjugates, giving rise to free enzyme [Citation11]. Here, the reactivation of the oxono-and thiono phosphoramidate-AChE conjugates was examined using two conventional oximes, pralidoxime, (A) and obidoxime (B).
Materials and methods
All reactions for the synthesis of compounds 1 and 2, were carried out under an argon atmosphere. Melting points were determined on a Gallenkamp apparatus. 1H, 13C and 31P NMR spectra were recorded on a Bruker (Avance DRS 500 MHZ spectrometer) and chemical shifts were determined relative to TMS and 85% H3PO4, respectively, as external standards. IR spectra (KBr) were obtained with a Shimadzu, IR–60 model spectrometer. Elemental analysis was performed using a Heraeus CHN-O-RAPID instrument and UV measurements were performed by a Shimadzu UV-2100 spectrometer. Pralidoxime and obidoxime were obtained from Aldrich. Other compounds and solvents for synthesis were obtained from Merck.
Human erythrocyte acetylcholinesterase (hAChE) from Sigma (50 units/785 μL) was diluted 50-fold in phosphate buffer (Na2HPO4, NaH2PO4, 70 mM, pH = 7.8). All other reagents for the enzymatic experiments were obtained from Fluka. Native human erythrocytes were obtained from the Institute for Blood Transfusion in Tehran- Iran. A crude extract of native erythrocytes was performed using the reported procedure [Citation13].
Synthesis
Benzoyl phosphoramidic dichloride, [Cl2P(O) NH(COC6H5)] [Citation12]
This was prepared by refluxing phosphorus pentachloride and benzamide in 1:1 molar ratio, in CCl4 for 2 h. Formic acid was then syringed drop-wise into the cold solution, and resulting colorless solution with a fine precipitate was stirred at 25°C for 30 min. Volatiles were then removed and the remaining white solid was dried in vacuo.
N-benzoyl N′,n′-(tert-butybenzyl) Phosphoramidic Chloride, [(C6h5ch2)(c(ch3)3)np(o)(cl)nhcoc6h5], 1
A mixture of 50 ml of acetonitrile and 1.19 g (5 mmol) of benzoyl phosphoramidic dichloride was cooled to − 10°C and a mixture of 0.815 g (5 mmol) N-benzyl-N-tert-butylamine and 0.506 g (5 mmol) triethylamine syringed dropwise into the cooled solution against an argon counterflow. The solution was then stirred at − 10°C for 30 min, warmed to 25 °C and further stirred at room temperature for an additional 90 min. Volatiles were removed and the remaining white solid was dried in vacuo. The white solid was washed with 40 ml of carbon tetrachloride. An analytical sample was obtained by recrystallization from ethanol. Yield (95%), m.p. = 133.2 °C; Anal.Calc. for C18H22ClN2O2P: C, 59.26; H, 6.08; N, 7.68. Found: C, 59.3; H, 6.1; N, 7.59%; 1H NMR (DMSO), δ (ppm): 1.36 (9H, s, C(CH3)), 4.067–4.088 (2H,m, CH2), 7.42–7.47 (10H, m, Ar), 9.47 (1H, d, 2JP-NH = 8.96 Hz); 13C NMR(DMSO), δ (ppm): 25.627 (s, CH3), 44.991 (s, CH2), 57.330 (s, C), 128.510 (s), 128.775 (s), 129.134 (s), 130.628 (s), 132.497 (s), 133.110 (s), 168.279 (s, CO); 31P {1H} NMR (DMSO), δ (ppm): − 4.331(s); 31P NMR (DMSO), δ (ppm): − 4.331 (d, 2JP-NH = 8.96 Hz); IR (KBr), ν (cm− 1): 451(m), 707 (s), 981 (w), 1024 (w), 1096 (s), 1235 (vs), 1419 (s), 1448 (s), 1580 (s), 2975 (w), 3370 (m).
N-benzoyl N′,n′-(tert-butybenzyl) Thiophosphoramidic Chloride, [(C6h5ch2)(c(ch3)3)np(s)(cl)nhcoc6h5], 2
A mixture of 0.847 g (5 mmol) thiophosphorylchloride (PSCl3), 0.605 g (5 mmol) benzamide and 50 ml of acetonitrile. The solution stirred at room temperature. After 5 days 0.815 g, (5 mmol) N-benzyl-N-tert-butylamine and 0.506 g (5 mmol) triethylamine were added to the mixture and the resulting yellow solution was cooled to 0°C for 60 min. Volatiles were removed and remaining solid was washed once with 20 ml of carbon tetrachloride and once with 10 ml of chloroform and purified by recrystallization from dry ethanol. Yield (70%), mp. = 124.7°C; Anal. Calc. for C18H22ClN2OPS: C, 56.76; H, 5.82; N, 7.35. Found: C, 56.69; H, 5.85; N, 7.37%; 1H NMR (DMSO), δ (ppm): 1.27 (9H, s, C(CH3)), 3.891–3.913 (2H, m, CH2), 7.21–7.26 (10H, m, Ar), 9.21 (1H, d, 2JP-NH = 9.23 Hz); 13C NMR(DMSO), δ (ppm): 21.472 (s, CH3), 41.251 (s, CH2), 53.991 (s), 124.411 (s), 124,676 (s), 125.038 (s), 126.529 (s), 128.391 (s), 129.014 (s), 162.951 (s, CO); 31P {1H } NMR (DMSO), δ (ppm): − 12.211(s); 31P NMR (DMSO), δ (ppm): − 12.211 (d, 2JP-NH = 9.23 Hz); IR (KBr), ν (cm− 1): 471(m), 581 (m), 710 (s), 980(s), 1018(s), 1420 (s), 1445(s), 1653(s), 2925(s), 3210(m).
Kinetic experiments
Acetylcholinesterase assay and inhibition experiments
The activity of the enzyme was measured at 25 °C by a modified Ellman's method [Citation14]. The reaction mixture for determination of IC50 value consisted of DTNB solution, 50 μL (10− 4M); inhibitors 1 and 2, × μL (5–150), ((0.112 × 10− 3 M – 3.358 × 10− 3 M) and (0.373 × 10− 3 M – 1.121 × 10− 2 M)); acetylthiocholine (ASCh) solution, 15 μL (1.35 × 10− 4M); phosphate buffer, (895-x) μL; hAChE solution, 40 μL. A solution containing all of the above materials except the inhibitor was used to determine the activity of enzyme as a control. The reaction mixture for determination of the inhibition mechanism and ki values was: DTNB and ASCh (same as above); a solution of the enzyme (40 μL) plus inhibitor 1 and 2 were x = 110 μL and x = 80 μL respectively; phosphate buffer (935-x) μL.
Reactivation of the Inhibited Enzyme
Human erythrocyte acetylcholinesterase, 25 μL, was incubated with 80 μL inhibitor (concentration adjusted to give about 80% hAChE inhibition) at 37°C. After 20 min, 50 μL of 0.1 M oxime solution in phosphate buffer was added to the above mixture which was incubated at 37°C for 30 min. hAChE activity in this sample was assayed by a modified Ellman's method [Citation14]. The percentage of reactivation was calculated according to the De Jong method [Citation15].
Results
Synthesis and Spectral Data
Benzoyl phosphoramidic dichloride reacted smoothly with N-Benzyl-N-tert-butylamine in the presence of triethylamine to afford N-Benzoyl N′, N′-(tert-butybenzyl) phosphoramidic chloride as a white powder in high yield (95%), after purification by recrystallization in ethanol. Benzamide reacts slowly with PSCl3 in carbon tetrachloride solvent a scaled reaction required more than 5 days to reach completion at room temperature). The crude product reacted with N-Benzyl-N-tert-buylamine in the presence of triethylamine to afford N-Benzoyl N′, N′-(tert-butybenzyl) thiophosphoramidic chloride 2 as a white powder in (70%) yield, after purification by recrystallization in benzene. These compounds are stable against hydrolysis in buffer solution and show only a single peak in the 31P NMR spectra.
Some spectroscopic data and melting points for these compounds are summarized in . 1H and 13C NMR spectroscopy showed approximately equivalent resonances for 1 and 2. The 1H NMR spectrum of compounds 1 and 2 indicated the couplings of NH hydrogen with phosphorus as 2JP-NH = 8.96 Hz and 2JP-NH = 9.23 Hz respectively according to similar phosphoramidate compounds [Citation16]. 31P {1H} NMR spectra showed that the phosphorus atom in compound 1 was deshielded with respect to that in 2 ().
Table I. The spectral and enzymatic data for compound 1 and 2.
IR spectra of compound 1 indicated a strong band at 1235 cm− 1 which belongs to P = O moiety which is absent in compound 2. A band at 581 cm− 1, attributed to P = S bond, compared with other reported data [Citation5]. Elemental analysis confirmed 1H, 13C, 31P NMR and IR spectroscopy data for the synthesized compounds.
Enzymatic measurements
The rates of inhibition of hAChE were determined with seven different concentrations of each phosphorus compounds. The IC50 values from of compounds 1 and 2 in reaction with hAChE (pure and crude) are shown in .
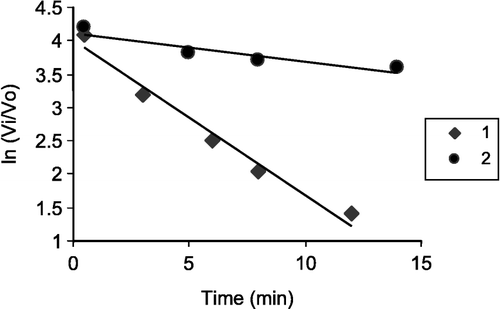
Figure 1 A plot of vi/v0 against log ([I] × 106) for inhibitors 1 and 2: vi and v0 are the activity of enzyme in the presence and absence of inhibitor, respectively and [I] is inhibitor concentration (mol/L).
![Figure 1 A plot of vi/v0 against log ([I] × 106) for inhibitors 1 and 2: vi and v0 are the activity of enzyme in the presence and absence of inhibitor, respectively and [I] is inhibitor concentration (mol/L).](/cms/asset/c8d77432-cb27-415c-ba7d-de2963d4400f/ienz_a_174815_f0001_b.gif)
shows the typical time-course of inhibition of human erythrocyte AChE by compounds 1 and 2 plotted as ln percent activity remaining (vi/v0) vs time the slopes of the graphs being linear. From these plots, the inactivation constant for reaction of compounds 1 and 2 with hAChE, ki, was calculated and were 0.271 and 0.0287 min− 1, respectively ().
The reactivating effectiveness of two oximes, pralidoxime, (A) and obidoxime (B) was investigated. In the effect of oximes on hAChE inhibited by compounds 1 and 2 are shown. Also the Km value for the enzyme was determined and is 108.20 × 10− 5 molL− 1 [Citation17].
Table II. Effects of oximes on hAChE inhibited by phosphoramidates
Discussion
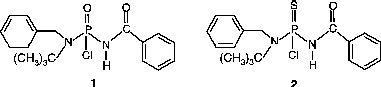
Two isosteric phosphoramidate compounds 1 and 2, with -(CO)NHP(X)- group, which have a likely β-diketone skeleton, were designed and synthesized.
The inhibitory potency of organophosphorus compounds is dependent upon the charge on the phosphorus atom, stereochemistry, the leaving group, reactivity and phosphorus atom substitutes [Citation18]. According to the Sussman model for the tetrahedral intermediate in AChE catalysis there is a covalent bond between Ser200 of the enzyme and the carbonyl carbon of acetylcholine (ACh) and also a hydrogen bond between His440 and the leaving group on ACh [Citation19]. Furthermore, it is known that the chlorine atom is a leaving group [Citation20] and this matter has been confirmed by thermochemical bond energies in phosphorus compounds [Citation21]. Hence, inhibition of hAChE by selected compounds probably resulted from the ability of the P-Cl bond that phosphorylates the active site of AChE by the phosphoryl residue [(C6H5CH2)(C(CH3)3)NP(X)NHCOC6H5], (X = O,S). This idea is confirmed by hydrogen bonding between His440 and the Cl atom and covalent bond between Ser200 and the phosphorus atom in compounds 1 and 2.
As shown in , the IC50 values indicate that the replacement of oxygen in the P = O moiety of compound 1 by sulfur decreased the affinity toward the active site of human erythrocyte AChE. These results are in accord with previously reported work with other organophosphorus compounds [Citation7,Citation8,Citation22]. An explanation for the greater inhibition of AChE by oxono compounds in comparison with thiono perhaps is the greater electronegativity of oxygen. This effect enhances the electrophilicity of phosphorus and thereby increases its reactivity toward nucleophiles, such as water or the active site serine of AChE [Citation8]. In this study, compound 2 exhibited a 31P NMR signal which is significantly shielded (8 ppm) relative to that of compound 1.
On the other hand, substitution of the ester moiety with an amino group of phosphoramidates decreased anticholinesterase activity as did replacement of a substituted alkyl group by an ester moiety (
[Citation23]. Therefore, the low value of IC50 for compounds 1 and 2 is comparable with that of other phosphoramidate compounds Citation23-25.
For simulation of the in vivo situation, the inhibitory potency of compounds 1 and 2 was measured using crude enzyme. In this case, IC50 values for compound 1 and 2 were 2.341 and 4.64 mM respectively, which are similar to other reported data [Citation26].
Phosphorus compounds are very potent inhibitors of AChE and usually react with the active site serine forming a very stable covalent phosphoryl-enzyme complex [Citation18,Citation23]. The biomolecular rate constants for compound 1 and 2 were 0.271 and 0.0287 min− 1, respectively, which indicate that both compounds are irreversible inhibitors. These results are in agreement with other reported values [Citation18,Citation23].
In the literature, there are many reports where oxime-induced reactivation of organophosphorus-AChE conjugate, were studied [Citation11,Citation15,Citation23]. Here, the reactivating potency of two conventional oximes was studied. The results showed that the reactivation effect of these oximes on hAChE inhibited by 1 and 2 was relatively low (), which was perhaps due to the steric effect of the tetrahedral phosphorus [Citation27].
In summary, two isosteric phosphoramidate compounds [(C6H5CH2)(C(CH3)3)NP(O)(Cl)NH(COC6H5)], (1) and [(C6H5CH2)(C(CH3)3)NP(S)(Cl)NH(COC6H5)], (2) were synthesized and characterized. The toxic properties of compounds 1 and 2 on hAChE were determined and the results showed low potency for these compounds, especially the thiono compound. IC50 values of compounds 1 and 2 in the reaction with crude hAChE were measured and were in agreement with those for the pure enzyme. The ki value indicated irreversible inhibition by compounds 1 and 2. Oxime-induced reactivation of hAChE inhibited by the two phosphoramidates was studied but reactivation was low using two different oximes.
References
- Taylor P. Anticholinesterase Agents7th ed. Mc Milam Press, New York 1985; 110–129
- Fukuto TR. Health Org 1971; 44: 31–41
- Eto M. Organophosphorus Pesticides, Organic and Biological Chemistry. CRC Press, Florida 1974
- Pardio VT, Ibarra N, Rodriguez MA. J Agric Food Chem 2001; 49: 6057–6062
- Quin LD. A Guide to Organophosphorus Chemistry. John Wiley & Sons. Inc, Publication, New York 2000
- Clothier B, Johnson MK, Reine E. Biochim Biophys Acta 1981; 660: 306–316
- Lee HS, Kim YA, Cho YA, Lee YT. Biosensor Chemosphere 2002; 46: 571–576
- Maxwell DM, Brecht KM. Chem Res Toxicol 1992; 5: 66–71
- Thompson CM, Frich JA, Natke BC, Hansen L. Chem. Res. Toxicol 1989; 2: 386–391
- Vilanova E, Johnson MK, Vicedo JL. Physiol 1987; 281: 224–238
- Langenberg JP, De Jong LPA, Otto MF, Benschop HP. Bull WHO 1988; 44: 31–42
- Bayiess AV, Millner OS. U.S. Patent 1980; 4: 182–881
- Sihotang K. Biochim Biophys Acta 1974; 370: 468–476
- Ellman GL, Courtney KD, Andres V, Featherstone RM. Biochem Pharmacol 1961; 7: 88
- De Jong LPA, Wolring GZ, Benschop HP. Arch Toxicol 1982; 49: 175–184
- Quin LD. Phosphorus-31 NMR Spectral Properties in Compound Characterization and Structural Analysis. VCH Publishers, Inc, New York 1994
- Gholivand K, Shariatinia Z, Ghadimi S, Mojahed F, Madani-Alizadegan A, Khajeh K, Naderi-manesh H. J Enz Inhib Med Chem 2004; 19: 403–407
- Thompson CM, Suarez AI, Rodriguez OP. Chem Res Toxicol 1996; 9: 1325–1332
- Sussman JL, Harel M, Frolow F, Oefner C, Gddman A, Toker L, Silman I. Science 1991; 253: 872–879, (Washington, D. C.)
- Ashani Y, Wins P, Wilson IB. Biochim Biophys Acta 1972; 284: 427–434
- Corbridge DEC. Phosphorus5th ed. Elsevier, Amsterdam 1995
- Ryu S, Lin J, Thompson CM. Chem Res. Toxicol 1991; 4: 517–520
- Jokanovic M, Maksimovic M, Kilibarda V, Jovanovic D. Toxicol Lett 1996; 85: 35–39
- Barak D, Ordentlich A, Kaplan D, Barak R, Mizrahi D, Kronman C, Segall Y, Velan B, Shafferman A. Biochemistry 2000; 39: 1156–1161
- Elhanay E, Ordentlich A, Dgany O, Kaplan D, Segall Y, Barak R, Velan B, Shafferman A. Chem Res Toxicol 2001; 14: 912
- Quistad GB, Zhang N, Spaks SE, Casida JE. Biochemistry 2000; 13: 652–657
- Wong L, Radie Bruggemann RIM, Hosea N, Berman HA, Taylor P. Biochemistry 2000; 39: 5750–5757