Abstract
The aim of this study was to investigate whether nicotine affects 6-phosphogluconate dehydrogenase (6PGD) enzyme activity in some rat tissues, and to see the modulatory effects of vitamin E on this effect in vivo. In addition, the effects of nicotine and vitamin E on 6PGD activity were also tested in vitro. The groups were: nicotine [0.5 mg/kg/day, intraperitoneal (i.p.)]; nicotine + vitamin E [75 mg/kg/day, intragastric (i.g.)]; and control group (receiving only vehicles). There were eight rats per group and supplementation period was 3 weeks. The results of in vivo study showed that nicotine activated the muscle, lungs, and testicular 6PGD enzyme activity but had no effect on heart and liver 6PGD activity. Also, nicotine + vitamin E activated the muscle, testicle, and liver 6PGD enzyme activity, while this combination had no effect on heart, and lungs in vivo. When nicotine is administered with vitamin E the increase in 6PGD enzyme activity in muscle and testicles were lower. On the other hand the increase in 6PGD enzyme activity was eliminated by vitamin E in lungs, while 6PGD enzyme activity was increased by vitamin E, which was not affected by nicotine only. In vitro results correlated well with in vivo experimental results. Our results suggest that vitamin E may favourably increase 6PGD enzyme activity in liver in nicotine treated rats, while it has negligible effects on this enzyme activity in other tissues.
Introduction
The third enzyme of pentose phosphate metabolic pathway, 6-phosphogluconate dehydrogenase (EC 1.1.1.44; 6PGD) catalyzes the conversion of 6-PGA to D-riboluse-5-phosphate in the presence of NADP+. This reaction yields NADPH, which protects the cell against the oxidant agents by producing reduced glutathione [Citation1,Citation2]. NADPH is also a coenzyme participating in the synthesis of a number of biomolecules such as fatty acids, steroids, and some amino acids [Citation3,Citation4]. When NADPH level decreases the concentration of reduced glutathione in living system declines, resulting in cell death. For this reason, 6PGD can be defined as an antioxidant enzyme [Citation5,Citation6].
There are many drugs which are commonly used by patients in the world. However, they have a lot of side effects on the activities of some enzymes such as G6PD, 6PGD, carbonic anhydrase (CA) and glutathione reductase (GR) in vitro and in vivo Citation7-16. If any drug inhibits 6PGD, the decreased NADPH and GSH will cause cell damage, resulting in severe health problems [Citation7,Citation8]. Effects of many chemicals and drugs on 6PGD enzyme activity have been investigated [Citation13,Citation17]. Vitamin C has been reported to stimulate 6PGD [Citation18] but no studies could be found on the in vivo effects of nicotine and vitamin E on rat tissue 6PGD enzyme.
The formation of the reactive oxygen species (ROS) in cells leads to the formation of free radicals in metabolic processes. These harmful species cause damages in many molecules such as lipids, proteins and nucleic acids. These harmful effects are controlled by antioxidant defense system in cells. The most important molecule in antioxidant defense system in various tissues of the body is glutathione Citation19-23. Furthermore, the enzymes such as superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), glutathione reductase (GR), glucose-6-phosphate dehydrogenase (G6PD) and 6PGD are necessary to remove these radicals and keep the cells stable. In normal conditions, the reductive and oxidative capacity of the cell (redox state) is in favor of oxidation Citation24-26. However, ROS produced in oxidative stress are removed by antioxidant defense system. Many researchers have determined that nicotine contributes ROS production Citation27-29. Consumption of cigarettes is common in many human societies. Two third of the American adults are addicted to alcohol and 30% of them are addicted to both cigarette and alchohol Citation30-33. Nicotine is a risk for various cardiovascular diseases and cancer. Nicotine is the major toxic component of cigarette smoke Citation34-37. Kessler et al [Citation38] have determined a marked increase in nicotine ratio in all kinds of cigarette in last decade in US. Shaw et al reported that one cigarette decreases lifespan 11 minutes [Citation39]. Nicotine is oxidized to its metabolite cotinine, which has a long half-life and may take part in vascular diseases Citation39-41. It is reported that chronic nicotine treatment decreases the cytochrom P450, and increases free radical formation and leads to oxidative damage in rats Citation29Citation31Citation37Citation42-44. Increased lipid peroxidation in blood of smokers has also been reported [Citation45].
Since cigarette smoking is common in many societies, we thought that the effect of nicotine on 6PGD activity is important. Therefore, we investigated the in vivo effects of nicotine and nicotine + vitamin E on rat muscle, heart, lungs, testicle and liver 6PGD activities. In addition, we also tested the effects of nicotine and vitamin E on 6PGD activity in vitro.
Materials and methods
Materials
NADP+, 6-phosphogluconate, protein assay reagent were purchased Sigma Chem. Co. All other chemicals used were analytical grade and purchased from either Sigma or Merck.
Animals
Twenty-four rats (Sprague-Dawley strain with a body weight of 225 ± 28 g), fed with standard laboratory chow and water, were used in the study. They were randomly divided into 3 groups (8 rats per group) and placed in separate cages during the study. The groups were as follows:
Group I: Nicotine (0.5 mg/kg/day, i.p.)
Group II: Nicotine (0.5 mg/kg/day, i.p.)+ Vitamin E (75 mg/kg/day, i.g.)
Group III: Control group (received only the same amounts of vehicles, 0.9% NaCl solution, i.p., and corn oil, i.g.). Supplementation period was 3 weeks. Animal experimentations were carried out in an ethically proper way by following guidelines as set by the Ethical Committee of the Ataturk University.
Preparation and administration of nicotine
Hydrogen tartrate salt of nicotine (Sigma N-5260) was dissolved in 0.9% NaCl solution to get a 0.15 mg/mL concentration of nicotine. Then, pH of the nicotine solution was adjusted to 7.4 by 0.1 N NaOH. Nicotine (0.5 mg/kg/day) was administered by intraperitoneal injection to groups 1, 2 and 3 for three weeks.
Preparation and administration of vitamin E
Vitamin E (Ephynal 300 capsule, Roche, France) was dissolved in corn oil (30 mg/mL) and administered orally by a stomach tube (approximately 75 mg/kg/day) to group 2 for 3 weeks.
Sample collection
At the end of the experiment, the animals were anesthetized with ketamine-HCl (Ketalar, 20 mg/kg, i.p.). The animals were killed by exsanguination by cardiac puncture after thoracotomy. Then, each tissue was carefully removed, rinsed in saline and stored in − 80°C until homogenization.
Preparation of homogenate
A piece of each tissue (approximately 300 mg) was homogenized by an OMNI TH International, model TH 220 (Warrenton, VA 20187 USA) homogenizer in 20 mM Tris-HCl, pH 7.4 (1/10 weight/volume) on ice for 10 in the first speed level. Then, the homogenates were centrifuged at 10.000 × g for 15 min at 4°C. The supernatants were stored at − 80°C in aliquots until biochemical measurements.
Ammonium sulphate fractionation and dialysis
Ammonium sulphate (20–60%) precipitation was made in homogenate. Ammonium sulphate was slowly added for completely dissolution. It was centrifuged at 5000 × g for 15 min and precipitate was dissolved in 20 mM Tris-HCl (pH 7.4), then dialysed at 4°C in 20 mM Tris-HCl (pH 7.4) for 2 h with two changes of buffer. Thus, partially purified total 6PGD was obtained by ammonium sulphate fractionation and dialysis from tissue homogenates (muscle, heart, lungs, testicle, and liver tissues).
In vitro studies
In vitro effects of nicotine (5 mM), nicotine (5 mM) + vitamin E (10 mM) on partially purified with ammonium sulphate fractionation total 6PGD from tissues homogenates were investigated. Activies were measured by adding 20, 40, 60, 80, and 100 μL of 5 mM nicotine, and nicotine (5 mM) + vitamin E (10 mM). Control cuvette activity was accepted as 100%.
In vivo studies and protein determination
The enzymatic activity was measured by Beutler's method [Citation46]. One EU was defined as the enzyme reducing 1 μmol NADP+ per min at 25°C and optimal pH (pH 8.0). Protein was determined by Bradford's method [Citation47] by using bovine serum albumin as a standard. The enzymatic activity and protein amount were measured one mL each sample of enzyme. Then, enzyme activity was determined as EU/mg protein.
Statistical analysis
One-way ANOVA with post-hoc LSD test was used to compare the group means and p < 0.05 was considered statistically significant. SPSS-for windows (version 10.0) was used for statistical analyses.
Results
Nicotine activated muscle, lungs, and testicle 6PGD enzyme activity in vivo. The differences in the mean values among the nicotine treatment group were statistically significant (p < 0.05) according to the Analysis of Variance (). However, it had no effect on the heart and liver 6PGD activity ().
Figure 1 In vivo effects of nicotine and nicotine + vitamin E on rat muscle, heart, lungs, testicle, and liver tissue 6-phosphogluconate dehydrogenase enzyme activity (Signicifant, *nicotine versus control, +nicotine + vitamin E versus control, ψnicotine + vitamin E versus nicotine, (Significant (*P, +P, and ψP < 0.05; **P, ++P < 0.02; ***P and ψψψP < 0.001, n = 8).
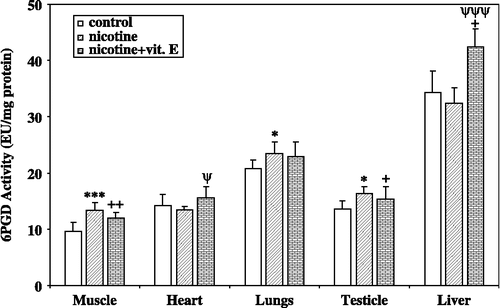
Nicotine + vitamin E had also activated the muscle, testicle, and liver 6PGD enzyme activity. The differences in the mean values among the nicotine treatment group were greater than would be expected by chance; there was a statistically significant difference (p < 0.05) according to the Analysis of Variance (). On the other hand, nicotine + vitamin E had no statistically significant effects on the heart, and lungs 6PGD enzyme activity compared with the control ().
The in vivo results showed that nicotine (0.5 mg/kg) activated the muscle, lungs, and testicle 6PGD enzyme activity by ∼38.8% (p < 0.001), ∼12.6% (p < 0.05), and ∼20.3% (p < 0.05) respectively, and nicotine had no effects on the heart and liver 6PGD activity in vivo. Also, nicotine + vitamin E (0.5 mg/kg + 75 mg/kg) activated the muscle, testicle, as well as liver 6PGD enzyme activity by ∼24.8% (p < 0.05), ∼12.68% (p < 0.05), and ∼23.7% (p < 0.001) respectively, and nicotine + vitamin E had no effect on the heart, and lungs 6PGD activity in vivo.
The results of the in vitro inhibition studies with nicotine (5 mM) and nicotine (5 mM) + vitamin E (10 mM) are shown in . As seen, nicotine activated 6PGD activity in vitro, in muscle, lungs, and testicle tissues and this activation was eliminated partly by vitamin E.
Table I. In vitro effects of nicotine and nicotine + vitamin E on 6-phosphogluconate dehydrogenase activity in the various rat tissues.
Discussion
There are many drugs and chemicals, which have adverse effects on living cells when they are used as therapeutics Citation19Citation27-29. These effects may be very important for health [Citation27]. For example in 1926, pamaquine used for malaria treatment caused severe adverse effects in the patients within several days, resulting in black urination, hyperbilirubinemia, dramatic decrease in blood hemoglobin levels, and finally death, which occurred in cases of severe G6PD deficiency [Citation30]. Similarly, acetazolamide inhibited carbonic anhydrase (CA), giving rise to severe diuresis [Citation31].
Glutathione levels in liver and testicles decrease markedly by chronic nicotine treatment [Citation16]. Nicotine is oxidized to its main metabolite cotinine in liver and causes the formation of free radicals in tissues. The formation of these radicals causes oxidative damage. The decrease in GSH in tissues leads to oxidative tissue damage. There are many studies about the effects of nicotine on the enzyme activities. For example, Gumustekin et al. [Citation48] have reported that nicotine has increased the activity of GSH-Px of brain while vitamin-E reversed this effect. Nicotine has also inhibited the brain glutathione S-transferase (GST) enzyme and this inhibition has been reversed by vitamin-E, too. In addition, Suleyman et al. [Citation49] have reported that nicotine has inhibited the activity of SOD of erythrocytes, however vitamin-E could not reverse this effect.
Activation of liver enzymes in rats, which were exposed to cigarette smoke [Citation50], and increased (2.5 fold) expression of enzymes of energy metabolism in nicotine treated rats [Citation51] were found. In addition, activation of adenylate cyclase enzyme in nicotine (6 mg/kg) treated rats was demonstrated by Abreu-Villaca et al. [Citation52].
As seen in , when nicotine is administered with vitamin E, the increase in 6PGD enzyme activity in muscle decreased from 38.8% to 24.8, while this increase in testicle decreased from 20.3% to 12.68%. On the other hand the increase in 6PGD enzyme activity was eliminated by vitamin E in lungs. In vitro 6PGD activation results generally correlated well with in vivo experimental results. Our results suggested that vitamin E may favorably increase 6PGD enzyme activity in the liver of nicotine treated rats, while it has negligible effects an other tissues.
References
- Lehninger AL, Nelson DL, Cox MM. Principles of biochemistry. Worth Publishers, New York 1993
- Bianchi D, Bertrand O, Haupt K, Coello N. Enzyme Microb Technol 2001; 28: 754–759
- Kahler SG, Kirkman HN. J Biol Chem 1983; 258: 717–718
- Takizawa T, Huang IY, Ikuta T, Yoshida A. Proc Natl Acad Sci U S A 1986; 83: 4157–4161
- Kozar RA, Weibel CJ, Cipolla J, Klein AJ, Haber MM, Abedin MZ, Trooskin SZ. Crit Care Med 2000; 28: 2486–2491
- Srivastava SK, Beutler E. Biochem J 1970; 119: 353–357
- Beutler E. Blood 1994; 84: 3613–3636
- Morse EE. Ann Clin Lab Sci 1988; 18: 13–18
- Jacobasch G, Rapoport SM. Mol Aspects Med 1996; 17: 143–170
- Beydemir S, M CI, Ozmen I, Buyukokuroglu ME, Ozdemir H, Kufrevioglu OI. Pharmacol Res 2000; 42: 187–191
- Ciftci M, Ozmen I, Buyukokuroglu ME, Pence S, Kufrevioglu OI. Clin Biochem 2001; 34: 297–302
- Ciftci M, Bilici D, Kufrevioglu OI. Pharmacol Res 2001; 44: 7–11
- Ciftci M, Beydemir S, Yilmaz H, Bakan E. Pol J Pharmacol 2002; 54: 275–280
- Ciftci M, Turkoglu V, Aldemir S. Vet Med-Czech 2002; 47: 283–288
- Erat M, Sakiroglu H, Ciftci M. Vet Med-Czech 2003; 48: 305–312
- Husain K, Scott BR, Reddy SK, Somani SM. Alcohol 2001; 25: 89–97
- Akyuz M, Erat M, Ciftci M, Gumustekin K, Bakan N. J Enz Inhib Med Chem 2004; 19: 361–365
- Puskas F, Gergely P, Jr, Banki K, Perl A. Faseb J 2000; 14: 1352–1361
- Ames BN, Shigenaga MK, Hagen TM. Proc Natl Acad Sci U S A 1993; 90: 7915–7922
- McCord JM. Clin Biochem 1993; 26: 351–357
- Bondy SC, Orozco J. Alcohol 1994; 29: 375–383
- Gul M, Kutay FZ, Temocin S, Hanninen O. Indian J Exp Biol 2000; 38: 625–634
- DeLeve LD, Kaplowitz N. Pharmacol Ther 1991; 52: 287–305
- Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. Clarendon Press, Oxford 1989
- Somani SM. Exercise, drugs and tissue specific antioxidant system. Pharmacology in exercise and sports, SM Somani. CRC Press, Boca Raton FL 1996; 57–95
- Rubin E. Alcohol Health Res World 1993; 17: 272–278
- Jenkins RR, Goldfarb A. Med Sci Sports Exerc 1993; 25: 210–212
- Guemouri L, Lecomte E, Herbeth B, Pirollet P, Paille F, Siest G, Artur Y. J Stud Alcohol 1993; 54: 626–629
- Leanderson P, Tagesson C. Chem Biol Interact 1992; 81: 197–208
- Mehta MC, Jain AC, Billie M. J Cardiovasc Pharmacol 1998; 31: 930–936
- Wetscher GJ, Bagchi M, Bagchi D, Perdikis G, Hinder PR, Glaser K, Hinder RA. Free Radic Biol Med 1995; 18: 877–882
- US Department of Health and Human Services. Seventh special report to the US congress on alcohol and health. National Institute on Alcohol Abuse and Alcoholism (US Department of Health and Human Services Publication ADM 90, 1656), Washington, DC 1990
- US Environmental Protection Agency. Respiratory health effects of passive smoking, lung cancer and other disorders. US Environmental Protection Agency (US Environmental Protection Agency Publication 600, 6-90/006F), Washington, DC 1992
- Del Boccio G, Lapenna D, Porreca E, Pennelli A, Savini F, Feliciani P, Ricci G, Cuccurullo F. Atherosclerosis 1990; 81: 127–135
- Maser E. Trends Pharmacol Sci 1997; 18: 270–275
- Hoffmann D, Rivenson A, Hecht SS. Crit Rev Toxicol 1996; 26: 199–211
- Pryor WA, Stone K. Ann N Y Acad Sci 1993; 686(12–27)27–18
- Kessler DA, Witt AM, Barnett PS, Zeller MR, Natanblut SL, Wilkenfeld JP, Lorraine CC, Thompson LJ, Schultz WB. N Engl J Med 1996; 335: 988–994
- Shaw M, Mitchell R, Dorling D. BMJ 2000; 320: 53
- Sastry BV, Gujrati VR. Ann N Y Acad Sci 1994; 714: 312–314
- Sastry BV, Chance MB, Singh G, Horn JL, Janson VE. Pharmacology 1995; 50: 128–136
- Anandatheerthavarada HK, Williams JF, Wecker L. J Neurochem 1993; 60: 1941–1944
- Ashakumary L, Vijayammal PL. J Ecotoxicol Environ Monit 1991; 1: 283–290
- Bhagwat SV, Vijayasarathy C, Raza H, Mullick J, Avadhani NG. Biochem Pharmacol 1998; 56: 831–839
- Altuntas I, Dane S, Gumustekin K. J Basic Clin Physiol Pharmacol 2002; 13: 69–72
- Beutler E. Red cell metabolism: A manual of biochemical method. Grune and Stration, New York 1984
- Bradford MM. Anal Biochem 1976; 72: 248–254
- Gumustekin K, Altinkaynak K, Timur H, Taysi S, Oztasan N, Polat MF, Akcay F, Suleyman H, Dane S, Gul M. Hum Exp Toxicol 2003; 22: 425–431
- Suleyman H, Gumustekin K, Taysi S, Keles S, Oztasan N, Aktas O, Altinkaynak K, Timur H, Akcay F, Akar S, Dane S, Gul M. Biol Pharm Bull 2002; 25: 1133–1136
- Vanscheeuwijck PM, Teredesai A, Terpstra PM, Verbeeck J, Kuhl P, Gerstenberg B, Gebel S, Carmines EL. Food Chem Toxicol 2002; 40: 113–131
- Hu D, Cao K, Peterson-Wakeman R, Wang R. Biochem Biophys Res Commun 2002; 297: 729–736
- Abreu-Villaca Y, Seidler FJ, Slotkin TA. Brain Res 2003; 988: 164–172