ABSTRACT
Introduction
Measles, mumps, and rubella incidence decreased drastically following vaccination programs’ implementation. However, measles and mumps’ resurgence was recently reported, outbreaks still occur, and challenges remain to control these diseases.
Areas covered
This qualitative narrative review provides an objective appraisal of the literature regarding current challenges in controlling measles, mumps, rubella infections, and interventions to address them.
Expert opinion
While vaccines against measles, mumps, and rubella (including trivalent vaccines) are widely used and effective, challenges to control these diseases are mainly related to insufficient immunization coverage and changing vaccination needs owing to new global environment (e.g. traveling, migration, population density). By understanding disease transmission peculiarities by setting, initiatives are needed to optimize vaccination policies and increase vaccination coverage, which was further negatively impacted by COVID-19 pandemic. Also, awareness of the potential severity of infections and the role of vaccines should increase. Reminder systems, vaccination of disadvantaged, high-risk and difficult-to-reach populations, accessibility of vaccination, healthcare infrastructure, and vaccination services management should improve. Outbreak preparedness should be strengthened, including implementation of high-quality surveillance systems to monitor epidemiology. While the main focus should be on these public health initiatives to increase vaccination coverage, slightly more benefits could come from evolution of current vaccines.
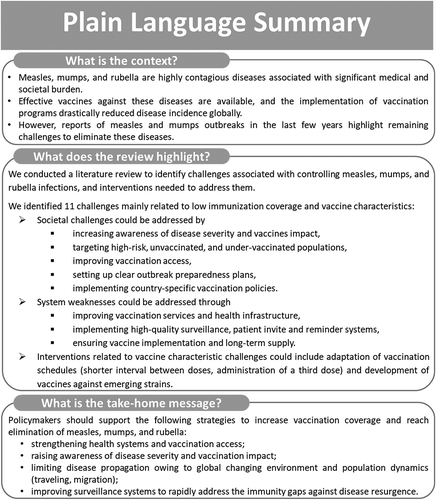
PLAIN LANGUAGE SUMMARY
What is the context?
Measles, mumps, and rubella are highly contagious diseases associated with significant medical and societal burden. Effective vaccines against these diseases are available, and the implementation of vaccination programs drastically reduced disease incidence globally. However, reports of measles and mumps outbreaks in the last few years highlight remaining challenges to eliminate these diseases.
What does the review highlight?
We conducted a literature review to identify challenges associated with controlling measles, mumps, and rubella infections, and interventions needed to address them. We identified 11 challenges mainly related to low immunization coverage and vaccine characteristics. Societal challenges could be addressed by increasing awareness of disease severity and vaccines impact, targeting high-risk, unvaccinated, and under-vaccinated populations, improving vaccination access, setting up clear outbreak preparedness plans, and implementing country-specific vaccination policies. System weaknesses could be addressed through improving vaccination services and health infrastructure, implementing high-quality surveillance, patient invite, and reminder systems, ensuring vaccine implementation and long-term supply. Interventions related to vaccine characteristic challenges could include adaptation of vaccination schedules (shorter interval between doses, administration of a third dose) and development of vaccines against emerging strains.
What is the take-home message?
Policymakers should support the following strategies to increase vaccination coverage and reach elimination of measles, mumps, and rubella: strengthening health systems and vaccination access; raising awareness of disease severity and vaccination impact; limiting disease propagation owing to global changing environment and population dynamics (traveling, migration); improving surveillance systems to rapidly address the immunity gaps against disease resurgence.
1. Introduction
Measles, mumps, and rubella (M, M, and R) are highly contagious viral infections causing a significant burden on the affected individuals and on healthcare systems. Measles is one of the most infectious human diseases, which can cause serious illness, lifelong complications, and death [Citation1,Citation2]. Although mortality for mumps is less than that for measles, it may result in complications such as orchitis, meningitis, encephalitis, and deafness [Citation3,Citation4]. Rubella infections cause a relatively mild disease for children, but infections in women during early pregnancy can affect the fetus, resulting in miscarriage, fetal death, or congenital rubella syndrome [Citation1].
In 2012, the World Health Assembly endorsed the Global Vaccine Action Plan, with the objective of eliminating measles, rubella, and congenital rubella syndrome in at least five of the six World Health Organization (WHO) regions by 2020 [Citation1,Citation5]. All WHO regions had measles elimination goals and two had rubella elimination goals [Citation6]. Between 2012 and 2020, the measles and rubella diseases burden was reduced, but the targets of the Global Vaccine Action Plan were not met [Citation7]. Therefore, the measles and rubella strategic framework 2021–2030 was developed by the Measles & Rubella Initiative within the umbrella of the Immunization Agenda 2030 structure with the objective of achieving a world without measles and rubella by 2030 [Citation7,Citation8]. Since another goal of the WHO is to achieve a high level of mumps control, the choice of implementing combined live attenuated vaccines against M, M, and R (MMR vaccines) in their national immunization programs has been made in 121 countries worldwide [Citation9–11]. The objective was and is to target simultaneously the three diseases and to potentially increase vaccination coverage through reductions in the number of visits and injections. Of note, different single and combined vaccines targeting M, M, and R are available [Citation12,Citation13].
The vaccination coverage rate needed to stop transmission depends on the basic reproductive rate (R0) of the infectious agent [Citation14] and on vaccine effectiveness. Since measles is the most contagious of all vaccine-preventable transmitted diseases (R0 of 12 to 18 [Citation15,Citation16]), immunization policies for measles-containing vaccines are driven by the measles coverage rate needed to reach the community immunity level of 90% to 95% required to prevent outbreaks [Citation10,Citation17,Citation18]. Sufficient coverage levels to provide community protection are more easily obtained for mumps (R0 of 4–7 [Citation9]) and rubella (R0 of 3–8 in Europe [Citation19] and up to 12 in developing countries [Citation20]). Since 2017, the WHO and Gavi, the Vaccine Alliance, have encouraged all countries to recommend a second dose of measles-containing vaccine in their childhood national immunization programs [Citation21,Citation22]. By the end of 2019, the inclusion of a second dose of measles-containing vaccine as part of routine immunization had been implemented in 178 countries, and 122 and 173 countries had introduced mumps and rubella vaccination, respectively [Citation23]. Between 2000 and 2019, estimated coverage rates for the first dose of measles-containing vaccine increased globally from 72% to 85% and for the second dose from 18% to 71% [Citation24].
Historically, after the implementation of vaccination programs, dramatic decreases in global annual incidence for M, M, and R were observed [Citation4,Citation25–28]. However, a resurgence of these diseases has recently occurred, especially for measles and mumps, possibly as a result of the changing environment and context [Citation25–27,Citation29–39]. For measles, the global annual incidence decreased by 83% and the number of measles deaths by 80% between 2000 and 2017 [Citation25]. However, since 2017, measles incidence has increased [Citation25,Citation40,Citation41], and large outbreaks have been reported in several areas, including European countries, in a wide range of contexts and due to diverse causes [Citation25,Citation29–33,Citation42]. Concerning the incidence of mumps, decreases from 100 to 1,000 cases per 10,000 population in the absence of immunization to <1 case per 100,000 population within 10 years after the introduction of immunization programs were observed in several countries [Citation4,Citation26,Citation27]. However, mumps outbreaks still occur, especially in older adolescents and young adults [Citation26,Citation27]. In contrast with measles outbreaks which mostly occur in unvaccinated individuals [Citation40], mumps outbreaks were also reported in countries with high vaccination coverage rates [Citation3,Citation34–39,Citation43,Citation44]. For rubella, the number of reported cases has declined by 97% after the introduction of the vaccination campaigns [Citation28]. Whereas previously the primary goal of rubella vaccination was to prevent congenital rubella syndrome cases by immunizing girls before childbearing age, it is now considered that this can be more easily reached through universal childhood vaccination, which may eventually also eliminate rubella infections [Citation28,Citation45].
The foregoing sections highlight that efforts to eliminate M, M, and R have not yielded the desired results, even in countries that implemented vaccination programs for M, M, and R to reach the required coverage and diseases control. The aim of this qualitative narrative review is to provide an objective appraisal of the literature regarding the current challenges associated with controlling M, M, and R infections, and an overview of the interventions needed to address them. These challenges are even more important today considering the negative impact of the COVID-19 pandemic on coverage rates for several vaccines [Citation46], including measles-containing vaccines [Citation47]. Considerations about strengths and limitations of the currently available trivalent MMR vaccines will be also discussed. In this review, the qualitative outcomes have been grouped under challenges related to societal factors, system weaknesses, and vaccine characteristics. A summary contextualizing the outcomes of this review is displayed in the Plain Language Summary () for the convenience of healthcare professionals.
2. Methods
This qualitative narrative review of the literature [Citation48] considers studies where challenges related to MMR vaccination coverage and vaccine characteristics were identified and interventions to address them were proposed. Priority was given to existing systematic reviews. Proposed interventions could be aimed at people (e.g. caregivers of children eligible for MMR vaccination) or at service providers (e.g. general practices, community primary care teams). The language was restricted to English. No restrictions on publication date were applied, but focus was on recent literature (published after 2005).
To ensure an objective review of the literature and reduce bias, we first undertook a preliminary search to identify published articles relevant to the field of interest. Then, we defined the topics of discussion based on titles and abstracts. The search was initiated in November 2019. We searched PubMed/MEDLINE to identify articles using either ‘MMR,’ ‘measles,’ ‘mumps,’ or ‘rubella’ in combination with each of the following search terms: ‘vaccination,’ ‘challenge,’ ‘outbreak,’ ‘control,’ ‘elimination,’ ‘eradication,’ ‘coverage,’ ‘early,’ ‘migrant,’ ‘healthcare worker,’ ‘traveler,’ ‘surveillance,’ ‘waning,’ ‘genotype,’ ‘effectiveness,’ ‘mandatory,’ ‘third dose,’ ‘guidance,’ and ‘position.’ On a second reading, we collated the challenges and interventions into 11 topics, which were categorized into societal factors (awareness on the value of vaccination, unvaccinated or under-vaccinated individuals, access threats and constraints, outbreak preparedness, and epidemiology), system weaknesses (vaccination infrastructure and management, surveillance systems, and vaccine supply), and vaccine characteristics (primary vaccination failure, secondary vaccination failure, and vaccine strains and new genotypes) (). Of note, some important articles published before 2005 and after November 2019 were included during the development of the review even if these were not identified during the literature search.
Figure 2. Summary of reported challenges related to the control of measles, mumps, and rubella infections and the most important interventions to address them that are described in this publication* [Citation49]
![Figure 2. Summary of reported challenges related to the control of measles, mumps, and rubella infections and the most important interventions to address them that are described in this publication* [Citation49]](/cms/asset/fa8190b5-4835-4667-bac1-a755794039e0/ierv_a_1927722_f0002_oc.jpg)
3. Challenges and interventions
3.1. Societal factors
3.1.1. Awareness on the value of vaccination
Measles outbreaks occur mostly in unvaccinated or under-vaccinated individuals [Citation40,Citation50]. One of the explanations for the resurgence of measles outbreaks in several countries with available vaccination services is the increasing proportion of individuals who are delaying or refusing vaccination for themselves or their children. This phenomenon is known as vaccine hesitancy according to the Strategic Advisory Group of Experts (SAGE) [Citation51,Citation52], and is influenced by complacency, convenience, and confidence [Citation51–54]. In the case of M, M, and R, the perception of the disease risks by the caregivers is affected because severe complications have relatively disappeared out of the public attention as a consequence of the successful vaccination programs in many regions [Citation10,Citation30,Citation55]. Moreover, unsubstantiated concerns about the safety of the combined MMR vaccines continue to cause vaccine hesitancy and threaten community immunity in many countries, even after the safety of this vaccine has been demonstrated [Citation9,Citation12,Citation55]. Although data on the effectiveness of interventions to address vaccine hesitancy are limited [Citation56,Citation57], a systematic review has shown that strategies should be multifactorial since various reasons may cause resistance to immunization, should include dialog-based approaches, and should be specific for the target population and context [Citation56].
In some countries, mandatory vaccination strategies were undertaken in response to decreased vaccination coverage and measles resurgence due to hesitancy and anti-vaccination movement [Citation58]. In Italy, where poor results were observed with improved public immunization services and communication campaigns, MMR vaccination was added to the list of mandatory vaccines in 2017, and proof of vaccination is required to attend kindergarten, nurseries, and elementary or middle school [Citation31,Citation59]. Compulsory vaccination had a positive impact on vaccine uptake, with a vaccination coverage for the first dose of MMR vaccine at 24 months of age increasing from 87.3% in 2016 to 94.1% in 2018 [Citation59]. As from March 2020, mandatory measles vaccination has been implemented in Germany for hospital personnel, children and teachers in schools and nurseries, and community facilities personnel [Citation60]. In the United States (USA), the introduction of compulsory vaccination is still under discussion, and the decision to impose vaccination by school entry varies from state to state [Citation61]. In Australia, incentives are used, and parents only receive financial benefits when their children are up to date with their immunizations [Citation62].
3.1.2. Unvaccinated or under-vaccinated individuals
The remaining pockets of unvaccinated or under-vaccinated individuals, who may be responsible for outbreaks and transmission, should be identified and targeted [Citation40].
A first pocket of individuals potentially responsible for disease transmission are short-term travelers with incomplete or unknown vaccination status who can be infected and import the disease to their country of origin, as observed in Italy and Costa Rica [Citation40,Citation63,Citation64]. While community protection is important for unvaccinated individuals and vaccinated individuals who did not develop protective antibody levels, it does not protect unvaccinated individuals when they are traveling to regions where the diseases are endemic [Citation40]. Therefore, national health authorities should encourage people to check their measles vaccination status before traveling abroad to avoid importation of measles by returning travelers and reduce the risk of outbreaks. Pre-travel health advice concerning measles vaccination should be given by general practitioners, especially for young adults [Citation65]. Travel medicine consultations should include measles immunization status checks, and measles vaccination should be recommended before traveling to endemic countries [Citation66].
Unvaccinated or under-vaccinated migrants and refugees may also play a role in disease transmission if they come from countries affected by endemic infections since they can be carriers in subclinical stages of the disease [Citation67]. They usually have lower immunization rates due to limited access to vaccination in their country of origin [Citation65,Citation68,Citation69]. Therefore, specific guidance is needed, and catch-up vaccination should be proposed for these vulnerable populations [Citation68,Citation70].
Unvaccinated or under-vaccinated healthcare workers (HCWs) are another pocket of individuals with a high potential for transmission, especially for measles infection as the estimated risk of acquiring this disease is 2 to 19 times higher for susceptible HCWs than for the general population [Citation71–75]. Nosocomial measles infection represents a risk for unvaccinated HCWs as well as for vulnerable patient groups, especially those with compromised immunity [Citation76]. Although vaccination of HCWs is important to achieve measles elimination goals and to limit the spread from healthcare settings to the community [Citation73,Citation74,Citation77], vaccination coverage rates remain too low in this potentially exposed population, especially among young HCWs [Citation78–80]. While educational interventions, improved access to vaccination, and personal engagement were shown to be useful to increase vaccine uptake [Citation81], global recommendations and policies should be established for the immunization of HCWs [Citation71–74]. In some countries (e.g. Finland and a few states in the USA), mandatory vaccination has been implemented to increase the proportion of HCWs who receive two doses of the MMR vaccine [Citation73,Citation79,Citation82]. Strategies should also be established to assess immune status during recruitment procedures (vaccination record and, if needed, antibody screening), monitor vaccination coverage rates, and exclude susceptible HCWs from high-risk settings [Citation78,Citation80].
Pockets of unvaccinated individuals also persist in countries with a high routine childhood vaccination coverage due to the unawareness that some individuals could or should be vaccinated [Citation40]. Measles may be transmitted by older individuals who are still susceptible because of suboptimal vaccination coverage in the past. This is especially important for 30- to 40-year-old adults among whom the proportion of individuals who are neither vaccinated nor naturally immune against measles is higher than in other age groups. Since the administration of the second MMR vaccine dose has only been implemented in immunization schedules in the 2000s, a large proportion of compliant young adults have only received one dose [Citation10]. In European countries, catch-up vaccination programs (vaccination of unvaccinated older children and adults) have been recommended for individuals who did not receive the first or second dose of the MMR vaccine, but these were not efficiently conducted or accepted [Citation10].
Individuals with contra-indications, such as specific immunodeficiency diseases, or a history of confirmed anaphylactic reaction to a previous vaccine dose or to a component of the vaccine, and children born to a mother who received immunosuppressive biological therapy during pregnancy constitute other pockets of vulnerable unvaccinated individuals [Citation4,Citation21,Citation83]. Even if MMR vaccines cannot be administered to severely immunocompromised persons, human immunodeficiency virus (HIV)-infected children with a CD4 T-cell count above severe immunosuppression thresholds and asymptomatic HIV-infected children could receive MMR vaccination according to routine childhood schedules [Citation84–86]. Two doses of measles-containing vaccine administered at 6 and 12 months of age were shown to be similarly safe and immunogenic in HIV-unexposed and HIV-exposed, uninfected children [Citation87]. However, HIV-infected children and adolescents may need additional MMR vaccine doses to achieve sufficient levels of immunity, especially if antiretroviral therapy has been interrupted [Citation88]. MMR vaccination may also be considered in patients with other immune deficiencies for whom the benefits of vaccination outweigh the risks, such as patients with immunoglobulin G (IgG) subclass deficiencies, congenital neutropenia, chronic granulomatous disease, and complement deficiency diseases [Citation86]. Of note, MMR vaccines should be used cautiously in patients with significant immunologic dysfunctions, as a few cases of severe or even fatal vaccine strain infections have been reported in this population [Citation89–91].
3.1.3. Access threats and constraints
Military conflict, civil unrest, and political and economic crises have been responsible for low vaccination coverage rates and increased risks of outbreaks in many countries [Citation32,Citation92,Citation93].
A recent review has shown that between 2010 and 2015, 39% of measles cases were reported in 16 conflict-affected countries [Citation92]. Moreover, according to estimates made by the United Nations International Children’s Fund (UNICEF) in 2016, about two-thirds of all unvaccinated children lived in conflict-affected countries [Citation94]. Conflicts cause several adverse effects on health parameters, including disruption of immunization supply chains and vaccine logistics (e.g. disruption of the cold chain and vaccine delivery), destruction of health infrastructure, depletion of human resources, and forced migration of communities to areas with limited access to immunization services [Citation92]. Civil unrest limiting vaccine delivery was also identified as a major challenge in achieving rubella elimination goal [Citation28,Citation95]. There are several strategies that may be used in conflict-affected countries to increase vaccination coverage, including the organization of vaccination campaigns (vaccination catch-up) and the use of outreach services (vaccination of children in remote locations on scheduled dates known by the communities) in collaboration with local communities and the military or security personnel to guarantee a safe passage and security for HCWs [Citation92,Citation94]. Such a post-conflict catch-up vaccination campaign has been successfully conducted in the Central African Republic [Citation96]. Gavi, the Vaccine Alliance, has requested that governments support the costs of civil society organizations for the development of vaccine stockpiles for humanitarian emergencies, the strengthening of healthcare systems, and the improvement of the access and affordability of vaccines in conflict-affected areas [Citation92,Citation97].
As social and military crises, public health crises of high magnitude, such as the COVID-19 pandemic, have also the potential to disrupt the delivery of basic healthcare and immunization services, including routine vaccination campaigns. Despite the WHO recommendations to maintain essential health services during outbreaks [Citation98], several countries have recorded significant decreases in vaccination coverage due to the COVID-19 pandemic, leading to increased risk for outbreaks of several vaccine-preventable diseases [Citation99,Citation100]. Disparities in this decline in vaccination coverage were observed, and low- to middle-income countries were particularly affected [Citation46]. The main factors contributing to this decline included the fear of being exposed to the virus at healthcare facilities, restrictions in terms of population movements, shortage of HCWs, and overloaded healthcare systems [Citation46]. Among others, measles vaccination campaigns were paused or postponed in several countries to limit the propagation of COVID-19 [Citation47]. In these regions, it is particularly important to track the children who did not receive measles-containing vaccines and to ensure that they are immunized as quickly as possible when the safety of communities and HCWs can be ensured.
In summary, the common goal of all interventions aimed at restoring disrupted vaccination coverage due to the abovementioned reasons is to ensure that all communities and individuals who were not vaccinated through routine immunization services receive their vaccines. The organization of supplementary immunization activities (SIAs; vaccination campaigns during which the targeted populations are immunized regardless of their vaccination history) and catch-up campaigns, as well as the strengthening of outreach services are major initiatives to reach this objective [Citation1,Citation25,Citation101–104]. In 2017, approximately 205 million doses of measles-containing vaccine were administered during 53 SIAs in 39 countries with a low vaccination coverage rate and a high measles burden [Citation25,Citation102]. Additional solutions to improve vaccination service arrangements in low- to middle-income countries include home visits and inter- and intra-facility referral linkages, which are particularly important when families move between vaccinations [Citation104].
3.1.4. Outbreak preparedness
Because measles is still endemic in many countries, its importation into measles-free areas can occur and new outbreaks can happen if the level of population immunity is insufficient [Citation105]. In these settings, many susceptible persons are already infected before an outbreak is recognized because of the very high contagiousness of measles [Citation105]. Immunization levels that are sufficient to eliminate the transmission of measles are particularly difficult to reach in densely populated areas [Citation106]. During outbreaks, infection prevention and control practices should be strengthened, infections among HCWs should be prevented, and transmission in healthcare settings and spread to vulnerable populations should be reduced [Citation29,Citation49]. The WHO recommends that all suspected measles cases be reported to public health authorities, non-severe measles cases be isolated at home, and patients with clinically suspected measles or other clinical warning signs be admitted to a treatment facility with isolation capacity [Citation49]. Exposed HCWs, patients, and visitors should be evaluated for evidence of measles immunity and should receive post-exposure prophylaxis if necessary [Citation49].
Another unsolved problem remains the prevention of mumps outbreaks, which also happen in countries with high vaccination coverage rates [Citation3,Citation34–39] and mainly involve adolescents and young adults who were previously immunized during childhood [Citation107]. Mumps outbreaks in vaccinated populations may be caused by changes in the circulating strains, which have different genotypes than those included in the vaccine [Citation107], but a major cause is the waning immunity after vaccination due to low persistence of neutralizing antibodies and poor B-cell memory [Citation108]. In many countries, including the USA, the administration of a third dose of MMR vaccine to adolescents and adults is recommended during outbreaks to increase anti-mumps antibody levels and avoid the propagation of mumps disease [Citation36,Citation109,Citation110]. This intervention is useful to control mumps outbreaks but the increases in anti-mumps antibody levels induced by a third dose of MMR vaccine were shown to be only transient [Citation111,Citation112].
Since both measles and mumps outbreaks are still important public health concerns with heavy consequences on elimination goals, other strategic interventions are needed to reduce their impact. Preparedness plans, taking into account the long timelines for the manufacturing and the release of vaccines, should be established to reduce the risk of vaccine shortage during outbreaks and the associated elevated level of societal panic [Citation113]. In preparedness plans, vaccination of all HCWs and of populations who are at increased risk of contracting the disease, such as adolescents and young adults residing or working in institutions, should be considered [Citation7,Citation26,Citation40,Citation105,Citation49,Citation107]. During outbreaks, policy-makers should also consider a temporary revision of the national childhood immunization schedule to include an early dose of the MMR vaccine for infants from 6 months of age and shorten the interval between doses [Citation114–116]. Infants who receive an early dose of MMR vaccine should subsequently receive two additional doses according to the recommended national schedule to achieve long-term protection [Citation21,Citation114,Citation117]. Early MMR vaccination was shown to have a favorable safety profile and to protect infants aged 6–14 months against measles during an outbreak in the Netherlands [Citation118].
3.1.5. Epidemiology
To achieve regional elimination goals for measles and rubella and to reduce mumps outbreaks, effective and well-organized surveillance systems are needed [Citation6,Citation9,Citation119]. National vaccination programs should be adapted to the epidemiological profile of the country, which can vary depending on current and past immunization programs (e.g. catch-up campaign following implementation) [Citation40] and on changes in natural immunity [Citation103]. The time of administration of the first and second dose of MMR vaccine may differ between countries, and even within a country, according to the population needs, and there is variation in the literature on the optimal timing. According to the WHO, vaccination against measles should be initiated at 9 months of age in countries where incidence and mortality rates are high in the first year of life, while it can be delayed until 12–15 months of age in countries where infections occur later in life [Citation21]. The age of first vaccination with a measles-containing vaccine should be a compromise between providing protection to young infants and avoiding suboptimal immunity due to interferences with maternal antibodies, i.e. blunting [Citation21,Citation120]. Tailored interventions may also be needed in low-income settings, where mortality and morbidity were shown to decrease following administration of a first dose of measles-containing vaccine at 18 weeks of age, in the presence of maternal antibodies [Citation121,Citation122].
3.2. System weaknesses
3.2.1. Vaccination infrastructure and management
The wide varying quality of vaccination services between and within countries in terms of infrastructure and strengths is one of the remaining challenges to achieve measles and rubella elimination [Citation6,Citation119,Citation123]. The main issues related to immunization service arrangements in low- and middle-income countries include long travel and waiting times, lack of transportation, limited opening hours of vaccination clinics, unwelcoming healthcare environment, sessions not held as planned, lack of commitment by some HCWs, and financial costs of attending vaccination visits (loss of earnings through time off work and travel costs) [Citation1,Citation5,Citation104,Citation124]. In particular, people with limited resources living in rural/inaccessible areas with a weak healthcare system constitute pockets of unvaccinated individuals [Citation5]. In high-income countries, restrictions in healthcare budget (e.g. prevention programs, pharmaceuticals, staff, and equipment) may induce increasing costs of vaccination visits for patients and can also impact vaccination coverage [Citation41,Citation124,Citation125]. In Italy, reductions of the health budget led to a drop in vaccine expenditure and a significant decrease in MMR vaccination coverage, which most likely contributed significantly to the resurgence of measles infections [Citation41]. In the United Kingdom, the declining healthcare workforce and increasing healthcare usage have resulted in restricted access and the ability to make and keep vaccination appointments [Citation125]. In these countries, improvements are needed in terms of number of appointments available for vaccination, in particular after-school appointments, and flexibility in time arrangements [Citation126,Citation127].
Another issue in terms of vaccination management is the unawareness of a proportion of children or their caregivers that recommended vaccinations are due or overdue [Citation128]. The implementation of strategies to remind caregivers of upcoming immunization for their children and to re-engage those whose children are considered overdue for a vaccination (call-recall systems) may increase MMR vaccine uptake in a population by up to 5–20% [Citation128–134]. While call-recall systems are ideally suited to reach caregivers who may forget appointments and encourage hesitant caregivers to vaccinate their child, they are not easy to implement and present several hurdles (e.g. inaccuracy of the recorded patient details and immunization history, unavailability of financial and human resources, patient factors) [Citation128,Citation134]. In some countries, such as the United Kingdom, call-recall strategies are recommended, but their implementation varies from one general practice to the other, showing that clarifications are needed on how to apply these systems in routine [Citation135]. A good strategy is to send an automatic initial invitation and subsequent reminder to caregivers of all children eligible for vaccination, to spend more time to engage those who are more hesitant and harder-to-reach, and to send reminders to caregivers of children who missed vaccinations [Citation134,Citation136]. Therefore, the correct patient contact details should be collected and the vaccination status of children should be tracked [Citation128,Citation137]. Finally, initiatives including routine immunization with monetary incentives were shown to moderately increase immunization rates in low- to middle-income countries [Citation138].
Day care and school entry could also be used to check the immunization status of children, and routine vaccines could be administered during school hours to the identified unvaccinated children [Citation139]. A previous review has shown that among the 174 countries for which data were available, vaccines against measles, mumps, and rubella were provided according to a school-based approach in 41, 36, and 39 countries, respectively [Citation139]. Since school attendance is low in some countries, mainly in poor and rural communities, this approach should be complemented by other activities to reach out-of-school children. In high-income countries, factors influencing the successful implementation of school-based immunization activities included program leadership and governance, organizational models, workforce capacity, and roles (school nurse), communication with caregivers and students, and clinic organization and delivery [Citation140].
3.2.2. Surveillance systems
While surveillance systems belong to the vaccination management, a separate section was added to discuss this important challenge. Indeed, the lack of an appropriate surveillance system can impede measles and rubella elimination [Citation141]. A recent review has identified several challenges related to the determination of vaccination coverage rates (e.g. inaccurate estimates of the target population and number of doses received) and numbers of susceptible people (accounting for immunity from vaccination, natural infection, and passively acquired maternal antibodies) [Citation103]. One solution could be to use geospatial statistical modeling of data from high-quality surveys to monitor vaccination coverage, risk of transmission, and population susceptibility [Citation103]. Case-based or population-based surveillance with laboratory confirmation at the regional or national level is critical for the early detection of outbreaks, the monitoring of elimination processes, the evaluation of outbreak responses, and the identification of unvaccinated or under-vaccinated populations and of geographic areas where vaccination programs require further support [Citation6,Citation141]. As measles outbreaks can occur in countries achieving a sufficient vaccination coverage, surveillance should be implemented at highly localized scales to identify clusters of under-vaccinated persons [Citation40]. Case-based surveillance systems for measles exist in 189 (97%) of 194 countries, but their quality is highly variable [Citation6]. Although rubella and measles surveillance are most often integrated, surveillance is less likely to detect rubella, which is generally a milder disease with subclinical infections in 30–50% of the cases [Citation28]. Therefore, specific surveillance for congenital rubella syndrome should also be implemented [Citation6]. When possible, parallel or consecutive testing for rubella and measles suspected cases could provide useful evidence [Citation6]. Concerning the surveillance of mumps, the most useful action should be to add mumps to the list of notifiable diseases in countries where routine vaccination is performed [Citation4].
3.2.3. Vaccine supply
Due to the complex manufacturing and lengthy control and release processes for each batch of vaccine, it is difficult to respond in the short term to unexpected increases in vaccine demand (e.g. outbreaks), especially for viral vaccines because of the variability in yield of live viral bulk antigen production [Citation113]. Such unpredictable increases in global demand may lead to disruptions in the delivery of certain vaccines [Citation113]. Regulatory requirements and the lack of knowledge/communication of countries’ immunization program plans ahead of time bring additional difficulties in planning and production of vaccines [Citation113]. Local problems with supplies and logistics can also make the vaccine unavailable during a certain period [Citation142]. To allow a more effective short-term response to unexpected increases in vaccine demand, the communication between authorities, scientific experts, and vaccine manufacturers should be improved [Citation113]. Vaccines should be requested regularly, and mechanisms should be available to avoid vaccine shortage due to logistic or supply problems [Citation142].
3.3. Vaccine characteristics
Since the currently available combined trivalent vaccines against M, M, and R are more advanced than monovalent and bivalent vaccines, are widely used, and are recommended in several countries [Citation9–11], this section will specifically focus on their characteristics and their potential improvements.
3.3.1. Primary vaccination failure
Two doses of the MMR vaccine are well-tolerated and effective, with seroresponse rates of approximately 98% against measles, and are currently recommended by the WHO [Citation4,Citation21,Citation83,Citation143–145]. After only one dose of MMR vaccine, primary measles-vaccine failures (failure to seroconvert) may happen in children vaccinated at a young age due to the persistence of maternal antibodies, highlighting the importance of the second dose of MMR vaccine [Citation146]. The proportion of children who do not seroconvert after the first dose of measles-containing vaccine is 10–15% among infants vaccinated at 9 months of age and 5–10% among those vaccinated at 12 months of age [Citation146,Citation147]. Of note, the vast majority of patients with measles during outbreaks had not received the two recommended doses of childhood MMR vaccine [Citation10].
3.3.2. Secondary vaccination failure
Secondary measles vaccine failures (waning and/or incomplete immunity) have also been reported [Citation148]. There is uncertainty concerning the ability of MMR vaccines to provide long-term protection against measles in regions with a low risk of exposure to measles (in the absence of boosting immunity resulting from asymptomatic infections) [Citation149]. The relevance of secondary vaccine failures for measles at the population level is therefore unclear [Citation73,Citation149,Citation150]. For rubella, vaccine-induced immunity is generally assumed to be lifelong, even if antibodies may fall below detectable levels [Citation83].
Secondary mumps vaccine failures have been frequently reported during mumps outbreaks in highly vaccinated populations. Mumps outbreaks mainly affecting vaccinated individuals were reported in several countries, including Australia [Citation35,Citation151], European countries [Citation152], and the USA [Citation3,Citation34,Citation39,Citation153]. Of note, the incidence of severe mumps cases was lower in highly vaccinated populations, suggesting that vaccination still offered protection against severe disease [Citation26,Citation35,Citation39].
Evidence on the long-term effectiveness of one or two doses of MMR vaccines has shown that the levels of antibodies against measles and rubella declined moderately, but were still well above the seropositivity thresholds after 10 years, and anti-mumps antibody levels remained relatively stable [Citation143]. In contrast, another study has shown that 15 years after the second MMR dose, seropositivity rates for measles and rubella have continued to be high, whereas they substantially decreased for mumps [Citation154]. Therefore, concerns of possible waning immunity after MMR vaccination has been proposed as a key contributing factor to mumps resurgence, especially in highly vaccinated populations [Citation3,Citation26,Citation152,Citation155,Citation156]. Waning immunity for measles has been well-established in HIV-infected children, in particular if antiretroviral therapy has been interrupted [Citation88]. For rubella, previous studies have shown that following two doses of MMR vaccine during childhood, the seropositivity rates remained high in young adults but antibody concentrations declined below protective levels in some individuals, which could have implications for the prevention of rubella in pregnancy [Citation157–159].
While secondary vaccination failures are classically measured by humoral immunity, cellular immunity is also important to fully characterize immune responses induced by vaccination. Indeed, cellular immunity against rubella and mumps seems to contribute to disease protection following vaccination [Citation160–165].
3.3.3. Vaccine strains and new genotypes
A total of eight measles clades containing 24 genotypes have been identified [Citation166]. All available measles-containing vaccines belong to clade A, which is not associated with endemic transmission anymore since 2008 [Citation40,Citation166–168]. In the last decade, new measles genotypes have emerged in many parts of the world [Citation40,Citation77] while other genotypes seem to be disappearing [Citation168]. Evidence shows that the emergence of new measles genotypes seems to have a limited impact on vaccine effectiveness as genotypes not belonging to clade A are also neutralized by vaccine-induced antibodies [Citation10]. Moreover, measles cases in previously vaccinated individuals have a limited contribution to virus transmission, and vaccination remains essential to prevent measles outbreaks [Citation169,Citation170].
In contrast, the influence of different genotypes on vaccine effectiveness seems more evident for mumps than for measles. Mumps outbreaks can be caused by waning immunity after vaccination, the presence of unvaccinated individuals, but also differences between circulating genotype variants and the vaccine strains [Citation107]. The mumps strains included in the currently available vaccines belong to genotype A (Jeryl Lynn and RIT 4385 vaccines), to a lesser extent to genotype B (Urabe-AM9 vaccines), or still have to be assigned to a genotype (Leningrad-Zagreb vaccines). In many parts of the world, outbreaks have been attributed to different genotype variants (genotypes I, J, F, G, C, H, K) as compared to the vaccine strains [Citation26,Citation37,Citation107,Citation155,Citation171–181]. In particular, recent outbreaks of mumps caused by the genotype G virus have been reported in several countries, such as the USA, Canada, Norway, Scotland, and Belgium [Citation37,Citation155,Citation177–181]. While differences between vaccine and circulating strains may induce vaccine escape and favor outbreaks, the major cause of mumps outbreaks seems to be the waning immunity after vaccination and contradicting literature exists as to the importance of the mumps genetic variants [Citation107,Citation155]. A previous study has shown that although neutralizing antibody titers were lower against some mumps virus strains than others, genetically diverse strains were neutralized in the sera obtained from children 6 weeks after the MMR vaccine administration [Citation155]. Other studies conducted during mumps outbreaks have shown that the currently used genotype A vaccines provide suboptimal protection against genotype G, even if MMR vaccination was associated with less severe disease [Citation37], and induce significantly lower neutralization titers against wild-type genotypes than against the vaccine strain [Citation177,Citation182]. Moreover, molecular evidence suggested that emergent mumps virus strains have different antigenic epitopes than vaccine strains [Citation183].
Although virus circulation and genetic drift could be limited if high vaccination coverage rates with available vaccines are reached, the molecular epidemiology of circulating strains should be evaluated to determine if new multivalent vaccines are needed. The availability of the whole genome sequencing, reverse vaccinology [Citation184], and platform technologies (including recently developed mRNA platforms [Citation185]) could potentially reduce the time and costs required to develop and produce new vaccines against emerging strains and to prepare vaccine stockpiles that could be deployed in case of outbreaks [Citation186]. While new and more effective MMR vaccines could help to reduce primary and secondary vaccine failure rates [Citation179], the potential need is particularly important for mumps since highly vaccinated populations also experience outbreaks, which may be caused by vaccine escape, mismatch between the vaccine and wild-type virus, and waning immunity. Additional studies are needed to evaluate if new multivalent mumps vaccines should be developed or if a third dose of the existing MMR vaccine would have a stronger effect to avoid mumps outbreaks [Citation107,Citation183].
4. Conclusion
This review shows that many challenges remain in controlling M, M, and R infections despite the availability of effective vaccines. For measles, the remaining challenges in reaching the elimination goal are mainly related to the suboptimal vaccination coverage that creates pockets of unvaccinated or under-vaccinated individuals that fuel outbreaks. For mumps, outbreaks have been reported in highly vaccinated populations and major causes include waning of the immune response and emergence of genotypes not included in the vaccines. Governments and policy-makers should support the implementation of country-specific vaccination policies, raise awareness of disease severity and vaccination impact, improve the provision of vaccination services, health infrastructure, and vaccination access, and strengthen outbreak preparedness. Surveillance systems should be improved to avoid delayed detection of outbreaks and identify any potential escape variants. Additional benefits could come from the evolution of the current MMR vaccines and more tailored vaccination schedules.
5. Expert opinion
The resurgence of measles and mumps in recent years proves that despite two doses of the MMR vaccine being effective and recommended in routine immunization programs for children in several countries, several gaps need to be addressed to eliminate these diseases [Citation7,Citation9,Citation10,Citation28,Citation40,Citation107]. For measles, challenges are mainly related to low immunization coverage that leaves pools of susceptible individuals and to the changing global environment (e.g. increased population density, traveling, and migration) which facilitates disease propagation [Citation40]. Mumps outbreaks have also been reported in vaccinated populations and besides the low vaccination coverage, additional causes of their resurgence include the waning immunity after vaccination and the emergence of new genotypes [Citation107]. A reviewed approach for measles and rubella elimination has been the focus of the recent update of the measles and rubella strategic framework 2021–2030 [Citation7].
Importantly, strategies to improve vaccination coverage and prevent outbreaks should be tailored to the local context of each country. While insufficient funding and sociopolitical issues are important barriers in many low-income countries [Citation5], increases in healthcare usage and declines in healthcare workforce may also contribute to inadequate compliance to completing the vaccination schedule in high-income countries [Citation125]. To increase the proportion of children who receive two MMR vaccine doses, governments should improve access to vaccination clinics, strengthen immunization service arrangements, and ensure availability of vaccines in the long term. Therefore, the number of appointments available for vaccination at healthcare facilities and the flexibility in time arrangements should increase [Citation104,Citation126,Citation127], call-recall systems should be implemented [Citation134–136], and outreach services should be used to immunize children living in rural or inaccessible areas [Citation1]. If additional vaccination points and workforce are needed, government and policy-makers could also authorize additional vaccinators, such as pharmacists, to administer vaccines [Citation187]. In addition to routine childhood immunizations, authorities should also develop clear strategies to immunize unvaccinated older children and adults, such as the organization of catch-up activities [Citation10] or SIAs [Citation188–190]. Of note, as SIAs are costly, their cost-effectiveness versus other immunization strategies should be assessed [Citation188–190]. Catch-up activities are also needed when public health emergencies, such as the current COVID-19 pandemic, are disrupting the implementation of recommended vaccination programs and induce reductions in coverage for several pediatric and adult vaccines, including vaccines against M, M, and R [Citation191]. Specific policies are also needed for the immunization of other pockets of individuals who may play a role in disease transmission if they are unvaccinated or under-vaccinated, such as travelers [Citation66], HCWs [Citation74], and immunocompromised individuals [Citation84–86]. Governments and international organizations (e.g. WHO, United Nations Development Program, and the African Union) should also ensure access to vaccination services for disadvantaged and vulnerable populations, such as migrants and refugees who need catch-up vaccination [Citation68,Citation70]. Immunization programs should also be strengthened in conflict-affected countries and conflict-displaced populations to help rebuild the systems (cold chain and network delivery are often disrupted in case of political threats) and to protect children in the future [Citation94,Citation192].
While very often neglected, high-quality surveillance systems are needed to identify vaccination gaps and to guide M, M, and R control and elimination efforts. Poor surveillance systems can create the false impression of a good disease control, resulting in delayed responses to outbreaks and further spread. Even if surveillance systems to monitor the molecular epidemiology of circulating viruses have a high cost and are time-consuming, they are important and exclusive to identify patterns of transmission, imported viruses, and consequent outbreaks [Citation40], as well as any potential escape variants. Since patterns of measles transmission vary between regions due to differences in demographic context and vaccination programs implementation, countries should carefully analyze local data to adapt strategies [Citation103]. In particular, childhood immunization schedules should be adapted to local epidemiological evidence. Although the second dose (or third dose for early vaccinated children) of measles-containing vaccine is usually provided in the second year of life or at school entry, the timing of its administration varies across countries and regions based on several considerations [Citation10]. It has been previously suggested that increasing the time between the pediatric doses of MMR vaccine to 4–8 years could decrease the risk of mumps outbreaks [Citation193] and improve the durability of protection [Citation35]. Additionally, since immune immaturity affects antibody responses in infants, the second dose given at an older age might induce antibodies of better quality [Citation194]. However, administrating the second dose of the MMR vaccine later in life increases the risk of accumulating a high proportion of children with primary vaccine failure [Citation21,Citation146]. Therefore, another solution could be to administer the second dose of the MMR vaccine in the second year of life [Citation21], followed by a third dose in adolescents/young adults. There is no medical evidence suggesting that a short interval between the two first doses of MMR vaccine (i.e. second dose given in the second year of life) would cause any harm [Citation195]. This schedule (two doses during the first two years of life followed by a third dose) would have several advantages, including a decreased impact of lower immune responses arising from either primary vaccine failure and/or interference with transferred maternal antibodies, as well as a prolongation of the duration of protection. Completing the two-dose schedule earlier in life reduces vaccination failure concerns and the number of second doses missed in countries where their administration is recommended after the standard pediatrician’s follow-up period [Citation196].
To obtain the vaccination coverage rates required to achieve the diseases elimination goals, the wider societal and system challenges should be balanced with changes in public perspectives concerning vaccination. MMR vaccines are victims of their own success as the perceived balance between benefit and risk of vaccination is distorted toward observing fewer severe complications of these infectious diseases after the introduction of MMR vaccines in routine vaccination programs, eventually leading to increased vaccine hesitancy. Parents’, caregivers‘, and even HCWs’ knowledge of the role and safety of MMR vaccines, as well as their understanding of vaccination schedules and operational aspects of immunization service arrangements, should be improved through education [Citation104,Citation197]. Emphasis should be on the responsibility of each person in contributing to public health interventions and protecting individuals and the community, and on the role of vaccines as effective tools to reduce the morbidity and mortality associated with infectious diseases [Citation198]. Educating future healthcare providers on the importance of MMR vaccination and on specific strategies for communicating with vaccine hesitant parents can also be considered [Citation199]. Efforts are also needed to improve the relationship between HCWs and caregivers since many parents, especially in developing countries, have negative views about HCWs and do not contact them to get advice regarding how their children can receive a missed dose [Citation104].
Although somewhat criticized and pushed-back, the introduction of mandatory MMR vaccination for eligible individuals was shown to be a successful intervention in some areas. Although it may be considered as acceptable because caregivers deciding not to vaccinate their child are putting other people’s health at risk, this decision could lead to undecided caregivers becoming more vaccine-resistant and increasing distrust in vaccination programs [Citation52,Citation197]. The SAGE working group on vaccine hesitancy concluded that mandating immunization to address vaccine hesitancy should be done carefully and cautiously as negative consequences could outweigh potential benefits. Therefore, other options should be considered before this step, such as solving infrastructure problems and investigating other obstacles to vaccine uptake [Citation197].
Article highlights
Many challenges remain in controlling measles, mumps, and rubella infections despite the availability of effective vaccines.
These challenges vary considerably between regions and can be related to societal factors, system weaknesses, or vaccine characteristics.
Interventions to address societal challenges include increasing awareness of the potential severity of measles, mumps and rubella, and the role of vaccination, building strategies to target high-risk, unvaccinated and under-vaccinated populations, improving access to vaccination, prioritizing outbreak preparedness, and developing clear vaccination policies.
Interventions to address system weaknesses include improving the provision of vaccination services and health infrastructures, implementing high-quality surveillance and reminder systems, and developing clear action plans for vaccine implementation and long-term supply.
Measures to address the challenges related to vaccine characteristics include, first, the possibility to adapt vaccination schedules to make current vaccines more effective (shorter interval between vaccine doses, administration of a third dose for waning mumps efficacy); then, if needed, the development of new vaccines to target emerging strains.
Declaration of interest
F. Kauffmann, M. O. C. Ota, V. Vetter, and G. Casabona are employees of the GSK group of companies and hold shares in the GSK group of companies. F. Meurice was employed by the GSK group of companies during the manuscript development and holds shares in the GSK group of companies. C. Heffernan works for Public Health England and NHS England advising on how to improve vaccination uptake and coverage of national immunization programs in London. She received no financial compensation from GSK. None of the authors have any non-financial interest to declare. Medical writing support, provided by Claire Verbelen (Modis), and manuscript development and editorial support, provided by Julie Mellery (Modis), were utilized in the production of this manuscript and funded by GlaxoSmithKline Biologicals SA. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Reviewer disclosures
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
Authors’ contributions
All authors contributed to the interpretation of the data and a critical review of the paper for important intellectual content. All authors are in agreement with the content of the final article and have approved it for submission and publication.
Acknowledgments
Authors thank the Modis platform for editorial assistance and manuscript coordination, on behalf of GSK. Claire Verbelen provided medical writing support and Julie Mellery coordinated the manuscript development and editorial support.
Additional information
Funding
References
- World Health Organization. Global measles and rubella strategic plan: 2012-2020. World Health Organization, Geneva, 2012. [cited 2020 Aug 19]. Available from https://apps.who.int/iris/bitstream/handle/10665/44855/9789241503396_eng.pdf?sequence=1
- Keja K, Chan C, Hayden G, et al. Expanded programme on immunization. World health Stat Q. 1988;41(2):59–63.
- Rasheed MAU, Hickman CJ, McGrew M, et al. Decreased humoral immunity to mumps in young adults immunized with MMR vaccine in childhood. Proc Natl Acad Sci U S A. 2019;116(38):19071–19076.
- World Health Organization. Mumps virus vaccines. Wkly Epidemiol Rec. 2007;82(7):51–60.
- World Health Organization. Global vaccine action plan 2011–2020. WHO Library Cataloguing-in-Publication Data. 2013. [cited 2020 Mar 12]. Available from https://www.who.int/immunization/global_vaccine_action_plan/GVAP_doc_2011_2020/en/
- Orenstein WA, Cairns L, Hinman A, et al. Measles and Rubella Global Strategic Plan 2012-2020 midterm review report: background and summary. Vaccine. 2018;36(Suppl 1):A35–A42.
- Measles & Rubella Initiative. The measles and rubella strategic framework 2021-2030. [cited 2021 Jan 6]. Available from https://measlesrubellainitiative.org/measles-rubella-strategic-framework-2021-2030/.
- World Health Organization. Immunization agenda 2030: a global strategy to leave no one behind. [cited 2021 Jan 05]. Available from https://www.who.int/teams/immunization-vaccines-and-biologicals/strategies/ia2030
- Bankamp B, Hickman C, Icenogle JP, et al. Successes and challenges for preventing measles, mumps and rubella by vaccination. Curr Opin Virol. 2019;34:110–116.
- Holzmann H, Hengel H, Tenbusch M, et al. Eradication of measles: remaining challenges. Med Microbiol Immunol. 2016;205(3):201–208.
- World Health Organization. WHO vaccine-preventable diseases: monitoring system. 2019 global summary. [cited 2020 Mar 13]. Available from http://apps.who.int/immunization_monitoring/globalsummary/schedules
- Demicheli V, Rivetti A, Debalini MG, et al. Vaccines for measles, mumps and rubella in children. Cochrane Database Syst Rev. 2012(2):CD004407
- Sood A, Mitra M, Joshi HA, et al. Immunogenicity and safety of a novel MMR vaccine (live, freeze-dried) containing the Edmonston-Zagreb measles strain, the Hoshino mumps strain, and the RA 27/3 rubella strain: results of a randomized, comparative, active controlled phase III clinical trial. Hum Vaccin Immunother. 2017;13(7):1523–1530.
- Hartfield M, Alizon S. Introducing the outbreak threshold in epidemiology. PLoS Pathog. 2013;9(6):e1003277.
- Guerra FM, Bolotin S, Lim G, et al. The basic reproduction number (R 0) of measles: a systematic review. Lancet Infect Dis. 2017;17(12):e420–e428.
- Delamater PL, Street EJ, Leslie TF, et al. Complexity of the basic reproduction number (R0). Emerg Infect Dis. 2019;25(1):1–4.
- Funk S, Knapp JK, Lebo E, et al. Combining serological and contact data to derive target immunity levels for achieving and maintaining measles elimination. BMC Med. 2019;17(1):180.
- Roberts L. Is measles next? Science. 2015;348(6238):958–961, 963.
- Edmunds WJ, Gay NJ, Kretzschmar M, et al. The pre-vaccination epidemiology of measles, mumps and rubella in Europe: implications for modelling studies. Epidemiol Infect. 2000;125(3):635–650.
- Lambert N, Strebel P, Orenstein W, et al. Rubella. Lancet. 2015;385(9984):2297–2307.
- Measles vaccines: WHO position paper – April 2017. Wkly Epidemiol Rec. 2017;92(17):205–227.
- Gavi. Application guidelines: gavi’s support to countries. [cited 2020 Mar 13]. Available from https://www.gavi.org/sites/default/files/document/support/Application%20guidelines%20for%20all%20types%20of%20Gavi%20Support.pdf
- World Health Organization. Immunization coverage. [cited 2021 Jan 06]. Available from https://www.who.int/en/news-room/fact-sheets/detail/immunization-coverage
- Patel MK, Goodson JL, Alexander JP Jr, et al. Progress Toward Regional Measles Elimination — worldwide, 2000–2019. MMWR Morb Mortal Wkly Rep. 2020;69(45):1700–1705. DOI: 10.15585/mmwr.mm6945a6.
- Dabbagh A, Laws RL, Steulet C, et al. Progress Toward Regional Measles Elimination — worldwide, 2000–2017. MMWR Morb Mortal Wkly Rep. 2018;67(47):1323–1329.
- Marshall HS, Plotkin S. The changing epidemiology of mumps in a high vaccination era. Lancet Infect Dis. 2019;19(2):118–119.
- Beleni A-I, Borgmann S. Mumps in the vaccination age: global epidemiology and the situation in Germany. Int J Environ Res Public Health. 2018;15(8):1618.
- Grant GB, Reef SE, Patel M, et al. Progress in Rubella and Congenital Rubella Syndrome Control and Elimination — worldwide, 2000 – 2016. MMWR Morb Mortal Wkly Rep. 2017;66(45):1256–1260 .
- Zimmerman LA, Muscat M, Singh S, et al. Progress toward measles elimination — European region, 2009–2018. MMWR Morb Mortal Wkly Rep. 2019;68(17):396–401. DOI:10.15585/mmwr.mm6817a4.
- Coombes R. Europe steps up action against vaccine hesitancy as measles outbreaks continue. BMJ. 2017;359:j4803.
- Filia A, Bella A, Del Manso M, et al. Ongoing outbreak with well over 4,000 measles cases in Italy from January to end August 2017 − what is making elimination so difficult? Euro Surveill. 2017;22(37):30614.
- Cousins S. Measles: a global resurgence. Lancet Infect Dis. 2019;19(4):362–363.
- Grammens T, Schirvel C, Leenen S, et al. Ongoing measles outbreak in Wallonia, Belgium, December 2016 to March 2017: characteristics and challenges. Euro Surveill. 2017;22(17):30524.
- Lewnard JA, Grad YH. Vaccine waning and mumps re-emergence in the United States. Sci Transl Med. 2018;10(433):eaao5945.
- Westphal DW, Eastwood A, Levy A, et al. A protracted mumps outbreak in Western Australia despite high vaccine coverage: a population-based surveillance study. Lancet Infect Dis. 2019;19(2):177–184.
- Marlow M, Even S, Hoban MT, et al. Universities’ experience with mumps outbreak response and use of a third dose of MMR vaccine. J Am Coll Health. 2021;69(1):53-58.
- Veneti L, Borgen K, Borge KS, et al. Large outbreak of mumps virus genotype G among vaccinated students in Norway, 2015 to 2016. Euro Surveill. 2018;23(38):1700642.
- Aasheim ET, Inns T, Trindall A, et al. Outbreak of mumps in a school setting, United Kingdom, 2013. Hum Vaccin Immunother. 2014;10(8):2446–2449.
- Barskey AE, Schulte C, Rosen JB, et al. Mumps outbreak in Orthodox Jewish communities in the United States. N Engl J Med. 2012;367(18):1704–1713.
- Javelle E, Colson P, Parola P, et al. Measles, the need for a paradigm shift. Eur J Epidemiol. 2019;34(10):897–915.
- Toffolutti V, McKee M, Melegaro A, et al. Austerity, measles and mandatory vaccination: cross-regional analysis of vaccination in Italy 2000–14. Eur J Public Health. 2019;29(1):123–127.
- Chen RT, Moses JM, Markowitz LE, et al. Adverse events following measles—mumps—rubella and measles vaccinations in college students. Vaccine. 1991;9(5):297–299.
- Briss PA, Fehrs LJ, Parker RA, et al. Sustained transmission of mumps in a highly vaccinated population: assessment of primary vaccine failure and waning vaccine-induced immunity. J Infect Dis. 1994;169(1):77–82.
- Hersh BS, Fine PEM, Kent WK, et al. Mumps outbreak in a highly vaccinated population. J Pediatr. 1991;119(2):187–193.
- Robertson SE, Cutts FT, Samuel R, et al. Control of rubella and congenital rubella syndrome (CRS) in developing countries, Part 2: vaccination against rubella. Bull World Health Organ. 1997;75(1):69–80.
- Lassi ZS, Naseem R, Salam RA, et al. The impact of the COVID-19 pandemic on immunization campaigns and programs: a systematic review. Int J Environ Res Public Health. 2021;18(3):988.
- Centers for Disease Control and Prevention. COVID-19’s impact on measles vaccination coverage. Page last reviewed: November 11, 2020. [cited 2021 Mar 09]. Available from https://www.cdc.gov/globalhealth/measles/news/covid-impact-on-measles-vaccination.html
- Green BN, Johnson CD, Adams A. Writing narrative literature reviews for peer-reviewed journals: secrets of the trade. J Chiropr Med. 2006;5(3):101–117.
- World Health Organization. Guide for clinical case management and infection prevention and control during a measles outbreak. License: CC BY-NC-SA 3.0 IGO. [cited 2021 Feb 03]. Available from https://apps.who.int/iris/handle/10665/331599
- Phadke VK, Bednarczyk RA, Salmon DA, et al. Association between vaccine refusal and vaccine-preventable diseases in the United States: a review of measles and pertussis. JAMA. 2016;315(11):1149–1158.
- MacDonald NE. SAGE working group on vaccine hesitancy. vaccine hesitancy: definition, scope and determinants. Vaccine. 2015;33(34):4161–4164.
- World Health Organization. Report of the SAGE Working Group on Vaccine Hesitancy. Oct 2014.[cited 2020 Mar 16]. Available from http://www.who.int/immunization/sage/meetings/2014/october/1_Report_WORKING_GROUP_vaccine_hesitancy_final.pdf
- Larson HJ, Clarke RM, Jarrett C, et al. Measuring trust in vaccination: a systematic review. Hum Vaccin Immunother. 2018;14(7):1599–1609.
- Larson HJ, Jarrett C, Eckersberger E, et al. Understanding vaccine hesitancy around vaccines and vaccination from a global perspective: a systematic review of published literature, 2007–2012. Vaccine. 2014;32(19):2150–2159.
- Brown KF, Kroll JS, Hudson MJ, et al. Factors underlying parental decisions about combination childhood vaccinations including MMR: a systematic review. Vaccine. 2010;28(26):4235–4248.
- Jarrett C, Wilson R, O’Leary M, et al. Strategies for addressing vaccine hesitancy - A systematic review. Vaccine. 2015;33(34):4180–4190.
- Sadaf A, Richards JL, Glanz J, et al. A systematic review of interventions for reducing parental vaccine refusal and vaccine hesitancy. Vaccine. 2013;31(40):4293–4304.
- Montalti M, Kawalec A, Leoni E, et al. Measles immunization policies and vaccination coverage in EU/EEA countries over the last decade. Vaccines (Basel). 2020;8(1):86.
- D’Ancona F, D’Amario C, Maraglino F, et al. The law on compulsory vaccination in Italy: an update 2 years after the introduction. Euro Surveill. 2019;24(26):1900371.
- Torjesen I. German parliament votes to make measles vaccination mandatory. BMJ. 2019;367:l6558.
- Cantor JD. Mandatory measles vaccination in New York City - reflections on a bold experiment. N Engl J Med. 2019;381(2):101–103.
- Yang YT, Studdert DM. Linking immunization status and eligibility for welfare and benefits payments: the Australian “No Jab, No Pay” legislation. JAMA. 2017;317(8):803–804.
- Magurano F, Baggieri M, Mazzilli F, et al. Measles in Italy: viral strains and crossing borders. Int J Infect Dis. 2019;79:199–201.
- Rovida F, Brianese N, Piralla A, et al. Outbreak of measles genotype H1 in Northern Italy originated from a case imported from Southeast Asia, 2017. Clin Microbiol Infect. 2019;25(4):526–528.
- Heywood AE. Measles: a re-emerging problem in migrants and travellers. J Travel Med. 2018;25(1). https://doi.org/10.1093/jtm/tay118.
- European Centre for Disease Prevention and Control. Who is at risk for measles in the EU/EEA? Identifying susceptible groups to close immunity gaps towards measles elimination. Stockholm: ECDC; 2019. [cited 2020 Apr 16]. Available from https://www.ecdc.europa.eu/sites/default/files/documents/RRA-Measles-EU-EEA-May-2019.pdf
- Barnett ED, Walker PF. Role of immigrants and migrants in emerging infectious diseases. Med Clin North Am. 2008;92(6):1447–1458.
- Mipatrini D, Stefanelli P, Severoni S, et al. Vaccinations in migrants and refugees: a challenge for European health systems. A systematic review of current scientific evidence. Pathog Glob Health. 2017;111(2):59–68.
- Abbas M, Aloudat T, Bartolomei J, et al. Migrant and refugee populations: a public health and policy perspective on a continuing global crisis. Antimicrob Resist Infect Control. 2018;7:113.
- Hargreaves S, Nellums LB, Ravensbergen SJ, et al. Divergent approaches in the vaccination of recently arrived migrants to Europe: a survey of national experts from 32 countries, 2017. Euro Surveill. 2018;23(41):1700772.
- George F, Valente J, Augusto GF, et al. Measles outbreak after 12 years without endemic transmission, Portugal, February to May 2017. Euro Surveill. 2017;22(23):30548.
- Currie J, Davies L, McCarthy J, et al. Measles outbreak linked to European B3 outbreaks, Wales, United Kingdom, 2017. Euro Surveill. 2017;22(42):17–00673.
- Hahné SJM, Nic Lochlainn LM, Van Burgel ND, et al. Measles outbreak among previously immunized healthcare workers, the Netherlands, 2014. J Infect Dis. 2016;214(12):1980–1986.
- Fiebelkorn AP, Seward JF, Orenstein WA. A global perspective of vaccination of healthcare personnel against measles: systematic review. Vaccine. 2014;32(38):4823–4839.
- Botelho-Nevers E, Cassir N, Minodier P, et al. Measles among healthcare workers: a potential for nosocomial outbreaks. Euro Surveill. 2011;16(2):19764.
- Haviari S, Benet T, Saadatian-Elahi M, et al. Vaccination of healthcare workers: a review. Hum Vaccin Immunother. 2015;11(11):2522–2537.
- Melenotte C, Zandotti C, Gautret P, et al. Measles: is a new vaccine approach needed? Lancet Infect Dis. 2018;18(10):1060–1061.
- European Centre for Disease Prevention and Control. Review of outbreaks and barriers to MMR vaccination coverage among hard-to-reach populations in Europe. Stockholm: ECDC; 2013. [cited 2020 Feb 26]. Available from https://www.ecdc.europa.eu/sites/default/files/media/en/publications/Publications/MMR-vaccination-hard-to-reach-population-review-2013.pdf
- Maltezou HC, Theodoridou K, Ledda C, et al. Vaccination of healthcare workers: is mandatory vaccination needed? Expert Rev Vaccines. 2019;18(1):5–13.
- Bianchi FP, Mascipinto S, Stefanizzi P, et al. Prevalence and management of measles susceptibility in healthcare workers in Italy: a systematic review and meta-analysis. Expert Rev Vaccines. 2020;19(7):611–620.
- Kopsidas J, Maroudi S, Tsopela GC, et al. Increasing healthcare workers’(HCW) uptake of seasonal influenza vaccination (SIV) in a tertiary pediatric hospital in Greece (ESP17-0994). 35th Annual Meeting of the European Society for Paediatric Infectious Diseases (ESPID 2017), Madrid, Spain, May 23-27, 2017.
- Hiller U, Mankertz A, Köneke N, et al. Hospital outbreak of measles - Evaluation and costs of 10 occupational cases among healthcare worker in Germany, February to March 2017. Vaccine. 2019;37(14):1905–1909.
- Rubella vaccines: WHO position paper. Wkly Epidemiol Rec. 2011;86(29):301–316.
- Centers for Disease Control and Prevention. Recommendations of the Advisory Committee on Immunization Practices (ACIP): use of vaccines and immune globulins for persons with altered immunocompetence. MMWR Recomm Rep. 1993;42(RR–4):1–18.
- Menson EN, Mellado MJ, Bamford A, et al. Guidance on vaccination of HIV-infected children in Europe. HIV Med. 2012;13(6):333–336.
- Summary of product characteristics PRIORIX. [cited 2020 Jul 16]. Available from https://gskpro.com/content/dam/global/hcpportal/en_MT/PDF/Homepage/Products/productlisting/priorix/PRIORIX-SPC-PL-PIL-JUN15.pdf
- Mutsaerts EAML, Nunes MC, Bhikha S, et al. Immunogenicity and safety of an early measles vaccination schedule at 6 and 12 months of age in human immunodeficiency virus (HIV)-unexposed and HIV-exposed, uninfected South African children. J Infect Dis. 2019;220(9):1529–1538.
- Mutsaerts EAML, Nunes MC, Van Rijswijk MN, et al. Measles immunity at 4.5 years of age following vaccination at 9 and 15-18 months of age among human immunodeficiency virus (HIV)-infected, HIV-exposed-uninfected, and HIV-unexposed children. Clin Infect Dis. 2019;69(4):687–696.
- Angel JB, Walpita P, Lerch RA, et al. Vaccine-associated measles pneumonitis in an adult with AIDS. Ann Intern Med. 1998;129(2):104–106.
- Duncan CJA, Mohamad SMB, Young DF, et al. Human IFNAR2 deficiency: lessons for antiviral immunity. Sci Transl Med. 2015;7(307):307ra154.
- Hambleton S, Goodbourn S, Young DF, et al. STAT2 deficiency and susceptibility to viral illness in humans. Proc Natl Acad Sci U S A. 2013;110(8):3053–3058.
- Grundy J, Biggs BA. The impact of conflict on immunisation coverage in 16 countries. Int J Health Policy Manag. 2019;8(4):211–221.
- Page KR, Doocy S, Reyna Ganteaume F, et al. Venezuela’s public health crisis: a regional emergency. Lancet. 2019;393(10177):1254–1260.
- Nandy R Immunization under fire. Unicef Connect. 2016 Apr 25. [cited 2020 Feb 24]. Available from https://blogs.unicef.org/blog/immunization-under-fire/
- Orenstein WA, Hinman A, Nkowane B, et al. Measles and rubella global strategic plan 2012–2020 midterm review. 2016. [cited 2020 Mar 16]. Available from http://www.who.int/immunization/sage/meetings/2016/october/1_MTR_Report_Final_Color_Sept_20_v2.pdf?ua=1
- Peyraud N, Quere M, Duc G, et al. A post-conflict vaccination campaign, Central African Republic. Bull World Health Organ. 2018;96(8):540–547.
- Gavi. On the frontline: gavi’s support to fragile states. [cited 2020 Mar 16]. Available from https://www.gavi.org/news/media-room/frontline-gavis-support-fragile-states
- World Health Organization. Maintaining essential health services: operational guidance for the COVID-19 context interim guidance. [cited 2020 Jul 06]. Available from https://www.who.int/publications/i/item/covid-19-operational-guidance-for-maintaining-essential-health-services-during-an-outbreak
- Bramer CA, Kimmins LM, Swanson R, et al. Decline in child vaccination coverage during the COVID-19 pandemic - Michigan Care Improvement Registry, May 2016-May 2020. MMWR Morb Mortal Wkly Rep. 2020;69(20):630–631.
- Saxena S, Skirrow H, Bedford H. Routine vaccination during covid-19 pandemic response. BMJ. 2020;369:m2392.
- Marziano V, Poletti P, Trentini F, et al. Parental vaccination to reduce measles immunity gaps in Italy. Elife. 2019;8:e44942.
- Portnoy A, Jit M, Helleringer S, et al. Impact of measles supplementary immunization activities on reaching children missed by routine programs. Vaccine. 2018;36(1):170–178.
- Cutts FT, Dansereau E, Ferrari MJ, et al. Using models to shape measles control and elimination strategies in low- and middle-income countries: a review of recent applications. Vaccine. 2020;38(5):979–992.
- Zewdie A, Letebo M, Mekonnen T. Reasons for defaulting from childhood immunization program: a qualitative study from Hadiya zone, Southern Ethiopia. BMC Public Health. 2016;16(1):1240.
- Immunization Unit of the Pan American Health Organization. Measles elimination: field guide. Washington, D.C.: PAHO, 2005. (Scientific and Technical Publication No. 605). [cited 2020 Feb 26]. Available from http://new.paho.org/hq/dmdocuments/2010/FieldGuide_Measles_2ndEd_e.pdf
- Orenstein WA, Strebel PM, Papania M, et al. Measles eradication: is it in our future? Am J Public Health. 2000;90(10):1521–1525.
- Principi N, Esposito S. Mumps outbreaks: a problem in need of solutions. J Infect. 2018;76(6):503–506.
- Plotkin SA. Mumps: a Pain in the Neck. J Pediatric Infect Dis Soc. 2018;7(2):91–92.
- Cardemil CV, Dahl RM, James L, et al. Effectiveness of a third dose of MMR vaccine for mumps outbreak control. N Engl J Med. 2017;377(10):947–956.
- Center for Disease Control and Prevention (CDC). CDC guidance for public health authorities on use of a 3rd dose of MMR vaccine during mumps outbreaks. [cited 2019 Nov 21]. Available from https://www.cdc.gov/mumps/health-departments/MMR3.html
- Kaaijk P, Wijmenga-Monsuur AJ, Van Houten MA, et al. A third dose of measles-mumps-rubella vaccine to improve immunity against mumps in young adults. J Infect Dis. 2020;221(6):902–909.
- Fiebelkorn AP, Coleman LA, Belongia EA, et al. Mumps antibody response in young adults after a third dose of measles-mumps-rubella vaccine. Open Forum Infect Dis. 2014;1(3):ofu094.
- Vaccines Europe. From vaccines shortages to sustainable vaccine supply: vaccines Europe Position. [cited 2019 Nov 20]. Available from http://www.vaccineseurope.eu/wp-content/uploads/2016/06/VE-Paper_shortagesFIN-2.pdf
- Brinkman ID, De Wit J, Smits GP, et al. Early measles vaccination during an outbreak in the Netherlands: short-term and long-term decreases in antibody responses among children vaccinated before 12 months of age. J Infect Dis. 2019;220(4):594–602.
- Vashishtha VM, Yewale VN, Bansal CP, et al. IAP perspectives on measles and rubella elimination strategies. Indian Pediatr. 2014;51(9):719–722.
- Javed N, Saqib MAN, Hassan Bullo MM, et al. Seroprevalence of transplacentally acquired measles antibodies in unvaccinated infants at nine months of age and its relation to the feeding practices. BMC Infect Dis. 2019;19(1):587.
- Principi N, Esposito S. Early vaccination: a provisional measure to prevent measles in infants. Lancet Infect Dis. 2019;19(11):1157–1158.
- van der Maas, Woudenberg T, Hahne SJ, et al. Tolerability of early measles-mumps-rubella vaccination in infants aged 6–14 months during a measles outbreak in the Netherlands in 2013–2014. J Infect Dis. 2016;213(9):1466–1471.
- Hardt K, Bonanni P, King S, et al. Vaccine strategies: optimising outcomes. Vaccine. 2016;34(52):6691–6699.
- Hansen JS, Thysen SM, Rodrigues A, et al. Is early measles vaccination associated with stronger survival benefits than later measles vaccination? BMC Public Health. 2018;18(1):984.
- Do VA, Biering-Sorensen S, Fisker AB, et al. Effect of an Early Dose of Measles Vaccine on Morbidity Between 18 Weeks and 9 Months of Age: a Randomized, Controlled Trial in Guinea-Bissau. J Infect Dis. 2017;215(8):1188–1196.
- Smits G, Stabell Benn C, Whittle H, et al. Maternal measles antibodies and their influence on all-cause mortality following measles vaccination: an alternative to measure very low maternal antibody levels. Clin Infect Dis. 2019;68(10):1758–1760.
- Esposito S, Principi N, Cornaglia G. Barriers to the vaccination of children and adolescents and possible solutions. Clin Microbiol Infect. 2014;20:25–31.
- Expert Panel on effective ways of investing in Health (EXPH). Preliminary report on vaccination programmes and health systems in Europe, 2018 Sept 26. [cited 2020 Aug 17]. Available from https://ec.europa.eu/health/sites/health/files/expert_panel/docs/020_vaccinationpgms_en.pdf
- The King’s Fund. Understanding pressures in general practice. 2016. [cited 2020 Feb 27]. Available from https://www.kingsfund.org.uk/sites/default/files/field/field_publication_file/Understanding-GP-pressures-Kings-Fund-May-2016.pdf
- Wighton K Access to children’s GP appointments linked to use of emergency departments. Imperial College London News. Published on 2016 Jan 20. [cited 2020 Feb 27]. Available from https://www.imperial.ac.uk/news/170333/access-childrens-gp-appointments-linked-emergency/
- The National Institute for Health and Care Excellences (NICE). Quality standard topic: vaccine uptake in under 19s. Date of Quality Standards Advisory Committee meeting. 2016 Jun 29th. [cited 2020 Feb 27]. Available from https://www.nice.org.uk/guidance/qs145/documents/briefing-paper
- Pereira JA, Quach S, Heidebrecht CL, et al. Barriers to the use of reminder/recall interventions for immunizations: a systematic review. BMC Med Inform Decis Mak. 2012;12:145.
- Chachou MJ, Mukinda FK, Motaze V, et al. Electronic and postal reminders for improving immunisation coverage in children: protocol for a systematic review and meta-analysis. BMJ Open. 2015;5(10):e008310.
- Betsch C, Böhm R, Chapman GB. Using behavioral insights to increase vaccination policy effectiveness. Policy Insights Behav Brain Sci. 2015;2(1):61–73.
- Harvey H, Reissland N, Mason J. Parental reminder, recall and educational interventions to improve early childhood immunisation uptake: a systematic review and meta-analysis. Vaccine. 2015;33(25):2862–2880.
- Jacobson Vann JC, Szilagyi P. Patient reminder and patient recall systems to improve immunization rates. Cochrane Database Syst Rev. 2005;3:Cd003941.
- Williams N, Woodward H, Majeed A, et al. Primary care strategies to improve childhood immunisation uptake in developed countries: systematic review. JRSM Short Reports. 2011;2(10):81. DOI:10.1258/shorts.2011.011112.
- NHS England. Optimising your invite-reminder systems for childhood immunisations. 2017. [cited 2019 Feb 12]. Available from https://www.england.nhs.uk/london/wp-content/uploads/sites/8/2018/06/Good-Practice-Immunisation-Invite-Reminder-Guide.pdf
- Lonergan K, Latif A, Beaney T, et al. Implementing the evidence: are call/recall systems for immunisations feasible in general practice? Vaccine Immunol Open Access J. 2018;1:11–15.
- Crocker-Buque T, Edelstein M, Mounier-Jack S. Interventions to reduce inequalities in vaccine uptake in children and adolescents aged 19 years: a= systematic=/19years. J Epidemiol Community Health. 2017;71(1):87–97.
- Oyo-Ita A, Wiysonge CS, Oringanje C, et al. Interventions for improving coverage of childhood immunisation in low- and middle-income countries. Cochrane Database Syst Rev. 2016;7(7):CD008145.
- Driessen J, Olson ZD, Jamison DT, et al. Comparing the health and social protection effects of measles vaccination strategies in Ethiopia: an extended cost-effectiveness analysis. Soc Sci Med. 2015;139:115–122.
- Vandelaer J, Olaniran M. Using a school-based approach to deliver immunization—Global update. Vaccine. 2015;33(5):719–725.
- Perman S, Turner S, Ramsay AIG, et al. School-based vaccination programmes: a systematic review of the evidence on organisation and delivery in high income countries. BMC Public Health. 2017;17(1):252.
- Patel MK, Gibson R, Cohen A, et al. Global landscape of measles and rubella surveillance. Vaccine. 2018;36(48):7385–7392.
- Mokdad AH, Gagnier MC, Colson KE, et al. Missed opportunities for measles, mumps, and rubella (MMR) immunization in Mesoamerica: potential impact on coverage and days at risk. PLoS One. 2015;10(10):e0139680.
- Carryn S, Feyssaguet M, Povey M, et al. Long-term immunogenicity of measles, mumps and rubella-containing vaccines in healthy young children: a 10-year follow-up. Vaccine. 2019;37(36):5323–5331.
- Gillet Y, Steri GC, Behre U, et al. Immunogenicity and safety of measles-mumps-rubella-varicella (MMRV) vaccine followed by one dose of varicella vaccine in children aged 15 months–2 years or 2–6 years primed with measles-mumps-rubella (MMR) vaccine. Vaccine. 2009;27(3):446–453.
- Johnson CE, Kumar ML, Whitwell JK, et al. Antibody persistence after primary measles-mumps-rubella vaccine and response to a second dose given at four to six vs. eleven to thirteen years. Pediatr Infect Dis J. 1996;15(8):687–692.
- Nic Lochlainn LM, De Gier B, Van Der Maas N, et al. Immunogenicity, effectiveness, and safety of measles vaccination in infants younger than 9 months: a systematic review and meta-analysis. Lancet Infect Dis. 2019;19(11):1235–1245.
- World Health Organization. WHO immunological basis for immunization series. Module 7: Measles. Update 2020. [cited 2021 Feb 12]. Available from https://apps.who.int/iris/bitstream/handle/10665/331533/9789241516655-eng.pdf?ua=1
- Paunio M, Hedman K, Davidkin I, et al. Secondary measles vaccine failures identified by measurement of IgG avidity: high occurrence among teenagers vaccinated at a young age. Epidemiol Infect. 2000;124(2):263–271.
- Haralambieva IH, Kennedy RB, Ovsyannikova IG, et al. Current perspectives in assessing humoral immunity after measles vaccination. Expert Rev Vaccines. 2019;18(1):75–87.
- Bitzegeio J, Majowicz S, Matysiak-Klose D, et al. Estimating age-specific vaccine effectiveness using data from a large measles outbreak in Berlin, Germany, 2014/15: evidence for waning immunity. Euro Surveill. 2019;24(17):1800529.
- Bangor‐Jones RD, Dowse GK, Giele CM, et al. A prolonged mumps outbreak among highly vaccinated Aboriginal people in the Kimberley region of Western Australia. Med J Aust. 2009;191(7):398–401.
- Vygen S, Fischer A, Meurice L, et al. Waning immunity against mumps in vaccinated young adults, France 2013. Euro Surveill. 2016;21(10):30156.
- Livingston KA, Rosen JB, Zucker JR, et al. Mumps vaccine effectiveness and risk factors for disease in households during an outbreak in New York City. Vaccine. 2014;32(3):369–374.
- Davidkin I, Jokinen S, Broman M, et al. Persistence of measles, mumps, and rubella antibodies in an MMR-vaccinated cohort: a 20-year follow-up. J Infect Dis. 2008;197(7):950–956.
- Rubin SA, Link MA, Sauder CJ, et al. Recent mumps outbreaks in vaccinated populations: no evidence of immune escape. J Virol. 2012;86(1):615–620.
- Hamami D, Cameron R, Pollock KG, et al. Waning immunity is associated with periodic large outbreaks of mumps: a mathematical modeling study of Scottish data. Front Physiol. 2017;8:233.
- Crooke SN, Haralambieva IH, Grill DE, et al. Seroprevalence and durability of rubella virus antibodies in a highly immunized population. Vaccine. 2019;37(29):3876–3882.
- Davidkin I, Peltola H, Leinikki P, et al. Duration of rubella immunity induced by two-dose measles, mumps and rubella (MMR) vaccination A 15-year follow-up in Finland. Vaccine. 2000;18(27):3106–3112.
- Kung W-J, Shih C-T, Shih Y-L, et al. Faster waning of the rubella-specific immune response in young pregnant women immunized with MMR at 15 months. Am J Reprod Immunol. 2020;84(4):e13294.
- Hanna‐Wakim R, Yasukawa LL, Sung P, et al. Immune responses to mumps vaccine in adults who were vaccinated in childhood. J Infect Dis. 2008;197(12):1669–1675.
- Hyöty H, Räsänen L, Lehto M, et al. Cell-mediated and humoral immunity to mumps virus antigen. Acta Pathol Microbiol Immunol Scand C. 1986;94(5):201–206.
- Terada K, Hagihara K, Oishi T, et al. Cellular and humoral immunity after vaccination or natural mumps infection. Pediatr Int. 2017;59(8):885–890.
- Vandermeulen C, Clement F, Roelants M, et al. Evaluation of cellular immunity to mumps in vaccinated individuals with or without circulating antibodies up to 16 years after their last vaccination. J Infect Dis. 2009;199(10):1457–1460.
- Lambert ND, Haralambieva IH, Ovsyannikova IG, et al. Characterization of humoral and cellular immunity to rubella vaccine in four distinct cohorts. Immunol Res. 2014;58(1):1–8.
- O’Shea S, Best JM, Banatvala JE. Viremia, Virus excretion, and antibody responses after challenge in volunteers with low levels of antibody to rubella virus. J Infect Dis. 1983;148(4):639–647.
- Center for Disease Control and Prevention (CDC). Measles. Genetic analysis of measles virus. Lab Tools. Page last reviewed: 2020 Nov 5. [cited 2021 Mar 09]. Available from https://www.cdc.gov/measles/lab-tools/genetic-analysis.html
- Bellini WJ. Genetic diversity of wild-type measles viruses: implications for global measles elimination programs. Emerg Infect Dis. 1998;4(1):29–35.
- Rota PA, Brown K, Mankertz A, et al. Global distribution of measles genotypes and measles molecular epidemiology. J Infect Dis. 2011;204suppl 1:S514–523.
- Gastañaduy PA, Funk S, Lopman BA, et al. Factors associated with measles transmission in the United States during the postelimination era. JAMA Pediatr. 2020;174(1):56–62.
- Pacenti M, Maione N, Lavezzo E, et al. Measles virus infection and immunity in a suboptimal vaccination coverage setting. Vaccines (Basel). 2019;7(4):199.
- Cui A, Zhu Z, Hu Y, et al. Mumps epidemiology and mumps virus genotypes circulating in Mainland China during 2013-2015. PLoS One. 2017;12(1):e0169561.
- Gouma S, Sane J, Gijselaar D, et al. Two major mumps genotype G variants dominated recent mumps outbreaks in the Netherlands (2009–2012). J Gen Virol. 2014;95(5):1074–1082.
- Hindiyeh MY, Aboudy Y, Wohoush M, et al. Characterization of large mumps outbreak among vaccinated Palestinian refugees. J Clin Microbiol. 2009;47(3):560–565.
- Malayan J, Warrier A, Ramanan PV, et al. Unnoticeable mumps infection in India: does MMR vaccine protect against circulating mumps virus genotype C? Int J Biotechnol Bioeng. 2012;6:1011–1017.
- Park SH. Resurgence of mumps in Korea. Infection & Chemotherapy. 2015;47(1):1–11.
- Peltola H, Kulkarni PS, Kapre SV, et al. Mumps outbreaks in Canada and the United States: time for new thinking on mumps vaccines. Clin Infect Dis. 2007;45(4):459–466.
- Vermeire T, Barbezange C, Francart A, et al. Sera from different age cohorts in Belgium show limited cross-neutralization between the mumps vaccine and outbreak strains. Clin Microbiol Infect. 2019;25(7):907.e901–907.e906.
- Willocks LJ, Guerendiain D, Austin HI, et al. An outbreak of mumps with genetic strain variation in a highly vaccinated student population in Scotland. Epidemiol Infect. 2017;145(15):3219–3225.
- Dayan GH, Rubin S. Mumps outbreaks in vaccinated populations: are available mumps vaccines effective enough to prevent outbreaks? Clin Infect Dis. 2008;47(11):1458–1467.
- L’Huillier AG, Eshaghi A, Racey CS, et al. Laboratory testing and phylogenetic analysis during a mumps outbreak in Ontario, Canada. Virol J. 2018;15(1):98.
- Wei Y, Wilkinson K, Rusk R, et al. Large community mumps outbreak in Manitoba, Canada, September 2016–December 2018. Canada Commun Dis Rep. 2020;46(4):70–76.
- Gouma S, Ten Hulscher HI, Schurink-van ‘T Klooster TM, et al. Mumps-specific cross-neutralization by MMR vaccine-induced antibodies predicts protection against mumps virus infection. Vaccine. 2016;34(35):4166–4171. DOI:10.1016/j.vaccine.2016.06.063.
- May M, Rieder CA, Rowe RJ. Emergent lineages of mumps virus suggest the need for a polyvalent vaccine. Int J Infect Dis. 2018;66:1–4.
- Rappuoli R. Reverse vaccinology, a genome-based approach to vaccine development. Vaccine. 2001;19(17–19):2688–2691.
- Corbett KS, Edwards DK, Leist SR, et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature. 2020;586(7830):567–571.
- Gilbert SC, Warimwe GM. Rapid development of vaccines against emerging pathogens: the replication-deficient simian adenovirus platform technology. Vaccine. 2017;35(35):4461–4464.
- Poudel A, Lau ETL, Deldot M, et al. Pharmacist role in vaccination: evidence and challenges. Vaccine. 2019;37(40):5939–5945.
- Bishai D, Johns B, Nair D, et al. The cost-effectiveness of supplementary immunization activities for measles: a stochastic model for Uganda. J Infect Dis. 2011;204Suppl 1(Suppl 1):S107–S115.
- Dabral M. Cost-effectiveness of supplementary immunization for measles in India. Indian Pediatr. 2009;46(11):957–962.
- Kaucley L, Levy P. Cost-effectiveness analysis of routine immunization and supplementary immunization activity for measles in a health district of Benin. Cost Eff Resour Alloc. 2015;13:14.
- World Health Organization. Mitigating the impact of COVID-19 on control of vaccine-preventable diseases: a health risk management approach focused on catch-up vaccination. Copenhagen: WHO Regional Office for Europe; 2020. Licence: CC BY-NC-SA 3.0 IGO. [cited 2021 Mar 03]. Available from https://apps.who.int/iris/bitstream/handle/10665/334248/WHO-EURO-2020-1086-40832-55187-eng.pdf?sequence=1&isAllowed=y
- Senessie C, Gage GN, Von Elm E. Delays in childhood immunization in a conflict area: a study from Sierra Leone during civil war. Conflict and Health. 2007;1:14.
- Eriksen J, Davidkin I, Kafatos G, et al. Seroepidemiology of mumps in Europe (1996–2008): why do outbreaks occur in highly vaccinated populations? Epidemiol Infect. 2013;141(3):651–666.
- Andersson U, Bird G, Britton S. A sequential study of human B lymphocyte function from birth to two years of age. Acta Paediatr Scand. 1981;70(6):837–842.
- Herrera OR, Thornton TA, Helms RA, et al. MMR vaccine: when is the right time for the second dose? J Pediatr Pharmacol Ther. 2015;20(2):144–148.
- Braeckman T, Theeten H, Roelants M, et al. Can Flanders resist the measles outbreak? Assessing vaccination coverage in different age groups among Flemish residents. Epidemiol Infect. 2018;146(8):1043–1047.
- Draeger E, Bedford HE, Elliman DAC. Should measles vaccination be compulsory? BMJ. 2019;365:l2359.
- Dubé E, Laberge C, Guay M, et al. Vaccine hesitancy: an overview. Hum Vaccines Immunother. 2013;9(8):1763–1773.
- Schnaith AM, Evans EM, Vogt C, et al. An innovative medical school curriculum to address human papillomavirus vaccine hesitancy. Vaccine. 2018;36(26):3830–3835.