Antimicrobial drug resistance, partly due to inappropriate use of antimicrobials, is a global health problem that causes high morbidity and mortality and inflicts enormous economic burden worldwide [Citation1]. In 2013, the estimated yearly cost related to the United States of America’s antimicrobial resistance was 55 USD billion, causing around 23,000 annual deaths [Citation2]. In the era of COVID-19, antimicrobial resistance poses an additional challenge to the therapeutic medicine, particularly in the vulnerable elderly population [Citation3]. A review published in the August 2020 of ‘Expert Review of Anti-Infective Therapy’ has highlighted the importance of incorporating antimicrobial stewardship programs in medical and allied health professional curricula [Citation4]. The authors have fittingly emphasized the necessity of providing education and training to future physicians, nurses, and other allied health professionals to increase the awareness of antimicrobial resistance [Citation4]. Recent publications have also elaborated on how antimicrobial stewardship programs are likely to equip future medical professionals to improve their understanding of antibiotics’ rational use to reduce antimicrobial resistance and improve patients’ overall outcome [Citation3,Citation4].
The antimicrobial stewardship is an integrated and multidisciplinary approach to select appropriate drugs for appropriate patients for a proper duration to minimize the risk of developing antimicrobial resistance [Citation5]. Furthermore, enhanced surveillance to implement prescribing guidelines and providing continuing medical education has been encouraged in the stewardship program. Many institutes have started to introduce the antimicrobial stewardship programs for acute care facilities to safeguard the proper use of antimicrobials to avoid eventual adverse effects. Of policy-making importance, the requirement of antimicrobial stewardship programs is being appropriated by the Centers for Disease Control and Prevention (C.D.C.). In 2015, the U.S. National Action Plan provided a five-year plan to decrease antibiotic resistance by (1) encouraging innovation and research for generating new prevention strategies, (2) increasing and enhancing surveillance, and (3) adopting evidence-based stewardship strategies [Citation6]. The main goals of the ‘National Action Plan’ were to reduce the alarming rise of resistant bacteria and to slow down the spread of resistant infections. To combat antimicrobial resistance, the emphasis was given to focus on strengthening national health surveillance. The use of innovative diagnostic tests for rapid identification and characterization of resistant bacteria was another goal set by the ‘National Action Plan.’ The importance of basic and applied research for developing new therapeutics and vaccines, and improving international collaboration was part of the ‘National Action Plan’ goals to minimize the development of antimicrobial resistance [Citation6]. In the 2016 fiscal year, U.S. Congress provided 160 USD million to C.D.C. to achieve the goals set by the ‘National Action Plan.’ To continue fighting against the antimicrobial resistance, the funding was increased to 170 USD million in the fiscal 2020 year, signifying the continued U.S. governmental priority to reduce the health burden due to antimicrobial resistance.
Antibiotics are highly prescribed drugs in clinical practice, and it is estimated that around 50% of antibiotic use in the hospitals (in both the outpatient and inpatient) is not prescribed correctly, either not necessary or are not optimally effective as recommended to the patients [Citation7]. After implementing antimicrobial stewardship programs in the emergency department, a markedly reduced number of unnecessary antimicrobial prescriptions (25.6% before the program vs. 17.4% after the program) were found in the outpatient [Citation8]. Such improper clinical use of antimicrobial agents resulted in increased resistance with higher numbers of deaths from treatable and curable infections before the emergence of resistance [Citation9,Citation10]. For example, the conventional treatment for the sexually transmitted disease gonorrhea is azithromycin and ceftriaxone. However, due to antimicrobial resistance, gonorrhea is becoming a difficult disease to treat, and the C.D.C. categorized it as an ‘urgent threat’ with the likelihood of becoming more prevalent [Citation11,Citation12]. For the clinicians needing updated information to prescribe antibiotics rationally, the World Health Organization (WHO) and Europe jointly launched a free online course accessible via the OpenWHO platform [Citation13]. Besides improving knowledge, attitude, and practices regarding antimicrobial resistance, WHO has also established a curricular guide for the health workers [Citation14]. As explained above, antimicrobial stewardship programs are becoming vital to clinical medicine, particularly in subspecialties like infectious diseases and medical microbiology, emphasizing competency-based medical education. Many of the current educational strategies devoted to antimicrobial stewardship programs are targeted toward practicing physicians.
In the hospital setting, the junior level resident physicians are responsible for providing patient care, usually prescribe the highest numbers of antimicrobials [Citation15]. Despite such an active role in prescribing antimicrobial agents, there appears to be inadequate training that affects the confidence among the junior level physicians while prescribing antimicrobials [Citation16]. Studies have found that online-spaced education is a useful tool that can improve junior doctors’ understanding of infectious diseases and enhance their confidence in antimicrobial prescription [Citation17]; however, it is important to note that antimicrobial stewardship programs should be incorporated into undergraduate medical and dental school course curricula for better and greater future benefits. In many countries, medical and other allied health professional curricula, a clinical pharmacology course does not adequately address the varied aspects of safe antimicrobial prescriptions [Citation18]. Theoretical education and hands-on training are fundamental in implementing successful antimicrobial stewardship programs. Future health professionals (medicine, nursing, pharmacy, dentistry, midwifery, and veterinary medicine) need to be aware of the misuse and overuse of antimicrobial therapy from their undergraduate level studies. Forums, seminars, and workshops need to be offered to the students to understand the rationalization of antimicrobial prescriptions. Incorporating applied aspects of clinical pharmacology in a modified curriculum and implementing such training in medical students’ clinical years will reduce the misuse of antimicrobial agents [Citation14,Citation19]. The pedagogic instruction in sensible antimicrobial prescribing skills should be developed among the future medical professionals, while still students. In the U.K., antimicrobial stewardship principles are incorporated in many undergraduate healthcare and veterinary degree programs in the U.K. [Citation20]. However, finetuning the curriculum and national standardization will strengthen the antimicrobial stewardship programs.
Various academic institutes in the U.S.A. incorporate antimicrobial stewardship programs to teach their healthcare professional students to precisely identify the patient cohort who will need antimicrobial therapy. This is to ensure that they receive the ideal choice of antimicrobial agents with optimal dose duration to minimize the emergence of antimicrobial resistance (). For instance, the Wake Forest School of Medicine of North Carolina, in collaboration with the C.D.C. and the Association of American Medical Colleges (AAMC) has introduced the ‘Antibiotic Stewardship Curriculum’ for medical students to enhance their understanding of antimicrobial resistance, prevention, and control. The curriculum for the medical students is comprised of (1) three didactic lectures (with facilitator notes and audio recordings) with nine corresponding examination questions and (2) five small group activities (facilitator guided). The students’ didactic lectures are allotted in the pre-clinical years while the small group activities are conducted during their clinical clerkship years [Citation21]. Again, the need for the new curriculum arises from the fact that the existing allied health professional curricula resulted in low levels of confidence and competencies amongst the students that prevent them from selecting the right antibiotics for the right patient for the suitable duration [Citation22]. A South African study conducted on final year medical students to assess the knowledge, attitudes, and perceptions on antibiotic prescribing rationale found that graduating physicians are not sufficiently equipped with knowledge and training to prescribe antibiotics efficiently [Citation23]. Similar observations are also noted among the fourth-year medical students in the U.S.A [Citation24]. Until recently, enough emphasis was not put on healthcare degree programs on teaching antimicrobial prescribing and stewardship. It appears more likely that the antimicrobial stewardship program will be more effective and meaningful if it is conducted at the medical school level, when the knowledge and attitudes of the future physicians are being shaped [Citation25].
Figure 1. The schematic diagram is showing the main steps needed to be implemented to minimize the antimicrobial drug resistance
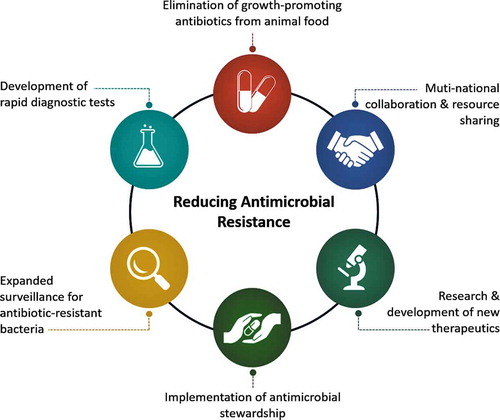
It is not just the prescribed antibiotics, but also the antibiotics used to promote animal growth that causes antimicrobial resistance. Such undesirable practice needs to be phased out to minimize the antimicrobial resistance. Whether consuming the antibiotics-fed animal meat can directly inflict harmful effects in humans is a controversial topic. However, it is generally agreed that the overuse of antibiotics in meat-producing animals can lead to the emergence of drug-resistant bacteria, which is a substantial public health concern. Of note, roughly 70%-80% of all antibiotics with clinical importance and significance are sold for animal use in the U.S.A [Citation26].
The WHO considers the antimicrobial drug resistance as one of the top five threats to human health. Incorporating antimicrobial stewardship in the healthcare system through surveillance, conducting basic and applied research, and developing newer-generation antimicrobial agents to prevent and control the antimicrobial drug resistance will significantly reduce the disease burden related to drug-resistant microorganisms. Since antimicrobial drug resistance is a global health concern, better coordination and collaboration among various countries’ regulatory bodies through standardized antimicrobial stewardship programs will yield better health outcomes. It needs to be emphasized that the alternative to the antimicrobial stewardship program will be a human disaster of unimaginable magnitude. The annual global deaths due to antimicrobial resistance are around 700,000 and if the enhanced rate of bacterial resistance is not addressed urgently through antimicrobial stewardship programs, an estimated annual death of 10 million people per year is predicted by 2050 [Citation27]. Such human tragedy will be amalgamated with 60 USD to 100 USD trillion in global economic loss [Citation27]. According to an estimation by the World Bank, more than 28 million additional people would be pushed into extreme poverty by 2050, if antibiotic resistance is not addressed adequately to prevent disease burden [Citation28]. Educating future healthcare professionals by antimicrobial stewardship programs on antimicrobials’ responsible use can improve the drugs’ current effectiveness and provide future sustainability [Citation29]. There is an urgent need for governing bodies’ involvement in the antimicrobial stewardship programs, not only at the urban hospitals but also in rural community healthcare centers [Citation30,Citation31].
Declaration of interest
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Reviewer disclosures
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
Acknowledgments
I want to express my sincere gratitude to Dr. Nuraly Akimbekov (Al-Farabi Kazakh National University, Kazakhstan) for help in drawing the illustration. I also wish to thank Mr. M. Muhit Razzaque and Dr. Margo Wolfe for carefully reading the manuscript and providing useful suggestions.
Additional information
Funding
References
- Cosgrove SE. The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis. 2006 Jan 15;42(Suppl 2):S82–9.
- CDC. Antibiotic resistance threats in the United States, 2013. US Department of Health & Human Services; 2006. https://www.cdc.gov/drugresistance/Threat-Report-2013/pdf/ar-Threats-2013-508.pdf
- Langford BJ, So M, Raybardhan S, et al. Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis. Clin Microbiol Infect. 2020 Jul 22; DOI:10.1016/j.cmi.2020.07.016
- Majumder MAA, Singh K, Hilaire MG, et al. Tackling antimicrobial resistance by promoting antimicrobial stewardship in medical and allied health professional curricula. Expert Rev Anti Infect Ther. 2020 Aug 17:1–14. DOI:10.1080/14787210.2020.1796638
- Moody J, Cosgrove SE, Olmsted R, et al. Antimicrobial stewardship: a collaborative partnership between infection preventionists and healthcare epidemiologists. Infect Control Hosp Epidemiol. 2012 Apr;33(4):328–330.
- CDC. U.S. national action plan for combating antibiotic-resistant bacteria (national action plan). US Department of Health & Human Services; 2015. https://www.cdc.gov/drugresistance/us-activities/national-action-plan.html
- Pulcini C, Cua E, Lieutier F, et al. Antibiotic misuse: a prospective clinical audit in a French university hospital. Eur J Clin Microbiol Infect Dis. 2007 Apr;26(4):277–280.
- Dinh A, Duran C, Davido B, et al. Impact of an antimicrobial stewardship programme to optimize antimicrobial use for outpatients at an emergency department. J Hosp Infect. 2017 Nov;97(3):288–293.
- Maduna LD, Kock MM, van der Veer B, et al. Antimicrobial resistance of Neisseria gonorrhoeae isolates from high risk men in Johannesburg, South Africa. Antimicrob Agents Chemother. 2020 Aug 31. DOI:10.1128/AAC.00906-20
- Bodie M, Gale-Rowe M, Alexandre S, et al. Addressing the rising rates of gonorrhea and drug-resistant gonorrhea: there is no time like the present. Can Commun Dis Rep. 2019;45(2–3):54–62.
- CDC. Drug-resistant N. gonorrhoeae pathogen page. US Department of Health & Human Services; 2019. https://www.cdc.gov/drugresistance/biggest-threats.html
- Lahra MM, Shoushtari M, George CRR, et al. Australian gonococcal surveillance programme annual report, 2019. Commun Dis Intell (2018). 2020 Jul 15; 44. DOI:10.33321/cdi.2020.44.58
- WHO. Antimicrobial stewardship: a competency-based approach. World Health Organization; 2015. https://openwho.org/courses/AMR-competency
- WHO. Health workers’ education and training on antimicrobial resistance: curricula guide. Geneva: World Health Organization; 2019. Available from: https://apps.who.int/iris/bitstream/handle/10665/329380/9789241516358-eng.pdf
- Jagsi R, Kitch BT, Weinstein DF, et al. Residents report on adverse events and their causes. Arch Intern Med. 2005 Dec 12-26;165(22):2607–2613.
- Phillips CJ, McKinnon RA, Woodman RJ, et al. Junior doctors’ preparedness to prescribe, monitor, and treat patients with the antibiotic vancomycin in an Australian teaching hospital. J Educ Eval Health Prof. 2017;14:13.
- Menon V, Chubaty A, Clezy K, et al. Infectious diseases and antimicrobial prescribing: online spaced education for junior doctors. J Glob Antimicrob Resist. 2020 Sep;22:226–230.
- Brinkman DJ, Tichelaar J, Graaf S, et al. Do final-year medical students have sufficient prescribing competencies? A systematic literature review. Br J Clin Pharmacol. 2018 Apr;84(4):615–635.
- Courtenay M, Lim R, Castro-Sanchez E, et al. Corrigendum to “Development of consensus-based national antimicrobial stewardship competencies for UK undergraduate healthcare professional education”. J Hosp Infect. 2019 Mar;101(3):366.
- Castro-Sanchez E, Drumright LN, Gharbi M, et al. Mapping antimicrobial stewardship in undergraduate medical, dental, pharmacy, nursing and veterinary education in the United Kingdom. PLoS One. 2016;11(2):e0150056.
- Wake Forest School of Medicine. Antimicrobial stewardship curriculum. Wake Forest School of Medicine. https://school.wakehealth.edu/Departments/Internal-Medicine/Infectious-Diseases/CAUSE/Antimicrobial-Stewardship-Curriculum.
- Dyar OJ, Pulcini C, Howard P, et al. European medical students: a first multicentre study of knowledge, attitudes and perceptions of antibiotic prescribing and antibiotic resistance. J Antimicrob Chemother. 2014 Mar;69(3):842–846.
- Wasserman S, Potgieter S, Shoul E, et al. South African medical students’ perceptions and knowledge about antibiotic resistance and appropriate prescribing: are we providing adequate training to future prescribers? S Afr Med J. 2017 Apr 25;107(5):405–410.
- Abbo LM, Cosgrove SE, Pottinger PS, et al. Medical students’ perceptions and knowledge about antimicrobial stewardship: how are we educating our future prescribers? Clin Infect Dis. 2013 Sep;57(5):631–638.
- Pulcini C, Gyssens IC. How to educate prescribers in antimicrobial stewardship practices. Virulence. 2013 Feb 15;4(2):192–202.
- FDA. Antimicrobials sold or distributed for use in food-producing animals. Food and Drug Administration; 2014. https://www.fda.gov/media/79581/download
- O’Neill J. Tackling drug-resistant infections globally: final report and recommendations. Government of the United Kingdom; 2016 Jan. https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf.
- World Bank. Drug-resistant infections: a threat to our economic future World Bank. 2017. http://documents.worldbank.org/curated/en/323311493396993758/final-report.
- Mendelson M, Balasegaram M, Jinks T, et al. Antibiotic resistance has a language problem. Nature. 2017 May 3;545(7652):23–25.
- Anderson DJ, Watson S, Moehring RW, et al. Feasibility of core antimicrobial stewardship interventions in community hospitals. JAMA Network Open. 2019;2(8): e199369..
- Buckel WR, Veillette JJ, Vento TJ, et al. Antimicrobial stewardship in community hospitals. Med Clin North Am. 2018 Sep;102(5):913–928.
