Asymmetric division plays a crucial role during oogenesis, characterized by extrusion of a small polar body in the first meiosis.Citation1 Oocytes preserve most of their maternal stores by expelling a minimal cytoplasmic content with half chromosomes, which is essential for early embryonic development by maintaining nutrition and energy resources due to extremely asymmetric division.Citation2 Spindle positioning is important for the asymmetric or symmetric division in cell cycle.Citation3 Cytoplasmic actin is involved in chromosome gathering and spindle positioning in mitosis, because randomization of spindle positioning occurs after disassembling actin with disrupting drugs.Citation4 Also, astral microtubules emanating from centrosomes interact with cortical anchor proteins decide the spindle position along the longest cell axis, which helps the symmetric division.Citation5 However, this rule is not suitable for oocytes because they are round and lack of centrosome and astral microtubules. In contrast to symmetric mitotic divisions, oocytes undergo asymmetric divisions with half homologous chromosomes segregated to the small polar body during anaphase of the first meiotic division. To compensate for the lack of centrosome and astral microtubules, actin assembly is very important for the spindle positioning by transmitting forces over long distances during oocytes meiosis. A recent paper in Cell Cycle describes a key factor, Formin-like 1 (FMNL1), associated with RhoA signal pathway, regulates asymmetric division by controlling actin assembly and spindle positioning in mouse oocytes (Fig. 1).Citation6
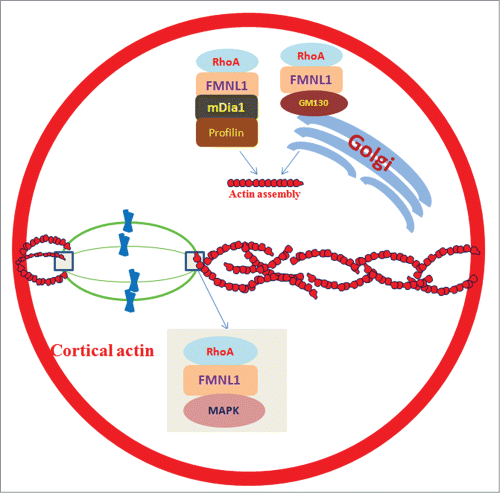
FMNL1 is a member of Formin family proteins which are the actin nucleators. It has been shown that FMNL1 is essential for cell adhesion, cytokinesis, cell polarization and migration in mitosis. The authors discovered the functions of FMNL1 in meiotic mouse oocytes by using specific morpholino microinjection and live cell imaging. FMNL1 knockdown caused disrupted asymmetric division, characterized by the large polar bodies and low rate of polar body extrusion. Immunofluorescent staining showed that in addition to its cytoplasmic distribution, FMNL1 was primarily localized at the spindle poles after germinal vesicle breakdown (GVBD), which implies that it may be involved in spindle organization and positioning. Next, the authors found various morphologically defective spindles in FMNL1 knockdown oocytes by co-staining FMNL1, α-tubulin and DNA. The wrong localization and lower expression level of phospho-p44/42 mitogen-activated protein kinase (p-MAPK) further confirmed the FMNL1s roles in spindle organization, because MAPK has been previously shown to be required for proper spindle formation. In addition, same results were observed in mammalian diaphanous1 (mDia1) knockdown oocytes. FMNL1 depletion resulted in reduced mDia1 expression, which implied that FMNL1 might be upstream to mDia1.Citation7
Subsequently, time-lapse microscopic and immunofluorescence intensity analysis demonstrated that the wrong spindle positioning was due to the aberrant actin assembly and expression level in FMNL1 knockdown oocytes. Similar results have been observed in other members of the Formin family members. Meantime, cortical polarity was disrupted as shown by a loss of actin cap and cortical granule-free domain (CGFD) formation. Because spindle migration is actin-dependent, FMNL1 may regulate spindle positioning by its effect on actin nucleation, which can further affect oocyte cortical polarity formation. Also, FMNL1 depletion resulted in aberrant localization and expression patterns of a cis-Golgi marker protein, GM130, which caused disrupted Golgi apparatus.Citation6 Because Golgi mediates the long-range transport of vesicles, FMNL1 may modulate vesicle-based mechanism of actin network for asymmetric division by regulating Golgi complex. In addition, mDia1 and Profilin1 had similar localization patterns in mouse oocytes and mDia1 knockdown resulted in reduced Profilin1 expression, which also partially explained the wrong actin assembly.Citation7
Inhibition of the small GTPase RhoA activity resulted in decreased FMNL1 expression, which indicates that RhoA may be the upstream molecule of FMNL1.Citation6 However, RhoA inhibition did not alter mDia1 expression, which indicates that there is a FMNL1-mDia1-Profilin1 signaling pathway. Taken together, there are RhoA-FMNL1/GM130 and RhoA-FMNL1-dMia1- Profilin1 pathways for the spindle positioning and actin assembly during mouse oocyte meiosis.Citation7
In summary, the identification of FMNL1 as a key regulator for spindle positioning and actin assembly in the current study strongly implies that the actin-based spindle positioning is vital for asymmetric division during oocyte maturation. These recent studies, on one hand clarify the mechanism for asymmetric division in mouse oocytes, and on the other hand show a RhoA-FMNL1 pathway. It will contribute to understanding how physical parameters including cell size and shape are controlled in the cell cycle.
Figure 1. FMNL1 regulates asymmetric division by controlling actin assembly and spindle positioning in mouse oocytes. Actin network modulation is essential for the asymmetric positioning of meiotic spindle. FMNL1 is important for spindle organization with the help of MAPK. FMNL1-GM130 is involved in the Golgi apparatus function and actin assembly. Also, FMNL1-mDia1-Profilin1 pathway is involved in actin assembly and consequent asymmetric spindle positioning.
References
- Brunet S, Verlhac MH. Hum Reprod Update 2011; 17(1):68-75; PMID:20833637; http://dx.doi: 10.1093/humupd/dmq044.
- Gillies TE, Cabernard C. Curr Biol. 2011; 21(15):R599-609; PMID:21820628; http://dx.doi:10.1016/j.cub.2011.06.055.
- Morin X, Bellaïche Y. Dev Cell 2011; 21(1):102-19; PMID:21763612; http://dx.doi:10.1016/j.devcel.2011.06.012.
- Toyoshima F, Nishida E. EMBO J 2007; 26(6):1487-98; PMID:17318179; http://dx.doi:10.1038/sj.emboj.7601599.
- Almonacid M, et al. J Cell Sci 2014; 127, 477-483; PMID:24413163; http://dx.doi:10.1242/jcs.142711.
- Wang F, et al. Cell Cycle 2015; 14(17):2835-43; PMID:26083584; http://dx.doi.org/10.1080/15384101.2015.1031438
- Zhang Y, et al. Cell Cycle 2015; Biochim Biophys Acta 2015; 1853(2):317-27; PMID:25447542; http://dx.doi:10.1016/j.bbamcr.
