Abstract
Chronic obstructive pulmonary disease (COPD) is one of the leading causes of morbidity and mortality worldwide. In Finland, the current prevalence of COPD is unknown. This population-based study aimed to assess the prevalence of COPD in Finland in adults 21–70 years of age by using internationally accepted criteria for COPD, and to evaluate risk factors for COPD. A random sample (n = 959) of questionnaire responders was invited to participate in a clinical study, which included a structured interview, skin-prick tests, and spirometry. Complete data were received from 683 subjects. According to the criteria of the British Thoracic Society and the Global Initiative for Chronic Obstructive Lung Disease, the prevalence of COPD was 5.4% and 9.4% respectively. The risk of COPD increased significantly with age, amount of smoking, and family history of obstructive airways disease, but not with skin-prick positive allergy, recreational skiing, or outdoor work. The smoking-adjusted risk of clinically relevant COPD did not differ significantly between men and women. Among subjects with a smoking history of more than 40 pack-years, the prevalence of COPD was over 50%. Early start age of smoking was a strong risk factor.
Key words: :
Introduction
According to common opinion, chronic obstructive pulmonary disease (COPD) is associated with smoking in 80–90% of cases, but only 10–20% of smokers develop clinically significant COPD Citation[1]. Pharmacological treatment may relieve symptoms of COPD Citation[2&3], but does not improve prognosis or slow down the decline of ventilatory function Citation[4&5].
The vast burden of COPD has recently been recognized globally Citation[6-8]. Standards for the diagnosis and care of patients with COPD have been updated Citation[9], and international Citation[10&11] and national Citation[12-14] COPD programs and initiatives have been started. Recent studies have shown some progress in the treatment of COPD Citation[15-17], and new epidemiological data are consistently emerging. The age-adjusted COPD mortality rate in the United States from 1979 to 1993 has increased by 47.3% Citation[18]. In Europe, the morbidity and mortality rates vary substantially between countries, but according to a recent review Citation[19], 4–6% of the adult population in Europe suffers from clinically relevant COPD. A population-based multi-center study in Spain Citation[20] reported that the prevalence of COPD in ever-smoking men in the age group 40–49 years was 8.6% and 30.6% in the age group 60–69 years. The corresponding prevalence rates in ever-smoking women were 4.3% and 6.1%. The majority of COPD cases had not been diagnosed previously. The prevalence of COPD, estimated in the United States and in Europe by using age-specific and smoking-specific rates, revealed that in the United States only 14–46% of all COPD cases had been formerly diagnosed Citation[21].
In Finland, the prevalence of emphysema with airflow limitation, reported over 40 years ago, was 8.7% in men and 3.7% in women Citation[22]. Another pioneer Finnish study found the prevalence of emphysema (with FEV1/FVC ratio < 0.70) to be 10.0% among men and 2.3% among women aged 40–64 years Citation[23]. Knowledge about the current COPD prevalence in Finland is, however, lacking. A report did assess the prevalence of COPD among elderly people Citation[24], but the criteria used did not conform to published guidelines Citation[11-14]. In a recent Finnish study among a general adult population aged 30 years and older, airways obstruction defined as FEV1/FVC ≤ 0.69 was found in 11% of men and 5% of women Citation[25].
The main aim of this study was to assess the prevalence of COPD in a general adult population in Northern Finland by using the Global Initiative for Chronic Obstructive Lung Disease (GOLD) Citation[11] and the British Thoracic Society (BTS) Citation[12&13] criteria. The associations between COPD and age, gender, smoking as cumulative life-time pack-years number, skin prick positive allergy to common allergens, outdoor work, regular cross-country skiing, passive smoking during childhood, patient-reported chronic bronchitis and emphysema, and family history of obstructive airways disease (OAD) were also evaluated.
Methods
Study Area and Population
During the winter 1995–96 a postal questionnaire study was conducted in Lapland, the northernmost province of Finland. A random sample of 7,937 subjects aged 20–69 years was invited to participate in a postal questionnaire study; 6,633 responded. The results of this study have been reported Citation[26-30]. Of the responders in the southern part of the study area (n = 3420) a random sample of 959 subjects was invited to a clinical study. This study included a structured interview, skin-prick tests, and pulmonary function measurements. At the time of the clinical study, the participants were 21–70 years of age.
Questionnaire and Structured Interview
Both the Finnish FinEsS questionnaire Citation[26]Citation[31] and FinEsS interview used in this study were developed from the OLIN questionnaire Citation[32], and contain questions originating from the BMRC Citation[33], IUATLD Citation[34] and ECRHS questionnaires Citation[35]. Pack-years data for the clinical study participants was taken from the interview, while all other data about allergy, heredity, respiratory symptoms, asthma, chronic bronchitis, symptoms under special circumstances, use of asthma medication, profession, outdoor work, and skiing habits were obtained from the postal questionnaire Citation[26]. For example, working conditions were assessed by the question “Are you nowadays working mainly at a) outdoor or b) indoor work?,” and skiing habits by the question “Are You, or have You been a regular recreational skier?”
Lung Function Tests
Spirometric measurements, using a flow-volume spirometer (Sensor-medics VMAX 22), included flow-volume spirometry, a bronchodilation test, and slow vital capacity. At least three successive measurements were performed according to the following American Thoracic Society (ATS) standards Citation[36]:
exhalation time at least 6 seconds,
the largest FVC and the largest FEV1 may come from different curves, but all other parameters from the curve that has largest sum of FVC plus FEV1 (best curve method),
the two largest FVC within 200 ml of each other,
the two largest FEV1 within 200 ml of each other.
The Finnish reference values were used Citation[37]. The spirometer was calibrated daily. Nose clips and standard bacterial filters were used. Bronchodilation was assessed 10 minutes after inhalation of salbutamol 2 × 200 mcg (Ventoline MDI 200 mcg/puff with Volumatic, Glaxo Pharmaceuticals).
Skin Prick Tests
The standard duplicate method Citation[38] was used to test skin prick reactivity to 12 allergens: Dermatophagoides pteronyssinus, Acarus siro, cat, dog, horse, cow, birch, timothy, mugwort, Cladosporium herbarum, latex, and reindeer. A positive (histamine HCL, 10 mg/ml, in 50% glycerol) and a negative control (50% glycerol) were included. The reactions were read 15 minutes after application of allergens on the forearm. A reaction was considered positive if the mean wheal diameter ([d1 + d2]/2) was at least 3 mm.
Definitions and Diagnostic Criteria
Airway obstruction in COPD is defined as FEV1/VC < 0.7 by BTS and FEV1/FVC < 0.7 by GOLD criteria, post-bronchodilator results. The BTS definition of COPD states: “Unlike asthma, airflow limitation in COPD as measured by the FEV1can never be returned to normal values.” Thus only those who also had post-bronchodilator FEV1< 80% of predicted were included in BTS COPD cases. According to the BTS criteria, FEV1 ranges (of predicted) in mild disease from 50% to 80%, in moderate from 30% to 49% and in severe COPD FEV1 is below 30%, whereas GOLD considers FEV1≥ 80% as mild COPD, 50% ≤ FEV1< 80% as moderate, 30% ≤ FEV1< 50% as severe, and FEV1< 30% as very severe.
In order to detect clinically relevant COPD cases among subjects with a smoking history ≤ 20 pack-years, they were classified with the following arbitrary criteria:
other respiratory conditions as they were reported
asthmatics: those who had BOTH a self-reported asthma or had a positive skin-prick test AND had reported attacks of respiratory obstruction or wheezing.
subjects with clinically relevant COPD: those who had nothing listed above but had post-bronchodilator FEV1 < 80% of predicted
chronic bronchitis: those who had nothing listed above but had reported chronic cough or chronic productive cough
healthy if subject had nothing listed above
Analyses
To distinguish COPD from pure asthma, the following adjustment was performed: those who had both physician-diagnosed asthma and smoking history of less than 10 pack-years were not included in the COPD group. Chi-square test was used for bi-variate comparisons. One-way ANOVA (analysis of variance) was used to test trends. Odds ratios (OR) for various independent variables were calculated using multiple logistic regression analysis. Variables included gender, age, smoking habits, allergy, family history of OAD, smoking parent, outdoor work, and recreational skiing. An interaction analysis between age, smoking, and family history of OAD was performed by using an additive model with multiple logistic regression analysis.
Results
Participation and Smoking Habits
Responses to the questionnaire study and participation in the clinical study with lung function results and smoking habits by age and gender are shown in . A total of 959 subjects were invited to the clinical study; 695 attended. Spirometry was repeated for 63 subjects, 51 of them were accepted after repetition, but for 12 subjects the result did not meet the criteria and they were excluded from the final analysis; thus 683 subjects (71.2% of invited) remained in the study sample. The total participation rate of the clinical study was 60.1% (0.844 × 0.712). Men in the youngest age group had the lowest participation rate both in the questionnaire and in the clinical study.
Table 1. Response, participation, and smoking habits in the questionnaire and clinical study, and accepted spirometries by age and gender
Distribution of age, gender, asthma, chronic bronchitis, smoking habits, and respiratory symptoms among the questionnaire respondents (n = 3420), those who were invited (n = 959), or those who participated (n = 683) in the clinical study are shown in . The proportion of ex-smokers was higher among the clinical study participants than among the questionnaire responders or the invited subjects. Differences in respiratory diseases and symptoms between the clinical study participants and the questionnaire respondents or the invited persons were, however, small and not significant.
Table 2. Age, gender, respiratory conditions, and smoking habits (% of group) among postal questionnaire respondents (n=3420), subjects invited to the clinical study (n=959), and participants with accepted lung function (n=683).
Among the clinical study participants, male current smokers (n = 112) had an average smoking history of 20.8 pack-years, and ex-smokers (n = 138) 17.7 pack-years. The corresponding figures for females were 11.7 (n = 95) and 6.9 (n = 75) pack-years. The average smoking history for ever-smoking (ex- or current smokers) men was 19.1 pack-years, and for women 9.6 pack-years.
Prevalence of COPD and Severity Classification
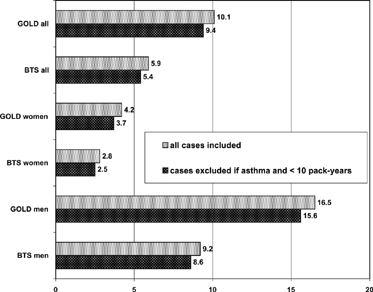
The overall prevalence of COPD was 5.4% by BTS criteria and 9.4% by GOLD criteria. In men, it was 8.6% according to BTS and 15.6% according to GOLD criteria, and in women 2.5% and 3.7%, respectively (). Adjustment of prevalence by excluding those with both physician-diagnosed asthma and smoking history of less than 10 pack-years had only a minor effect on prevalence (). The GOLD criteria classified 50% of the COPD cases as mild, 44% as moderate, and 6% as severe.
Table 3. COPD cases in the study population (n=683) by BTS or GOLD criteria and pack-years classification.
Effect of Smoking Habits on Prevalence of COPD
The prevalence of COPD increased with age and number of pack-years. Among subjects who were lifelong non-smokers or had a smoking history of 1 pack-year or less, the prevalence of COPD was 1.7% according to BTS and 5.3% according to GOLD criteria; but among those with a smoking history of more than 20 pack-years the respective prevalence was 24.1% and 35.3% ().
COPD was associated with a smoking history of at least one pack-year in 75% of GOLD cases and in 86% of BTS cases. By GOLD criteria, the proportion of non-smokers was 41% among the mild COPD cases and 16% among moderate cases. In mild COPD as defined by GOLD criteria, the mean amount of smoking was 18.8 pack-years. The corresponding figures in moderate and severe COPD according to GOLD criteria were 31.6 pack-years and 71.5 pack-years.
Of the 567 subjects with a smoking history of ≤ 20 pack-years, 27 had spirometric obstruction. Nine of them fulfilled the BTS criteria for COPD and 22 fulfilled the GOLD criteria. The analysis of these 27 subjects revealed that according to our arbitrary criteria:
all 7 healthy subjects were classified as healthy by BTS, but GOLD labeled all of them as having COPD
both BTS and GOLD found 5 of the 8 subjects having COPD
of 8 subjects having asthma, 3 were classified as COPD by BTS and 6 by GOLD
of the 9 cases having COPD by the BTS criteria, 4 might be false positives (3 asthmatics and 1 sequelae after pulmonary tuberculosis), but of the 22 cases having COPD by the GOLD criteria, only 5 may have a clinically relevant COPD.
Multivariate Relationships and Interaction Analysis
According to multiple logistic regression analysis, the odds ratios for COPD increased significantly with age and amount of smoking (). COPD was more prevalent among men than women. However, when adjusted for age and smoking, the risk of clinically relevant COPD (BTS criteria) did not differ significantly between men and women. Family history of obstructive airways disease was associated with a significantly higher risk for COPD. Positive skin-prick test, smoking parents, outdoor work, and recreational cross-country skiing did not affect the odds ratios significantly.
Table 4. Risk factors for COPD by BTS or GOLD criteria calculated by multiple logistic regression analysis
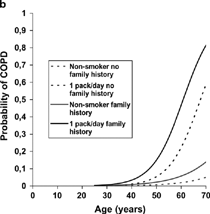
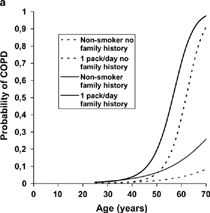
The probability of COPD by BTS and GOLD criteria in relation to age and family history of OAD was calculated for non-smokers and current smokers with the assumption that smoking 1 pack per day was started at the age of 18 years (). After 50 years of age, the probability of COPD increased considerably in subjects who continued to smoke or had a family history of OAD. The probability at the age of 60 years was 2 times higher among continuing smokers with a family history of OAD than among those with no family history of OAD.
Figure 2 Probability of COPD in correlation to age, family history of obstructive airways disease, and number of pack-years.
A logistic regression analysis was performed among ever-smokers (all current and ex-smokers included), with COPD (by BTS criteria) as the dependent variable, and age, gender, and starting age of smoking as independent variables. A significant increase in the risk of COPD was present with earlier start of smoking (p = 0.0182). The risk of COPD increased by 14% per year (95% CI 2–28%), while the corresponding increase per pack-year was 7% (95% CI 4%–9%).
COPD in Relation to Patient-Reported Previous Diagnoses or Symptoms
The prevalence of COPD according to GOLD criteria was 27.9% among subjects who reported having asthma and 40.0% among those reporting physician-diagnosed chronic bronchitis or emphysema. According to BTS criteria, the corresponding figures were 23.3% and 32.0%. In subjects with chronic productive cough, the prevalence of COPD by GOLD criteria was 21.1%, and by BTS criteria 12.6%. Men generally had higher prevalence rates than women.
Only 15.6% of GOLD COPD cases and 21.6% of BTS COPD cases reported having physician-diagnosed chronic bronchitis or emphysema, and 31.2% and 32.4%, respectively, had chronic productive cough. Among COPD cases diagnosed by GOLD criteria, 64.1% had smoked more than 20 pack-years, and 26.6% had a smoking history of one pack-year or less. The corresponding figures for cases diagnosed by BTS criteria were 75.7% and 13.5%.
Discussion
The results revealed that age, family history of obstructive airways disease, and number of pack-years were independent risk factors for COPD, and their effect was additive. The prevalence of COPD in Northern Finland in the general population aged 21–70 years was 9.4% by the GOLD and 5.4% by the BTS criteria. These prevalence rates are in line with recent international studies and reviews Citation[18-20]Citation[39]Citation[40]Citation[41]. Men had a higher prevalence of COPD than women, but according to multivariate analysis by the BTS criteria, the risk for COPD did not differ significantly between the sexes. The common opinion of higher risk of COPD among men may merely reflect the difference in smoking habits between men and women. In Finland, a population-based study assessing the prevalence of COPD in a general adult population was published about four decades ago Citation[23], but little current data on this topic have been published. Moreover, divergence from internationally accepted COPD criteria weakens the validity of recent Finnish studies. In a study among elderly Finns, COPD was defined as FEV1/FVC ratio ≤ 0.65 Citation[24], which may be clinically relevant but is not in agreement with national or international guidelines Citation[11-14].
In our study, BTS criteria found 37 COPD cases (28 men and 9 women), and GOLD criteria found 64 cases (51 men and 13 women). Low numbers in sub-groups weaken the precision and significance, therefore an extensive statistical analysis in was considered not meaningful. The original random sample was 4054 subjects. Of those 3420 subjects responded in the postal questionnaire, 959 subjects were invited to the clinical study and results were obtained from 683 subjects. Thus these 683 participants represent 20.0% of those responding in the questionnaire or 16.8% of the original sample. We feel that our results are well representative for the original study population because, firstly, standard study methods and criteria were used; secondly, all population samples were randomised; thirdly, the participation rates were high: 84.4% in the questionnaire study and 71.2% in the clinical study; fourthly, the proportion of subjects with respiratory symptoms and conditions did not differ between the questionnaire and the clinical study; and finally, our separate non-response analysis Citation[28] revealed no significant difference in the prevalence rates of respiratory diseases or symptoms in this study population. Thus, in spite of low numbers, prevalence rates shown in this report may apply in the general adult population in Northern Finland.
The cross-sectional design in our study could also bring about some limitations. For example, at the time of quitting smoking no data of atopy, lung function or symptoms are available and subjects with respiratory symptoms or allergy to pollen could have changed from working outdoor to indoor. Moreover, in our statistical analysis all pack-years were considered equal, but it may be argued that a certain number of pack-years is more harmful for a current smoker than for an ex-smoker. However, our interaction analysis for the risk of COPD (including both current and ex-smokers) revealed that the effect of an earlier start of smoking by one year, a 14% increase, was two times stronger than the effect of 1 pack-year.
The prevalence rates and distribution of severity of COPD in our study have a good correlation with recent studies in both Europe Citation[42] and the United States Citation[43]. The prevalence of COPD among smokers with a smoking history of more than 40 pack-years was 53% by BTS and 66% by GOLD criteria. Our results support recent findings of high prevalence of COPD among elderly smokers Citation[21]Citation[42]Citation[44]. However, it should be kept in mind that both BTS and GOLD use a fixed cut-off value of 0.7 for FEV1/VC or FEV1/FVC ratio, which may result in an overestimation of airway obstruction with advancing age Citation[45].
We have previously reported a comparison of asthma, chronic bronchitis, and respiratory symptoms between Northern and Southern Finland Citation[29]. According to those results, the prevalence of chronic productive cough was approximately the same in Northern (11.0%) and Southern (12.1%) Finland and the prevalence rates of asthma and chronic bronchitis did not differ significantly. Differences in smoking habits were small; in Northern Finland 36.6% of adults were current smokers vs. 37.7% in Southern Finland (not significant), for ever-smokers, the corresponding figures were 53.8% and 54.3% (not significant). Based on these findings and the homogeneity of the Finnish population, we assume that our findings of COPD in Northern Finland apply also to other parts of the country.
The BTS and GOLD guidelines consider COPD to be distinct from asthma, but the differentiation between COPD and chronic asthma with persistent airflow limitation is difficult. A consensus exists in international guidelines concerning the definition of COPD: “chronic obstructive pulmonary disease is a disease state characterized by airflow limitation that is not fully reversible. The airflow limitation is usually both progressive and associated with an abnormal inflammatory response of the lungs to noxious particles or gases” Citation[11]. However, despite overall agreement, the wording used between different COPD guidelines varies, and more importantly, the limit values for spirometry results differ Citation[11-14]. In practice, the cases classified as mild COPD by GOLD criteria are not labelled as COPD at all by BTS. This is an important point if early diagnosis and intervention are considered essential for COPD management.
On the other hand, among COPD cases found by the GOLD criteria, many false positives may be included. This was underlined by our sub-analysis among the COPD cases having a smoking history ≤ 20 pack-years. The results revealed that many asthmatics, subjects with other respiratory conditions and also apparently healthy subjects may be included among the COPD cases found by the GOLD criteria. In comparison to the other COPD criteria that include declined FEV1, the GOLD criteria may more often include other conditions among the COPD cases, e.g., undiagnosed asthma. Thus the clinical significance and prognostic value of stage 1 (mild) and stage 0 (“at risk”) COPD by the GOLD criteria may be questioned Citation[46].
We have earlier reported that the risk for chronic bronchitis and associated symptoms is higher among outdoor than indoor workers Citation[29&30]. However, pack-year data were not available in these reports. The multivariate analysis in the present study shows that the risk for COPD increases with age, pack-years, and family history for OAD, but not with positive skin prick test, outdoor work, or cross-country skiing.
Less than 25% of the COPD cases had an earlier diagnosis of chronic bronchitis or emphysema. The prevalence of COPD in subjects with chronic productive cough was 16–28% in men and 6–9% in women. Two-thirds of the COPD cases did not report chronic productive cough. This is supported by a recent paper that revealed that only 12% of subjects reporting a diagnosis of chronic bronchitis met the criteria of chronic bronchitis Citation[47]. These findings underline the diagnostic difficulties: many subjects with COPD do not report chronic symptoms and only a small proportion of subjects with chronic productive cough hve COPD. Women report less chronic productive cough than men, and thus under-diagnosis seems to be high in women, as reported by others Citation[48].
It does not seem justified to assess the prevalence of COPD by using patient-reported diagnosis or respiratory symptoms alone as has been done in some studies Citation[49]. Spirometric measurements have been recommended as the best means to find COPD cases at an early stage Citation[50] and to reduce the risk of under-diagnosis and gender bias Citation[48]. The problems with clinical practice guidelines targeting COPD have been recognised: the methodological quality is limited, and there are disparities among many of their recommendations Citation[51]. Valid comparisons of COPD prevalence between different studies are not possible unless smoking data are given in pack-years and similar spirometric and diagnostic criteria are employed. Obviously, a more specified international consensus about COPD criteria is needed. The assessment of COPD prevalence should be based on both relevant patient information Citation[47] and lung function measurements which pay attention to the normal distribution of lung function in different populations and age groups Citation[45].
When smoking started earlier in life, the risk of COPD increased considerably. Young teenagers may have a higher biological susceptibility to harmful effects of tobacco smoke than adults. The probability of COPD increased very steeply after the age of 50 years in subjects with a smoking history of more than 30 pack-years who continued to smoke, and in those who had a positive family history of OAD and a smoking history of more than 20 pack-years. Thus, the development of new COPD cases could be effectively diminished if starting age of smoking increases and if most smokers quit before 20 pack-years.
In conclusion, a high prevalence of COPD was found in a general adult population of smoking men and women in Finland. Among those having smoked more than 20 pack-years, the prevalence of COPD was 24% by BTS and 35% by GOLD criteria. The risk for COPD rose with increasing age and number of pack-years, especially if smoking was started early in life. Also a family history of OAD was a risk factor for COPD. Although outdoor work in a cold climate may increase respiratory symptoms and chronic bronchitis, it seemed not to have an impact on the risk for COPD.
Acknowledgments
The authors thank Länsi-Pohja Central Hospital for assistance in data collection, and Statistician Anders Odén, Sweden, and Docent Matti Kataja, Tampere, Finland, for help in performing statistical analyses.
References
- MacNee W. Chronic bronchitis and emphysema. In: Seaton A, Seaton D, Leitch A G, eds. Crofton and Douglas's Respiratory Diseases. London: Blackwell Science Ltd., 2000:616–695.
- Burge P S, Calverley P M, Jones P W, Spencer S, Anderson J A, Maslen T K. Randomised, double blind, placebo controlled study of fluticasone propionate in patients with moderate to severe chronic obstructive pulmonary disease: the ISOLDE trial.Br Med J 2000; 320:1297–1303. [CSA]
- The Lung Health Study Research Group. Effect of inhaled triamcinolone on the decline in pulmonary function in chronic obstructive pulmonary disease.N Engl J Med 2000; 343:1902–1909. [CSA], [CROSSREF]
- Pauwels R A, Löfdahl C G, Laitinen L A, Schouten J P, Postma D S, Pride N B, Ohlsson S V. Long-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. European Respiratory Society Study on Chronic Obstructive Pulmonary Disease.N Engl J Med 1999; 340(25):1948–1953. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Vestbo J, Sørensen T, Lange P, Brix A, Torre P, Viskum K. Long-term effect of inhaled budesonide in mild and moderate chronic obstructive pulmonary disease: a randomised controlled trial.Lancet 1999; 353:1819–1823. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Murray C J, Lopez A D. Alternative projections of mortality and disability by cause 1990–2020: global burden of disease study.Lancet 1997; 349(9064):1498–1504. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Hurd S. The impact of COPD on lung health worldwide: epidemiology and incidence.Chest Feb., 2000; 117(2 Suppl):1S–4S. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Gulsvik A. The global burden and impact of chronic obstructive pulmonary disease worldwide.Monaldi Arch Chest Dis 2001; 56(3):261–264. [PUBMED], [INFOTRIEVE], [CSA]
- American Thoracic Society. European Respiratory Society: Standards for the Diagnosis and Management of Patients with COPD. www.thoracic.org/copd.
- Siafakas N M, Vermeire P, Pride N B, Paoletti P, Gibson J, Howard P, Yernault J C, Decramer M, Higenbottam T, Postma D S, et al. Optimal assessment and management of chronic obstructive pulmonary disease (COPD). The European Respiratory Society Task Force.Eur Respir J 1995; 8:1398–1420. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease. In: NHLBI/WHO Workshop Report. NIH Publication 2701, Bethesda: U.S. Department of Health and Human Services, April 2001, (Updated 2003).
- BTS guidelines for the management of chronic obstructive pulmonary disease.Thorax 1997; 52(Suppl 5):S1–S28. [CSA]
- NICE Guideline No. 12: Chronic Obstructive Pulmonary Disease. National clinical guideline on management of chronic obstructive pulmonary disease in adults in primary and secondary care.Thorax 2004; 59(Suppl 1):i1–i232. [CSA], [CROSSREF]
- Laitinen L A, Koskela K. Chronic bronchitis and chronic obstructive pulmonary disease: Finnish National Guidelines for Prevention and Treatment 1998–2007.Respir Med May, 1999; 93(5):297–332. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Mahler D A, Wire P, Horstman D, Chang C N, Yates J, Fischer T, Shah T. Effectiveness of fluticasone propionate and salmeterol combination delivered via the diskus device in the treatment of chronic obstructive pulmonary disease.Am J Respir Crit Care Med Oct. 15, 2002; 166(8):1084–1091. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Szafranski W, Cukier A, Ramirez A, Menga G, Sansores R, Nahabedian S, Peterson S, Olsson H. Efficacy and safety of budesonide/formoterol in the management of chronic obstructive pulmonary disease.Eur Respir J Jan., 2003; 21(1):74–81. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Calverley P, Pauwels R, Vestbo J, Jones P, Pride N, Gulsvik A, Anderson J, Maden C. Trial of inhaled steroids and long-acting beta2 agonists study group: combined salmeterol and fluticasone in the treatment of chronic obstructive pulmonary disease: a randomised controlled trial.Lancet Feb. 8, 2003; 361(9356):449–456., [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Mannino D M, Brown C, Giovino G A. Obstructive lung disease deaths in the United States from 1979 through 1993. An analysis using multiple-cause mortality data.Am J Respir Crit Care Med Sep., 1997; 156(3 Pt 1):814–818. [PUBMED], [INFOTRIEVE], [CSA]
- Gulsvik A. Mortality in and prevalence of chronic obstructive pulmonary disease in different parts of Europe.Monaldi Arch Chest Dis 1999; 54(2):160–162. [PUBMED], [INFOTRIEVE], [CSA]
- Pena V S, Miravitlles M, Gabriel R, Jimenez-Ruiz C A, Villasante C, Masa J F, Viejo J L, Fernandez-Fau L. Geographic variations in prevalence and underdiagnosis of COPD: results of the IBERPOC multicentre epidemiological study.Chest 2000; 118(4):981–989. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Stang P, Lydick E, Silberman C, Kempel A, Keating E T. The prevalence of COPD: using smoking rates to estimate disease frequency in the general population.Chest 2000; 117(5 Suppl 2):354S–359S. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Järvinen K A, Pätiälä J, Thomander K. Frequency of obstructive pulmonary emphysema.Ann Med Intern Fenn 1960; 49:301–306. [CSA]
- Huhti E. Prevalence of respiratory symptoms, chronic bronchitis and pulmonary emphysema in a Finnish rural population. Field survey of age group 40–64 in the Harjavalta area.Acta Tuberc Pneumol Scand 1965, Suppl 61):1–111. [CSA]
- Isoaho R, Puolijoki H, Huhti E, Kivela S L, Laippala P, Tala E. Prevalence of chronic obstructive pulmonary disease in elderly Finns.Respir Med 1994; 88(8):571–580. [PUBMED], [INFOTRIEVE], [CSA]
- von Hertzen L, Reunanen A, Impivaara O, Mälkiä E, Aromaa A. Airway obstruction in relation to symptoms in chronic respiratory disease—a nationally representative population study.Respir Med 2000; 94(4):356–363. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Kotaniemi J.-T, Lundbäck B, Nieminen M M, Sovijärvi A R.A, Laitinen L A. Increase of asthma in adults in northern Finland? A report from the FinEsS study.Allergy 2001; 56:169–174. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Lindström M, Kotaniemi J, Jönsson E, Lundbäck B. Smoking, respiratory symptoms, and diseases. A comparative study between northern Sweden and northern Finland: report from the FinEsS study.Chest 2001; 119:852–861. [CSA], [CROSSREF]
- Kotaniemi J.-T, Hassi J, Kataja M, Jönsson E, Laitinen L.-A, Sovijärvi A R.A, Lundbäck B. Does non-responder bias have a significant effect on the results in a postal questionnaire study?Eur J Epidemiol 2002; 17:809–817. [CSA], [CROSSREF]
- Kotaniemi J.-T, Pallasaho P, Sovijärvi A R.A, Laitinen L A, Lundbäck B. Respiratory symptoms and asthma in relation to cold climate, inhaled allergens and irritants.J Asthma 2002; 39(7):649–658. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Kotaniemi J T, Latvala J, Lundbäck B, Sovijärvi A, Hassi J, Larsson K. Does living in a cold climate or recreational skiing increase the risk for obstructive respiratory diseases or symptoms?Int J Circumpolar Health May, 2003; 62(2):142–157. [PUBMED], [INFOTRIEVE], [CSA]
- Pallasaho P, Lundbäck B, Läspä S L, Jönsson E, Kotaniemi J, Sovijärvi A R, Laitinen L A. Increasing prevalence of asthma but not of chronic bronchitis in Finland? Report from the FinEsS-Helsinki Study.Respir Med Nov., 1999; 93(11):798–809. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Lundbäck B. Asthma, chronic bronchitis and respiratory symptoms: prevalence and important determinants. The obstructive lung disease in Northern Sweden Study I.Umeå Univ Med Diss 1993; 387:1–118, Appendix pp. I–xii. [CSA]
- Medical Research Council's Committee on the Aetiology of Chronic Bronchitis. Standardized questionaries on respiratory symptoms.Br Med J 1960; ii1665, [CSA]
- Burney P G.J, Laitinen L A, Perdrizet S, Huckauf H, Tattersfield A E, Chinn S, Poisson N, Heeren A, Britton J R, Jones T. Validity and repeatability of the IUATLD (1984) Bronchial Symptoms Questionnaire: an international comparison.Eur Respir J 1989; 2:940–945. [PUBMED], [INFOTRIEVE], [CSA]
- European Community Respiratory Health Survey. Variations in the prevalence of respiratory symptoms, self-reported asthma attacks, and use of asthma medication in the European Community Respiratory Health Survey (ECRHS).Eur Respir J 1996; 9:687–695. [CSA], [CROSSREF]
- American Thoracic Society. Standardization of spirometry. 1994 update.Am J Respir Crit Care Med 1995; 152:1107–1136. [CSA]
- Viljanen A. Reference values for spirometric, pulmonary diffusing capacity and body pletysmografic studies.Scand J Clin Lab Invest 1982; 42(Suppl 159):5–20. [CSA]
- Dreborg S. The skin prick test: methodological studies and clinical applications. In: Medical Dissertation No. 239. 1987:1–148, Linköping, Sweden.
- Whittemore A, Perlin S, DiCiccio Y. Chronic obstructive pulmonary disease in lifelong nonsmokers: results from NHANES.Am J Public Health 1995; 85(5):702–706. [PUBMED], [INFOTRIEVE], [CSA]
- Anto J M, Vermeire P, Vestbo J, Sunyer J. Epidemiology of chronic obstructive pulmonary disease.Eur Respir J 2001; 17(5):982–994. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Viegi G, Scognamiglio A, Baldacci S, Pistelli F, Carrozzi L. Epidemiology of chronic obstructive pulmonary disease (COPD).Respiration 2001; 68(1):4–19. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Lundbäck B, Lindberg A, Lindström M, Rönmark E, Jonsson A C, Jönsson E, Larsson L G, Andersson S, Sandström T, Larsson K. Obstructive lung disease in Northern Sweden studies: not 15 but 50% of smokers develop COPD?—Report from the Obstructive Lung Disease in Northern Sweden Studies.Respir Med 2003; 97(2):115–122. [CSA], [CROSSREF]
- Mannino D M, Gagnon R C, Petty T L, Lydick E. Obstructive lung disease and low lung function in adults in the United States: data from the National Health and Nutrition Examination Survey, 1988–1994.Arch Intern Med Jun. 12, 2000; 160(11):1683–1689. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Tylen U, Boijsen M, Ekberg-Jansson A, Bake B, Löfdahl C G. Emphysematous lesions and lung function in healthy smokers 60 years of age.Respir Med 2000; 94(1):38–43. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Hardie J A, Buist A S, Vollmer W M, Ellingsen I, Bakke P S, Mørkve O. Risk of over-diagnosis of COPD in asymptomatic elderly never-smokers.Eur Respir J Nov., 2002; 20(5):1117–1122. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Vestbo J, Lange P. Can GOLD Stage 0 provide information of prognostic value in chronic obstructive pulmonary disease?Am J Respir Crit Care Med 2002; 166:329–332. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Bobadilla A, Guerra S, Sherrill D, Barbee R. How accurate is the self-reported diagnosis of chronic bronchitis?Chest 2002; 122:1234–1239. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Chapman K R, Tashkin D P, Pye D J. Gender bias in the diagnosis of COPD.Chest Jun., 2001; 119(6):1691–1695. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Halbert R J, Isonaka S, George D, Iqbal A. Interpreting COPD prevalence estimates: what is the true burden of disease?Chest 2003; 123:1684–1692. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Zieliñski J, Bednarek M. Early detection of COPD in a high-risk population using spirometric screening.Chest 2001; 119:731–736. [CSA], [CROSSREF]
- Lacasse Y, Ferreira I, Brooks D, Newman T, Goldstein R S. Critical appraisal of clinical practice guidelines targeting chronic obstructive pulmonary disease.Arch Intern Med Jan. 8, 2001; 161(1):69–74. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]