Abstract
Chronic obstructive pulmonary disease (COPD) is characterized by high cardiovascular risk, which is further amplified during acute COPD exacerbations (AECOPD). Endothelial dysfunction has been previously suggested as one of the potential pathogenetic mechanisms. In order to study the effects of AECOPD on endothelial function assessed by available functional methods, we performed a literature search involving Pubmed and Scopus databases. Eligible studies were those that included adult patients with COPD and evaluated endothelial damage via semi-invasive or noninvasive functional methods, during AECOPD and after recovery or in stable condition. Newcastle-Ottawa Scale was applied to evaluate the quality of retrieved studies. Endothelial function was significantly impaired during AECOPD compared to recovery/stable condition (SMD: −0.87, 95%CI [−1.19, −0.55]). Patients during AECOPD presented a significantly worse response in endothelium-dependent (flow-mediated dilatation WMD: −2.59, 95%CI [−3.75, −1.42]) and independent vasodilation (nitroglycerine-mediated dilatation WMD: −3.13, 95%CI [−5.18, −1.09]) compared to recovery. Sensitivity analyses confirmed the above results. In conclusion, endothelium-dependent and independent vasodilation is worse during AECOPD compared to the stable condition. Endothelial dysfunction could play a role in the high cardiovascular risk during AECOPD.
Introduction
During the last decades, morbidity and mortality rates of chronic obstructive pulmonary disease (COPD) have risen exponentially and according to the World Health Organization, it is now considered to be the third leading cause of death worldwide [Citation1]. Concurrent cardiovascular disease (CVD) plays a central role in COPD prognosis, as it is highly prevalent and it is associated with long-term mortality in this population [Citation2]. Patients with COPD frequently have unhealthy habits and co-morbid conditions that are established risk factors of CVD, including smoking, hypertension, obesity and diabetes mellitus [Citation3,Citation4]. Apart from the above, a number of underlying pathogenetic mechanisms such as hypoxia, systemic inflammation, oxidative stress, arterial stiffness and endothelial dysfunction are suggested to play important roles in the relationship between COPD and CVD [Citation5].
Endothelial dysfunction is characterized by an imbalance between endothelium-derived relaxing factors and endothelium-derived contracting factors [Citation6]. Several animal and human studies suggest that endothelial dysfunction is linked through complex associations, in some cases of bi-directional nature, with the aforementioned major cardiovascular risk factors [Citation7–10]. In addition, endothelial dysfunction is shown to be independently associated with cardiovascular events and mortality in the general population [Citation11] and various patient groups, including patients with hypertension, diabetes mellitus and coronary artery disease [Citation12–14]. Accumulated evidence suggests that the degree of endothelial dysfunction in patients with COPD is related to COPD severity [Citation15]. Previous works in the field demonstrated that stable COPD patients had markedly declined endothelium-dependent and endothelium-independent vasodilation of the brachial artery compared to smoking and nonsmoking healthy controls, and that this decline was proportionally associated with the degree of airway obstruction [Citation16,Citation17]. Apart from impaired endothelial function in the conduit arteries, COPD individuals showed to have damaged endothelium in the microvasculature, that was again related to disease severity [Citation18]. Of note, preliminary evidence suggests that endothelial function in COPD patients significantly decreases over time [Citation19].
AECOPD are a major source of morbidity and mortality in COPD [Citation20]. Several studies demonstrated that AECOPD increase the risk of acute coronary syndromes, cardiovascular and all-cause mortality [Citation21–23]. Among the main factors that were previously suggested to be pathogenetically involved in the increased risk of cardiovascular events and death during AECOPD, are acute deteriorations of systemic inflammation, hypoxia and endothelial injury [Citation3]. In this regard, it was shown that markers of endothelial injury (e.g. microalbuminuria, fibrinogen, endothelin-1, etc.) increase during AECOPD and this is related to AECOPD severity [Citation24,Citation25]. Furthermore, recent studies tried to examine whether endothelial function deteriorates during AECOPD, however with non-uniform results. Thus, the aim of this systematic review and meta-analysis is to investigate endothelial function during AECOPD versus stable condition of COPD patients, including studies evaluating endothelial impairment with available functional methods.
Materials and methods
This systematic review and meta-analysis was conducted in accordance with the Preferred-Reporting-Items-for-Systematic Reviews-and-Meta-Analyses (PRISMA) and the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines. All research was conducted according to a protocol submitted in PROSPERO database (CRD42021225807).
Search strategy and eligibility criteria
Two authors (MT, DB) independently searched in two major databases (PubMed/MEDLINE and Scopus) from database inception up to 25 November 2020, using a combination of free text terms and relevant Medical Subject Headings (MeSH). An example of our search strategy and the keywords used are presented in Table 1, supplementary material. Reference lists of retrieved articles and reports, including relevant reviews and meta-analyses, were scrutinized for other potentially relevant articles. We searched for observational studies (cohorts, case-controls and cross-sectional studies) assessing endothelial function in COPD patients during AECOPD and at recovery/stable condition. All types of semi-invasive and noninvasive functional methods based on Doppler ultrasound, plethysmography, Laser Doppler, Near-infrared spectroscopy (NIRS) or novel, based on optical coherence techniques for assessment of endothelial damage [flow-mediated dilatation (FMD), nitroglycerine-mediated dilatation (NMD), peripheral arterial tonometry (PAT), laser-doppler flowmetry (LDF), venous occlusion plethysmography (VOP), laser-speckle contrast imaging/analysis (LSCI/LASCA), nailfold capillaroscopy, NIRS, arterial glycocalyx] were included. The search strategy was developed with English language restriction. Preclinical studies, studies with non-adult patients, studies evaluating endothelial dysfunction via biomarkers or invasive methods, and studies comparing endothelial function during acute exacerbation to healthy controls were excluded.
Study selection and data extraction
All retrieved studies were imported into reference management software for duplicate removal. Two reviewers (MT, DB) screened title/abstracts of records retrieved throughout search and subsequently performed independently full text assessment to identify eligible studies. A data extraction form designed according to the Cochrane checklist of Items, containing fields for all important data on study design, demographics, outcome measurements and details relevant to quality assessment was completed for each eligible study by the two authors (MT, DB). All disagreements on study selection and data collection were solved by a third senior reviewer (MA). In case of missing data, study authors were contacted by e-mail to try to retrieve original data.
Quality assessment tool
Assessment of the quality of the eligible studies was performed independently by two reviewers (MT, DB) according to Newcastle-Ottawa-Scale (NOS) for case-control studies, a tool developed for quality assessment of non-randomized studies [Citation26]. NOS is a 9-point scale that involves the appraisal of methodological issues and their reporting. The scoring system encompasses three major domains (participant selection, group comparability and ascertainment of exposure); scores range from 0 to 9, with scores ≥7 indicating high quality studies (Table 2, supplementary material).
Statistical analyses
Data were analyzed using Review Manager (RevMan) Version 5.3. Endothelial function is reported as a continuous outcome, based on the respective method used for the assessment. For studies assessing endothelial function using the same method, the weighted mean between-group difference (WMD) was calculated with pertinent 95% confidence intervals (95%CI) when data were expressed in the same measurement scale (i.e. proportional change from baseline). When pooling all available data from all types of assessment, the respective standardized-mean-difference (SMD, with 95%CI) was used. For the total of the studies, subgroup analysis was performed based on a) hospitalization and b) the presence of overt CVD [(i.e. coronary artery disease (CAD), peripheral arterial disease (PAD), stroke]. Finally, we planned to explore robustness of our findings by means of a sensitivity analysis excluding studies judged as of poor quality (NOS < 7). For studies reporting median and range or interquartile range values, we calculated mean and SDs values based on relevant formulas [Citation27].
We evaluated statistical heterogeneity across studies using the Cochran’s Q-test (p<0.1 indicating existence of heterogeneity) along with the I2 statistic (with a result >50% suggesting significant heterogeneity). Funnel plot of all studies assessing endothelial function was examined for presence of asymmetry. The random-effects meta-analytic model was used to combine our data, due to the existence of clinical and methodological high between-study heterogeneity; the inverse variance method was used to estimate study weights.
Results
Search results and study characteristics
Study selection process is presented as flow diagram in . The initial searches identified in total 1,040 reports; after removing duplicates (n = 390), studies were screened at a title/abstract level and 696 were rejected. Overall, 5 studies enrolling totally 279 patients met inclusion/exclusion criteria and were included in this systematic review [Citation28–32]. From those, only 4 studies (with 111 participants) [Citation28–30,Citation32] were included in the quantitative analysis, since available data for 1 study were inadequate [Citation31]. Authors were contacted by email requesting supplemental data, with none of them responding.
Of the 5 studies included in this systematic review, 3 studies evaluated endothelial function via FMD/NMD [Citation28,Citation30,Citation32] and 2 via PAT [Citation29,Citation31]. Two studies included patients who required hospitalization for AECOPD [Citation30,Citation32]. shows the characteristics of the included studies.
Table 1. Studies included in this systematic review.
Quality assessment
NOS for case-control studies was used to assess the quality of retrieved studies. The overall study quality assessment for studies included in this analysis is depicted in Table 2, supplementary material. According to the NOS score, 3 studies where classified as high quality (NOS ≥ 7) [Citation28,Citation29,Citation32] and the remaining 1 study as low quality [Citation30].
Publication bias
As presented in Figure 1, supplementary material, a light asymmetry in the funnel plot suggests that some small studies with non-significant results might be missing, indicating that the possibility of publication bias could not be excluded.
Endothelial function assessment
Across 3 studies evaluating endothelial function by measuring FMD of the brachial artery, a significantly lower endothelium-dependent vasodilatation of WMD −3.20% (95%CI −4.35 to −2.06) was observed in patients during AECOPD, compared to patients in recovery/stable condition, with very low heterogeneity across studies (I2 = 0%, p %, ss 3 s ).
Figure 2. Forest plots of the difference in (A) FMD%, (B) RHI (PAT), among patients during AECOPD and patients at recovery/stable condition. AECOPD, acute exacerbation of chronic obstructive pulmonary disease; FMD, flow-mediated dilatation; RHI, reactive hyperemia index; PAT, peripheral arterial tonometry.
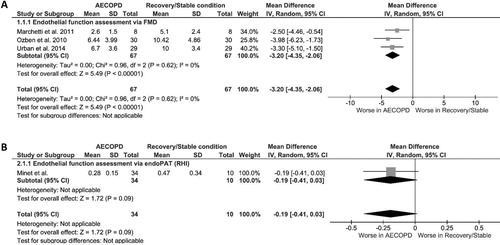
In the one study explored endothelial function via PAT in COPD patients during AECOPD and recovery, a marginally significant lower reactive hyperemia index (RHI) in the former was showed ().
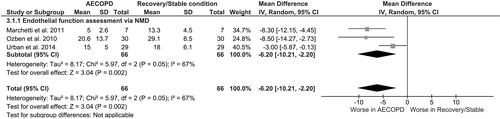
Similarly to FMD, across 3 studies evaluating endothelium-independent vasodilation by the use of NMD, a significantly worse response by WMD −6.20% (95%CI −10.21 to −2.20) was observed in COPD patients during AECOPD compared to recovery/stable condition, with moderate heterogeneity (I2 = 67%, p 7%, arly t ).
Endothelial function assessment via all methods (pooled analysis)
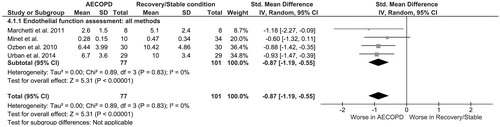
When pooling all studies together, regardless of the type of method used for assessment of vascular reactivity, more pronounced endothelial dysfunction was observed in AECOPD compared to recovery/stable condition (SMD: −0.87, 95%CI −1.19 to −0.55), with very low heterogeneity (I2 = 0%, p %, pooli ).
Subgroup analysis
In order to explore the heterogeneity across the included studies, subgroup analysis comparing endothelial function according to hospitalization during AECOPD was performed (Figure 2, supplementary material). No significant differences between the two study subgroups were observed.
A second subgroup analysis including only studies enrolling patients without overt cardiovascular disease is presented in Figure 3, supplementary material. As depicted in the Figure, in the 2 studies including participants without overt cardiovascular disease, a pronounced endothelial function was again worse during AECOPD compared to recovery/stable condition (WMD: −2.93, 95%CI −4.26 to −1.61), with very low heterogeneity (I2 = 0%, p %, depic Figure 3, supplementary material).
Sensitivity analysis
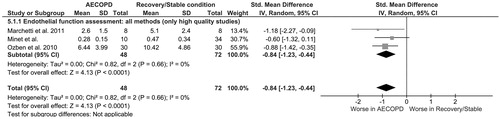
We repeated the main analysis by including only the high quality studies (NOS score ≥ 7) in order to explore the robustness of our findings. Of interest, after excluding one study classified as of low quality, presence of a similarly impaired endothelial function in ΑΕCOPD compared to recovery/stable condition was confirmed (SMD: −0.84, 95%CI [−1.23, −0.44]) ().
Figure 5. Sensitivity analysis (included studies with NOS score ≥7): Forest plot of the difference in endothelial function among patients during AECOPD and patients at recovery/stable condition (all methods). AECOPD, acute exacerbation of chronic obstructive pulmonary disease; NOS, Newcastle-Ottawa-Scale.
Discussion
This is the first systematic review of the assessment of peripheral endothelial function in COPD patients during exacerbation, indicating that endothelial function is significantly worse during AECOPD compared to recovery/stable condition. Both endothelium-dependent, but mainly endothelium-independent vasodilation of the brachial artery, are markedly decreased during AECOPD, suggesting a combination of impaired endothelial and vascular smooth cell function. These observations strengthen the aspect that endothelial damage is a significant factor contributing to the increased cardiovascular risk during or immediately after the AECOPD.
Endothelial dysfunction is a systemic disorder characterized by an imbalance between factors with vasodilating, antimitogenic, and antithrombogenic properties (endothelium-derived relaxing factors) and factors with vasoconstricting, prothrombotic, and proliferative properties (endothelium-derived contracting factors) [Citation33]. Several functional methods and biomarkers have been utilized to assess endothelial damage [Citation6], but FMD and PAT are the most commonly used in COPD [Citation31]. Flow-mediated dilatation is a nitric oxide (NO)-mediated response of the vascular walls after a blood flow occlusion period (reactive hyperemia) that reflects endothelial function [Citation6,Citation33]; on the other hand, NMD reflects endothelium-independent function, that is the relaxation ability of vascular smooth muscle [Citation34]. Both FMD and NMD are strong predictors of cardiovascular outcomes [Citation33,Citation35].
Several studies showed that the severity of FMD and NMD impairment is associated with inflammatory markers in stable COPD [Citation36]. AECOPD is characterized by intensification of inflammation and oxidative stress, hypoxia and sympathetic nervous system overdrive [Citation37]; all these factors are known to significantly affect endothelial function. In this meta-analysis, we demonstrated that endothelium-dependent vasodilation is markedly reduced during AECOPD compared to recovery/stable condition, suggesting impaired NO-bioavailability and endothelial dysfunction. Moreover, pooled analysis of NMD showed that patients suffering from AECOPD have significantly decreased endothelium-independent vasodilation, indicating not only the presence of endothelial damage, but also an additional impairment of vascular smooth muscle sensitivity to exogenous NO [Citation35]. It should be noted here, that concurrence of acute hypercapnia may, theoretically, have impaired the vascular capacity to respond to stimuli such FMD/NMD and confound the study results [Citation38]; however, as noted in the primary studies, either PaCO2 levels were within normal limits [Citation30] or there were not observed differences between baseline (during AECOPD) and recovery brachial artery diameter, making it unlikely that the impaired FMD and NMD was due to vasodilatation from hypercapnia [Citation28,Citation32].
PAT is a noninvasive, operator-independent technique assessing changes in pulse wave amplitude before and after reactive hyperemia. RHI, an indicator of endothelial function and vascular reactivity in the microvasculature, is a more modern index used to assess vascular function in populations at high cardiovascular risk [Citation34], and is strongly correlated with classical cardiovascular risk factors in various populations [Citation33]. In stable COPD, RHI is significantly lower compared to healthy individuals [Citation18] and is associated with worse exercise capacity [Citation39]. In the prospective study of Minet et al. [Citation29], approximately 50% of study participants presented markedly impaired endothelial function (RHI ≤ 0.40), that deteriorated further during AECOPD [Citation29]. In contrast with the aforementioned observations, in a prospective study in the field which investigated the associations of endothelial dysfunction measured by PAT with disease severity and adverse outcomes, no differences in endothelial function was noted between AECOPD and stable condition in a subgroup of participants (n . In contrast with the aforemenCitation31].
Accumulated evidence suggested that severe AECOPD requiring hospital management is associated with increased mortality [Citation40,Citation41], independently of other classical prognostic factors (i.e. age, FEV1, BMI) [Citation41]. Indeed, in a previous study from Soler-Cataluña et al. increased mortality was particularly observed in patients with AECOPD requiring hospitalization and less so in patients requiring emergency visits without admission to hospital; inflammation triggered during AECOPD was suggested as the main underlying mechanism [Citation41]. Interestingly, the present analysis demonstrated that even less severe AECOPD (not requiring hospitalization) carries also some important risks, as it is also associated with impaired endothelial function compared to stable/recovery condition. However, larger case-control studies are needed in the future to confirm these interesting observations.
Our meta-analysis has several limitations that have to be acknowledged. First, there was considered to be high clinical heterogeneity across the included studies; we attempted to minimize the extent that it might affect our results by using the random-effects model and by performing subgroup analyses. Second, it included only 4 studies, but to our knowledge, the present systematic review and meta-analysis is the first work examining endothelial function during AECOPD; moreover, it followed a careful literature search, including all the available functional methods for endothelial damage assessment and a rigorous methodology. Fourth, our search was restricted in English-language journals, hence results from small studies published in non-English journals might have been missed. Finally, although we extensively tried to retrieve missing data by contacting authors of the primary studies, we could not use data from a few studies due to missing values.
In conclusion, the present meta-analysis showed that COPD patients have significantly impaired endothelium-dependent and endothelium-independent vasodilation during AECOPD compared to the stable condition. Given that endothelial dysfunction is independently associated with cardiovascular events in various populations, as well as that the incidence of cardiovascular events is higher during AECOPD, these observations suggest that endothelial damage might be an important factor contributing to the increased cardiovascular risk during or immediately after the AECOPD. Future studies should further delineate these complex interactions for the benefit of COPD patients.
Declaration of interest
All authors disclose that they do not have any financial or other relationships which might lead to a conflict of interest
References
- WHO. Causes of death [Internet]. [cited 2020 Nov 24]. Available from: https://www.who.int/data/maternal-newborn-child-adolescent/monitor.
- Sin DD, Man SFP. Why are patients with chronic obstructive pulmonary disease at increased risk of cardiovascular diseases? The potential role of systemic inflammation in chronic obstructive pulmonary disease. Circulation. 2003;107(11):1514–1519. DOI:https://doi.org/10.1161/01.cir.0000056767.69054.b3
- Brassington K, Selemidis S, Bozinovski S, et al. New frontiers in the treatment of comorbid cardiovascular disease in chronic obstructive pulmonary disease. Clin Sci (Lond). 2019;133(7):885–904. DOI:https://doi.org/10.1042/CS20180316
- Barnes PJ, Celli BR. Systemic manifestations and comorbidities of COPD. Eur Respir J. 2009;33(5):1165–1185. DOI:https://doi.org/10.1183/09031936.00128008
- Morgan AD, Zakeri R, Quint JK. Defining the relationship between COPD and CVD: what are the implications for clinical practice? Ther Adv Respir Dis. 2018;12:1753465817750524. DOI:https://doi.org/10.1177/1753465817750524
- Theodorakopoulou MP, Schoina M, Sarafidis P. Assessment of endothelial and microvascular function in CKD: older and newer techniques, associated risk factors, and relations with outcomes. Am J Nephrol. 2020;51(12):931–949. doi: https://doi.org/10.1159/000512263.
- Boulanger CM. Secondary endothelial dysfunction: hypertension and heart failure. J Mol Cell Cardiol. 1999;31(1):39–49. DOI:https://doi.org/10.1006/jmcc.1998.0842
- Sarafidis PA, Bakris GL. Review: insulin and endothelin: an interplay contributing to hypertension development? J Clin Endocrinol Metab. 2007;92(2):379–385. DOI:https://doi.org/10.1210/jc.2006-1819
- Wilkes JJ, Hevener A, Olefsky J. Chronic endothelin-1 treatment leads to insulin resistance in vivo. Diabetes. 2003;52(8):1904–1909. DOI:https://doi.org/10.2337/diabetes.52.8.1904
- Messner B, Bernhard D. Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb Vasc Biol. 2014;34(3):509–515. DOI:https://doi.org/10.1161/ATVBAHA.113.300156
- Shechter M, Issachar A, Marai I, et al. Long-term association of brachial artery flow-mediated vasodilation and cardiovascular events in middle-aged subjects with no apparent heart disease. Int J Cardiol. 2009;134(1):52–58. DOI:https://doi.org/10.1016/j.ijcard.2008.01.021
- Perticone F, Ceravolo R, Pujia A, et al. Prognostic significance of endothelial dysfunction in hypertensive patients. Circulation. 2001;104(2):191–196. DOI:https://doi.org/10.1161/01.cir.104.2.191
- Bruzzi P, Predieri B, Patianna VD, et al. Longitudinal evaluation of endothelial function in children and adolescents with type 1 diabetes mellitus: a long-term follow-up study. Pediatr Int. 2014;56(2):188–195. DOI:https://doi.org/10.1111/ped.12220
- Fichtlscherer S, Breuer S, Zeiher AM. Prognostic value of systemic endothelial dysfunction in patients with acute coronary syndromes: further evidence for the existence of the “vulnerable” patient. Circulation. 2004;110(14):1926–1932. DOI:https://doi.org/10.1161/01.CIR.0000143378.58099.8C
- Polverino F, Celli BR, Owen CA. COPD as an endothelial disorder: endothelial injury linking lesions in the lungs and other organs? (2017 Grover Conference Series). Pulm Circ. 2018;8(1):2045894018758528. DOI:https://doi.org/10.1177/2045894018758528
- Ambrosino P, Lupoli R, Iervolino S, et al. Clinical assessment of endothelial function in patients with chronic obstructive pulmonary disease: a systematic review with meta-analysis. Intern Emerg Med. 2017;12(6):877–885. DOI:https://doi.org/10.1007/s11739-017-1690-0
- Vaes AW, Spruit MA, Theunis J, et al. Endothelial function in patients with chronic obstructive pulmonary disease: a systematic review of studies using flow mediated dilatation. Expert Rev Respir Med. 2017;11:1–1031. DOI:https://doi.org/10.1080/17476348.2017.1389277
- Malerba M, Radaeli A, Nardin M, et al. Endothelial dysfunction assessment by noninvasive peripheral arterial tonometry in patients with chronic obstructive pulmonary disease compared with healthy subjects. Clin Respir J. 2018;12(4):1466–1472. DOI:https://doi.org/10.1111/crj.12686
- Clarenbach CF, Sievi NA, Kohler M. Annual progression of endothelial dysfunction in patients with COPD. Respir Med. 2017;132:15–20. DOI:https://doi.org/10.1016/j.rmed.2017.09.005
- Donaldson GC, Wedzicha JA. COPD exacerbations .1: epidemiology. Thorax. 2006;61(2):164–168. DOI:https://doi.org/10.1136/thx.2005.041806
- Zvezdin B, Milutinov S, Kojicic M, et al. A postmortem analysis of major causes of early death in patients hospitalized with COPD exacerbation. Chest. 2009;136(2):376–380. DOI:https://doi.org/10.1378/chest.08-2918
- Donaldson GC, Hurst JR, Smith CJ, et al. Increased risk of myocardial infarction and stroke following exacerbation of COPD. Chest. 2010;137(5):1091–1097. DOI:https://doi.org/10.1378/chest.09-2029
- Campo G, Guastaroba P, Marzocchi A, et al. Impact of COPD on long-term outcome after ST-segment elevation myocardial infarction receiving primary percutaneous coronary intervention. Chest. 2013;144(3):750–757. DOI:https://doi.org/10.1378/chest.12-2313
- Polatli M, Cakir A, Cildag O, et al. Microalbuminuria, von Willebrand factor and fibrinogen levels as markers of the severity in COPD exacerbation. J Thromb Thrombolysis. 2008;26(2):97–102. DOI:https://doi.org/10.1007/s11239-007-0073-1
- Roland M, Bhowmik A, Sapsford RJ, et al. Sputum and plasma endothelin-1 levels in exacerbations of chronic obstructive pulmonary disease. Thorax. 2001;56(1):30–35. DOI:https://doi.org/10.1136/thorax.56.1.30
- Herzog R, Álvarez-Pasquin MJ, Díaz C, et al. Are healthcare workers' intentions to vaccinate related to their knowledge, beliefs and attitudes? A systematic review. BMC Public Health. 2013;13:154. DOI:https://doi.org/10.1186/1471-2458-13-154
- Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5(1):13. DOI:https://doi.org/10.1186/1471-2288-5-13
- Ozben B, Eryüksel E, Tanrikulu AM, et al. Acute exacerbation impairs endothelial function in patients with chronic obstructive pulmonary disease. Turk Kardiyoloji Dernegi arsivi. Turk Kardiyoloji Derneginin Yayin Organidir. 2010;38:1–7.
- Minet C, Vivodtzev I, Tamisier R, et al. Reduced six-minute walking distance, high fat-free-mass index and hypercapnia are associated with endothelial dysfunction in COPD. Respir Physiol Neurobiol. 2012;183(2):128–134. DOI:https://doi.org/10.1016/j.resp.2012.06.017
- Urban MH, Valipour A, Kiss D, et al. Soluble receptor of advanced glycation end-products and endothelial dysfunction in COPD. Respir Med. 2014;108(6):891–897. DOI:https://doi.org/10.1016/j.rmed.2014.03.013
- Scherr A, Schumann DM, Karakioulaki M, et al. Endothelial dysfunction is not a predictor of outcome in chronic obstructive pulmonary disease. Respir Res. 2020;21(1):90. DOI:https://doi.org/10.1186/s12931-020-01345-9
- Marchetti N, Ciccolella DE, Jacobs MR, et al. Hospitalized acute exacerbation of COPD impairs flow and nitroglycerin-mediated peripheral vascular dilation. COPD. 2011;8(2):60–65. DOI:https://doi.org/10.3109/15412555.2011.558541
- Flammer AJ, Anderson T, Celermajer DS, et al. The assessment of endothelial function: from research into clinical practice. Circulation. 2012;126(6):753–767. DOI:https://doi.org/10.1161/CIRCULATIONAHA.112.093245
- Lekakis J, Abraham P, Balbarini A, et al. Methods for evaluating endothelial function: a position statement from the European Society of Cardiology Working Group on Peripheral Circulation. Eur J Cardiovasc Prev Rehabil. 2011;18(6):775–789. DOI:https://doi.org/10.1177/1741826711398179
- Maruhashi T, Kihara Y, Higashi Y. Assessment of endothelium-independent vasodilation: from methodology to clinical perspectives. J Hypertens. 2018;36(7):1460–1467. DOI:https://doi.org/10.1097/HJH.0000000000001750
- Eickhoff P, Valipour A, Kiss D, et al. Determinants of systemic vascular function in patients with stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2008;178(12):1211–1218. DOI:https://doi.org/10.1164/rccm.200709-1412OC
- Vukic Dugac A, Ruzic A, Samarzija M, et al. Persistent endothelial dysfunction turns the frequent exacerbator COPD from respiratory disorder into a progressive pulmonary and systemic vascular disease. Med Hypotheses. 2015;84(2):155–158. DOI:https://doi.org/10.1016/j.mehy.2014.11.017
- de Matthaeis A, Greco A, Dagostino MP, et al. Effects of hypercapnia on peripheral vascular reactivity in elderly patients with acute exacerbation of chronic obstructive pulmonary disease. Clin Interv Aging. 2014;9:871–878.
- Vaes AW, Spruit MA, Theunis J, et al. Peripheral endothelial function is positively associated with maximal aerobic capacity in patients with chronic obstructive pulmonary disease. Respir Med. 2018;142:41–47. DOI:https://doi.org/10.1016/j.rmed.2018.07.013
- Almagro P, Calbo E, Ochoa de Echagüen A, et al. Mortality after hospitalization for COPD. Chest. 2002;121(5):1441–1448. DOI:https://doi.org/10.1378/chest.121.5.1441
- Soler-Cataluña JJ, Martínez-García MA, Román SP, et al. Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax. 2005;60(11):925–931. DOI:https://doi.org/10.1136/thx.2005.040527