Abstract
The distinct and overlapping roles of adhesion molecules belonging to the selectin and integrin families control the rate of leukocyte adhesion to stimulated vascular endothelial cells under hydrodynamic shear flow. Crystal structures have appeared for some of these interactions which complement molecular biology experiments, and clarify the molecular mechanism of the receptor-ligand binding interactions. Binding affinity data have also appeared using surface plasmon resonance and single-molecule biophysics experiments. These studies confirm and extend the predictions of previous experiments carried out in parallel-plate flow chambers, and cone and plate viscometers. This review discusses the current state of understanding on how molecular bond formation rates coupled with cellular and hydrodynamic features regulate leukocyte binding to endothelial cells.
INTRODUCTION
Cell adhesion molecules belonging to the selectin and integrin families facilitate the localization of leukocytes at sites of inflammation and injury (Citation25). These molecules also play an important role in leukocyte trafficking (Citation83), and they mediate the formation of homotypic and heterotypic cellular aggregates in suspension (Citation78). Three members of the selectin family expressed on blood leukocytes, platelets and the vascular endothelium contribute to cell adhesion (Citation34, 40, 89). These are L-selectin (also called LECAM-1, LAM-1, MEL-14, TQ-1, Leu-8, CD62L), E-selectin (ELAM-1, CD62E), and P-selectin (PADGEM, GMP-140, CD62P). Among the leukocyte integrins, members of the β1, β2, and β7 family have been shown to play a role in leukocyte adhesion (Citation29, 83).
While the classical paradigm of leukocyte interaction suggests that selectin mediated rolling on the vascular endothelial cell monolayer precedes cell activation and integrin mediated firm arrest (Citation42, 90), important exceptions have been noted. In particular, the α4-integrins on lymphocytes have been shown to support rolling under physiological flow conditions (Citation4, 7). In addition, knock-out mice experiments lacking the β2 integrin subunits (either LFA-1 [αLβ2], Mac-1 [αMβ2], or the common CD18/β2-integrin), suggest that these integrin subunits contribute to stabilizing cell rolling (Citation11, 27, 32) (). This contribution of CD18 integrins to rolling is supported by in vitro observations, which demonstrate that human neutrophils roll faster on stimulated Human Umbilical Vein Endothelial Cells (HUVECs) upon blocking the CD18 integrins (Citation21). Selectin independent cell adhesion has also been observed at low shear rates (below 400 s−1) during neutrophil homotypic aggregation experiments (Citation85) and in runs where stimulated neutrophils bound ICAM-1 transfected cells in suspension (Citation53).
1 Quantifying the frequency of rolling and arrest: Multistep pathway shows the overlapping and distinct roles of selectins and integrins in leukocyte-endothelial cell adhesion. It is possible to quantify the roles of selectins and integrins in terms of “efficiencies” or “frequencies” (Citation95, 96). The distance from the point the cell contacts the ligand bearing substrate to the point of capture is a measure of selectin on-rate. θfr or primary capture frequency is inversely related to this distance. θcc or cell-cell capture probability quantifies the fraction of collisions between cells in the free stream and previously recruited substrate-bound cells that result in secondary capture. As cells progress from capture to firm arrest there is progressive activation of integrins, which results in the reduction of cell rolling velocity. θra or firm-arrest frequency is inversely related to the distance taken for a cell to transition from rolling to arrest. θTM is a measure of the average time taken for transmigration. In addition to selectin molecular on/off-rates and integrin activation levels, cellular properties like cell-surface microvilli extension and deformation also play a prominent role in regulating cell rolling.
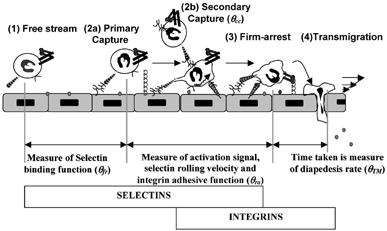
The overlapping roles of selectins and integrins suggest that there is a gradual shift in the molecular requirements as leukocytes transition from the free stream to initiate tethering, rolling and firm arrest. These changes in molecular requirements may be both due to the structural features controlling receptor-ligand affinities and other factors, like receptor number, topography and cytoskeletal attachment, which influence receptor-ligand avidity. In addition, since molecular recognition takes place under fluid flow conditions, an interplay between fluid hydrodynamic forces and adhesion molecule properties controls the rates of leukocyte-endothelial cell binding. The current review examines how this interplay may contribute to the functions of the selectins and integrins during leukocyte localization. Only selected recent results are reviewed in detail in this article, since a comprehensive discussion of all literature in this field is beyond the scope of a single study.
SELECTIN AND INTEGRIN LIGANDS
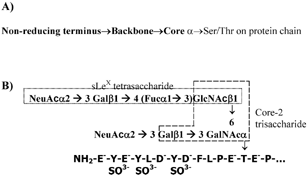
The structure of selectins and selectin-ligands are reviewed in detail elsewhere (Citation34, 40, 89). Briefly, all members of the selectin family of adhesion molecules consist of a common calcium-dependent C-type lectin domain followed by an epidermal growth factor (EGF) domain, variable numbers of short consensus repeats (SCRs), a transmembrane, and a cytoplasmic domain. Selectins bind diverse mucinous glycoproteins including Glycosylation-dependent Cell Adhesion Molecule-1 (GlyCAM-1), Mucosal Addressin Cell Adhesion Molecule-1 (MAdCAM-1), CD34, endoglycan, E-selectin ligand-1 (ESL-1), P-Selectin Glycoprotein Ligand-1 (PSGL-1, CD162), and CD24. Recently, P-selectin has also been shown to anchor newly released ultralarge von Willebrand factor multimers on endothelial cell surfaces (Citation58). While many of these ligands bear O-linked glycans displayed on a core-2 structure () (Citation88), N-glycans have also been implicated to bind the selectins especially in the case of E-selectin (Citation84). Among the carbohydrate structures on these glycosylated proteins, the tetrasaccharide sialyl Lewis-X [sLeX, NeuAcα2-3Galβ1-4(Fucα1-3)GlcNAc] located at the terminal regions of glycans and related molecules has been implicated to recognize the lectin domain of the selectins in a calcium dependent fashion (Citation22). Glycolipids expressing sialyl Lewis-X at high densities have also been shown to support leukocyte rolling via selectins. While it is thought that the EGF domain does not directly recognize the carbohydrate ligands of selectins (Citation81), mutation studies of the EGF-domain demonstrate altered selectin-ligand recognition perhaps through an allosteric effect (Citation35). Sulfation is also thought to be important for selectin recognition. Sulfation occurs at the 6-position of Gal and/or GlcNac in some of the carbohydrate ligands especially those that bind L-selectin (Citation26, 33). Of these, sulfation at the 6-positon of GlcNac in sLeX, which results in the 6-sulfo sLeX moiety, binds L-selectin with higher affinity than does sLeX or 6′-sulfo sLeX (Citation68). Sulfation of tyrosine residues on the peptide chain is considered to be critical for some receptor-ligand recognition, especially for P-selectin and L-selectin binding to PSGL-1 under shear (Citation44, 62, 65)
2 O-glycans that act as selectin-ligands: (A) The complex chains of O-linked sugars found on mucinous glycoproteins consist of three distinct regions: core, backbone and non-reducing terminus. Here, the core structure forms the critical link between the protein chain and the sugar backbone. Among the known core structures, the core-2 structure Galβ1→3(GlcNAcβ1→6)GalNAcα-Ser/Thr, is prominent in selectin ligands (Citation14). The natural ligands for selectins including PSGL-1, CD34 and GlyCAM-1 bear core-2 sugars. (B) The carbohydrate structure of PSGL-1 that is thought to represent a physiological selectin ligand is di-sialylated at the non-reducing terminus and lacks a distinct backbone (Citation43). It is located near the N-terminus of the PSGL-1 peptide chain, which has sulfated tyrosine residues. The core-2 and sialyl Lewis-X structures are shown.
InterCellular Adhesion Molecule (ICAM-1, -2 and -3) and Vascular Cell Adhesion Molecule (VCAM), members of the Immunoglobulin supergene family (IgSF), act as prominent ligands for the integrins (Citation83). Recently, a family of molecules called Junctional Adhesion Molecules (JAM) that were previously implicated in cell-cell junction formation has also been shown to bind integrins (Citation13). It is suggested that JAM-1 is a counter-receptor for LFA-1 that is ideally situated to guide and control transmigration during leukocyte recruitment (Citation57). JAM-2 interacts with the VLA-4 antigen (Citation10). JAM-3 on human platelets is a counter receptor for the leukocyte integrin Mac-1 and mediates leukocyte-platelet interactions (Citation69). This molecule also acts as a counter-receptor for JAM-2 (Citation5). Additional molecules that recognize the β2− integrin subunit Mac-1 include iC3b, factor X, fibrinogen and serum albumin.
STRUCTURAL BASIS OF SELECTIN AND INTEGRIN BINDING
Detailed studies of the structural basis of selectin-carbohydrate recognition have been undertaken in various laboratories. These include studies of the crystal structure of selectins (Citation22, 81). These studies suggest that the binding of selectins by their ligands requires a network of few, but optimally disposed, interactions that include electrostatic interactions, hydrogen bonds, and hydrophobic forces. While the binding of sLeX to P-/E-selectin results in a contact area between the receptor and ligand of 501–549 Å2, the binding of the entire PSGL-1 (the peptide chain with the carbohydrate) to P-selectin excludes an area of 1641 Å2. It is suggested that L-, E- and P-selectin have common conserved residues in the binding pocket surrounding the calcium moiety in the lectin domain. Thus, all three selectins appear to recognize fucose on the sLeX moiety in a common fashion, and this may be the reason why all physiologically relevant selectin ligands identified to date are fucosylated. All of the selectins also have a conserved Tyrosine at the 94 position and Glutamic acid at the 92 position that binds Galactose. Distinct differences are however observed in the motif that binds the sialic acid residue. While this motif consists of a Arg97-Glu98-Lys99-Asp100 in E-selectin, it is replaced by Ser97-Pro98-Ser99-Ala100 in P-selectin and Arg97-Asn98-Lys99-Asp100 in L-selectin. Differences in this sequence may contribute to the distinct affinities of each of the selectins for sLeX. These structural differences may also contribute to the ability of some selectins to bind sulfated oligosaccharides in addition to sLeX. GlcNAc does not form a contact with the selectin in this model. Additional differences in the amino acid sequences that mediate tyrosine sulfate recognition during PSGL-1 binding to P-selectin versus E-selectin may account for the strict requirement of tyrosine sulfation in P-selectin but not E-selectin recognition (Citation45, 67).
The ability of integrins to bind their ligand has been attributed to molecular conformational changes, along with alterations in the pattern of adhesion molecule clustering (Citation29, 46, 47). The crystal structure of the recombinant I domain of the α-subunit of LFA-1 complexed with ICAM-1 reveals that 1250 Å2 of solvent accessible surface is buried in the I domain:ICAM-1 contact (Citation76). The area of contact is small and comparable to that of selectin-ligand interactions. In this study, compared to the unliganded “closed” conformation of the I domain, the “open” conformation exhibits a large 10 Å movement of the C-terminal α helix down the side of the domain, and a rearrangement in metal coordination at the metal ion-dependent adhesion site (MIDAS). Ligand binding involves the interaction of a Glutamic acid residue on the ICAM-1 domain 1 with the Mg2+ ion held in the LFA-1 I domain MIDAS sequence. Surrounding this Mg2+ ion are a ring of hydrophobic residues on the I domain that contact with similar hydrophobic residues on ICAM-1. Surrounding the hydrophobic ring are polar interactions involving hydrogen bonds and salt bridges. These interactions orient ICAM-1 for optimal contact with the I domain. Associated with these changes is a dramatic reorientation of a residue Glu-241 in the binding pocket, which enables the formation of a critical salt bridge with ICAM-1.
DIVERSE EXPERIMENTAL SYSTEMS APPLIED TO STUDY SELECTIN AND INTEGRIN BINDING
The observation that: (a) leukocytes are rapidly recruited from the flow stream via selectin-ligand interactions, (b) these interactions can mediate the rolling of cell, and (c) leukocyte rolling velocity is stable over a wide range of shear rates has suggested that selectins bind with a rapid on-rate and off-rate, and with high tensile strength (Citation31, 36, 42). Since few other eukaryotic receptor-ligand systems exist where cells tether onto substrates in this fashion, with the exception of platelets interacting with immobilized von Willebrand Factor (Citation66), it has been suggested that the mechanical properties of this class of adhesion molecules makes them unique among all others.
Studies have thus been undertaken in vitro to study the biomechanics of selectin and integrin binding to their ligand, using a variety of methods including surface plasmon resonance to measure binding rates of receptors to ligands (Citation51, 52, 76, 92), parallel-plate flow chamber experiments to study cell rolling in vitro in cellular and reconstituted systems (Citation42, 94) and cone-plate viscometry runs to study cell adhesion in suspension (Citation20, 53, 85). Atomic force microscopy (Citation18), micropipettes (Citation48, 75), and biomembrane force probes (Citation16) have also been applied to study single molecule biophysical properties. Molecular binding data has also been derived from NMR spectro-scopy (Citation61), fluorescence (Citation9) and ELISA based assays (Citation30, 37). We discuss findings from some, but not all, of the above experimental systems below.
In all these systems, the binding of the receptor to the ligand depends on two factors: the rate at which the receptor and ligand are brought together or the “transport features” of the system, and the reaction rates that controls bond formation when molecules are in “close” proximity. While the former transport features of the system include molecular diffusion effects in the case of ELISA based measurements, convective transport is the dominant feature during measurements involving surface plasmon resonance detection. More detailed discussion of transport features regulating cell adhesion in parallel-plate flow chambers and cone-plate viscometers are discussed later.
SURFACE PLASMON RESONANCE
Recently, several estimates of selectin and integrin binding kinetics and affinities have appeared utilizing surface plasmon resonance (SPR) methods. In these experiments, the binding of a molecule in suspension (analyte/receptor) to an immobilized ligand is measured based on changes in refractive index near the substrate when the ligand binds the analyte. Efforts are undertaken in these experiments to create monomeric selectins and integrins (without protein aggregates) so that the measurements may reflect the true binding affinities of the molecules.
In such experiments, the binding of selectin to ligand is typically very rapid and reaches saturation within seconds. Measurements of human P-selectin binding to PSGL-1 (Citation51), and P-selectin binding to the 19 N-terminal residues of PSGL-1 with three sulfated tyrosine and disialylated sLeX on the core-2 structure (Citation81) have emerged (, Data summarized in ). It is noted that the affinity of this P-selectin PSGL-1 interaction is higher and the off-rate is lower in comparison with rat L-selectin binding to mouse GlyCAM-1 (Citation52) and mouse E-selectin ESL-1 interaction (Citation92). The binding affinities of E-selectin to ESL-1 and L-selectin to GlyCAM-1 were comparable. Thus, it is evident that the binding kinetics and affinities of the selectins can vary over several orders of magnitude depending on the nature of the receptor and ligand. In studies of P-selectin PSGL-1 interaction (Citation51), the selectin off-rate was measured. Arguments are made that these represent the true off-rates, since the density of the immobilized receptors was kept low and the flow rate in the elution step was kept high to prevent/minimize rebinding. Even if some rebinding took place and diffusion effects in the SPR instrument hydrogel retarded the release of molecules, the off-rates measured in these studies likely represent the lower bounds of the selectin molecular off-rates. On-rates inferred from the dissociation constant are also presented in . These studies support the proposition that selectin bonds form and dissociate rapidly in subsecond timescales.
Receptor-ligand binding kinetics
Based on the above observations, it may be expected that the soluble, small-molecule, tetrasaccharide sLeX (tetrasaccharide) and its mimetics also bind and unbind selectins rapidly. In support of this, we have observed that a di-sLeX mimetic in our laboratory undergoes nearly instantaneous binding and unbinding in SPR experiments (unpublished data).
In comparison to the selectins, estimates of integrin-immunoglobulin binding kinetics and affinity using SPR reveal that the later molecular interactions take place with off-rates that are at least one order of magnitude smaller than that of selectins (). This table presents SPR data with focus on LFA-1 binding to ICAM-1. Here, it is noted that, receptor-ligand binding rates and magnitude vary depending on the conformation of the molecules and its activation state. Wild type LFA-1 or recombinant molecules in closed conformation bind ICAM-1 with lower affinity than activated molecules.
L-selectin and PSGL-1 are expressed on the microvillus tips of leukocytes (Citation8, 60), while the β2-integrin subunits LFA-1 and Mac-1 are expressed predominantly on the cell body in resting neutrophils (Citation15). Upon activation, Mac-1 expression is rapidly upregulated and it appears on the microvilli, cell surface ruffle, and on the cell body. Thus, it may be speculated that the distinct roles of selectins and integrins is due to their unique topography on leukocytes. However, while this may influence the roles of the molecules to some degree, the above data on binding kinetics using SPR also suggest that there are distinct differences in the binding affinities of the selectins versus the integrins.
ESTIMATING HYDRODYNAMIC FORCES APPLIED ON CELL ADHESION MOLECULES
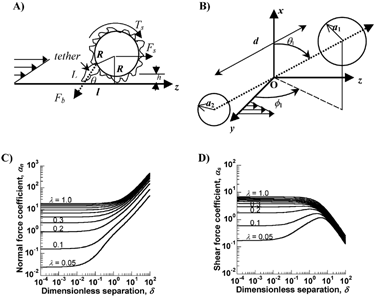
Selectin and integrin bond formation takes place in the presence of fluid flow. Hence, it is of interest to examine the effects of hydrodynamic forces on bond formation. For a cell of radius R that is rigidly bound to a substrate via a single contact, say a selectin bond at the tip of a microvilli of length L (), the applied shear force (Fs) and torque (Ts) are described by Eq. 3.7, 3.8 in (Citation19). For the case of neutrophils of radius 4.25 μm and microvilli length 0.4 μm, these equations can be written in the form of force and torque balances (Citation3):
(1)
(2)
3 Forces on inter-cellular bonds: (A) Force balance on an adherent cell of radius R which is separated from the substrate by one microvilli length, h. A single attachment/tether of length L is located at an angle θ with the substrate: θ = tan−1(R/l) + cos−1((L2+l2)/2L(R2+l2)1/2). Fb is the force on the adhesive bond, Fs the shear force, and Ts the applied torque. (B) A doublet cell aggregate composed of a pair of (un)equal-sized cells of radii a1 and a2 (a1 > a2) is shown. d is the separation distance between the sphere surfaces. z coincides with the direction of fluid flow, y is the direction of the velocity gradient and O is the origin, which lies at the midpoint of the line joining the centers of the two spheres. θ1 is the angle between the line joining the centers of the cells and the x axis. ϕ1 is the angle between the y-axis and the projection of the doublet centerline on the yz-plane. (C) and (D) provide values of force coefficients αn and αs as a function of the geometric features of the doublet Δ and λ. Here, the dimensionless separation distance between spheres is denoted Δ(= d/a1) and the radius ratio is λ(= a1/a2). Data on force coefficients can be applied in conjunction with equations in the main text of this article to determine forces on homotypic and heterotypic cell aggregates in suspension.
Here, μ is the media viscosity, G is the wall shear rate at the substrate and Fb is the force applied on the cell-substrate attachment. Thus, for experiments performed for isolated neutrophils at 37°C (μ = 0.7 cP) at a wall shear rate of 100 s−1, the wall shear stress is 0.7 dyn/cm2 and the shear force applied on the attachment is 40.52 pN. Now, if the cell contact is oriented at an angle of 55° with respect to the substrate (θ = 55° in ), then the force applied on the selectin attachment is 70 pN.
The above equations assume that the cell is stationary at a given instant, and that the hydrodynamic forces are balanced by forces applied along a single molecular contact. If the dynamic motion of cells is to be considered, more complex analysis strategies can be applied (Citation24). Such analysis can be made to account for cellular features like extensions to cell-surface microvilli, multiple bond formation and breakup, and time-dependent reorientation of bonds with respect to the substrate. As for most modeling methods, the increasing complexity in analysis is accompanied by the introduction of additional model parameters.
Analogous to the above methods that quantify forces applied on cells immobilized on substrates, methods for force calculations on cell doublets in suspension have also been developed (Citation74). depicts the prototypical doublet used for such calculations. This doublet is composed of two unequal cells of radius a1 and a2 (a1 > a2 by definition) separated by a surface-to-surface distance, d. Here, the normal (Fn) and shear forces (Fs) applied along the center-to-center line are quantified by:
(3)
(4)
αn and αs are force coefficients that are functions solely of the dumbbell geometric features, specifically the dimensionless separation distance Δ (= d/a1), and the radius ratio λ (= a2/a1) as shown in . Also, θ1 and ϕ1 describe the orientation of the dumbbell with respect to the flow (). When ϕ1 is between −90° to 0°, Fn is negative, and the force applied on dumbbell is compressive. These forces push the membranes of apposing cells together and provide an opportunity for the formation of intercellular bonds under unstressed conditions. When ϕ1 exceeds 0° (up to 90°), Fn becomes positive and tensile in nature. These tensile forces apply stress on intercellular bonds and attempt to break them. Thus, cell doublets tumbling in fluid shear flow experience cycles of compressive and tensile force loading. The nature and magnitude of these forces depends on the aggregate's dimension, rotation time-period and orientation with the flow field.
The maximum value of Fn is αnμGa12, and this occurs when ϕ1 = π/4 and θ1 = π/2, and the highest value of Fs is αs μGa12 at ϕ1 = π/2 and θ1 = π/2. Thus, for a doublet of equal sized neutrophils, the maximum normal force at G = 100s−1 is of magnitude 24.44 pN (αn = 19.33 for λ = 1 and Δ = 0.14). Peak shear forces during neutrophil homotypic binding are three times smaller than peak normal forces (since αs = 7.02 at λ = 1 and Δ = 0.14, ). In an extension to this logic, it is seen that the force applied on heterotypic aggregates composed of neutrophil-platelet doublets is ∼4 times smaller than neutrophil-neutrophil aggregates due to the smaller size of the platelet which results in a smaller surface area exposed to fluid flow (Citation74).
The peak force applied on neutrophil-neutrophil doublets is ∼40% (24.44 vs. 40.52 pN) lower than that applied on neutrophil-endothelial interactions at the same shear rate. For this reason, L-selectin mediated leukocyte-leukocyte aggregates can form in suspension at higher shear stresses (Citation85) than occur during leukocyte capture onto substrates bearing L-selectin ligands (Citation64).
In vivo, the force calculated in the above manner may be distributed over multiple bonds. Further, depending on the orientation of the bond and the cell-surface microvilli, at any instant, the force applied on intercellular bonds may be either tensile or compressive in nature, and it may be time-variant. These fluid forces may increase the unbinding or off-rates of molecular bonds. A common approach to describe the dissociation of bonds under tensile force loading is based on the exponential relationship (Citation6):
(5)
Here, k0off is the intrinsic off-rate in the absence of applied force (unstressed off-rate) while koff is the off-rate upon application of force of magnitude Fb.KB is the Boltzmann constant, T is the absolute temperature and γ represents the susceptibility of bonds to undergo force-dependent dissociation. While the above expression has been applied extensively to study selectin-ligand interactions (see next section), descriptions of the force-dependent dissociation of integrin-bonds during slow leukocyte rolling have not yet emerged. It is likely that similar exponential expressions with appropriate definition of bond susceptibility will fit the integrin data well also.
THE PARALLEL-PLATE FLOW CHAMBER
The parallel-plate flow chamber is applied to quantify the binding of leukocytes to endothelial cells and reconstituted ligand-bearing substrates under fluid-flow conditions that mimic the human vasculature. In this device, cells contact the substrate by settling under gravity while simultaneously being convected in the free stream. Due to the dominant role of cell settling in such experiments, the size of the cells and the difference between cell and media density are important features regulating the flux of cells to the substrate (Citation94). In flow chambers with typical geometries, only the cells that enter the flow chamber close to the bottom contact the substrate and a majority of cells exit in the free stream. Increasing wall shear stress by increasing fluid volumetric flow into the flow chamber is balanced by changes in the trajectory of cells in the device such that a ∼25% decrease in the number of cell-substrate collisions may be observed upon increasing wall shear stress from 0.5 to 10 dyn/cm2. The exact magnitude of this effect depends on the dimensions of the chamber and the properties (density, size) of the leukocyte.
While conventional methods quantify cell adhesion in this device in terms of the number of rolling and adherent cells, methods for estimating the contact times and “efficiencies” of cell binding have also emerged (Citation94, 95) (). Analysis of data on human neutrophils interacting with a substrate composed of E-selectin and ICAM-1 bearing cells (E/I cells) using this strategy reveals that, on average, cells contact the E-selectin bearing substrates for 4–8.5s before tethering and initiating rolling. Based on data published elsewhere (Citation64), it is estimated that the time taken for leukocyte tethering on P-selectin substrates at comparable ligand densities is 1/2 and for L-selectin it is 1/5–1/10 that of E-selectin. This suggests that the on-rates of selectins vary as L-selectin > P-selectin > E-selectin.
Adherent cells may influence the flow streams in their vicinity up to a distance of 2.5 cell diameters away (Citation94). This disturbance to flow along with the simultaneous expression of both L-selectin and its ligand PSGL-1 on neutrophils results in leukocyte-leukocyte secondary capture. This secondary tethering event may lead to cell capture at distances greater than one cell diameter. Approximately 5–7% of collisions between neutrophils in the free stream and previously recruited neutrophils bound on the substrate result in secondary capture in flow chamber experiments carried out at a wall shear stress of 2 dyn/cm2. While this cell-cell binding results in cells lining up in a “string of bead” arrangement in reconstituted systems with isolated immobilized selectins (Citation91), clusters of rolling cells may form in cellular systems (Citation94). Rapid transmigration of cells may limit the degree to which secondary capture contributes to tethering in experiments performed with isolated HUVEC.
Following the initiation of rolling, cells undergo activation via a variety of mechanisms including chemokine mediated signaling and selectin ligation (Citation79). On the E/I cell substrate, rolling results in slow activation of neutrophils via E-selectin mediated signaling, and the average cell takes ∼300s to transition to firm arrest via leukocyte β2-integrins (Citation94). In marked contrast, on IL-1β stimulated HUVEC, where both E-selectin and chemokines are present, transition from rolling to firm arrest typically takes place within 2–5 sec. at very high efficiency (Citation21). The subsecond activation of integrins following chemokine stimulation has been suggested (Citation2).
To examine the mechanical properties of selectin-ligand bonds under shear flow, in detail, parallel-plate flow chamber experiments were conducted where P-selectin was immobilized on a lipid bilayer at low density (Citation3). Transient tethering and release of leukocytes was observed on this substrate via P-selectin PSGL-1 interactions. This tethering lifetime could be modeled as a first-order process. Based on these measurements and extrapolation to zero shear, it was suggested that the unstressed off rates (k0off) of P-selectin is ∼1 s−1. Similar studies have been carried out to measure the binding of L-selectin to PNAd (Peripheral Node Addresins), and E-selectin to its ligands on leukocytes. High-speed video microscopy has also been applied in such experiments (Citation80). The extrapolated off-rates at zero stress from such experiments are summarized in .
The extrapolated zero-force off-rates estimated in the parallel plate flow chamber match well with off-rates obtained in the surface plasmon resonance experiments performed with monomeric molecules. This supports the proposition that flow chamber measurements are a result of unimolecular receptor-ligand bonds. At the same time, many of the receptor-ligand interactions studied in the flow chamber are mediated by the binding of immobilized ligand to dimeric molecules on the cell-surface, like PSGL-1. In this regard, while dimerization is not necessary for receptor-ligand interaction, it is recognized that dimerization of ligands like PSGL-1 and P-selectin stabilize tethering and rolling upon application of force, probably by increasing rebinding within a bond cluster (Citation65). Further, a recent article has suggested that high time-resolved videomicroscopy (2 millisec. resolution) can reveal brief L-selectin mediated tethers with lifetimes of 4 ms (Citation12). Such interactions were not previously detected using standard video microscopy, which has a resolution of 20-30 millisec. Thus, it remains to be fully clarified as to what extent the off-rates measured in the flow chamber experiments at low ligand density are influenced by multiple bond formation and rebinding effects.
CONE AND PLATE VISCOMETRY
Cone and plate viscometers have been applied in studies of platelet function and aggregation for several decades (Citation38). This device has also been applied to study leukocyte selectin and integrin function under fluid shear (Citation54, 85). In these experiments, human neutrophils were placed in suspension in the gap between the 1° cone and the plate of the viscometer, and fluid shear was applied by rotating the cone at a given angular velocity. Samples taken at particular time points were fixed, fluorescently labeled, and the extent of cell aggregation was quantified using flow cytometry analysis. Mathematical modeling was also applied to quantify the fraction of cell-cell collisions that result in adhesion (Citation28, 54). This fraction of effective collisions was termed “adhesion efficiency.” In some experiments, collisions between cells have also been visualized in real-time using video microscopy coupled with high-speed imaging (Citation20). Viscometry studies have been applied to examine neutrophil homotypic aggregation (Citation20, 54, 56, 85), and heterotypic aggregation where human neutrophils bind blood platelets and other transfected cell lines (Citation28, 53).
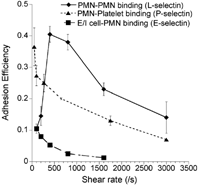
L-, E- and P-selectin mediated leukocyte adhesion has been studied in the viscometer over a range of shear rates (selected results in ). These studies reveal that L-selectin exhibits a prominent shear-threshold phenomenon with peak adhesion efficiency at 400/s (Citation85). While E- and P-selectin do not exhibit the shear threshold over a range of shear rates from 100–3000/s, a shear threshold for P-selectin PSGL-1 mediated adhesion has been observed in the viscometer at near stagnant flows below 100/s (H. L. Goldsmith and S. N., unpublished observations). In the viscometer, selectin bonds alone in the absence of integrins can hold transient leukocyte aggregates together over multiple rotations thus allowing sufficient time for integrins to engage and form stable cell aggregates (Citation20). The minimum time required for integrin bond formation in chemoattractant stimulated neutrophil homotypic aggregation experiments is estimated to be ∼6 millisec (Citation54). Increasing the cell-cell contact duration via selectin bonds beyond 6 millisec enhances the ability of leukocytes to engage integrin-ligands on apposing cells.
4 Adhesion efficiency of L-, P- and E-selectin mediated cell binding in suspension. Cell adhesion in the cone-plate viscometer was studied in three systems. (a) Isolated human neutrophils (PMNs) were stimulated with 1 μM FMLP and mixed in the viscometer over a range of shear rates. This adhesion process is dependent on L-selectin and β2-integrin (symbol: ♦). (b) Unstimulated isolated PMNs were mixed with human platelets, which were preactivated with 25 μM Thrombin Receptor Activating hexaPeptide (TRAP) for 3 min. prior to the viscometer experiment. Platelets stimulated with TRAP expressed high levels of P-selectin and bound PSGL-1 on neutrophils (symbol: ▴). (c) E/I cells described in the flow chamber studies (see main text (Citation94)) were mixed in suspension with 1 μM FMLP stimulated human PMNs (symbol: ▪). This cell adhesion process occurred via E-selectin and β2-integrins. As seen, L-selectin but not P- and E-selectin mediated cell adhesion exhibits a prominent shear threshold effect in these studies conduced in suspension.
The cone-plate viscometer is suited:
For rapid testing and quantitative evaluation of scarce inhibitors or other reagents under shear flow, since the volume of these experiments is small (∼100–250μ l) compared to flow chamber assays. As an example, this device has been used to study the ability of small-molecule carbohydrates to block neutrophil homotypic aggregation (Citation55). The application of flow cytometric data analysis in conjunction with viscometry enables averaging of cell adhesion data over thousands of collisions despite the small experiment volume.
For time resolved studies of cell function, since we can simultaneously stimulate all cells in suspension at a given instant, and then follow their fate by sampling the aggregating cell suspension over time. Rapid sampling is not possible in the flow chamber assay. Previous time resolved studies in the viscometer (Citation53, 56) reveal that following rapid activation of neutrophil β2-integrin affinity and avidity due to chemotactic stimulation, there is a gradual time-dependent decrease in cellular adhesivity via these integrins. The avidity of LFA-1 binding to ICAM-1 and ICAM-3 increases within seconds and then decays over a period of minutes after stimulation. Mac-1 mediated cell adhesion decreases over a four-fold longer time scale compared to LFA-1. These time-dependent changes in β2-integrin function may be an important feature regulating leukocyte firm arrest and transmigration rates.
For studying the effect of hydrodynamic forces on cellular processes since, unlike the flow chambers where the shear rate is maximum at the wall and minimum at the center, the shear rate in the viscometer is relatively uniform throughout the instrument [Effects of viscometer secondary flow on cellular phenomena are discussed elsewhere (Citation71, 72)]. While the viscometer has revealed novel information on selectin and integrin mediated cell adhesion mechanics, we note that its application in vascular biology has been limited since leukocyte-endothelial cell adhesion has been traditionally studied in the context of cells binding to substrates in flow chamber geometries. Further, as discussed below, a thorough understanding of the mechanics of cell collision and bond formation in this device is lacking currently.
In cell adhesion studies conducted in the cone and plate viscometer, the convective transport due to fluid flow induces cell-cell collision. Approaching cells in such experiments rotate in the same direction, along axes that have a common orientation. Thus, the surfaces of these approaching cells are moving in opposite directions relative to each other. Receptors and ligands on approaching cells sample each other and may form molecular bonds when they are in close proximity (within one bond length). While the relative surface velocity between the two approaching cells is largest when the cells are far apart, this velocity decreases upon approach and it falls to zero upon cell-cell contact. Analogous to our calculations of hydrodynamic forces applied on cells in suspension (), the relative velocity of the surface of approaching cells and the number of receptor-ligand encounters during individual cell rotation is a strong function of two parameters: (a) the radius ratio of the two colliding cells (λ), and (b) the distance of separation between the cells (Δ) (unpublished data). Following collision, the relative motion between the cells is zero and a rigid dumbbell is said to form. Interactions between receptors and ligands on the two cells that compose the dumbbell is now solely dependent on the diffusivity of the molecules on the cell membrane. The relative roles of surface diffusion and fluid convection in contributing to cell aggregation in suspension in the viscometer is not yet well defined, and this will be the subject of future investigations. Development of such analysis strategies will allow us to directly compare cell adhesion data measured in the viscometer with SPR and flow chamber results.
AN INTERPLAY BETWEEN CELLULAR AND MOLECULAR PROPERTIES REGULATE LEUKOCYTE ROLLING VELOCITY
Several lines of evidence point to the contribution of cellular properties in controlling leukocyte rolling mechanics. Rolling leukocytes exhibit deformation in a tear-drop shape (Citation87). Further, it is demonstrated that neutrophils interacting with immobilized, activated platelets via P-selectin PSGL-1 bonds extend thin membrane tethers that are pulled from neutrophils in ∼30% of the binding events (Citation70). In this study, tethers sometime stretched to lengths several times the cell diameter. The elongated tethers also snapped and were left behind in some instances suggesting that membrane failure may occur before bond failure in some instances. Evidence of possible leukocyte fragmentation is also suggested in vivo in an animal model of L-selectin dependent rolling (Citation82). Overall, leukocyte tethers may shield the receptor-ligand bond from hydrodynamic force loading and present additional ligands on the vascular endothelium. Other studies that compare cell rolling in microbead systems with cellular systems provide evidence that cell deformation is necessary for shear-resistant rolling (Citation59, 93). Unlike cells, the rolling velocity of ligand-bearing beads increases rapidly with increasing wall shear stress.
Another novel property of leukocyte rolling, which is prominent in the case of L-selectin, is the shear threshold phenomenon (Citation17, 41, 85). The physiological relevance of the shear threshold phenomenon is uncertain though it has been suggested that this may provide a means to limit inappropriate cell adhesion in regions of low shear. According to this feature, L-selectin mediated leukocyte rolling is negligible at both very low shear stresses below 0.5 dyn/cm2 and at the very higher shear stresses. It is maximum in the intermediate range. This property was observed for the case of neutrophils rolling on PNAd coated substrates (Citation17), and for the case of L-selectin mediated neutrophil homotypic aggregation (Citation85). In vivo also, this has been observed in the high endothelial venules Payer's patch where a reduction in L-selectin mediated neutrophil rolling has been observed at low shear stresses. The shear threshold phenomenon is more prominent for L-selectin, rather than E- or P-selectin, probably due its characteristic high binding on- and off-rate compared to the other selectins.
Potential mechanisms for the threshold phenomenon include:
An increase in the encounter rate between receptors and ligands at intermediate, but not low shear rates, due to an increase in the relative velocity between cells (Citation17). In support of this, the shear threshold phenomenon has been suggested in cell free systems where sLeX bearing beads roll on L-selectin bearing substrates (Citation23). Further, the threshold phenomenon is a strong function of receptor and ligand density (Citation41).
Increase in cell deformation and a consequent increase in the contact area between interacting cells at higher shears (Citation41). In support of this, in experiments carried out in viscometers, it has been shown that increasing media viscosity decreases the shear rate at which the shear threshold phenomenon occurs (Citation85).
Increased bond formation kinetics due to either the nature of bond loading, or the ability of shear forces to surmount energy barriers that otherwise limit receptor-ligand bond formation. This is supported by atomic force microscopy measurements, which lead the authors to suggest that P-selectin forms catch bonds (Citation50).
Finally, it has been shown that chemical modification of the L-selectin ligand abrogates the shear-threshold phenomenon (Citation63). This might potentially be due to a redefinition of the energy landscape for receptor-ligand interaction in this system. Overall, based on the above, it is likely that both the transport and reaction features of the selectins control the shear-threshold phenomenon. Together, these features may contribute to an increase in the lumped on rate (kon* number of ligand) for selectins over low shear stresses until the threshold shear is reached (Citation73). At higher shear stresses, the lumped on rate decreases with shear as the hydrodynamic forces limit the encounter-duration required for inter-cellular bond formation.
CONCLUSION
Studies of the molecular properties of selectin and integrin bonds during leukocyte-endothelial cell adhesion have been an active area of research, ever since these molecules were cloned over a decade ago. A better understanding of the effect of hydrodynamic forces on these molecular interactions along with the development of sophisticated instrumentation and mathematical models for data interpretation has greatly enhanced this research area. The ongoing cloning of glycosyltransferases that form selectin-ligands and the discovery of new molecules that mediate cell adhesion under flow is likely to continue to spur interest in this field. Studies of integrin bond mechanics also lag behind our current understanding of selectin biophysics, and this is likely to receive greater attention in the near future.
ACKNOWLEDGEMENTS
Grant support from the NIH (HL63014, H676211) and Whitaker Biomedical Engineering Foundation is gratefully acknowledged.
REFERENCES
- Alon R, Chen S, Puri K D, Finger E B, Springer T A. 1997. The kinetics of L-selectin tethers and the mechanics of selectin-mediated rolling. J Cell Biol. 138: 1169–1180. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Alon R, Feigelson S. 2002. From rolling to arrest on blood vessels: Leukocyte tap dancing on endothelial integrin ligands and chemokines at sub-second contacts. Semin Immunol. 14: 93–104. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Alon R, Hammer D A, Springer T A. 1995. Lifetime of the P-selectin-carbohydrate bond and its response to tensile force in hydrodynamic flow. Nature. 374: 539–542. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Alon R, Kassner P D, Carr M W, Finger E B, Hemler M E, Springer T A. 1995. The integrin VLA-4 supports tethering and rolling in flow on VCAM-1. J Cell Biol. 128: 1243–1253. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Arrate M P, Rodriguez J M, Tran T M, Brock T A, Cunningham S A. 2001. Cloning of human junctional adhesion molecule 3 (JAM3) and its identification as the JAM2 counter-receptor. J Biol Chem. 276: 45826–45832. [PUBMED], [INFOTRIEVE]
- Bell G I. 1978. Models for the specific adhesion of cells to cells. Science. 200: 618–627. [PUBMED], [INFOTRIEVE]
- Berlin C, Bargatze R F, Campbell J J, von, Andrian U H, Szabo M C, Hasslen S R, Nelson R D, Berg E L, Erlandsen S L, Butcher E C. 1995. Alpha 4 integrins mediate lymphocyte attachment and rolling under physiologic flow. Cell. 80: 413–422. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Bruehl R E, Moore K L, Lorant D E, Borregaard N, Zimmerman G A, McEver R P, Bainton D F. 1997. Leukocyte activation induces surface redistribution of P-selectin glycoprotein ligand-1. J Leukoc Biol. 61: 489–499. [PUBMED], [INFOTRIEVE]
- Croce K, Freedman S J, Furie B C, Furie B. 1998. Interaction between soluble P-selectin and soluble P-selectin glycoprotein ligand 1: Equilibrium binding analysis. Biochem. 37: 16472–16480
- Cunningham S A, Rodriguez J M, Arrate M P, Tran T M, Brock T A. 2002. JAM2 interacts with alpha4beta1. Facilitation by JAM3. J Biol Chem. 277: 27589–27592. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Dunne J L, Ballantyne C M, Beaudet A L, Ley K. 2002. Control of leukocyte rolling velocity in TNF-alpha-induced inflammation by LFA-1 and Mac-1. Blood. 99: 336–341. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Dwir O, Solomon A, Mangan S, Kansas G S, Schwarz U S, Alon R. 2003. Avidity enhancement of L-selectin bonds by flow: Shear-promoted rotation of leukocytes turn labile bonds into functional tethers. J Cell Biol. 163: 649–659. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Ebnet K, Suzuki A, Ohno S, Vestweber D. 2004. Junctional adhesion molecules (JAMs): More molecules with dual functions?. J Cell Sci. 117: 19–29. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Ellies L G, Tsuboi S, Petryniak B, Lowe J B, Fukuda M, Marth J D. 1998. Core 2 oligosaccharide biosynthesis distinguishes between selectin ligands essential for leukocyte homing and inflammation. Immunity. 9: 881–890. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Erlandsen S L, Hasslen S R, Nelson R D. 1993. Detection and spatial distribution of the beta-2 integrin (Mac-1) and L-selectin (LECAM-1) adherence receptors on human neutrophils by high-resolution field emission SEM. The J Histochem and Cytochem. 41: 327–333
- Evans E, Leung A, Hammer D, Simon S. 2001. Chemically distinct transition states govern rapid dissociation of single L-selectin bonds under force. Proc Natl Acad Sci USA. 98: 3784–3789. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Finger E B, Puri K D, Alon R, Lawrence M B, von, Andrian U H, Springer T A. 1996. Adhesion through L-selectin requires a threshold hydrodynamic shear. Nature. 379: 266–269. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Fritz J, Katopodis A G, Kolbinger F, Anselmetti D. 1998. Force-mediated kinetics of single P-selectin/ligand complexes observed by atomic force microscopy. Proc Natl Acad Sci USA. 95: 12283–12288. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Goldman A J, Cox R G, Brenner H. 1967. Slow viscous motion of a sphere parallel to a plane wall -II: Couette flow. Chem Eng Sci. 22: 653–660
- Goldsmith H L, Quinn T A, Drury G, Spanos C, McIntosh F A, Simon S I. 2001. Dynamics of neutrophil aggregation in couette flow revealed by videomicroscopy: Effect of shear rate on two-body collision efficiency and doublet lifetime. Biophys J. 81: 2020–2034. [PUBMED], [INFOTRIEVE]
- Gopalan P K, Burns A R, Simon S I, Sparks S, McIntire L V, Smith C W. 2000. Preferential sites for stationary adhesion of neutrophils to cytokine-stimulated HUVEC under flow conditions. J Leukoc Biol. 68: 47–57. [PUBMED], [INFOTRIEVE]
- Graves B J, Crowther R L, Chandran C, Rumberger J M, Li S, Huang K S, Presky D H, Familletti P C, Wolitzky B A, Burns D K. 1994. Insight into E-selectin/ligand interaction from the crystal structure and mutagenesis of the lec/EGF domains. Nature. 367: 532–538. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Greenberg A W, Brunk D K, Hammer D A. 2000. Cell-free rolling mediated by L-selectin and sialyl Lewis(x) reveals the shear threshold effect. Biophys J. 79: 2391–2402. [PUBMED], [INFOTRIEVE]
- Hammer D A, Apte S M. 1992. Simulation of cell rolling and adhesion on surfaces in shear flow: General results and analysis of selectin-mediated neutrophil adhesion. Biophys J. 63: 35–57. [PUBMED], [INFOTRIEVE]
- Harlan J M, Liu D Y, 1992; Adhesion: Its role in Inflammatory Disease. Breakthroughs in Molecular Biology. W. H. Freeman, New York
- Hemmerich S, Leffler H, Rosen S D. 1995. Structure of the O-Glycans in GlyCAM-1, an Endothelial-derived Ligand for L-Selectin. J Biol Chem. 270: 12035–12047. [PUBMED], [INFOTRIEVE]
- Henderson R B, Lim L H, Tessier P A, Gavins F N, Mathies M, Perretti M, Hogg N. 2001. The use of lymphocyte function-associated antigen (LFA)-1-deficient mice to determine the role of LFA-1, Mac-1, and alpha4 integrin in the inflammatory response of neutrophils. J Exp Med. 194: 219–226. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Hentzen E R, Neelamegham S, Kansas G S, Benanti J A, McIntire L V, Smith C W, Simon S I. 2000. Sequential binding of CD11a/CD18 and CD11b/CD18 define neutrophil capture and stable adhesion to ICAM-1. Blood. 95: 911–920. [PUBMED], [INFOTRIEVE]
- Hynes R O. 2002. Integrins: Bidirectional, allosteric signaling machines. Cell. 110: 673–687. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Jain R K, Piskorz C F, Huang B G, Locke R D, Han H L, Koenig A, Varki A, Matta K L. 1998. Inhibition of L- and P-selectin by a rationally synthesized novel core 2-like branched structure containing GalNAc-Lewisx and Neu5Acalpha2–3Galbeta1–3GalNAc sequences. Glycobiology. 8: 707–717. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Jones D A, Abbassi O, McIntire L V, McEver R P, Smith C W. 1993. P-selectin mediates neutrophil rolling on histamine-stimulated endothelial cells. Biophys J. 65: 1560–1569. [PUBMED], [INFOTRIEVE]
- Jung U, Norman K E, Scharffetter-Kochanek K, Beaudet A L, Ley K. 1998. Transit time of leukocytes rolling through venules controls cytokine-induced inflammatory cell recruitment in vivo. J Clin Invest. 102: 1526–1533. [PUBMED], [INFOTRIEVE]
- Kanamori A, Kojima N, Uchimura K, Muramatsu T, Tamatani T, Berndt M C, Kansas G S, Kannagi R. 2002. Distinct sulfation requirements of selectins disclosed using cells that support rolling mediated by all three selectins under shear flow. L-selectin prefers carbohydrate 6-sulfation totyrosine sulfation, whereas p-selectin does not. J Biol Chem. 277: 32578–32586. [PUBMED], [INFOTRIEVE]
- Kansas G S. 1996. Selectins and their ligands: Current concepts and controversies. Blood. 88: 3259–3287. [PUBMED], [INFOTRIEVE]
- Kansas G S, Saunders K B, Ley K, Zakrzewicz A, Gibson R M, Furie B C, Furie B, Tedder T F. 1994. A role for the epidermal growth factor-like domain of P-selectin in ligand recognition and cell adhesion. J of Cell Biol. 124: 609–618
- Kaplanski G, Farnarier C, Tissot O, Pierres A, Benoliel A M, Alessi M C, Kaplanski S, Bongrand P. 1993. Granulocyte-endothelium initial adhesion. Analysis of transient binding events mediated by E-selectin in a laminar shear flow. Biophys J. 64: 1922–1933. [PUBMED], [INFOTRIEVE]
- Koenig A, Jain R, Vig R, Norgard-Sumnicht K E, Matta K L, Varki A. 1997. Selectin inhibition: Synthesis and evaluation of novel sialylated, sulfated and fucosylated oligosaccharides, including the major capping group of GlyCAM-1. Glycobiology. 7: 79–93. [PUBMED], [INFOTRIEVE]
- Kroll M H, Hellums J D, McIntire L V, Schafer A L, Moake J L. 1996. Platelets and shear stress. Blood. 88: 1525–1541. [PUBMED], [INFOTRIEVE]
- Labadia M E, Jeanfavre D D, Caviness G O, Morelock M M. 1998. Molecular regulation of the interaction between leukocyte function-associated antigen-1 and soluble ICAM-1 by divalent metal cations. J Immunol. 161: 836–842. [PUBMED], [INFOTRIEVE]
- Lasky L A. 1995. Selectin-carbohydrate interactions and the initiation of the inflammatory response. Annual Review of Biochemistry. 64: 113–139. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Lawrence M B, Kansas G S, Kunkel E J, Ley K. 1997. Threshold levels of fluid shear promote leukocyte adhesion through selectins (CD62L,P,E). J Cell Biol. 136: 717–727. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Lawrence M B, Springer T A. 1991. Leukocytes roll on a selectin at physiologic flow rates: Distinction from and prerequisite for adhesion through integrins. Cell. 65: 859–873. [PUBMED], [INFOTRIEVE]
- Leppanen A, Penttila L, Renkonen O, McEver R P, Cummings R D. 2002. Glycosulfopeptides with O-glycans containing sialylated and polyfucosylated polylactosamine bind with low affinity to P-selectin. J Biol Chem. 277: 39749–39759. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Leppanen A, Yago T, Otto V I, McEver R P, Cummings R D. 2003. Model glycosulfopeptides from PSGL-1 require tyrosine sulfation and a core-2 branched Oglycan to bind to L-selectin. J Biol Chem. 278: 26391–26400. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Li F, Wilkins P P, Crawley S, Weinstein J, Cummings R D, McEver R P. 1996. Post-translational modifications of recombinant P-selectin glycoprotein ligand-1 required for binding to P- and E-selectin. J Biol Chem. 271: 3255–3264. [PUBMED], [INFOTRIEVE]
- Li R, Mitra N, Gratkowski H, Vilaire G, Litvinov R, Nagasami C, Weisel J W, De Lear J DGrado W F, Bennett J S. 2003. Activation of integrin alphaIIbbeta3 by modulation of transmembrane helix associations. Science. 300: 795–798. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Liddington R C, Ginsberg M H. 2002. Integrin activation takes shape. J Cell Biol. 158: 833–839. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Lomakina E B, Waugh R E. 2004. Micromechanical tests of adhesion dynamics between neutrophils and immobilized ICAM-1. Biophys J. 86: 1223–1233. [PUBMED], [INFOTRIEVE]
- Lupher M L, Jr, Harris E A, Beals C R, Sui L M, Liddington R C, Staunton D E. 2001. Cellular activation of leukocyte function-associated antigen-1 and its affinity are regulated at the I domain allosteric site. J Immunol. 167: 1431–1439. [PUBMED], [INFOTRIEVE]
- Marshall B T, Long M, Piper J W, Yago T, McEver R P, Zhu C. 2003. Direct observation of catch bonds involving cell-adhesion molecules. Nature. 423: 190–193. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Mehta P, Cummings R D, McEver R P. 1998. Affinity and kinetic analysis of P-selectin binding to P-selectin glycoprotein ligand-1. J Biol Chem. 273: 32506–32513. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Nicholson M W, Barclay A N, Singer M S, Rosen S D, van der, Merwe P A. 1998. Affinity and kinetic analysis of L-selectin (CD62L) binding to glycosylation-dependent cell-adhesion molecule-1. J Biol Chem. 273: 763–770. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Neelamegham S, Taylor A D, Burns A B, Smith C W, Simon S I. 1998. Hydrodynamics shear reveals distinct roles for LFA-1 and Mac-1 in neutrophil adhesion to ICAM-1. Blood. 92: 1626–1638. [PUBMED], [INFOTRIEVE]
- Neelamegham S, Taylor A D, Hellums J D, Dembo M, Smith C W, Simon S I. 1997. Modeling the reversible kinetics of neutrophil aggregation under hydrodynamic shear. Biophys J. 72: 1527–1540. [PUBMED], [INFOTRIEVE]
- Neelamegham S, Matta K L. Liposomes containing ligands. Binding specificity to selectins, in Methods Mol Biol. 2002. Humana Press, New York, pp. 175–191
- Neelamegham S, Taylor A D, Shankaran H, Smith C W, Simon S I. 2000. Shear and time dependent changes in Mac-1, LFA-1, and ICAM-3 binding regulate neutrophil homotypic adhesion. J of Immunol. 164: 3798–3805
- Ostermann G, Weber K S, Zernecke A, Schroder A, Weber C. 2002. JAM-1 is a ligand of the beta(2) integrin LFA-1 involved in transendothelial migration of leukocytes. Nat Immunol. 3: 151–158. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Padilla A, Moake J L, Bernardo A, Ball C, Wang Y, Arya M, Nolasco L, Turner N, Berndt M C, Anvari B, Lopez J A, Dong J F. 2004. P-selectin anchors newly released ultralarge von Willebrand factor multimers to the endothelial cell surface. Blood. 103: 2150–2156. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Park E Y, Smith M J, Stropp E S, Snapp K R, Di, Vietro J A, Walker W F, Schmidtke D W, Diamond S L, Lawrence M B. 2002. Comparison of PSGL-1 microbead and neutrophil rolling: Microvillus elongation stabilizes P-selectin bond clusters. Biophys J. 82: 1835–1847. [PUBMED], [INFOTRIEVE]
- Picker L J, Warnock R A, Burns A R, Doerschuk C M, Berg E L, Butcher E C. 1991. The neutrophil selectin LECAM-1 presents carbohydrate ligands to the vascular selectins ELAM-1 and GMP-140. Cell. 66: 921–933. [PUBMED], [INFOTRIEVE]
- Poppe L, Brown G S, Philo J S, Nikrad P V, Shah B H. 1997. Conformation of slex tetrasaccharide free in solution and bound to E-, P- and L-selectin. J of the Amer Chem Soc. 119: 1727–1736
- Pouyani T, Seed B.. 1995. PSGL-1 recognition of P-selectin is controlled by a tyrosine sulfation consensus at the PSGL-1 amino terminus. Cell. 83: 333–343. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Puri K D, Chen S, Springer T A. 1998. Modifying the mechanical property and shear threshold of L-selectin adhesion independently of equilibrium properties. Nature. 392: 930–933. [PUBMED], [INFOTRIEVE]
- Puri K D, Finger E B, Springer T A. 1997. The faster kinetics of L-selectin than of E-selectin and P-selectin rolling at comparable binding strength. J Immunol. 158: 405–413. [PUBMED], [INFOTRIEVE]
- Ramachandran V, Nollert M U, Qiu H, Liu W J, Cummings R D, Zhu C, McEver R P. 1999. Tyrosine replacement in P-selectin glycoprotein ligand-1 affects distinct kinetic and mechanical properties of bonds with P- and L-selectin. Proc Natl Acad Sci USA. 96: 13771–13776. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Ruggeri Z M. 2002. Platelets in atherothrombosis. Nat Med. 8: 1227–1234. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Sako D, Comess K M, Barone K M, Camphausen R T, Cumming D A, Shaw G D. 1995. A sulfated peptide segment at the amino terminus of PSGL-1 is critical for P-selectin binding. Cell. 83: 323–331. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Sanders W J, Katsumoto T R, Bertozzi C R, Rosen S D, Kiessling L L. 1996. L-selectin-carbohydrate interactions: Relevant modifications of the Lewis x trisaccharide. Biochem. 35: 14862–14867
- Santoso S, Sachs U J, Kroll H, Linder M, Ruf A, Preissner K T, Chavakis T. 2002. The junctional adhesion molecule 3 (JAM-3) on human platelets is a counterreceptor for the leukocyte integrin Mac-1. J Exp Med. 196: 679–691. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Schmidtke D W, Diamond S L. 2000. Direct observation of membrane tethers formed during neutrophil attachment to platelets or P-selectin under physiological flow. J Cell Biol. 149: 719–730. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Shankaran H, Alexandridis P, Neelamegham S. 2003. Aspects of hydrodynamic shear regulating shear-induced platelet activation and self-association of von Willebrand factor in suspension. Blood. 101: 2637–2645. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Shankaran H, Neelamegham S. 2001. Effect of secondary flow on biological experiments in the cone-plate viscometer: Methods for estimating collision frequency, wall shear stress and inter-particle interactions in non-linear flow. Biorheology. 30: 275–304
- Shankaran H, Neelamegham S. 2001. Nonlinear flow affects hydrodynamic forces and neutrophil adhesion rates in cone-plate viscometers. Biophys J. 80: 2631–2648. [PUBMED], [INFOTRIEVE]
- Shankaran H, Neelamegham S. 2004. Hydrodynamic forces and force loading rates applied on intercellular bonds, soluble molecules and cell surface receptors. Biophys J. 86: 576–588. [PUBMED], [INFOTRIEVE]
- Shao J Y, Ting-Beall H P, Hochmuth R M. 1998. Static and dynamic lengths of neutrophil microvilli. Proc Natl Acad Sci USA. 95: 6797–6802. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Shimaoka M, Xiao T, Liu J H, Yang Y, Dong Y, Jun C D, McCormack A, Zhang R, Joachimiak A, Takagi J, Wang J H, Springer T A. 2003. Structures of the alpha L I domain and its complex with ICAM-1 reveal a shape-shifting pathway for integrin regulation. Cell. 112: 99–111. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Shimaoka M, Lu C, Palframan R T, von, Andrian U H, McCormack A, Takagi J, Springer T A. 2001. Reversibly locking a protein fold in an active conformation with a disulfide bond: Integrin alphaL I domains with high affinity and antagonist activity in vivo. Proc Natl Acad Sci USA. 98: 6009–6014. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Simon S I, Neelamegham S, Taylor A, Smith C W. 1998. The multistep process of homotypic neutrophil aggregation: A review of the molecules and effects of hydrodynamics. Cell Adhes Commun. 6: 263–276. [PUBMED], [INFOTRIEVE]
- Smith C W. 2000. Possible steps involved in the transition to stationary adhesion of rolling neutrophils: A brief review. Microcirc. 7: 385–394
- Smith M J, Berg E L, Lawrence M B. 1999. A direct comparison of selectinmediated transient, adhesive events using high temporal resolution. Biophys J. 77: 3371–3383. [PUBMED], [INFOTRIEVE]
- Somers W S, Tang J, Shaw G D, Camphausen R T. 2000. Insights into the molecular basis of leukocyte tethering and rolling revealed by structures of P- and E-selectin bound to SLe(X) and PSGL-1. Cell. 103: 467–479. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Sperandio M, Smith M L, Forlow S B, Olson T S, Xia L, McEver R P, Ley K. 2003. P-selectin Glycoprotein Ligand-1 Mediates L-Selectin-dependent Leukocyte Rolling in Venules. J Exp Med. 197: 1355–1363. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Springer T A. 1995. Traffic signals on endothelium for lymphocyte recirculation and leukocyte emigration. Annu Rev Physiol. 57: 827–872. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Steegmaier M, Levinovitz A, Isenmann S, Borges E, Lenter M, Kocher H P, Kleuser B, Vestweber D. 1995. The E-selectin-ligand ESL-1 is a variant of a receptor for fibroblast growth factor. Nature. 373: 615–620. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Taylor A D, Neelamegham S, Hellums J D, Smith C W, Simon S I. 1996. Molecular dynamics of the transition from L-selectin- to beta 2-integrin-dependent neutrophil adhesion under defined hydrodynamic shear. Biophys J. 71: 3488–3500. [PUBMED], [INFOTRIEVE]
- Tominaga Y, Kita Y, Satoh A, Asai S, Kato K, Ishikawa K, Horiuchi T, Takashi T.. 1998. Affinity and kinetic analysis of the molecular interaction of ICAM-1 and leukocyte function-associated antigen-1. J Immunol. 161: 4016–4022. [PUBMED], [INFOTRIEVE]
- Tozeren A, Ley K. 1992. How do selectins mediate leukocyte rolling in venules?. Biophys J. 63: 700–709. [PUBMED], [INFOTRIEVE]
- Tsuboi S, Fukuda M. 2001. Roles of O-linked oligosaccharides in immune responses. Bioessays. 23: 46–53. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Vestweber D, Blanks J E. 1999. Mechanisms that regulate the function of the selectins and their ligands. Physiol Rev. 79: 181–213. [PUBMED], [INFOTRIEVE]
- von, Andrian U H, Chambers J D, McEvoy L M, Bargatze R F, Arfors K E, Butcher E C. 1991. Two step model of leukocyte-endothelial cell interaction in inflammation: Distinct roles for LECAM-1 and the leukocyte beta-2 integrins in vivo. Proc. of the Nati. Acad. of Sci., USA. 88: 7538–7542
- Walcheck B, Moore K L, McEver R P, Kishimoto T K. 1996. Neutrophilneutrophil interactions under hydrodynamic shear stress involve L-selectin and PSGL-1. A mechanism that amplifies initial leukocyte accumulation on P-selectin in vitro. J of Clin Investi. 98: 1081–1087
- Wild M K, Huang M C, Schulze-Horsel U, van der, Merwe P A, Vestweber D. 2001. Affinity, kinetics, and thermodynamics of E-selectin binding to E-selectin ligand-1. J Biol Chem. 276: 31602–31612. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Yago T, Leppanen A, Qiu H, Marcus W D, Nollert M U, Zhu C, Cummings R D, McEver R P. 2002. Distinct molecular and cellular contributions to stabilizing selectin-mediated rolling under flow. J Cell Biol. 158: 787–799. [CROSSREF], [PUBMED], [INFOTRIEVE]
- Zhang Y, Neelamegham S. 2002. Estimating the Efficiency of Cell Capture and Arrest in Flow Chambers: Study of Neutrophil Binding via E-selectin and ICAM-1. Biophys J. 83: 1934–1952. [PUBMED], [INFOTRIEVE]
- Zhang Y, Neelamegham S. 2003. PPLATE: A computer program for analysis of parallel-plate flow chamber experimental data. J of Immunol Meth. 278: 319–321
- Zhang Y, Neelamegham S. 2003. An analysis tool to quantify the efficiency of cell tethering and firm-adhesion in the parallel-plate flow chamber. J of Immunol Meth. 278: 305–317