ABSTRACT
The seedless barberry, ‘Bidaneh,’ is one of the most important cultivated small fruits in Iran. This cultivar is propagated only by suckers, and so, the establishment of large-scale orchards is not possible in a short period of time. During this research, different grafting methods were evaluated on the grafting success rate of ‘Bidaneh’ on wild types of barberry. More than 1000 grafts including cleft grafting (with hard shoots and green/semi-hard shoots), omega grafting, T-budding, and chip budding (at mid-spring and late summer) methods were performed for two consecutive years (2018 and 2019). The results showed that the grafting methods have significant effects on the percentage of callus formation and graft success (P < .01). The chip budding method resulted in the maximum callus formation (36.7%), and graft success (30%). The rate of callus formation and graft success in the cleft grafting with semi-hard shoots was 2% and 18%, respectively. No callus formation and graft success were found in the cleft in hard stem and omega grafting methods. The graft success was zero in T-budding, despite of 18.33% callus formation. The results revealed the success of grafting of ‘Bidaneh’ on wild barberry species for the first time. However, increasing the graft success rate requires further research.
Introduction
Iranian seedless barberry, Berberis integerrima ‘Bidaneh,’ Berberidaceae, is a vigorous shrub which grows in the low-input lands of the deserts in Iran (Alemardan et al., Citation2013). More than 15,000 ha of the seedless barberry are grown only in Iran, and over 97% of all the fields under B. integerrima ‘Bidaneh’ cultivation are located in Southern Khorasan province, Iran (Ahmadi Karim et al., Citation2018). The Seedless barberry cultivar, ‘Bidaneh,’ is used as dried fruit and also for food seasoning in Iran. Moreover, it is used in products, such as jam, marmalade, juice, and jelly (Alemardan et al., Citation2013). Various wild types of Berberis are grown naturally in mountainous regions of Iran and elsewhere in the world (Safari-Khuzani et al., Citation2020). B. integerrima Bunge is the most predominant wild species in Iran (Jannatizadeh and Khadivi-Khub, Citation2016). B. integerrima Bunge can improve the soil quality in semi-arid sites and high altitudes of mountainous areas, which are often considered fragile and sensitive ecosystems (Kooch and Noghre, Citation2020).
Separating the suckers from thorny shrubs, as the only commercial propagation method for Iranian seedless barberry cultivar, is laborious and costly. Furthermore, it restricts the production of large numbers of propagules for new orchard establishment. To date, no studies have been conducted on grafting to propagate seedless barberry, while wild types of barberries contain viable seeds which can germinate easily with cold scarification (Rezaei and Balandary, Citation2012). Also, some wild barberries have important properties, such as tolerance to environmental stresses (i.e., drought, cold, salinity) which can make them suitable to be used as rootstocks. The use of grafting techniques with wheat black-rust resistant barberry genotypes could be an alternative approach for susceptible black-rust barberry eradication programs in natural habitats in the world (Peterson, Citation2013).
Grafting is a horticultural technique used to combine two plants or pieces of plants as scions and rootstocks to raise a superior composite plant (Hartman and Kester, Citation1997). In most cases, the graft is more likely to be successful when the plants are genetically related. Establishing close contact between the rootstock and the scion and appropriate temperature conditions is important for grafting success and vascular cambium conductivity (Hartman and Kester, Citation1997). The environmental conditions have more effects on callus formation than the rootstock genotype (Vršič et al., Citation2015). In grafting approaches, the vascular cambium of the scion and rootstock should be tightly pressed together. The proper alignment and pressure encourage the tissues to join quickly, and allows nutrients and water to transfer from the rootstock to the scion (Baron et al., Citation2019). After grafting, the scion and rootstock produce callus and other wound-response tissues. In a successful graft, the cells surrounding the cut area differentiate to vascular tissue and vascular connections are re‐established (Baron et al., Citation2019).
One of the most important factors in graft success is the grafting method. In each crop, a specific grafting method will be more successful than other methods, such as patch budding in the walnut (Karadeniz, Citation2005), chip budding, and modified cleft grafting in grapes (ÇElİK, Citation2000; Sabir, Citation2011), tongue, and wedge grafting in the persimmon (Zenginbal, Citation2015), tongue grafting in the spur type apples (Kumar and Ananda, Citation2002) and top-cleft and tongue grafting in tea (Ranjith and Victor, Citation2017).
The main aim of this study was to find suitable grafting methods for seedless barberry cultivar ‘Bidaneh’ on B. integerrima Bunge by evaluating various grafting and budding techniques.
Materials and Methods
Plant Materials and Treatments
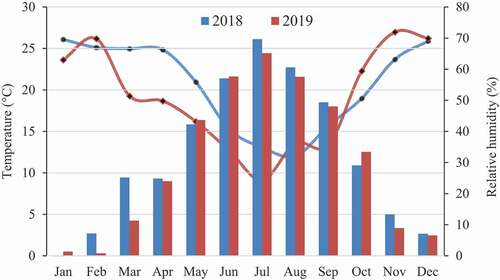
The grafting was carried out on shrubs of B. integerrima Bunge in barberry germplasm at the Agriculture Faculty, Shahrood University of Technology, Bastam, Iran, with geographical coordinates of latitude, 36ᵒ 48′ N; longitude, 54ᵒ 99′ E and 1325 m altitude with 250 mm annual rainfall. The monthly temperature and relative humidity of the place of experiment during 2018 and 2019 are presented in . Seven-year-old barberry shrubs raised from seedlings cultivated in 3 m × 2.5 m plots with regular irrigation and horticultural care were used as rootstocks. Scions for grafting and budding were provided freshly from a commercial orchard in the south Khorasan province, Iran.
Figure 1. The meteorological data during the experimental period (shahrood weather station, https://www.irimo.ir/)
In this experiment, the effects of five grafting techniques including cleft grafting (with hard shoots and green/semi-hard shoots), omega grafting, T-budding, and chip budding (at mid-spring and late summer) were assessed for two consecutive years. Treatments were based on a randomized complete block design (RCBD) with three replications. Each block included 25 shrubs and each treatment was performed on five seedlings of B. integerrima Bunge shrubs. In total, more than 1000 grafts (530 in 2018 and 475 in 2019) of seedless barberry cultivar were done. There were 187 cleft, 125 omega, 181 green cleft, 211 T-budding, 301 chip grafts (mid-spring and late summer) performed for two consecutive years.
Barberry is a very thorny shrub, and with the low-angle branches and many suckers, it is a very compact and dense bush. Removing excess suckers and reducing the shrub down to 3 to 5 main trunks were the most important pruning operations that done each year. Before grafting, the thorns in branches were removed for the convenience of the grafting operation.
Grafting Techniques
Cleft Grafting
The cleft grafting in Berberis was performed at the end of February and early March, 30 days before bud-breaking. The shrubs were severely pruned to three to five trunks, at the height of 40 cm above the ground for grafting (&b). The scions were collected from one-year-old hard shoots of ‘Bidaneh’ with the length of 12–20 cm. The process was completed by grafting one or two scions on suitable shoots of B. integerrima Bunge and a grafting wax-latex was used to maintain the moisture in graft union ().
Omega Grafting
At the same time as cleft grafting, Omega grafting was carried out using the omega-cut grafting tool (Zenport, ZJ67). The one-year-shoots of scion and rootstock with equal diameter, ~1 cm were prepared using the omega-cut machine, tangent together and fastened with adhesive grafting tape. The shoots were completely covered by tape except for one or two buds.
Cleft Grafting on Green Shoots
Cleft grafting was done in newly grown suckers at the base of barberry shrubs in spring, similar to the procedures outlined by Sabir (Citation2011) and Ranjith and Victor (Citation2017). The shoots were soft, non-lignified, and herbaceous (). The scions were provided from newly grown green shoots of ‘Bidaneh’ with 7–10 cm lengths. The basal end of each scion was cut into a gently sloping wedge about 2.5 cm with a razor. A rootstock with the same diameter of scion was selected and split up around 3 cm, and then the scion was inserted into the cleft of the rootstock. Then, the graft interface was tied and sealed with tapes (). To reduce water loss, the grafted unions were covered with a plastic bag for 1 week. Further, this grafting method was performed early in the morning or evening with the least sunlight ().
T-budding
The T-budding was done in the same manner as described in other fruit trees after the buds completely developed in the current season shoots in July or early August (Kümpers and Bishopp, Citation2015; Lewis and Alexander, Citation2008). In Berberis, the thorns in the shoots were eliminated before grafting operation.
Chip Budding
Chip budding was performed in mid to late spring and scions were provided from cold-stored bud twigs of seedless barberry. One-year dormant vegetative shoots were collected at the end of winter, before the bud breaking stage and stored in the refrigerator in 5ᵒC, while covered with a damp cloth for 30 days. Chip budding was done in a typical manner to other fruit trees with the exception of removing the thorns in barberry shoots before the grafting operation (Kümpers and Bishopp, Citation2015; Lewis and Alexander, Citation2008).
Measured Traits
Graft success and callus formation percentage was calculated in each plot. The shoot growth from the buds of scions was considered as the graft success. The percentage of grafting success was assessed 50 days after the grafting. The callus formation at the graft union was evaluated visually. The length of the scions was measured after 1 year.
Microscopic Pictures
Several cross-sections cuts were prepared from the current season and the one-year shoots of Berberis. After staining with methyl green- Alumal Carmen for 15 to 20 minutes, the related tissue was observed under an Olympus microscope (CX22).
Statistical Analysis
For the data analysis, first the zero treatments were removed. Data were analyzed separately in each year in RCBD. The statistical analysis was done using the SAS 9.1 software (SAS/STAT Software, Citation2004). The least significant differences (LSD) test was used for comparison of means (p < .01).
Results and Discussion
Grafting Success
Among the five grafting techniques in this research, chip budding and cleft grafting on green shoots were found to be successful (). The maximum success, 30%, was observed in chip grafting and followed by 18% in green cleft grafting during 2018 ( and ). The average temperature and humidity at the time of grafting operation were higher in 2018 than 2019 (). Climate conditions, temperature, and humidity are among the factors influencing callus growth and graft success (Karadeniz, Citation2005). None of the 523 grafts of T-budding, omega, and cleft was successful in the two consecutive years of the experiment ().
Table 1. The percent of callus formation and grafting success of seedless barberry cultivar grafted on Berberis integerrima rootstock with five grafting techniques
Figure 4. The successful chip budding of Iranian seedless barberry cultivar on B. integerrima Bunge after two, four and 12 weeks

In several fruit trees, numerous grafting techniques have been evaluated and the best grafting method has been introduced, but complete failure of other grafting approaches were rarely reported (El-Deen and El-Rhman, Citation2011; Kumar and Ananda, Citation2002; Rezaee et al., Citation2008; Stino et al., Citation2011; Zenginbal, Citation2015). The grafting techniques vary in many aspects, such as time of the grafting (dormant stage or active growth), type of the tissue (with wood or without wood), the amount of vascular cambium alignment, the quantity of phenolic compounds in scion or stock, and bud numbers in the scion (grafting vs. budding) (ÇElİK, Citation2000; Kumar and Ananda, Citation2002; Kümpers and Bishopp, Citation2015; Mng’omba et al., Citation2008; Zenginbal, Citation2015).
Environmental conditions including temperature and humidity, may also be vary among grafting and budding (Hartman and Kester, Citation1997). Similar to other fruit trees grafting, the cleft and omega grafting in Berberis in this study were done before bud breaking stage in the late winter, and the scion was a one-year-old softwood shoot with two or three dormant vegetative buds. At this time, the mean month temperature is low (below 10°C) (). Hartman and Kester (Citation1997) state that the best temperature for grafting success is 12°C to 32°C. On the other hand, dormant buds need more time to establish (Baron et al., Citation2019).
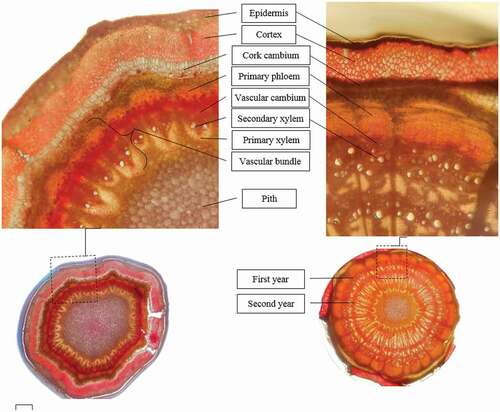
From an evolutionary standpoint, the Barberry family are closely related to monocotyledons (Loconte, Citation1993). Anatomically, Berberis shoots have a deeply-seated ring of cork cells with remarkably large lumina or hollow pore. The sclerotic ring increases considerably in the older shoots () (Loconte, Citation1993). The anatomical barriers may affect the callus formation and cambium vascular connection in Berberis. Andersen (Citation1986) stated that the failure of stem cuttings in Berberis species could be mostly attributed to the anatomical barriers and the absence of rooting co-factors. Incorrect anatomical tissue alignment between one-year-old scion and older stem of rootstock in Berberis at cleft grafting may be triggered as an anatomical barrier. On the other hand, barberries are famous for their phenolic compound such as Berberin in their roots and stems (Končić et al., Citation2010). Phenolic compounds are involved in the polysaccharides and lignin formation that leads to solid tissue in the graft area (Errea, Citation1998). Phenols caused poor callus formation at the grafting union, and hence implicated in graft incompatibility (Mng’omba et al., Citation2008).
In the chip budding method, the scion consisted of a bud with some stem parts (). Unlike other trees, in Berberis the vascular cambium is located in a deeper layer of the stem (Rathi et al., Citation2013) (). Both the scion and the rootstock in chip budding are one-year-old shoots with the same anatomical structure. More cambium alignment may occur in this budding. In addition, the phenolic compound berberin is lower in one-year-old shoots than in older tissue (Imenshahidi and Hosseinzadeh, Citation2019) which may result in more graft success in chip budding. For a successful graft, alignment between the tissues near the cambium is necessary to form a continuous connection (Kümpers and Bishopp, Citation2015).
Figure 5. Anatomy of cross section of Berberis stem stained with methyl green under the microscope view. The current growth shoot (left) and one years-old shoot (right
In this experiment, only the grafts in late spring and early summer, in active growing stages, were successful. This may be due to appropriate environmental conditions or more meristem activity in the rootstock and the scion during spring (Hartman and Kester, Citation1997; Sabir, Citation2011). The average monthly temperature from May to July ranged from 15.85 to 26.13 in 2018 and from 16.37 to 26.43 in 2019 (). Suitable temperature and meristem activity in grafting site facilitates the callus formation, and increases the success rate of grafting (Stino et al., Citation2011).
Callus Formation
The highest callus formation was observed in chip budding with 36% in 2018 and 28% in 2019. The lowest callus formation occured in the green semi-hard-cleft grafting with 2% in 2018 and 1.5% in 2019. No callus formation was observed in Omega and cleft methods. The callus tissue is the first communication tissue between the rootstock and the scion to transport water and nutrients. In the chip budding procedure, the callus formation, and vascular cambium connection and grafting success were high. In the T-budding grafting, despite 18% of callus formation, no bud growth or graft success was observed in both years ().
Failure of vascular cambium formation in T-budding could be due to the anatomical structure of stem in barberry (Rathi et al., Citation2013). The scion in T-budding includes a bud and a small amount of surrounding bark. This bark in Berberis includes cork and a thin layer of cortex () (Upwar et al., Citation2010). In the older stems, the cork comprises 12–16 layers of thick-walled highly suberized cells, and the secondary phloem and vascular cambium are located in the wide funnel-shaped medullary rays in the pith (). In the T-budding of Berberis, the separated bark may not have vascular cambium, and so, after the wound-induced callus, differentiation to vascular tissue did not occur. In cleft grafting on green semi-hard shoots, callus formation was only 1.8% while grafting success was around 18%. In accordance with our results, Sabir (Citation2011) in the study of grape showed that in the green cleft grafting, there was not enough time for callus formation. Inadequate tissue differentiation and high rate of cell division lead to faster healing in herbaceous tissue than wood, which is one of the factors that stimulate healing and increase success rate of grafting in herbaceous green tissues (Sabir, Citation2011).
The rate of the graft success depends on the cut-area in rootstock and scion. The green cleft grafting has large cut-area which results in rapid loss of water and so the wounded cells do not have enough time to heal and form callus (Sabir, Citation2011). On the other hand, some scientists report that callus formation is due to the wound healing process and is not related to grafting success (Hartman and Kester, Citation1997). The current growing shoots in Berberis, like herbaceous plants, have vascular bundles (). In the woody shoots, the continuous ring from the new tissues grows to form a cylinder of unspecialized meristem cells. For successful grafting, the vascular cambium of the rootstock and scion must be aligned, so they can grow together.
Shoot Growth
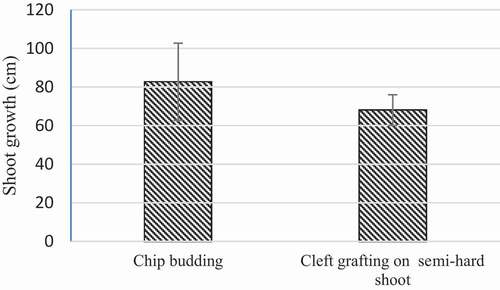
In the green cleft grafting, the growth of scion shoots ranged between 54 and 78 cm ( F), and in the chip budding method it was 47–132 cm. The average stem lengths in the green and chip grafting methods were 82 and 68 cm, respectively. These differences were not statistically significant ().
Conclusion
The results revealed that grafting can be used to propagate barberry. The rate of the graft success depends on the time and the method of grafting. The highest percentage of success was observed in chip budding in newly grown shoots at the end of spring and early summer. The green cleft grafting on semi-hard suckers in early spring was in second place in terms of graft success. The grafting methods with dormant buds, cleft, and omega grafts, were not successful. These results may be due to the anatomical barriers of barberry, phenolic compounds, environmental temperature, and dormant buds, which calls for future research in this area. Further studies on the effects of environmental conditions, and the use of substances that reduce the amount of phenolic compounds and antioxidants can increase our knowledge about Berberis graft success. The results of this study will be useful for fruit growers for the establishment of clonal orchards with commercially important scions cultivars.
Disclosure Statement
No potential conflict of interest was reported by the author(s).
References
- Ahmadi Karim, H.E., F. Hatami, R. Hosseinpour, and H. Abdshah. 2018. Irania agricultural-johad statistics. Volume iii: horticultural products. Ministry of Jihad Agric Iran. 3:13–16.
- Alemardan, A., W. Asadi, M. Rezaei, L. Tabrizi, and S. Mohammadi. 2013. Cultivation of Iranian seedless barberry (Berberis integerrima ‘Bidaneh’): A medicinal shrub. Ind Crops Prod. 50:276–287. doi: https://doi.org/10.1016/j.indcrop.2013.07.061.
- Andersen, A.S. 1986. Environmental influences on adventitious rooting in cuttings of non-woody species, New root formation in plants and cuttings, pp. 223–253. In: M.B. Jackson (ed.). New root formation in plants and cuttings. developments in plant and soil sciences. Vol. 20. Springer, Dordrecht. doi: https://doi.org/10.1007/978-94-009-4358-2_7.
- Baron, D., A.C.E. Amaro, A. Pina, and G. Ferreira. 2019. An overview of grafting re-establishment in woody fruit species. Sci. Hortic. 243:84–91. doi: https://doi.org/10.1016/j.scienta.2018.08.012.
- ÇElİK, H. 2000. The effects of different grafting methods applied by manual grafting units on grafting success in grapevines. Turkish J. Agri. Forestry 24:499–504.
- El-Deen, E.M.A.Z., and I.E.A. El-Rhman. 2011. Studies on grafting methods and dates of pistachio trees under supplemental irrigation in north sinai. Res. J. Agri. Biological. Sci. 7:456–463.
- Errea, P. 1998. Implications of phenolic compounds in graft incompatibility in fruit tree species. Sci. Hortic. 74(3):195–205. doi: https://doi.org/10.1016/S0304-4238(98)00087-9.
- Hartman, H., and D. Kester. 1997. Plant propagation: Principles and practices. New Jersey. Prentice-Hall, Inc.USA.
- Imenshahidi, M., and H. Hosseinzadeh. 2019. Berberine and barberry (Berberis vulgaris): A clinical review. Phytotherapy. Res 33(3):504–523. doi: https://doi.org/10.1002/ptr.6252.
- Jannatizadeh, A., and A. Khadivi-Khub. 2016. Morphologische Vielfalt von Berberis integerrima im Iran. Erwerbs-Obstbau 58(4):247–252. doi: https://doi.org/10.1007/s10341-016-0285-7.
- Karadeniz, T. 2005. Relationships between graft success and climatic values in walnut (Juglans regia L.). J. Central Eur. Agri. 6:631–634.
- Končić, M.Z., D. Kremer, K. Karlović, and I. Kosalec. 2010. Evaluation of antioxidant activities and phenolic content of berberis vulgaris L. and Berberis croatica Horvat. Food and Chemical Toxicology 48(8–9):2176–2180. doi: https://doi.org/10.1016/j.fct.2010.05.025.
- Kooch, Y., and N. Noghre. 2020. The effect of shrubland and grassland vegetation types on soil fauna and flora activities in a mountainous semi-arid landscape of Iran. Science of the Total Environment. 703:135497. doi: https://doi.org/10.1016/j.scitotenv.2019.135497.
- Kumar, R., and S.A. Ananda. 2002. Effects of grafting method and height on the growth of grafted plants and production of feathers in spur-type apple cultivars at nursery stage. Journal of Applied Horticulture 4(1):54–55. doi: https://doi.org/10.37855/jah.2002.v04i01.18.
- Kümpers, B.C., and A. Bishopp. 2015. Plant grafting: Making the right connections. Current Biology 25(10):R411–R413. doi: https://doi.org/10.1016/j.cub.2015.03.055.
- Lewis, W.J., and D. Alexander. 2008. Grafting and budding: A practical guide for fruit and nut plants and ornamentals. Landlinks Press, Collingwood, Australia.
- Loconte, H. 1993. Berberidaceae, p. 147–152. In: K. Kubitzki, J.G. Rohwer, and V. Bittrich (eds.). Flowering plants · dicotyledons. the families and genera of vascular plants. Springer, Berlin, Heidelberg. doi: https://doi.org/10.1007/978-3-662-02899-5_14.
- Mng’omba, S.A., E.S. du Toit, and F.K. Akinnifesi. 2008. The relationship between graft incompatibility and phenols in uapaca kirkiana müell arg. Sci. Hortic. 117(3):212–218. doi: https://doi.org/10.1016/j.scienta.2008.03.031.
- Peterson, P.D. 2013. The barberry or bread”: The public campaign to eradicate common barberry in the United States in the early 20th century. APS Features. doi: 10.
- Ranjith, K., and I.R. VictorJ. 2017. Impact of grafting methods, scion materials and number of scions on graft success, vigour and flowering of top worked plants in tea (Camellia spp.). Sci. Hortic. 220:139–146. doi: https://doi.org/10.1016/j.scienta.2017.03.039.
- Rathi, B., J. Sahu, S. Koul, and R.L. Kosha. 2013. Detailed pharmacognostical studies on Berberis aristata DC plant. Anc Sci Life 32(4):234. doi: https://doi.org/10.4103/0257-7941.131981.
- Rezaee, R., K. Vahdati, V. Grigoorian, and M. Valizadeh. 2008. Walnut grafting success and bleeding rate as affected by different grafting methods and seedling vigour. J. Hort.l Sci. Biotechnology 83(1):94–99. doi: https://doi.org/10.1080/14620316.2008.11512352.
- Rezaei, M., and A. Balandary. 2012. “Effects of gibberellic acid (GA3), stratification and acid scarification on the seed germination of three Iranian barberry genotypes.” J. Seed Sci. tech. 2 (3):11–18. In persian.
- Sabir, A. 2011. Comparison of green grafting techniques for success and vegetative development of grafted grape cultivars (Vitis spp.). International Journal of Agriculture and Biology 13:628–630.
- Safari-Khuzani, A., M. Rezaei, and A. Khadivi. 2020. Screening of the superior F1 segregating populations of barberry (Berberis spp.) for cultivation based on phenotypic characterizations. Ind Crops Prod. 158:113054. doi: https://doi.org/10.1016/j.indcrop.2020.113054.
- SAS, I. 2004. SAS/STAT® Software: Version 9. SAS Institute, Cary, NC, USA. www.support.sas.com/pubs.
- Stino, R.G., E.I. Ghoneim, I.D. Marwad, and T.R. Fadl. 2011. Performance of summer grafted superior seedless grapes grafts on different rootstock. J. Of Hort. Sci. And Ornament. Plants 3:86–90.
- Upwar, N., R. Patel, N. Waseem, and N.K. Mahobia. 2010. Pharmacognostic Evaluation of stem of Berberis aristata DC. Pharmacognosy Journal 2(17):5–9. doi: https://doi.org/10.1016/S0975-3575(10)80002-1.
- Vršič, S., B. Pulko, and L. Kocsis. 2015. Factors influencing grafting success and compatibility of grape rootstocks. Sci Hortic. 181:168–173. doi: https://doi.org/10.1016/j.scienta.2014.10.058.
- Zenginbal, H. 2015. The effects of grafting methods (by hand and with manual grafting unit) and grafting times on persimmon (Diospyros kaki L.) propagation. Acta Sci. Pol. Hortorum Cultus 14:39–50.