ABSTRACT
Abiotic stresses are significant environmental issues that restrict plant growth, productivity, and survival while also posing a threat to global food production and security. Plants produce compatible solutes known as osmolytes to adapt themselves in such changing environment. Osmolytes contribute to homeostasis maintenance, provide the driving gradient for water uptake, maintain cell turgor by osmotic adjustment, and redox metabolism to remove excess level of reactive oxygen species (ROS) and reestablish the cellular redox balance as well as protect cellular machinery from osmotic stress and oxidative damage. Perceiving the mechanisms how plants interpret environmental signals and transmit them to cellular machinery to activate adaptive responses is important for crop improvement programs to get stress–tolerant varieties. A large number of studies conducted in the last few decades have shown that osmolytes accumulate in plants and have strong associations with abiotic stress tolerance. Production of abundant osmolytes is needed for tolerance in many plant species. In addition, transgenic plants overexpressing genes for different osmolytes showed enhanced tolerance to various abiotic stresses. Many important aspects of their mechanisms of action are yet to be largely identified, especially regarding the relevance and relative contribution of specific osmolytes to the stress tolerance of a given species. Therefore, more efforts and resources should be invested in the study of the abiotic stress responses of plants in their natural habitats. The present review focuses on the possible roles and mechanisms of osmolytes and their association toward abiotic stress tolerance in plants. This review would help the readers in learning more about osmolytes and how they behave in changing environments as well as getting an idea of how this knowledge could be applied to develop stress tolerance in plants.
Introduction
Abiotic stresses on plants have been rising frequently and intensely due to rapid global climate change.Citation1,Citation2,Citation3 Plant cellular and developmental processes are disrupted by various abiotic stresses throughout their lifespan.Citation4–17 Several abiotic stresses such as salinity, drought, light, ultraviolet radiation, temperature, and heavy metal are responsible for the increase of reactive oxygen species (ROS) levels, peroxidation of lipids, activation of antioxidant system, as well as accumulation of compatible solutes.Citation18–20
Plant cells usually permit the influx, sequestering and synthesis of various solutes and accumulate them for maintaining homeostasis status and keeping the cell turgid for growth and development of plants during abiotic stress conditions.Citation21–26 In plants, osmotic adjustment mediated by the production of osmolytes has been found to protect the cellular machinery from stresses which could confer abiotic stress tolerance.Citation24,Citation27–30 The term osmolytes refers to various low molecular weight compounds or metabolites namely, sugars, polyamines, secondary metabolites, amino acids, and polyols.Citation31–36 These molecules also known as cytoptotectants due to their ability of protecting cell contents against abiotic stresses.Citation24,Citation37,Citation38 To cope with different stresses and protect themselves, plants have evolved complex and well–organized mechanisms.Citation39 Biosynthesis and accumulation of various osmolytes are considered one of the paramount responses of host plants for combating against oxidative as well as osmotic stress caused by various stressors. The increasing uncertainty of climate change definitely intensifies various abiotic stresses, in turn affecting a considerable damage to the agricultural world. Consequently, precise environmental adaptation approaches on target plants were developed to address the problems.Citation40 A number of studies have interpreted the relationship between osmolytes and their tolerance to various abiotic stresses in different crop plants either through exogenous application of osmolytes, natural accumulation, or through transgenic expression of osmolytes pathway genes that have appeared recently.Citation41–45
The overproduction of osmolytes is the result of various stress signaling pathways (phytohormones, mitogen activated protein (MAP) kinase, and calcium-signaling pathways). Several osmolytes and their biosynthesis have been studied in detail, such as proline, glycine betaine (GB) and mannitol.Citation28,Citation46–49 Moreover, several plants have been engineered metabolically for induced osmolyte biosynthesis and those transgenic plants have displayed better survival as well as tolerance of various abiotic stresses.Citation21,Citation23,Citation47,Citation48,Citation50–53
Therefore, controlling cellular osmotic balance and ion homeostasis, i.e., intracellular transportation of water, transportation of toxic ions inside the vacuole, synthesizing osmolytes in the cytoplasm, heat shock proteins, and activation of the enzymatic as well as non–enzymatic antioxidant systems are the most important conserved mechanism that confer stress tolerance in plants.Citation54–56 Considering the significant roles played by the osmolytes against different abiotic stresses, the present review gathers information related to this topic focusing on conferring abiotic stress tolerance in plants.
Biosynthesis and accumulation of osmolytes and their roles toward abiotic stress tolerance
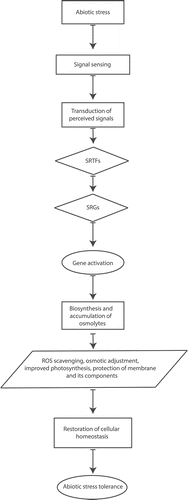
shows the processes involved in mitigating the adverse effects of abiotic stresses. Osmolytes mitigate the adverse effect induced from abiotic stresses directly or indirectly either by maintaining membrane structure or scavenging stress-induced ROS respectfully. It has been observed that various stress response signaling pathways such as MAP kinase signaling, calcium signaling, abscisic acid (ABA) signaling, and ROS signaling synergistically lead to increased biosynthesis and/or accumulation of different osmolytes for tolerance, acclimatization and detoxification under abiotic stress conditions.Citation57
Figure 1. Schematic representation of overall relation between abiotic stress and osmolytes. (Upon insight of abiotic stress signal, the related signaling pathway is initiated and results in the induction of stress-responsive transcription factors (SRTFs) which consequently upregulate stress-responsive genes (SRGs) related to biosynthesis and accumulation of osmolytes such as free amino acids and their derivatives, carbohydrates and soluble sugars, polyols, polyamines, free amines to protect and make plants tolerant of encountered abiotic stresses.)
Carbohydrates and soluble sugars
It is widely accepted that the production and collection of soluble sugars directly contribute to radical scavenging, osmotic adjustment, carbon storage, and stabilization of protein structures such as Ribulose-1,5-bisphosphate carboxylase–oxygenase (RuBisCo)Citation58 and thus increases sugar levels when exposed to stresses.Citation59,Citation60 Sugars are the main osmolytes for osmotic adjustment found in several plant species. Sugars function as osmoprotectants along with substrates for growth and regulators of gene expression during abiotic stress situations.Citation61 Under high saline environments, some rice cultivars and Medicago species showed an increase in the total soluble sugars.Citation62–63 In addition, Keunen, et al.Citation64 concluded that disaccharides and oligosaccharides are potentially involved in plant abiotic stress tolerance.
The increase in monosaccharides in plants is associated with the initial response to drought stress while the increase in fructan believed to be associated with the delayed response.Citation65 In aerial tissues of Craterostigma plantagineum, sucrose accumulation has been found to be linked during the survival phase of complete tissue dehydration.Citation66,Citation67 Trehalose, a disaccharide, has been found to be superior to other saccharides in conferring protection of plants under abiotic stress conditions, showed specific characteristics of reversible water absorption capacity against dehydration induced damages.Citation68,Citation69 Increased level of trehalose has been also observed in drought-stressed cowpea (Vigna sinensis)Citation41 Raffinose family oligosaccharides (RFOs) have been found to be involved in mitigating the effects including protecting cellular integrity during desiccation and imbibition and supplying substrates for energy generation during germination in response to harsh environmental conditions, namely, heat, cold, and dehydration.Citation70 The functions of RFOs have been examined in Arabidopsis thaliana plants under drought and cold stress conditions and mentioned that drought, high salinity, and cold-treated Arabidopsis plants gather a large amount of raffinose and galactinol, but not stachyose, proposed that raffinose and galactinol are involved in tolerance to drought, high salinity, and cold stresses, and they also act as signals to mediate abiotic stress responses.Citation71
Thus, it is acknowledged that sugars participate in specific signal transduction mechanisms and osmotic adaptation during abiotic stress environment.Citation72 It has been well documented that plants accumulate available sugars for proper metabolic functions, for example, in green gramCitation73 and pigeon peaCitation74 thus helping plants to tolerate waterlogged conditions. An experiment showed that sorbitol (5 and 10 mM) and trehalose (5 and 10 mM) exhibited protective roles in salt-sensitive cultivar of rice (Oryza sativa L. cv. KDML105) when exposed to salt stress (170 mM) situations.Citation75
Amino acids
Plant exposed to abiotic stress conditions has commonly been found to synthesize amino acids.Citation76,Citation77 Among various amino acids, proline, GB and Gamma–Aminobutyric Acid (GABA) are largely associated and therefore, became the very candidate genes for genetic manipulation for improved abiotic stress tolerance.Citation49,Citation78,Citation79
Proline
To protect plant cells during osmotic stress situation, proline serves as compatible soluteCitation80,Citation81 and being a molecular chaperone, proline also acts as an antioxidant as well as ROS scavenger.Citation82–84 Proline accumulation might be due to a novel synthesis or decrease in degradation process or both.Citation85 Generally, proline is synthesized from glutamate by Δ1-pyrroline-5-carboxylate (P5C) synthetase (P5CS) and P5C reductase (P5CR) enzymes and also from ornithine pathway which is transformed into P5C/GSA (glutamate-1-semialdehyde) via ornithine-δ-aminotransferase.Citation30,Citation86
In plants, under osmotic stress conditions, synthesis of proline from glutamine is the principal pathway while, under limited nitrogen, it is known that the ornithine approach works.Citation87 Researches have identified accelerated rate of proline biosynthesis during abiotic stress conditions which retained the low NADPH: NADP+ ratio, contributed to sustaining the electron flow between excitation centers of photosynthetic apparatus, stabilized the cell redox balance and lessen photoinhibition and damage of the photosynthetic apparatus in chloroplasts.Citation80 Proline is converted to Δ1-pyrroline-5–carboxylate (P5C) by proline dehydrogenase (PDH) and then to glutamate by P5C dehydrogenase (P5CDH) during recovery from stress. A very usual adaptive reaction to different abiotic stresses in halophytes is the increase accumulation of proline.
Being an osmoprotectant, the members of the Aizoaceae family also gather huge amounts of proline.Citation88–90 The salt tolerance features in Pancratium maritimum improved by stimulating the process of accumulation of the stress-protective proteins, proline.Citation91 It has been found that proline overproduction links with halophytic behavior in some plant species with a view to conferring tolerance against salinity but did not show the complete responses for all time when exposed to extreme abiotic stress conditions. Moreover, it has recently been documented that proline mitigates the effect of both drought and heat stress situations.Citation92 Under waterlogging conditions, accumulation of proline was reported and waterlogging tolerance in pigeon pea was found associated with proline accumulation.Citation93 Another experiment showed similar waterlogging tolerance in Vigna angularis.Citation94 Proline has been found to be associated to increase the salinity tolerance of tomato (Solanum lycopersicum L.),Citation95 soybean (Glycine max),Citation96,Citation97 Groundnut (Arachis hypogaea L.),Citation98 pea (Pisum sativum),Citation99 sainfoin (Onobrychis viciaefolia)Citation100 and mung bean (Vigna radiata L.).Citation101 Moreover, proline exhibited drought tolerance in lentil (Lens culinaris).Citation42,Citation102
In addition, proline exhibited both the salinity toleranceCitation103 and the heavy metal tolerance particularly selenium tolerance in common bean (Phaseolus vulgaris L).Citation104
Gamma-Aminobutyric Acid (GABA)
As a non-protein amino acid, Gamma-aminobutyric acid (GABA) is reported to play a substantial role in functioning of plant when exposed to abiotic stress conditions, due to their important roles in plant osmoregulation, antioxidant defense system and act as signaling molecule.Citation105,Citation106 α-ketoglutarate is the antecedent compound of GABA. Under abiotic stress conditions, α–ketoglutarate first altered by glutamate dehydrogenase to glutamate and then to GABA by glutamate decarboxylase.Citation106,Citation107
Moreover, GABA averts ROS accumulation.Citation108,Citation109 GABA accumulation has been found under salinity and flooding stress.Citation109–111 GABA alters the gene expression in various roots under saline condition. In addition, GABA activates various mechanisms associated with cascades signaling, regulation of protein degradation, hormones biosynthesis, and ROS production in Caragana intermedia (a legume shrub).Citation112 Under water deficit conditions, GABA preserved the water balance through osmotic adjustment in Nigella sativa L.Citation113 The application of GABA increased the protein content along with the activities of glutamate decarboxylase (GAD) and diamine oxidase (DAO) in rice cultivar CSR 43 (tolerant) contrasted with Pusa 44 (susceptible) at seedling stage under 200 mM saline–alkaline stress. Subsequently, it is recommended that the increased level of endogenous GABA by exogenous GABA treatment could improve salinity stress tolerance of rice seedlings associated with GABA regulation in improving DAO activity along with proline content.Citation114 Exogenous application of GABA positively facilitated polyamines biosynthesis and enhanced endogenous GABA level,Citation115 significantly reduced the salt damage indices and increased plant height, chlorophyll content and the dry and fresh weights of tomato plants in response to NaCl stress.Citation45 GABA treatment well accumulates the content of endogenous GABA and proline, which was found to be useful to protect peach fruit suffering from chilling injury.Citation116 Exogenous supply of GABA enhanced drought tolerance through the maintenance of membrane stability in perennial ryegrass (Lolium perenne) and black pepper.Citation117,Citation118
Glycine betaine
Glycine betaine, an important osmolyte, has widely been accumulated in plants and other microorganisms.Citation119,Citation120 Physiological studies have demonstrated that the increased level of accumulation of GB is linked with the degree of tolerance.Citation121 In case of osmoregulation, it significantly plays a vital role in effective protection against several abiotic stresses such as salinity, drought, and extreme heat stress.Citation28,Citation49,Citation120
Choline and glycine are the two substrates which produces GB.Citation122 In several plants, animals and microorganisms, there are two-enzyme pathway involved in the conversion of choline to GB. By the double step oxidation of choline through the toxic intermediate betaine aldehyde, GB is formed. These processes are catalyzed by choline monooxygenase (CMO) and NAD+–dependent betaine aldehyde dehydrogenase (BADH) in higher plants, and these enzymes are confined inside stroma of the chloroplasts.
The biosynthesis of GB can be induced with stress. The concentration of GB greatly lies between 40 and 400 μ mol (g DW)−1 depending on the plant species.Citation119,Citation120 In several crop plants, comprising sugar beet (Beta vulgaris), spinach (Spinacia oleracea), barley (Hordeum vulgare), wheat (Triticum aestivum) and sorghum (Sorghum bicolor), GB is known to add more GB than sensitive genotypes in response to abiotic stresses.Citation119,Citation123,Citation124 Transgenic plants with the increased level genes associated with GB synthesis, exhibited improved production of GB thus showed better tolerance to salt, cold, drought, ultraviolet light or high temperature stress.Citation119 GB might be involved in preventing accumulation of ROS, protecting membrane, and photosynthetic machinery, and activating several genes associated with stress.Citation120,Citation125 The ROS detoxification by GB as well as the reduced accumulation of ROS in transgenic plants under drought stress condition has been reported and even very lower concentration of GB provides tolerance to abiotic stress conditions.Citation126,Citation127 It is also mentioned that GB enhanced the activity of photosystem-II leading to improved abiotic stress tolerance.Citation126 It is reported that exogenous application of GB to lower- or non-accumulating plants helps in reducing adverse effects of abiotic stresses.Citation93 Moreover, GB improved photosynthesis and reduced oxidative damages with a view to conferring heavy metal stress particularly cadmium in Gossypium hirsutum L.Citation128
GB treatment enhanced the antioxidant enzyme activity, reduced ROS accumulation and thus exhibiting tolerance against chilling injury in Crataegus monogyna.Citation129 Foliar application of GB increased both the salinity and drought tolerance in soybean (Glycine max).Citation96,Citation97 Salinity tolerance in response to GB has also been observed in mung bean (Vigna radiata L.),Citation101 green bean (Phaseolus vulgaris L),Citation130 and soybean (Glycine max).Citation131,Citation132
Polyols (Sugar alcohols)
Polyols such as glycerol, mannitol, sorbitol, ononitol and pinitols etc. act as ROS scavengers, thus prevents lipid peroxidation and subsequently reduce cell damage. In general, myo-inositol, D-pinitol, and D-ononitol have the ability of scavenging hydroxyl radicals.Citation133 Under salinity stress, with the overexpression of enzymes like NADPH dependent mannitol 1–phosphate dehydrogenase in tobacco and Arabidopsis, plays essential role in the synthesis of mannitol thus provides potential tolerance.Citation134,Citation135 It is found that mannitol produced in a considerable amount in Apium graveolens under abiotic stress conditions.Citation136 Polyols have been found to synthesize with regard to freezing stress acting as osmoprotectants in halophytic plants, algae, and some insects.Citation136
Glucose-6-phosphate, on the other hand, serves as the precursor for the synthesis of several metabolites which provides protection against stresses revealed that polyols serve dual purposes both against stress protection as well as in maintenance of cells redox control.
Some stress tolerant plants showed a higher amount of cyclitols than mannitol and sorbitol. The genes involved in the synthesis of the cyclitols and modulation of their regulation can be important traits in developing abiotic stress-tolerant crops. It has been perceived that myoinositol converted into osmoprotectants D-ononitol and D-pinitol by a dual-step pathway which is primarily regulated by abiotic stress conditions.Citation137–140
Polyamines
Polyamines, a nitrogenous compounds, are ubiquitous in every alive cells and have been specified to take part in various cellular mechanisms.Citation141–148 Several types of polyamines such as diamine putrescine, triamine spermidine, and tetramine spermine play important role in the mitigating mechanism against extreme environmental stresses.Citation149–151 Accumulation of polyamines, for example, homospermine and cadaverine has been well documented in different plant tissues during normal developmental phases as well as during abiotic stresses. Their genetic actions have been ascribed to their cationic behavior which permits them to act together with all the negatively charged cellular components such as DNA, RNA, phospholipids and proteins.Citation145,Citation152–154 The beneficial role of polyamines is in their ability to block ion channels (hence, achieve osmotic adjustment by means of inorganic ions) and in ROS scavenging.
An elevation in the putrescine level has been reported during chilling stress in Arabidopsis.Citation155 Similar results has been reported in the case of wheat under cold hardening.Citation156 Exogenous spermidine treatment in both salt-sensitive and salt-tolerant rice cultivar shows recovery against salinity stress-induced plasma membrane injury.Citation157 Spermine on the other hand, inhibits DNA oxidative degradation carried out by hydroxyl radical in Mesembryanthemum crystallinum, thus ensuring the functions of polyamine as a potential ROS scavenger osmolytes.Citation158 Exogenous application of spermidine and putrescine increased the postharvest shelf‐life of Capsicum annuum L,Citation44 and improved grain filling and drought tolerance in wheat.Citation159 Putrescine treatment improved water content of leaf, dry matter accumulation, antioxidant activities and decreased cell injuries in Thymus vulgaris L. under water stress situations.Citation160 Putrescine, spermidine, and spermine were showed to increase drought toleranceCitation161 by increasing soluble protein content, relative water content, chlorophyll value, net photosynthetic rate and salinity tolerance in mung bean (Vigna radiata L.).Citation162 It is reported that putrescine exhibited salinity tolerance in pigeon pea (Cajanus cajan) by modulating anabolic and catabolic enzyme activities responsible for putrescine biosynthesis.Citation163
Bioengineering: Osmolyte induced stress tolerances
Bioengineering of osmolytes biosynthetic and metabolic genes has paved the pathway for comprehensive investigation regarding significance of osmolytes during abiotic stress responses.Citation38 The improved level of compatible nontoxic osmolytes, radical scavengers, and other transgene produces has been confirmed with the abiotic stress tolerance.Citation56 Advancement in the process of cellular and molecular biological tools has made it realistic to clone potential osmolyte biosynthetic genes and transfer them into required crop plants.Citation164 It is mentioned that overproduction of proline synthesis genes improved drought stress tolerance in tobacco plants.Citation50 A list of transgenic plants engineered to osmolyte-synthesizing or metabolic genes and their tolerance to stresses have been presented in . Overexpression of MfMIPS1 (Medicago falcate myo-inositol phosphate synthase) in tobacco resulted enriched resistance to abiotic stresses namely, cold, drought and salinity in transgenic tobacco plants.Citation165 Similar tolerances has been reported in the transgenic sweet potato plant which expresses betaine aldehyde dehydrogenase (BADH) gene.Citation166 Transgenic Brassica plants containing choline oxidase (codA) gene showed improved resistance to multiple abiotic stresses due to increased glycinebetaine accumulation.Citation164 In addition, several studies stated that otsA (trehalose-6-phosphate synthase) and otsB (trehalose-6-phosphate phosphatase) genes from E. coli showed more drought tolerance than controls in raising transgenic tobacco plants.Citation69,Citation167
Table 1. Overexpression of osmolytes biosynthetic genes in transgenic plants showing stress tolerance
Using the yeast, TPS1 gene in tomato plants had shown improved tolerance possibly by the carbohydrate alterations generated by trehalose biosynthetic pathway.Citation171 Altered cultured tobacco cell lines with ectA, ectB and ectC showed higher resistance to mannitol-induced osmotic stress with the control of the constitutive CaMV 35S promoterCitation182 and showed enhanced nitrogen supply to leaves by enhancing transpiration and by protecting RuBisCO proteins.Citation183 mentioned that transgenic plant lines of Petunia hybrida showed improved tolerance to drought stress which have been developed by 1-pyrroline-5-carboxylate synthetase genes (AtP5CS gene taken from Arabidopsis thaliana L. or OsP5CS gene from Oryza sativa L.) and they accumulated higher proline.Citation184 The transgenic wheat plants (VaP5CS from Vigna aconitifolia) showed increased tolerances to drought stress as well.Citation185 Abiotic stress conditions namely, salt, poly ethylene glycine, abscisic acid, and heat stresses showed overexpression of P5CR in Arabidopsis. Moreover, overexpression of proline P5CR increased proline accumulation in soybean (Glycine max L. Merr. cv. Ibis) exhibiting better drought tolerance.Citation186,Citation187 It is documented that maize inbred line DH4866 which was transformed with the betA gene encoding choline dehydrogenase accumulated increased level of GB and showed improved drought stress tolerance.
The potential parameters for drought tolerance are improved osmotic adjustment, reduced ion leakage, lipid membrane peroxidation, and improved level of relative water content when plants exposed to stress conditions.Citation188 Experiment shows that transgenic cotton developed by introducing CMO gene (AhCMO) replicated from Atriplex hortensis accumulated higher GB than those of non-transgenic plants in both at general and salinity stress (150 mM NaCl). Transformed wheat with the betA gene has shown tolerance to salt stress.Citation189 Moreover, overexpression of betaine by the influence of betaine aldehyde dehydrogenase (BADH) through chloroplast genetic engineering demonstrate to be a vital approach with a view to conferring salt tolerance on preferred crops.Citation190,Citation191 In addition, tobacco plants which were introduced with BADH gene for betaine aldehyde dehydrogenase showed the capability of producing GB in chloroplasts conferring salinity stress.
Transgenic plants resulted significantly better seedling growth having greater ribulose 1, 5 bisphosphate carboxylase, and phosphoenolpyruvate compared to the wild type.Citation179 Under salinity stress, transgenic rice plants overexpress trehalose synthesis genes (otsA and otsB), accumulated higher trehalose content and showed higher photosynthetic capacity, decreased photooxidative damage, as well as improved ion uptake and partitioning during salt stress.Citation192 In transgenic potato plant using the yeast TPS1 gene showed improved tolerance against drought stress compared to the wild type.Citation193 Rice OsTPP1 and OsTPP2 were transiently induced for conferring chilling, salinity and drought stress and external applications of ABA enhanced abiotic stress tolerance of rice plants.Citation194,Citation195 Several studies stated that transgenic Arabidopsis plants overexpressing TPS1Citation196 and tomato transgened with yeast TPS1 gene revealed to be drought tolerant.Citation171,Citation197 Abebe, et al.Citation170 reported that ectopic expression of the mtlD gene from Escherichia coli for the accumulation of mannitol ensured improved tolerance to water as well as salinity stress in wheat Similar results has been observed under salinity and drought conditions which meaningfully increased mannitol transport activity and OeMaT1 expression, thus conferring olive trees to survive under salinity and drought by coordinating mannitol transport with cellular metabolism.Citation198
Research showed that engineered Nicotiana tabacum and Populus tomentosa accumulates mannitol by introducing Escherichia coli mannitol-1-phosphate dehydrogenase (mltD) resulted in better salt-tolerant plants.Citation199,Citation200 Research also showed that tobacco plant transformed with imt1 added significantly higher D–ononitol and showed enhanced tolerance against drought and salt stress conditions.Citation201 Tobacco plant engineered with sacB gene for levansucrase has been showed enhanced drought tolerance.Citation202 In transgenic rice with overexpressed ADC gene from Datura stramonium accumulates higher amount of putrescine exhibiting tolerant to drought stress.Citation203,Citation204 It is mentioned that SAMDC gene of tritordeum was introduced in rice plants which accelerated a three- to fourfold increase in the level of spermidine and thus conferring salt tolerance.Citation144 found that overexpressed SAMDC gene of Arabidopsis in tobacco plants exhibit tolerance toward multiple stress.Citation205 Overexpression of SPDS (spermidine synthase) from Cucurbita ficifolia in Arabidopsis manifested a wide spectrum of tolerance toward chilling, freezing, salinity, hyperosmosis, drought stress.Citation206 Overexpression of SAMDC from Dianthus caryophyllus in transgenic tobacco manifested a wide spectrum of tolerance toward salinity, chilling and oxidative stress.Citation206
It has been found that overexpression of apple MdSPDS1 gene in European pear (Pyrus communis L.) increases the tolerance to multiple abiotic stresses by altering polyamine level.Citation207 Similarly, transgenic eggplants with oat ADC gene displayed an upsurge in tolerance toward drought, salinity, low, high temperature, and heavy metal stress.Citation208 Thus, to mitigate the ever-increasing demand of food crops across the world, transgenic approach involving osmolytes biosynthetic genes is considered as a proficient strategy to improve crop tolerance to stress.
Conclusions and perspectives
This review article highlighted various positive influences of osmolytes on plants together with the ability to induce abiotic stress tolerance. The synthesis and stress mitigation roles of osmolytes could be considered as a promising tool to improve the quality and quantity of agricultural crops which ultimately contribute to achieving sustainable development goal number two, that is, zero hunger. According to several studies, the biosynthesis and regulatory genes from different sources have been successfully exploited for generating stress-tolerant transgenic plants which have better capability to rapidly accumulate satisfactory amounts of osmolytes. The different candidate genes for the osmolyte biosynthesis pathway can be utilized for better survival of several crop plants. Due to rapid global climate change, researches need to focus on utilizing the benefits of osmolyte-mediated crop improvement to contribute in global food security. Advancements in gene insertion and manipulation might improve the generation of super crops using super genes from different sources by boosting their ability to accumulate osmolytes for better yield as well as ensure survival under various stress conditions. Many approaches including biochemical and transgenic have been put forward to develop stress tolerance or resistance in plants, although most of them have been found partially successful. Further researches need to be conducted for exploring the actual signaling behavior of different osmolytes in many cases where it is still largely unrevealed for increasing our knowledge of the actual mechanisms for plant abiotic stress tolerance and adaptation. With the exogenous application of osmolytes, the frequency of field research for a wide range of globally important crops needs to be substantially increased along with cost-effectiveness in mitigating various abiotic stresses.
Acknowledgments
Authors acknowledge Ministry of Science and Technology, People’s Republic of Bangladesh for financial support regarding this study.
Disclosure statement
Authors declare no conflict of interest.
Additional information
Funding
References
- Mittler R, Blumwald E. Genetic engineering for modern agriculture: challenges and perspectives. Annu Rev Plant Biol. 2010;1–13.
- Surabhi G-K. Update in root proteomics with special reference to abiotic stresses: achievements and challenges. J Proteins Proteomics. 2018;9:31–35.
- Ali S, Eum H-I, Cho J et al. Assessment of climate extremes in future projections downscaled by multiple statistical downscaling methods over Pakistan. Atmos Res. 2019;222:114–133.
- Boyer JS. Plant Productivity and Environment. Science. 1982;218(4571):443–448. doi:https://doi.org/10.1126/science.218.4571.443.
- Atkinson NJ, Urwin PE. The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot. 2012;63(10):3523–3543. doi:https://doi.org/10.1093/jxb/ers100.
- Suzuki N, Rivero RM, Shulaev V, Blumwald E, Mittler R. Abiotic and biotic stress combinations. New Phytol. 2014;203(1):32–43. doi:https://doi.org/10.1111/nph.12797.
- Singh S, Srivastava PK, Kumar D, Tripathi DK, Chauhan DK, Prasad SM. Morpho-anatomical and biochemical adapting strategies of maize (Zea mays L.) seedlings against lead and chromium stresses. Biocatal Agric Biotechnol. 2015;4(3):286–295. doi:https://doi.org/10.1016/j.bcab.2015.03.004.
- Zhu J-K. Abiotic Stress Signaling and Responses in Plants. Cell. 2016;167(2):313–324. doi:https://doi.org/10.1016/j.cell.2016.08.029.
- Jeandroz S, Lamotte O. Editorial: plant Responses to Biotic and Abiotic Stresses: lessons from Cell Signaling. Front Plant Sci. 2017;8:81772. doi:https://doi.org/10.3389/fpls.2017.01772.
- Tripathi DK, Singh S, Singh S, Chauhan DK, Dubey NK, Prasad R. Silicon as a beneficial element to combat the adverse effect of drought in agricultural crops. Water Stress and Crop Plants. West Sussex, UK: John Wiley & Sons, Ltd; 2016. p.682–694. https://:10.1002/9781119054450.ch39
- Tripathi A, Tripathi DK, Chauhan DK, Kumar N. Chromium (VI)-induced phytotoxicity in river catchment agriculture: evidence from physiological, biochemical and anatomical alterations in Cucumis sativus(L.) used as model species. Chem Ecol. 2016;32(1):12–33. doi:https://doi.org/10.1080/02757540.2015.1115841.
- Tripathi A, Liu S, Singh PK, et al. Differential phytotoxic responses of silver nitrate (AgNO3) and silver nanoparticle (AgNps) in Cucumis sativus L. Plant Gene. 2017;11255–11264. doi:https://doi.org/10.1016/j.plgene.2017.07.005.
- Tripathi DK, Shweta SS, Yadav V. Silicon: a potential element to combat adverse impact of UV‐B in plants. UV-B radiation: from environmental stressor to regulator of plant growth. 2017;1:175–195.
- Cheng C, Pei L, Yin T, Zhang K. Seed treatment with glycine betaine enhances tolerance of cotton to chilling stress. J Agric Sci. 2018;156(3):323–332. doi:https://doi.org/10.1017/S0021859618000278.
- Choudhary V, Chander S, Chethan C, Kumar B, Hasanuzzaman M, Fujita M, et al. Effect of seed priming on abiotic stress tolerance in plants. Plant Tolerance Environ Stress Role Phytoprotectants. 2019;29–46.
- Dutta T, Neelapu NR, Wani SH, Challa S. Compatible solute engineering of crop plants for improved tolerance toward abiotic stresses. Biochemical, physiological and molecular avenues for combating abiotic stress tolerance in plants: UK: Elsevier; 2018. p. 221–254.
- Khan MIR, Reddy PS, Ferrante A, Khan NA. Plant signaling molecules: role and regulation under stressful environments. Woodhead Publishing; 2019.
- Arif N, Yadav V, Singh S et al. Assessment of Antioxidant Potential of Plants in Response to Heavy Metals. Plant Responses to Xenobiotics: Springer Singapore; 2016. p. 97–125. https://:10.1007/978-981-10-2860-1_5
- Arif N, Yadav V, Singh S, Singh S, Ahmad P, Mishra RK, Mishra RK. Influence of High and Low Levels of Plant-Beneficial Heavy Metal Ions on Plant Growth and Development. Frontiers Environ Sci. 2016;4:4. doi:https://doi.org/10.3389/fenvs.2016.00069.
- Singh S, Tripathi DK, Singh S, et al. Toxicity of aluminium on various levels of plant cells and organism: a review. Environ Exp Bot. 2017;137177–137193. doi:https://doi.org/10.1016/j.envexpbot.2017.01.005.
- Bohnert HJ, Jensen RG. Strategies for engineering water-stress tolerance in plants. Trends Biotechnol. 1996;14(3):89–97. doi:https://doi.org/10.1016/0167-7799(96)80929-2.
- Kuznetsov VV, Rakitin VYU, Zholkevich VN. Effects of preliminary heat-shock treatment on accumulation of osmolytes and drought resistance in cotton plants during water deficiency. Physiol Plant. 1999;107(4):399–406. doi:https://doi.org/10.1034/j.1399-3054.1999.100405.x.
- Parvanova D, Ivanov S, Konstantinova T, Karanov E, Atanassov A, Tsvetkov T, Alexieva V, Djilianov D. Transgenic tobacco plants accumulating osmolytes show reduced oxidative damage under freezing stress. Plant Physiol Biochem. 2004;42(1):57–63. doi:https://doi.org/10.1016/j.plaphy.2003.10.007.
- Yancey PH. Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. J Exp Biol. 2005;208(15):2819–2830. doi:https://doi.org/10.1242/jeb.01730.
- Sharma SS, Dietz K-J. The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot. 2006;57(4):711–726. doi:https://doi.org/10.1093/jxb/erj073.
- Burg MB, Ferraris JD. Intracellular organic osmolytes: function and regulation. J Biol Chem. 2008;283(12):7309–7313. doi:https://doi.org/10.1074/jbc.R700042200.
- Ashraf M, Harris PJC. Potential biochemical indicators of salinity tolerance in plants. Plant Sci. 2004;166(1):3–16. doi:https://doi.org/10.1016/j.plantsci.2003.10.024.
- Ashraf M, Foolad MR. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot. 2007;59(2):206–216. doi:https://doi.org/10.1016/j.envexpbot.2005.12.006.
- Chen H, Jiang J-G. Osmotic adjustment and plant adaptation to environmental changes related to drought and salinity. Environl Rev. 2010;18(NA):309–319. doi:https://doi.org/10.1139/a10-014.
- Liang X, Zhang L, Natarajan SK, Becker DF. Proline mechanisms of stress survival. Antioxid Redox Signal. 2013;19(9):998–1011. doi:https://doi.org/10.1089/ars.2012.5074.
- Flowers TJ, Colmer TD. Salinity tolerance in halophytes*. New Phytol. 2008;179(4):945–963. doi:https://doi.org/10.1111/j.1469-8137.2008.02531.x.
- Türkan I, Demiral T. Recent developments in understanding salinity tolerance. Environ Exp Bot. 2009;67(1):2–9. doi:https://doi.org/10.1016/j.envexpbot.2009.05.008.
- Roychoudhury A, Chakraborty M. Biochemical and molecular basis of varietal difference in plant salt tolerance. Annu Res Rev Biol. 2013;422–454.
- Roychoudhury A, Das K. Functional role of polyamines and polyamine-metabolizing enzymes during salinity, drought and cold stresses. In: Plant Adaptation to Environmental Change: significance of Amino Acids And Their Derivatives. Wallingford (UK): CAB International Publishers; 2014. p. 141–156.
- Roychoudhury A, Banerjee A. Endogenous glycine betaine accumulation mediates abiotic stress tolerance in plants. Trop Plant Res. 2016;3:105–111.
- Hussain TM, Hazara M, Sultan Z, Saleh BK, Gopal GR. Recent advances in salt stress biology a review. Biotechnol Mol Biol Rev. 2008;3:8–13.
- Khan SH, Ahmad N, Ahmad F, Kumar R. Naturally occurring organic osmolytes: from cell physiology to disease prevention. IUBMB Life. 2010;62(12):891–895. doi:https://doi.org/10.1002/iub.406.
- Groppa M, Benavides M. Polyamines and abiotic stress: recent advances. Amino Acids. 2008;34(1):35. doi:https://doi.org/10.1007/s00726-007-0501-8.
- Munns R, Tester M. Mechanisms of Salinity Tolerance. Annu Rev Plant Biol. 2008;59(1):651–681. doi:https://doi.org/10.1146/annurev.arplant.59.032607.092911.
- Beebe S, Ramirez J, Jarvis A, Rao IM, Mosquera G, Bueno JM, et al. Genetic improvement of common beans and the challenges of climate change. Crop Adapt Clim Change. 2011;356–369.
- Khater M, Dawood MG, Sadak MS, Shalaby M, El-Awadi M, El-Din KG. Enhancement the performance of cowpea plants grown under drought conditions via trehalose application. Middle East J Agric Res. 2018;7:782–800.
- Bekka S, Abrous-belbachir O, Djebbar R. Effects of exogenous proline on the physiological characteristics of Triticum aestivum L. and Lens culinaris Medik. under drought stress. Acta agriculturae Slovenica. 2018;111(2):477–491. doi:https://doi.org/10.14720/aas.2018.111.2.20.
- Priya M, Sharma L, Kaur R, Bindumadhava H, Nair RM, Siddique K, Nayyar H. GABA (γ-aminobutyric acid), as a thermo-protectant, to improve the reproductive function of heat-stressed mungbean plants. Sci Rep. 2019;9(1):1–14. doi:https://doi.org/10.1038/s41598-019-44163-w.
- Patel N, Gantait S, Panigrahi J. Extension of postharvest shelf-life in green bell pepper (Capsicum annuum L.) using exogenous application of polyamines (spermidine and putrescine). Food Chem. 2019;275:681–687.
- Wu X, Jia Q, Ji S, Gong B, Li J, Lü G, Gao H. Gamma-aminobutyric acid (GABA) alleviates salt damage in tomato by modulating Na+ uptake, the GAD gene, amino acid synthesis and reactive oxygen species metabolism. BMC Plant Biol. 2020;20(1):1–21. doi:https://doi.org/10.1186/s12870-020-02669-w.
- Yoshiba Y, Kiyosue T, Nakashima K, Yamaguchi-Shinozaki K, Shinozaki K. Regulation of Levels of Proline as an Osmolyte in Plants under Water Stress. Plant Cell Physiol. 1997;38(10):1095–1102. doi:https://doi.org/10.1093/oxfordjournals.pcp.a029093.
- Hare PD, Cress WA, Van Staden J. Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environ. 1998;21(6):535–553. doi:https://doi.org/10.1046/j.1365-3040.1998.00309.x.
- Karakas B, Ozias-Akins P, Stushnoff C, Suefferheld M, Rieger M. Salinity and drought tolerance of mannitol-accumulating transgenic tobacco. Plant Cell Environ. 1997;20(5):609–616. doi:https://doi.org/10.1111/j.1365-3040.1997.00132.x.
- Giri J. Glycinebetaine and abiotic stress tolerance in plants. Plant Signal Behav. 2011;6(11):1746–1751. doi:https://doi.org/10.4161/psb.6.11.17801.
- Kishor PBK, Hong Z, Miao GH, Hu CAA, Verma DPS. Overexpression of [delta]-Pyrroline-5-Carboxylate Synthetase Increases Proline Production and Confers Osmotolerance in Transgenic Plants. Plant Physiol. 1995;108(4):1387–1394. doi:https://doi.org/10.1104/pp.108.4.1387.
- Shen B, Jensen RG, Bohnert HJ. Increased resistance to oxidative stress in transgenic plants by targeting mannitol biosynthesis to chloroplasts. Plant Physiol. 1997;113(4):1177–1183. doi:https://doi.org/10.1104/pp.113.4.1177.
- Sakamoto A, Murata N. The role of glycine betaine in the protection of plants from stress: clues from transgenic plants. Plant Cell Environ. 2002;25(2):163–171. doi:https://doi.org/10.1046/j.0016-8025.2001.00790.x.
- Yang Q, Chen -Z-Z, Zhou X-F, Yin H-B, Li X, Xin X-F, Hong X-H, Zhu J-K, Gong Z. Overexpression of SOS (Salt Overly Sensitive) genes increases salt tolerance in transgenic Arabidopsis. Mol Plant. 2009;2(1):22–31. doi:https://doi.org/10.1093/mp/ssn058.
- Zhu J-K. Plant salt tolerance. Trends Plant Sci. 2001;6(2):66–71. doi:https://doi.org/10.1016/s1360-1385(00)01838-0.
- Munns R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002;25(2):239–250. doi:https://doi.org/10.1046/j.0016-8025.2001.00808.x.
- Vinocur B, Altman A. Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Curr Opin Biotechnol. 2005;16(2):123–132. doi:https://doi.org/10.1016/j.copbio.2005.02.001.
- Jogawat A. Osmolytes and their role in abiotic stress tolerance in plants. Mol Plant Abiotic Stress Biol Biotechnol. 2019;91–104.
- Dubey RS, Singh AK. Salinity Induces Accumulation of Soluble Sugars and Alters the Activity of Sugar Metabolising Enzymes in Rice Plants. Biologia Plantarum. 1999;42(2):233–239. doi:https://doi.org/10.1023/a:1002160618700.
- Strand A, Hurry V, Henkes S, Huner N, Gustafsson P, Gardeström P, Stitt M. Acclimation of Arabidopsis leaves developing at low temperatures. Increasing cytoplasmic volume accompanies increased activities of enzymes in the Calvin cycle and in the sucrose-biosynthesis pathway. Plant Physiol. 1999;119(4):1387–1398. doi:https://doi.org/10.1104/pp.119.4.1387.
- Gill PK, Sharma AD, Singh P, Bhullar SS. Effect of various abiotic stresses on the growth, soluble sugars and water relations of sorghum seedlings grown in light and darkness. Bulg J Plant Physiol. 2001;27:72–84.
- Koch KE. Carbohydrate-Modulated Gene Expression in Plants. Annu Rev Plant Physiol Plant Mol Biol. 1996;47(1):509–540. doi:https://doi.org/10.1146/annurev.arplant.47.1.509.
- López M, Tejera NA, Iribarne C, Lluch C, Herrera-Cervera JA. Trehalose and trehalase in root nodules of Medicago truncatula and Phaseolus vulgaris in response to salt stress. Physiol Plant. 2008;134(4):575–582. doi:https://doi.org/10.1111/j.1399-3054.2008.01162.x.
- Cha-Um S, Charoenpanich A, Roytrakul S, Kirdmanee C. Sugar accumulation, photosynthesis and growth of two indica rice varieties in response to salt stress. Acta Physiologiae Plantarum. 2009;31(3):477–486. doi:https://doi.org/10.1007/s11738-008-0256-1.
- Keunen ELS, Peshev D, Vangronsveld J, Van Den Ende WIM, Cuypers ANN. Plant sugars are crucial players in the oxidative challenge during abiotic stress: extending the traditional concept. Plant Cell Environ. 2013;36(7):1242–1255. doi:https://doi.org/10.1111/pce.12061.
- Kerepesi I, Galiba G. Osmotic and Salt Stress-Induced Alteration in Soluble Carbohydrate Content in Wheat Seedlings. Crop Sci. 2000;40(2):482–487. doi:https://doi.org/10.2135/cropsci2000.402482x.
- Norwood M, Truesdale MR, Richter A, Scott P. Photosynthetic carbohydrate metabolism in the resurrection plant Craterostigma plantagineum. J Exp Bot. 2000;51(343):159–165. doi:https://doi.org/10.1093/jexbot/51.343.159.
- Whittaker A, Bochicchio A, Vazzana C, Lindsey G, Farrant J. Changes in leaf hexokinase activity and metabolite levels in response to drying in the desiccation-tolerant species Sporobolus stapfianus and Xerophyta viscosa. J Exp Bot. 2001;52(358):961–969. doi:https://doi.org/10.1093/jexbot/52.358.961.
- Rontein D, Basset G, Hanson AD. Metabolic Engineering of Osmoprotectant Accumulation in Plants. Metab Eng. 2002;4(1):49–56. doi:https://doi.org/10.1006/mben.2001.0208.
- Delorge I, Janiak M, Carpentier S, Van Dijck P. Fine tuning of trehalose biosynthesis and hydrolysis as novel tools for the generation of abiotic stress tolerant plants. Front Plant Sci. 2014: 5147–147. doi:https://doi.org/10.3389/fpls.2014.00147.
- ElSayed AI, Rafudeen MS, Golldack D, Weber A. Physiological aspects of raffinose family oligosaccharides in plants: protection against abiotic stress. Plant Biol. 2013;16(1):1–8. doi:https://doi.org/10.1111/plb.12053.
- Valluru R, Van Den Ende W. Myo-inositol and beyond– emerging networks under stress. Plant Sci. 2011;181(4):387–400. doi:https://doi.org/10.1016/j.plantsci.2011.07.009.
- Radomiljac JD, Whelan J, Van Der Merwe M. Coordinating metabolite changes with our perception of plant abiotic stress responses: emerging views revealed by integrative-omic analyses. Metabolites. 2013;3(3):761–786. doi:https://doi.org/10.3390/metabo3030761.
- Kumutha D, Sairam R, Meena R. Role of root carbohydrate reserves and their mobilization in imparting waterlogging tolerance in green gram (Vigna radiata (L.) Wilczek) genotypes. Indian J Plant Physiol. 2008;13:339–346.
- Sheokand S. Effect of waterlogging, salinity and their interaction on growth, oxidative and carbohydrate metabolism in pigeonpea (Cajanus cajan L. Millsp.) genotypes. CCSHAU; 2016.
- Theerakulpisut P, Gunnula W. Exogenous Sorbitol and Trehalose mitigated salt stress damage in salt-sensitive but not salt-tolerant rice seedlings. Asian J Crop Sci. 2012;4(4):165–170. doi:https://doi.org/10.3923/ajcs.2012.165.170.
- Rhodes D, Handa S, Bressan RA. Metabolic changes associated with adaptation of plant cells to water stress. Plant Physiol. 1986;82(4):890–903. doi:https://doi.org/10.1104/pp.82.4.890.
- Lugan R, Niogret M-F, Leport L, Guégan J-P, Larher FR, Savouré A, et al. Metabolome and water homeostasis analysis of Thellungiella salsuginea suggests that dehydration tolerance is a key response to osmotic stress in this halophyte. The Plant Journal 2010; 64(2):215–229. https://doi:10.1111/j.1365-313x.2010.04323.x
- Huang J, Hirji R, Adam L, Rozwadowski KL, Hammerlindl JK, Keller WA, et al. Genetic engineering of glycinebetaine production toward enhancing stress tolerance in plants: metabolic limitations. Plant Physiol. 2000;122(3):747–756. https://doi:10.1104/pp.122.3.747
- Suprasanna P, Rai AN, HimaKumari P, Kumar SA, Kavi Kishor P. Modulation of proline: implications in plant stress tolerance and development. In: Anjum NA, Gill SS, Gill R, editors. Plant Adaptation to Environmental Change. UK: CABI Publishers; 2014. p. 68–93.
- Hare P, Cress W. Metabolic implications of stress-induced proline accumulation in plants. Plant Growth Regul. 1997;21(2):79–102. doi:https://doi.org/10.1023/A:1005703923347.
- Kavi Kishor PB, Sreenivasulu N. Is proline accumulation per se correlated with stress tolerance or is proline homeostasis a more critical issue? Plant Cell Environ. 2013;37(2):300–311. doi:https://doi.org/10.1111/pce.12157.
- Smirnoff N, Cumbes QJ. Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry. 1989;28(4):1057–1060. doi:https://doi.org/10.1016/0031-9422(89)80182-7.
- Mohanty P, Matysik J. Effect of proline on the production of singlet oxygen. Amino Acids. 2001;21(2):195–200. doi:https://doi.org/10.1007/s007260170026.
- Matysik J, Alia BB, Mohanty P. Molecular mechanisms of quenching of reactive oxygen species by proline under stress in plants. Curr Sci. 2002;525–532.
- Dos Reis SP, Lima AM, De Souza CRB. Recent molecular advances on downstream plant responses to abiotic stress. Int J Mol Sci. 2012;13(7):8628–8647. doi:https://doi.org/10.3390/ijms13078628.
- Rai AN, Penna S. Molecular evolution of plant P5CS gene involved in proline biosynthesis. Mol Biol Rep. 2013;40(11):6429–6435. doi:https://doi.org/10.1007/s11033-013-2757-2.
- Delauney A, Hu C, Kishor P, Verma D. Cloning of ornithine delta-aminotransferase cDNA from Vigna aconitifolia by trans-complementation in Escherichia coli and regulation of proline biosynthesis. J Biol Chem. 1993;268(25):18673–18678. doi:https://doi.org/10.1016/S0021-9258(17)46682-8.
- Slama I, Abdelly C, Bouchereau A, Flowers T, Savouré A. Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann Bot. 2015;115(3):433–447. doi:https://doi.org/10.1093/aob/mcu239.
- Lokhande VH, Nikam TD, Patade VY, Ahire ML, Suprasanna P. Effects of optimal and supra-optimal salinity stress on antioxidative defence, osmolytes and in vitro growth responses in Sesuvium portulacastrum L. Plant Cell, Tissue and Organ Culture (PCTOC). 2011;104(1):41–49. doi:https://doi.org/10.1007/s11240-010-9802-9.
- Deuschle K, Funck D, Hellmann H, Däschner K, Binder S, Frommer WB. A nuclear gene encoding mitochondrial Δ1-pyrroline-5-carboxylate dehydrogenase and its potential role in protection from proline toxicity. Plant J. 2001;27(4):345–356. doi:https://doi.org/10.1046/j.1365-313X.2001.01101.x.
- Khedr AHA. Proline induces the expression of salt-stress-responsive proteins and may improve the adaptation of Pancratium maritimum L. to salt-stress. J Exp Bot. 2003;54(392):2553–2562. doi:https://doi.org/10.1093/jxb/erg277.
- Cvikrová M, Gemperlová L, Martincová O, Vanková R. Effect of drought and combined drought and heat stress on polyamine metabolism in proline-over-producing tobacco plants. Plant Physiol Biochem. 2013: 737–15. doi:https://doi.org/10.1016/j.plaphy.2013.08.005.
- Singh VP, Srivasatava J, Bansal R. Biochemical responses as stress indicator to water logging in pigeon pea (Cajanus cajan L.). 2017.
- Ullah I, Waqas M, Khan MA, Lee I-J, Kim W-C. Exogenous ascorbic acid mitigates flood stress damages of Vigna angularis. Appl Biol Chem. 2017;60:603–614.
- Kahlaoui B, Hachicha M, Misle E, Fidalgo F, Teixeira J. Physiological and biochemical responses to the exogenous application of proline of tomato plants irrigated with saline water. J Saudi Soc Agric Sci. 2018;17(1):17–23. doi:https://doi.org/10.1016/j.jssas.2015.12.002.
- El Sabagh A, Sorour S, Ragab A, Islam MS, Barutçular C, Ueda A, et al. Alleviation of adverse effects of salt stress on soybean (Glycine max. L.) by using osmoprotectants and organic nutrients. Int J Agric Biol Eng. 2015;9(9):1014–1018.
- El Sabagh A, Sorour S, Omar A, Islam M, Ueda A, Saneoka H, et al. Soybean (Glycine Max L.) Growth Enhancement under Water Stress Conditions. International Conference on Chemical, Agricultural and Biological Sciences Istanbul, Turkey. Int J Agric Biol Eng. 2015:144–148.
- Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A. Role of proline under changing environments: a review. Plant Signal Behav. 2012;7(11):1456–1466.
- Shahid MA, Balal RM, Pervez MA, Abbas T, Aqeel MA, Javaid MM, Garcia-Sanchez F. Exogenous proline and proline-enriched Lolium perenne leaf extract protects against phytotoxic effects of nickel and salinity in Pisum sativum by altering polyamine metabolism in leaves. Turkish J Bot. 2014;38(5):914–926. doi:https://doi.org/10.3906/bot-1312-13.
- Wu G, Feng R, Li S, Du Y. Exogenous application of proline alleviates salt-induced toxicity in sainfoin seedlings. J Anim Plant Sci. 2017;27:246–251.
- Hossain MA, Hasanuzzaman M, Fujita M. Coordinate induction of antioxidant defense and glyoxalase system by exogenous proline and glycinebetaine is correlated with salt tolerance in mung bean. Frontiers Agric China. 2011;5(1):1–14. doi:https://doi.org/10.1007/s11703-010-1070-2.
- Molla MR, Ali MR, Hasanuzzaman M, Al-Mamun MH, Ahmed A, Nazim-Ud-Dowla M, Rohman MM. Exogenous Proline and Betaine-induced Upregulation of Glutathione Transferase and Glyoxalase I in Lentil (Lens culinaris) under Drought Stress. Notulae Botanicae Horti Agrobotanici Cluj-Napoca. 2014;42(1):73–80. doi:https://doi.org/10.15835/nbha4219324.
- Abdelhamid MT, Rady MM, Osman AS, Abdalla MA. Exogenous application of proline alleviates salt-induced oxidative stress in Phaseolus vulgaris L. plants. J Hortic Sci Biotechnol. 2013;88(4):439–446. doi:https://doi.org/10.1080/14620316.2013.11512989.
- Aggarwal M, Sharma S, Kaur N, Pathania D, Bhandhari K, Kaushal N, Kaur R, Singh K, Srivastava A, Nayyar H. Exogenous proline application reduces phytotoxic effects of selenium by minimising oxidative stress and improves growth in bean (Phaseolus vulgaris L.) seedlings. Biol Trace Elem Res. 2011;140(3):354–367. doi:https://doi.org/10.1007/s12011-010-8699-9.
- Nayyar H, Kaur R, Kaur S, Singh R. γ-Aminobutyric acid (GABA) imparts partial protection from heat stress injury to rice seedlings by improving leaf turgor and upregulating osmoprotectants and antioxidants. J Plant Growth Regul. 2014;33(2):408–419. doi:https://doi.org/10.1007/s00344-013-9389-6.
- Michaeli S, Fromm H. Closing the loop on the GABA shunt in plants: are GABA metabolism and signaling entwined? Front Plant Sci. 2015;6:419.
- Sita K, Kumar V. Role of Gamma Amino Butyric Acid (GABA) against abiotic stress tolerance in legumes: a review. Plant Physiol Rep. 2020;1–10.
- Barbosa JM, Singh NK, Cherry JH, Locy RD. Nitrate uptake and utilization is modulated by exogenous γ-aminobutyric acid in Arabidopsis thaliana seedlings. Plant Physiol Biochem. 2010;48(6):443–450. doi:https://doi.org/10.1016/j.plaphy.2010.01.020.
- Renault H, Roussel V, El Amrani A, Arzel M, Renault D, Bouchereau A, et al. The Arabidopsis pop2-1 mutant reveals the involvement of GABA transaminase in salt stress tolerance. BMC Plant Biol. 2010; 1020–20. doi:https://doi.org/10.1186/1471-2229-10-20.
- Kinnersley AM, Turano FJ. Gamma Aminobutyric Acid (GABA) and Plant Responses to Stress. CRC Crit Rev Plant Sci. 2000;19(6):479–509. doi:https://doi.org/10.1080/07352680091139277.
- Zhang J, Zhang Y, Du Y, Chen S, Tang H. Dynamic Metabonomic Responses of Tobacco (Nicotiana tabacum) Plants to Salt Stress. J Proteome Res. 2011;10(4):1904–1914. doi:https://doi.org/10.1021/pr101140n.
- Shi S-Q, Shi Z, Jiang Z-P, Qi L-W, Sun X-M, Li C-X, Liu J-F, Xiao W-F, Zhang S-G. Effects of exogenous GABA on gene expression ofCaragana intermedia roots under NaCl stress: regulatory roles for H2O2and ethylene production. Plant Cell Environ. 2010;33(2):149–162. doi:https://doi.org/10.1111/j.1365-3040.2009.02065.x.
- Rezaei-Chiyaneh E, Seyyedi SM, Ebrahimian E, Moghaddam SS, Damalas CA. Exogenous application of gamma-aminobutyric acid (GABA) alleviates the effect of water deficit stress in black cumin (Nigella sativa L.). Ind Crops Prod. 2018;112:741–748.
- Kaur R, Zhawar VK, Kaur G, Asthir B. Gamma-amino butyric acid effect in the alleviation of saline–alkaline stress conditions in rice. Cereal Res Commun. 2020;1–9.
- Hu X, Xu Z, Xu W, Li J, Zhao N, Zhou Y. Application of γ-aminobutyric acid demonstrates a protective role of polyamine and GABA metabolism in muskmelon seedlings under Ca (NO3) 2 stress. Plant Physiol Biochem. 2015; 92:1–10.
- Shang H, Cao S, Yang Z, Cai Y, Zheng Y. Effect of exogenous γ-aminobutyric acid treatment on proline accumulation and chilling injury in peach fruit after long-term cold storage. J Agric Food Chem. 2011;59(4):1264–1268. doi:https://doi.org/10.1021/jf104424z.
- Krishnan S, Laskowski K, Shukla V, Merewitz EB. Mitigation of drought stress damage by exogenous application of a non-protein amino acid γ–aminobutyric acid on perennial ryegrass. J Am Soc Hortic Sci. 2013;138(5):358–366. doi:https://doi.org/10.21273/JASHS.138.5.358.
- Vijayakumari K, Puthur JT. γ-Aminobutyric acid (GABA) priming enhances the osmotic stress tolerance in Piper nigrum Linn. plants subjected to PEG-induced stress. Plant Growth Regul. 2016;78(1):57–67. doi:https://doi.org/10.1007/s10725-015-0074-6.
- Rhodes D, Hanson AD. Quaternary Ammonium and Tertiary Sulfonium Compounds in Higher Plants. Annu Rev Plant Physiol Plant Mol Biol. 1993;44(1):357–384. doi:https://doi.org/10.1146/annurev.pp.44.060193.002041.
- Chen THH, Murata N. Glycinebetaine: an effective protectant against abiotic stress in plants. Trends Plant Sci. 2008;13(9):499–505. doi:https://doi.org/10.1016/j.tplants.2008.06.007.
- Saneoka H, Nagasaka C, Hahn DT, Yang WJ, Premachandra GS, Joly RJ, Rhodes D. Salt Tolerance of Glycinebetaine-Deficient and -Containing Maize Lines. Plant Physiol. 1995;107(2):631–638. doi:https://doi.org/10.1104/pp.107.2.631.
- Chen THH, Murata N. Enhancement of tolerance of abiotic stress by metabolic engineering of betaines and other compatible solutes. Curr Opin Plant Biol. 2002;5(3):250–257. doi:https://doi.org/10.1016/s1369-5266(02)00255-8.
- McCue K, Hanson A. Drought and salt tolerance: towards understanding and application. Trends Biotechnol. 1990:8358–8362. doi:https://doi.org/10.1016/0167-7799(90)90225-m.
- Yang W-J, Rich PJ, Axtell JD, Wood KV, Bonham CC, Ejeta G, Mickelbart MV, Rhodes D. Genotypic Variation for Glycinebetaine in Sorghum. Crop Sci. 2003;43(1):162. doi:https://doi.org/10.2135/cropsci2003.1620.
- Chen TH, Murata N. Glycinebetaine protects plants against abiotic stress: mechanisms and biotechnological applications. Plant Cell Environ. 2011;34(1):1–20. doi:https://doi.org/10.1111/j.1365-3040.2010.02232.x.
- Al-Taweel K, Iwaki T, Yabuta Y, Shigeoka S, Murata N, Wadano A. A bacterial transgene for catalase protects translation of d1 protein during exposure of salt-stressed tobacco leaves to strong light. Plant Physiol. 2007;145(1):258–265. doi:https://doi.org/10.1104/pp.107.101733.
- Takahashi S, Murata N. How do environmental stresses accelerate photoinhibition? Trends Plant Sci. 2008;13(4):178–182. doi:https://doi.org/10.1016/j.tplants.2008.01.005.
- Farooq M, Ali S, Hameed A, Bharwana S, Rizwan M, Ishaque W, et al. Cadmium stress in cotton seedlings: physiological, photosynthesis and oxidative damages alleviated by glycinebetaine. S Afr J Bot. 2016;10461–10468.
- Razavi F, Mahmoudi R, Rabiei V, Aghdam MS, Soleimani A. Glycine betaine treatment attenuates chilling injury and maintains nutritional quality of hawthorn fruit during storage at low temperature. Sci Hortic (Amsterdam). 2018;233188–233194.
- Abdelmotlb N, Abdel-All F, Abd EL-Hady S, EL-Miniawy S, Ghoname A. Glycine betaine and sugar beet extract ameliorated salt stress adverse effect on green bean irrigated with saline water. Middle East J Agric Res. 2019;91:42–54.
- Rezaei MA, Kaviani B, Masouleh AK. The effect of exogenous glycine betaine on yield of soybean [Glycine max (L.) Merr.] in two contrasting cultivars Pershing and DPX under soil salinity stress. Plant Omics. 2012;5:87.
- Malekzadeh P. Influence of exogenous application of glycinebetaine on antioxidative system and growth of salt-stressed soybean seedlings (Glycine max L.). Physiol Mol Biol Plants. 2015;21(2):225–232. doi:https://doi.org/10.1007/s12298-015-0292-4.
- Smirnoff N. Plant resistance to environmental stress. Curr Opin Biotechnol. 1998;9(2):214–219. doi:https://doi.org/10.1016/s0958-1669(98)80118-3.
- Tarczynski MC, Jensen RG, Bohnert HJ Expression of a bacterial mtlD gene in transgenic tobacco leads to production and accumulation of mannitol. Proceedings of the National Academy of Sciences of the United States of America 1992; 89( 7):2600–2604. https://doi.org/10.1073/pnas.89.7.2600.
- Thomas JC, Sepahi M, Arendall B, Bohnert HJ. Enhancement of seed germination in high salinity by engineering mannitol expression in Arabidopsis thaliana. Plant Cell Environ. 1995;18(7):801–806. doi:https://doi.org/10.1111/j.1365-3040.1995.tb00584.x
- Yancey P, Clark M, Hand S, Bowlus R, Somero G. Living with water stress: evolution of osmolyte systems. Science. 1982;217(4566):1214–1222. doi:https://doi.org/10.1126/science.7112124.
- Vernon DM, Bohnert HJ. A novel methyl transferase induced by osmotic stress in the facultative halophyte Mesembryanthemum crystallinum. Embo J. 1992;11(6):2077–2085. doi:https://doi.org/10.1002/j.1460-2075.1992.tb05266.x.
- Adams P, Thomas JC, Vernon DM, Bohnert HJ, Jensen RG. Distinct cellular and organismic responses to salt stress. Plant Cell Physiol. 1992;33:1215–1223.
- Rammesmayer G, Pichorner H, Adams P, Jensen RG, Bohnert HJ. Characterization of IMT1, myo-Inositol O-methyltransferase, from Mesembryanthemum crystallinum. Arch Biochem Biophys. 1995;322(1):183–188. doi:https://doi.org/10.1006/abbi.1995.1450.
- Nelson DE, Rammesmayer G, Bohnert HJ. Regulation of cell-specific inositol metabolism and transport in plant salinity tolerance. Plant Cell. 1998;10(5):753–764. doi:https://doi.org/10.1105/tpc.10.5.753.
- Galston AW, Kaur-Sawhney R, Altabella T, Tiburcio AF. Plant Polyamines in Reproductive Activity and Response to Abiotic Stress*. Botanica Acta. 1997;110(3):197–207. doi:https://doi.org/10.1111/j.1438-8677.1997.tb00629.x.
- Thomas* T, Thomas TJ. Polyamines in cell growth and cell death: molecular mechanisms and therapeutic applications. Cell Mol Life Sci. 2001;58(2):244–258. doi:https://doi.org/10.1007/pl00000852.
- Paschalidis KA, Roubelakis-Angelakis KA. Spatial and temporal distribution of polyamine levels and polyamine anabolism in different organs/tissues of the tobacco plant. Correlations with age, cell division/expansion, and differentiation. Plant Physiol. 2005;138(1):142–152. doi:https://doi.org/10.1104/pp.104.055483.
- Paschalidis KA, Roubelakis-Angelakis KA. Sites and regulation of polyamine catabolism in the tobacco plant. Correlations with cell division/expansion, cell cycle progression, and vascular development. Plant Physiol. 2005;138(4):2174–2184. doi:https://doi.org/10.1104/pp.105.063941.
- Liu JH. Polyamine biosynthesis of apple callus under salt stress: importance of the arginine decarboxylase pathway in stress response. J Exp Bot. 2006;57(11):2589–2599. doi:https://doi.org/10.1093/jxb/erl018.
- Zhao H, Yang H. Exogenous polyamines alleviate the lipid peroxidation induced by cadmium chloride stress in Malus hupehensis Rehd. Sci Hortic (Amsterdam). 2008;116(4):442–447. doi:https://doi.org/10.1016/j.scienta.2008.02.017.
- Kusano T, Yamaguchi K, Berberich T, Takahashi Y. Advances in polyamine research in 2007. J Plant Res. 2007;120(3):345–350. doi:https://doi.org/10.1007/s10265-007-0074-3.
- Kusano T, Yamaguchi K, Berberich T, Takahashi Y. The polyamine spermine rescues Arabidopsis from salinity and drought stresses. Plant Signal Behav. 2007;2(4):251–252. doi:https://doi.org/10.4161/psb.2.4.3866.
- De Oliveira LF, Elbl P, Navarro BV, Macedo AF, Dos Santos AL, Floh EI, Cooke J. Elucidation of the polyamine biosynthesis pathway during Brazilian pine (Araucaria angustifolia) seed development. Tree Physiol. 2017;37(1):116–130. doi:https://doi.org/10.1093/treephys/tpw107.
- Reis RS, De Moura Vale E, Heringer AS, Santa-Catarina C, Silveira V. Putrescine induces somatic embryo development and proteomic changes in embryogenic callus of sugarcane. J Proteomics. 2016;130:170–179.
- Mustafavi SH, Badi HN, Sękara A, Mehrafarin A, Janda T, Ghorbanpour M, Rafiee H. Polyamines and their possible mechanisms involved in plant physiological processes and elicitation of secondary metabolites. Acta Physiologiae Plantarum. 2018;40(6):1–19. doi:https://doi.org/10.1007/s11738-018-2671-2.
- Kaur-Sawhney R, Tiburcio AF, Altabella T, Galston AW. Polyamines in plants: an overview. J Cell Mol Biol. 2003; 2:1–12.
- Pang X-M, Zhang Z-Y, Wen X-P, Ban Y, Moriguchi T. Polyamines, all-purpose players in response to environment stresses in plants. Plant Stress. 2007;1:173–188.
- Kusano T, Berberich T, Tateda C, Takahashi Y. Polyamines: essential factors for growth and survival. Planta. 2008;228(3):367–381. doi:https://doi.org/10.1007/s00425-008-0772-7.
- Cuevas JC, López-Cobollo R, Alcázar R, Zarza X, Koncz C, Altabella T, Salinas J, Tiburcio AF, Ferrando A. Putrescine is involved in Arabidopsis freezing tolerance and cold acclimation by regulating abscisic acid levels in response to low temperature. Plant Physiol. 2008;148(2):1094–1105. doi:https://doi.org/10.1104/pp.108.122945.
- Nadeau P, Delaney S, Chouinard L. Effects of Cold Hardening on the Regulation of Polyamine Levels in Wheat (Triticum aestivum L.) and Alfalfa (Medicago sativa L.). Plant Physiol. 1987;84(1):73–77. doi:https://doi.org/10.1104/pp.84.1.73.
- Roy P, Niyogi K, SenGupta DN, Ghosh B. Spermidine treatment to rice seedlings recovers salinity stress-induced damage of plasma membrane and PM-bound H+-ATPase in salt-tolerant and salt-sensitive rice cultivars. Plant Sci. 2005;168(3):583–591. doi:https://doi.org/10.1016/j.plantsci.2004.08.014.
- Kuznetsov VV, Shevyakova NI. Polyamines and stress tolerance of plants. Plant Stress. 2007;1:50–71.
- Liu Y, Liang H, Lv X, Liu D, Wen X, Liao Y. Effect of polyamines on the grain filling of wheat under drought stress. Plant Physiol Biochem. 2016;100113–100129.
- Mohammadi H, Ghorbanpour M, Brestic M. Exogenous putrescine changes redox regulations and essential oil constituents in field-grown Thymus vulgaris L. under well-watered and drought stress conditions. Ind Crops Prod. 2018;122:119–132.
- Sadeghipour O. Polyamines protect mung bean [Vigna radiata (L.) Wilczek] plants against drought stress. Biologia Futura. 2019;70(1):71–78. doi:https://doi.org/10.1556/019.70.2019.09.
- Nahar K, Hasanuzzaman M, Rahman A, Alam M, Mahmud J-A, Suzuki T, et al. Polyamines confer salt tolerance in mung bean (Vigna radiata L.) by reducing sodium uptake, improving nutrient homeostasis, antioxidant defense, and methylglyoxal detoxification systems. Front Plant Sci. 2016;7:1104.
- Garg N, Sharma A. Role of putrescine (Put) in imparting salt tolerance through modulation of put metabolism, mycorrhizal and rhizobial symbioses in Cajanus cajan (L.) Millsp. Symbiosis. 2019;79(1):59–74. doi:https://doi.org/10.1007/s13199-019-00621-7.
- Wang W, Vinocur B, Altman A. Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta. 2003;218(1):1–14. doi:https://doi.org/10.1007/s00425-003-1105-5.
- Tan J, Wang C, Xiang BIN, Han R, Guo Z. Hydrogen peroxide and nitric oxide mediated cold- and dehydration-induced myo-inositol phosphate synthase that confers multiple resistances to abiotic stresses. Plant Cell Environ. 2012;36(2):288–299. doi:https://doi.org/10.1111/j.1365-3040.2012.02573.x.
- Fan W, Zhang M, Zhang H, Zhang P, Park S. Improved tolerance to various abiotic stresses in transgenic sweet potato (Ipomoea batatas) expressing spinach betaine aldehyde dehydrogenase. PloS One. 2012;7(5):e37344–e37344. doi:https://doi.org/10.1371/journal.pone.0037344.
- Pilon-Smits EAH, Terry N, Sears T, Kim H, Zayed A, Hwang S, Van Dun K, Voogd E, Verwoerd TC, Krutwagen RWHH. Trehalose-producing transgenic tobacco plants show improved growth performance under drought stress. J Plant Physiol. 1998;152(4–5):525–532. doi:https://doi.org/10.1016/s0176-1617(98)80273-3.
- Su J, Hirji R, Zhang L, He C, Selvaraj G, Wu R. Evaluation of the stress-inducible production of choline oxidase in transgenic rice as a strategy for producing the stress-protectant glycine betaine. J Exp Bot. 2006;57(5):1129–1135. doi:https://doi.org/10.1093/jxb/erj133.
- Kathuria H, Giri J, Nataraja KN, Murata N, Udayakumar M, Tyagi AK. Glycinebetaine-induced water-stress tolerance incodA-expressing transgenicindicarice is associated with up-regulation of several stress responsive genes. Plant Biotechnol J. 2009;7(6):512–526. doi:https://doi.org/10.1111/j.1467-7652.2009.00420.x.
- Abebe T, Guenzi AC, Martin B, Cushman JC. Tolerance of Mannitol-Accumulating Transgenic Wheat to Water Stress and Salinity. Plant Physiol. 2003;131(4):1748–1755. doi:https://doi.org/10.1104/pp.102.003616.
- Cortina C, Culiáñez-Macià FA. Tomato abiotic stress enhanced tolerance by trehalose biosynthesis. Plant Sci. 2005;169(1):75–82. doi:https://doi.org/10.1016/j.plantsci.2005.02.026.
- Mattioli R, Marchese D, D’Angeli S, Altamura MM, Costantino P, Trovato M. Modulation of intracellular proline levels affects flowering time and inflorescence architecture in Arabidopsis. Plant Mol Biol. 2008;66(3):277–288. doi:https://doi.org/10.1007/s11103-007-9269-1.
- Miranda JA, Avonce N, Suárez R, Thevelein JM, Van Dijck P, Iturriaga G. A bifunctional TPS–TPP enzyme from yeast confers tolerance to multiple and extreme abiotic-stress conditions in transgenic Arabidopsis. Planta. 2007;226(6):1411–1421. doi:https://doi.org/10.1007/s00425-007-0579-y.
- Yeo E-T, Kwon H-B, Han S-E, Lee J-T, Ryu J-C, Byu M. Genetic engineering of drought resistant potato plants by introduction of the trehalose-6-phosphate synthase (TPS1) gene from Saccharomyces cerevisiae. Mol Cells. 2000;10:263–268.
- Lilius G, Holmberg N, Bülow L. Enhanced NaCl Stress Tolerance in Transgenic Tobacco Expressing Bacterial Choline Dehydrogenase. Nat Biotechnol. 1996;14(2):177–180. doi:https://doi.org/10.1038/nbt0296-177.
- Holmström KO, Somersalo S, Mandal A, Palva TE, Welin B. Improved tolerance to salinity and low temperature in transgenic tobacco producing glycine betaine. J Exp Bot. 2000;51(343):177–185. doi:https://doi.org/10.1093/jexbot/51.343.177.
- Zhang J, Tan W, Yang X-H, Zhang H-X. Plastid-expressed choline monooxygenase gene improves salt and drought tolerance through accumulation of glycine betaine in tobacco. Plant Cell Rep. 2008;27(6):1113–1124. doi:https://doi.org/10.1007/s00299-008-0549-2.
- Hasthanasombut S, Ntui V, Supaibulwatana K, Mii M, Nakamura I. Expression of Indica rice OsBADH1 gene under salinity stress in transgenic tobacco. Plant Biotechnol Rep. 2009;4(1):75–83. doi:https://doi.org/10.1007/s11816-009-0123-6.
- Yang X, Liang Z, Wen X, Lu C. Genetic engineering of the biosynthesis of glycinebetaine leads to increased tolerance of photosynthesis to salt stress in transgenic tobacco plants. Plant Mol Biol. 2007;66(1–2):73–86. doi:https://doi.org/10.1007/s11103-007-9253-9.
- Holmström K-O, Mäntylä E, Welin B, Mandal A, Palva ET, Tunnela OE, et al. Drought tolerance in tobacco. Nature. 1996;379(6567):683–684. doi:https://doi.org/10.1038/379683a0.
- Karim S, Aronsson H, Ericson H, Pirhonen M, Leyman B, Welin B, Mäntylä E, Palva ET, Van Dijck P, Holmström K-O. Improved drought tolerance without undesired side effects in transgenic plants producing trehalose. Plant Mol Biol. 2007;64(4):371–386. doi:https://doi.org/10.1007/s11103-007-9159-6.
- Nakayama H, Yoshida K, Ono H, Murooka Y, Shinmyo A. Ectoine, the compatible solute of Halomonas elongata, confers hyperosmotic tolerance in cultured tobacco cells. Plant Physiol. 2000;122(4):1239–1247. doi:https://doi.org/10.1104/pp.122.4.1239.
- Moghaieb REA, Tanaka N, Saneoka H, Murooka Y, Ono H, Morikawa H, Nakamura A, Nguyen NT, Suwa R, Fujita K. Characterization of salt tolerance in ectoine-transformed tobacco plants (Nicotiana tabaccum): photosynthesis, osmotic adjustment, and nitrogen partitioning. Plant Cell Environ. 2006;29(2):173–182. doi:https://doi.org/10.1111/j.1365-3040.2005.01410.x.
- Yamada M, Morishita H, Urano K, Shiozaki N, Yamaguchi-Shinozaki K, Shinozaki K, Yoshiba Y. Effects of free proline accumulation in petunias under drought stress. J Exp Bot. 2005;56(417):1975–1981. doi:https://doi.org/10.1093/jxb/eri195.
- Vendruscolo ECG, Schuster I, Pileggi M, Scapim CA, Molinari HBC, Marur CJ, Vieira LGE. Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J Plant Physiol. 2007;164(10):1367–1376. doi:https://doi.org/10.1016/j.jplph.2007.05.001.
- Simon-Sarkadi L, Kocsy G, Varhegyi A, Galiba G, Ronde JA. Stress-induced changes in the free amino acid composition in transgenic soybean plants having increased proline content. Biologia Plantarum. 2006;50(4):793–796. doi:https://doi.org/10.1007/s10535-006-0134-x.
- Mwenye OJ, Van Rensburg L, Van Biljon A. Van der Merwe R. The role of proline and root traits on selection for drought-stress tolerance in soybeans: a review. S Afr J Plant Soil. 2016;33(4):245–256. doi:https://doi.org/10.1080/02571862.2016.1148786.
- Lv S, Yang A, Zhang K, Wang L, Zhang J. Increase of glycinebetaine synthesis improves drought tolerance in cotton. Mol Breed. 2007;20(3):233–248. doi:https://doi.org/10.1007/s11032-007-9086-x.
- He C, Yang A, Zhang W, Gao Q, Zhang J. Improved salt tolerance of transgenic wheat by introducing betA gene for glycine betaine synthesis. Plant Cell, Tissue and Organ Culture (PCTOC). 2010;101(1):65–78. doi:https://doi.org/10.1007/s11240-009-9665-0.
- Kumar S, Dhingra A, Daniell H. Plastid-expressed betaine aldehyde dehydrogenase gene in carrot cultured cells, roots, and leaves confers enhanced salt tolerance. Plant Physiol. 2004;136(1):2843–2854. doi:https://doi.org/10.1104/pp.104.045187.
- Fitzgerald TL, Waters DLE, Henry RJ. Betaine aldehyde dehydrogenase in plants. Plant Biol. 2009;11(2):119–130. doi:https://doi.org/10.1111/j.1438-8677.2008.00161.x.
- Garg AK, Kim J-K, Owens TG, Ranwala AP, Choi YD, Kochian LV, et al. Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proceedings of the National Academy of Sciences of the United States of America 2002; 99( 25):15898–15903. https://doi.org/10.1073/pnas.252637799.
- Kondrák M, Marincs F, Antal F, Juhász Z, Bánfalvi Z. Effects of yeast trehalose-6-phosphate synthase 1 on gene expression and carbohydrate contents of potato leaves under drought stress conditions. BMC Plant Biol. 2012: 1274–74. doi:https://doi.org/10.1186/1471-2229-12-74.
- Shima S, Matsui H, Tahara S, Imai R. Biochemical characterization of rice trehalose-6-phosphate phosphatases supports distinctive functions of these plant enzymes. Febs J. 2007;274(5):1192–1201. doi:https://doi.org/10.1111/j.1742-4658.2007.05658.x.
- Habibur Rahman Pramanik M, Imai R. Functional Identification of a Trehalose 6-phosphate Phosphatase Gene that is Involved in Transient Induction of Trehalose Biosynthesis during Chilling Stress in Rice. Plant Mol Biol. 2005;58(6):751–762. doi:https://doi.org/10.1007/s11103-005-7404-4.
- Avonce N, Leyman B, Mascorro-Gallardo JO, Van Dijck P, Thevelein JM, Iturriaga G. The Arabidopsis trehalose-6-P synthase AtTPS1 gene is a regulator of glucose, abscisic acid, and stress signaling. Plant Physiol. 2004;136(3):3649–3659. doi:https://doi.org/10.1104/pp.104.052084.
- Romero C, Bellés JM, Vayá JL, Serrano R, Culiáñez-Macià FA. Expression of the yeast trehalose-6-phosphate synthase gene in transgenic tobacco plants: pleiotropic phenotypes include drought tolerance. Planta. 1997;201(3):293–297. doi:https://doi.org/10.1007/s004250050069.
- Conde A, Silva P, Agasse A, Conde C, Geros H. Mannitol Transport and Mannitol Dehydrogenase Activities are Coordinated in Olea europaea Under Salt and Osmotic Stresses. Plant Cell Physiol. 2011;52(10):1766–1775. doi:https://doi.org/10.1093/pcp/pcr121.
- Hu L, Lu H, Liu Q, Chen X, Jiang X. Overexpression of mtlD gene in transgenic Populus tomentosa improves salt tolerance through accumulation of mannitol. Tree Physiol. 2005;25(10):1273–1281. doi:https://doi.org/10.1093/treephys/25.10.1273.
- Tarczynski MC, Jensen RG, Bohnert HJ. Stress Protection of Transgenic Tobacco by Production of the Osmolyte Mannitol. Science. 1993;259(5094):508–510. doi:https://doi.org/10.1126/science.259.5094.508.
- Sheveleva E, Chmara W, Bohnert HJ, Jensen RG. Increased Salt and Drought Tolerance by D-Ononitol Production in Transgenic Nicotiana tabacum L. Plant Physiol. 1997;115(3):1211–1219. doi:https://doi.org/10.1104/pp.115.3.1211.
- Pilon-Smits EAH, Ebskamp MJM, Paul MJ, Jeuken MJW, Weisbeek PJ, Smeekens SCM. Improved Performance of Transgenic Fructan-Accumulating Tobacco under Drought Stress. Plant Physiol. 1995;107(1):125–130. doi:https://doi.org/10.1104/pp.107.1.125.
- Capell T, Bassie L, Christou P Modulation of the polyamine biosynthetic pathway in transgenic rice confers tolerance to drought stress. Proceedings of the National Academy of Sciences of the United States of America 2004; 101( 26):9909–9914. https://doi.org/10.1073/pnas.0306974101.
- Peremarti A, Bassie L, Christou P, Capell T. Spermine facilitates recovery from drought but does not confer drought tolerance in transgenic rice plants expressing Datura stramonium S-adenosylmethionine decarboxylase. Plant Mol Biol. 2009;70(3):253–264. doi:https://doi.org/10.1007/s11103-009-9470-5.
- Roy M, Wu R. Overexpression of S-adenosylmethionine decarboxylase gene in rice increases polyamine level and enhances sodium chloride-stress tolerance. Plant Sci. 2002;163(5):987–992. doi:https://doi.org/10.1016/s0168-9452(02)00272-8.
- Franceschetti M, Fornalè S, Tassoni A, Zuccherelli K, Mayer MJ, Bagni N. Effects of spermidine synthase overexpression on polyamine biosynthetic pathway in tobacco plants. J Plant Physiol. 2004;161(9):989–1001. doi:https://doi.org/10.1016/j.jplph.2004.02.004.
- Wen X-P, Pang X-M, Matsuda N, Kita M, Inoue H, Hao Y-J, Honda C, Moriguchi T. Over-expression of the apple spermidine synthase gene in pear confers multiple abiotic stress tolerance by altering polyamine titers. Transgenic Res. 2007;17(2):251–263. doi:https://doi.org/10.1007/s11248-007-9098-7.
- Prabhavathi VR, Rajam MV. Polyamine accumulation in transgenic eggplant enhances tolerance to multiple abiotic stresses and fungal resistance. Plant Biotechnol. 2007;24(3):273–282. doi:https://doi.org/10.5511/plantbiotechnology.24.273.
