Abstract
Background It is well known that anterior cruciate ligament (ACL) injuries are commoner in female athletes. Accordingly, we hypothesized that serum estrogen may play some role in this sex difference. We evaluated the relationship between serum estrogen levels and the mechanical properties of the ACL in rabbits.
Animals and methods In 40 ovariectomized rabbits, the serum estrogen levels (SEL) were controlled by intramuscular injection of 17β-estradiol. The mean SEL in each rabbit was defined as the average of 5 determinations done at 1, 2, 3, 4 and 5 weeks after ovariectomy. The animals were divided into 4 groups according to the dose of estradiol administered (low, medium, high and control: L, M, H and C, respectively) and into 2 groups according to the mean SEL (high-SEL group and low-SEL group). The medial portions of ACL attached to both femur and tibia harvested at 5 weeks after ovariectomy were examined mechanically.
Results The mean serum estrogen levels in groups C, L, M and H were 37, 50, 60 and 231 pg/mL. Statistically significant differences in the mean serum estrogen levels were seen among the 4 groups, except between groups L and M. Statistically significant differences were found between groups M and H in both the ultimate tensile stress and linear stiffness. In the comparison between 2 groups using the mean SEL value, both ultimate tensile stress and linear stiffness were lower in the high-SEL group. In all animals, a positive correlation was found between ultimate tensile stress and linear stiffness.
Interpretation Our findings suggest that high SEL might be one of the factors in the multifactorial pathogenesis of ACL rupture.
Epidemiological studies have shown that female athletes suffer from anterior cruciate ligament (ACL) injuries more frequently than male athletes (Roos et al. Citation1995). In 1997, Bjordal reported that the ACL injury rate in female soccer players was almost twice as high as that in males (Bjordal et al. Citation1997). Among high school basketball players, the incidence of ACL injury was reported to be more than 3 times higher in female athletes than in male athletes (Messina et al. Citation1999). A number of pathogenetic factors have been proposed to explain this sex difference (Costill et al. Citation1976, Schantz et al. Citation1983, Grace et al. Citation1984, LaPrade and Burnett Citation1994, Hutchinson and Ireland 1995, Levy et al. Citation1997, Wojtys et al. Citation1998). Gwinn et al. (Citation2000) divided these factors into two groups: (1) intrinsic factors consisting of the lower limb alignment, shape of the intercondylar notch, joint laxity, hormonal effects, ligament size and body weight, and extrinsic factors including muscle strength and conditioning, skill level, experience and technique, field conditions and unequal access to adequate training facilities. However, the principal pathogenetic factor for the sex difference still remains unclear.
Liu et al. (Citation1996) investigated the distribution of estrogen receptors in human ACL tissue, and found that estrogen receptors were evident in the nuclei of synoviocytes, fibroblasts and cells in the blood vessel walls in the ACL. They concluded that estrogen had some effects on both the cell composition and the histological structure of the ACL. Moreover, they found that both the synthesis of collagen and the proliferation of the fibroblasts were reduced with increased estrogen concentrations (Liu et al. Citation1997).
Slauterbeck et al. (Citation1999) established an ovariectomized rabbit model to investigate the relationship between estrogen and the mechanical properties of the ACL. They divided the animals into two groups: no estradiol administered after ovariectomy (group O), and 0.25 mg of 17β-estra-diol crystals inserted after ovariectomy (group E). The serum estrogen concentration was significantly higher in group E (52 pg/mL) than in group O (16 pg/mL) prior to killing. The ultimate failure load in group E (446N) was significantly lower than that in group O (503N). From these results, they proposed that estrogen might alter the strength of the ligament. However, the relationship between serum estrogen level and the mechanical properties of the ACL was still unclear since there were only two groups in their study.
In contrast to the work of Slauterbeck et al. (Citation1999), Strickland et al. (Citation2003) and Rau et al. (Citation2005) recently reported that estrogen might not alter the material properties of the ACL, especially within a normal physiological level.
Given these controversies, we attempted to explore the hormonal effect of estrogen on the ACL at various doses. We also sought to determine the relationship between the serum estrogen level (SEL) and the mechanical properties of the ACL.
Animals and methods
Animal model
The experimental procedures were approved by the committee for animal experimentation, Tohoku University Graduate School of Medicine.
40 age-matched female Japanese White rabbits were used. The age of all animals was 32 weeks and their average body weight was 4.3 (SD 0.43) kg. All were ovariectomized under general anesthesia using an intramuscular injection of ketamine hydrochloride (0.5 mg/kg) and xylazine (1.0 mg/kg). Each rabbit was housed in a cage individually under the same conditions, including room temperature, humidity, food and water. To prevent postoperative infection, enrofloxacin (5 mg/kg) was injected once a day into the back muscle, for 3 days postoperatively.
The animals were divided into 4 groups (10 in each group) according to the dose of estradiol administered (low, medium, high and control: L, M, H and C). During surgery, 1 rabbit in Group M died due to an anesthetic problem. This animal was excluded from further analysis. No postoperative infections were seen in the other 39 rabbits. Intramuscular injection of 17β-estradiol (Ovahormon depot; Teikoku Hormone Manufacturing Co. Ltd., Japan) was performed 1, 2, 3 and 4 weeks after surgery. The doses of 17β-estradiol in groups L, M, and H were 50, 100, and 500 μg/kg. For group C, which served as control, neither estradiol nor any types of vehicles were administered. No significant difference was found in the body weight among the 4 groups at the time of killing ().
Table 1. The body weight of the rabbit and the cross-sectional area of the middle portion of the mACL among the 4 groups divided according to the dose of estradiol administered. Data are expressed as mean (SD). No significant differences were found either in body weight or in the cross-sectional area among the 4 groups at the time of killing
Collection and preparation of ACL specimens
All rabbits were killed with an overdose of pentobarbital sodium 5 weeks after the ovariectomy. The right hind-limb was disarticulated at the hip, wrapped in physiological saline-soaked gauze and stored at –20°C until the time of the mechanical testing (Woo et al. Citation1992, Danto and Woo Citation1993).
The specimens were thawed gradually at room temperature and the knees were dissected to expose the ACL. All the soft tissues except the ACL were removed. Both the femoral and the tibial insertions of the ACL were preserved. The femur and the tibia were cut transversely 7 cm apart from the knee joint. According to Woo et al. (Citation1992) and Danto and Woo (Citation1993), the ACL was divided into the lateral and the medial portions (mACL). The lateral portion of the ACL was then resected. For the mechanical testing, the femur-mACL-tibia complex was used (). Care was taken to keep the specimens moist with physiological saline during the testing procedure.
Measurement of the cross-sectional area of the mACL
The cross-sectional area of the middle portion of the mACL was measured using an area micrometer (Digimatic Indicator, ID-C125,1025; Mitutoyo, Japan), which has a 1.5-mm wide rectangular slot (Yamamoto and Hayashi Citation1998). The thickness of the mACL was measured 6 times with a constant pressure of 0.6 kg/cm2. All 6 measurements were performed at the same location (mid-portion of the mACL). For the first 3 times, the thickness of the mACL was measured in the frontal plane. The next 3 measurements were performed at the same location but the direction of the measurement was changed 90 degrees to the sagittal plane. The mean value of these 6 measurements was determined as the mean mACL thickness. The cross-sectional area of the mACL was then determined using the following equation:
Cross-sectional area (mm2) = mean mACL thickness (mm) × 1.5 (mm)
Mechanical testing
Both the femur and the tibia were fixed in metal pots with bismuth alloy (Sano et al. Citation1997). 2 screws were inserted into both the femur and the tibia to prevent the bones from slipping out of the pots during the testing procedure. The specimens were then mounted on a table-top Instron material testing machine (Instron, Tokyo, Japan).
To make sure that the fibers of the mACL were aligned along the long axis of the testing machine, the knee was flexed 50° and the tibia was internally rotated 45°. A 0.5 N preload was applied to the specimen and cyclic preconditioning from 0.0 to 0.3 mm of extension was performed (Woo et al. Citation1992, Danto and Woo Citation1993). The tensile load to failure was applied at a constant rate, 5 mm/sec, and the load-elongation curve was recorded. From this curve, the ultimate failure load and the linear stiffness were determined. Then, the ultimate tensile stress was calculated as the ultimate failure load divided by the cross-sectional area. The whole procedure of the tensile testing was videotaped to determine the failure mode of the femur-mACL-tibia complexes.
Measurement of serum estrogen level (SEL)
The serum estrogen levels (pg/mL) were measured immediately prior to ovariectomy (0 week) and 1, 2, 3, 4, and 5 weeks after surgery. The serum estrogen assay was performed using an Immulite immunoassay system (DPC, Los Angeles, CA).
The average SEL of each group was defined as the average SEL at each time point after ovariectomy of all rabbits contained in that group. The SELs sometimes differed even in the animals given the same dose of estradiol. Thus, the mean SEL in each rabbit was defined as the average of all 5 SELs (1, 2, 3, 4 and 5 weeks after surgery). Both the dose of estradiol administered and the mean SEL were separately compared to the mechanical properties of the ACL.
According to the report the report by Challis et al. (Citation1973), the highest value of serum estradiol in rabbits is 83 pg/mL on day 6 of gestation. To clarify whether non-physiologically high levels of SEL alter the material properties of the ACL, all 39 ovariectomized rabbits were divided into 2 groups using the mean SEL value (high-SEL group: < 83 pg/mL; low-SEL group: < 83 pg/mL).
Data analysis
To clarify the relationship between estrogen and the mechanical properties of the ACL, the following 5 separate comparisons were performed.
I. Comparisons of SEL among the groups with the dose of estradiol: the average SEL at each time point after ovariectomy was compared among the 4 groups (L, M, H and C) to validate our animal models. Then, the mean SEL was also compared among all 4 groups.
II. Comparisons of the mechanical properties among the groups with the dose of e : the ultimate tensile stress and the linear stiffness were compared among all 4 groups (L, M, H and C) to determine the relationship between the dose of estradiol administered and the mechanical properties of the ACL.
III. Relationships between SEL and the mechanical properties: in all 39 ovariectomized rabbits, a regression analysis was performed to determine the relationship between the mean SEL and the ultimate tensile stress or the linear stiffness.
IV. Relationships between the ultimate tensile stress and the stiffness: a regression analysis was also performed between the ultimate tensile stress and the linear stiffness in all 39 ovariectomized rabbits.
V. Comparison between the low-SEL group and the high-SEL group: the ultimate tensile stress and the linear stiffness were compared between the low-SEL group and the high-SEL group.
Statistics
The mean SEL and all mechanical data were reported as mean and 95% CI. SPSS version 11.0.1J for Windows was used to develop the database and to perform the statistical analyses.
The Dunnett T3 test was used to determine the differences in the SEL, the ultimate tensile stress, the linear stiffness, body weight and cross-sec-tional area among the 4 groups. In each group, the paired t-test was used to determine the differences in the average SEL before and after surgery. The unpaired t-test was used to compare mechanical data between low-SEL and high-SEL groups. Correlations between each category were analyzed with Pearson’s correlation coefficient. Differences were judged as being statistically significant when the p-value was less than 0.05.
Results
During the mechanical testing, neither fracture of the femur-mACL-tibia complex nor slipping of the specimen from the bismuth alloy was observed. Macroscopically, failure occurred around the tibial insertion of the mACL in all 39 rabbits. No avulsed bony fragments were seen around the distal stump of the mACL.
The cross-sectional area of the mid-portion of the mACL in groups C, L, M and H was 2.3 (SD 0.3), 1.8 (0.4), 2.1 (0.4) and 2.1 (0.4) mm2, respectively. No significant differences in the average cross-sectional area were seen among these 4 groups ().
Comparisons of SEL among the groups with the dose of estradiol
In group C, the average SEL at both the fourth and fifth week was significantly lower than that at week 0 (prior to the ovariectomy). On the other hand, the average SEL generally increased 2 weeks after surgery in groups L, M and H. In groups L and M, the average SEL at the second and fifth week was significantly higher than that at week 0. In group H, the average SEL from the second to the fifth week was significantly higher than that at week 0. For the mean SEL, statistically significant differences were seen among all 4 groups, except between groups L and M (p = 0.1) ().
Table 2. The average serum estrogen level (SEL) at each time point and mean SEL among the 4 groups divided according to the dose of estradiol administered. Data of the average SEL are expressed as mean (SD). Data of the mean SEL are expressed as mean (95% CI)
Comparisons of mechanical properties among the groups with estradiol
A statistically significant difference was found only between groups M and H regarding the ultimate tensile stress (p = 0.04). A significant difference in linear stiffness was also found between groups M and H only (p = 0.02) ().
Table 3. Mechanical properties of the femur-mACL-tibia complex among the 4 groups divided according to the dose of estradiol administered. Data are expressed as mean (CI)
Relationship between SEL and mechanical properties
In all 39 rabbits, there was a general trend for both ultimate tensile stress and linear stiffness to decrease with increase in the mean SEL. However, the correlation between mean SEL and the ultimate tensile stress was not statistically significant (r = –0.29, p = 0.07). Similarly, the correlation between mean SEL and linear stiffness was not statistically significant (r = –0.31, p = 0.06).
Relationship between ultimate tensile stress and stiffness
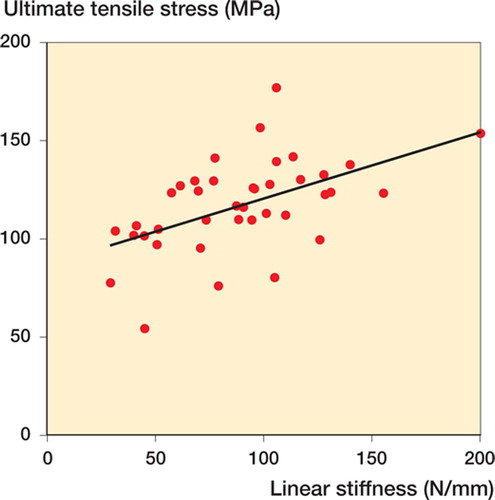
A positive correlation was found between the linear stiffness and the ultimate tensile stress (r = 0.51, p = 0.001) (), indicating that the ACL tissue with lower stiffness showed a lower value of ultimate tensile stress.
Comparison between the low-SEL group and the high-SEL group
Statistically significant differences were found in ultimate tensile stress (p = 0.02) and in linear stiffness (p = 0.01) between the low-SEL group and the hugh-SEL group ().
Table 4. The mechanical properties in 2 groups divided according to the mean SEL. Data are expressed as mean (CI)
Discussion
The pathogenetic role of estrogen in ACL rupture has not been elucidated. It has been reported that both collagen synthesis and fibroblast proliferation become reduced with an increase in serum estrogen concentration (Liu et al. 1997, Yu et al. 1999). Moreover, recent clinical studies have revealed that the incidence of ACL injuries shows a close correlation to the menstrual cycle (Lebrun Citation1994, Heitz et al. Citation1999, Wojtys et al. Citation2002). All these findings correspond well with the higher incidence of ACL injuries in female athletes.
Estrogen has been thought to affect the mechanical properties of the ACL. In particular, it has been assumed that an increased estrogen concentration might have a negative effect on the mechanical properties of the ACL. Slauterbeck et al. (Citation1999) clearly showed that administration of estradiol weakens the tensile load of the ACL. However, there were several problems in their study. Firstly, they showed the relationship between the serum estrogen level and the mechanical properties in only two groups (with or without estradiol injection after ovariectomy). Secondly, the testing speed (0.5 mm/sec) was too low to simulate the clinical setting, and it was reported that the material properties might change with the testing speed. Thirdly, the authors did not normalize their testing data with the cross-sectional area of the ACL. Individual variations such as size of the ligament or body weight may have affected their results.
In contrast to these reports, Strickland et al. (Citation2003) reported that estrogen may not affect the material properties of the sheep ACL. They used both estradiol and raloxifene, the agonist of an estrogen receptor. The serum estrogen level in their model was approximately 2 pg/mL, which simulates the luteal phase of the estrous cycle. More recently, Rau et al. (Citation2005) reported that normal physiological fluctuations in estrogen during the estrous cycle did not affect the failure properties of the rat ACL.
We used a rabbit model with 4 different doses of estradiol (0, 50, 100, and 500 μg/kg) to examine the relationship between SEL and mechanical properties in detail. In group C (controls), the SELs measured at 4 and 5 weeks after ovariectomy were lower than that measured just prior to the ovariectomy (week 0). In groups L, M and H, SEL was higher at the time of killing than that at week 0. We assumed that the SEL in groups L, M and H was successfully controlled by the estradiol administered. During the mechanical testing, we chose 5 mm/sec as the testing speed to simulate the clinical situation of ACL injury. We also measured the cross-sectional area of the ligament using an area micrometer to standardize the testing data. For further comparisons, we used the ultimate tensile stress instead of the ultimate failure load.
In the multiple comparison of the average SEL at 5 weeks after ovariectomy among the 4 groups, statistically significant differences were found among all groups except between groups L and M. We believe that the difference in the dose administered between groups L and M was too small to have resulted in a statistically significant difference.
In the comparison of the mechanical properties of the ACL among the 4 groups, group H showed significantly lower values—both for the ultimate tensile stress and for the linear stiffness—than those in group M. However, there was no significant difference in the ultimate tensile stress or the linear stiffness between any estradiol-administered groups and group C.
Since we assumed that the mean SEL might reflect the hormonal circumstances more accurately than the dose administered, we investigated the relationship between the mean SEL and the mechanical properties in all 39 rabbits. The ultimate tensile stress showed a positive correlation with the linear stiffness. It appeared that ligaments with lower linear stiffness showed lower ultimate tensile stress. Although we could not determine any significant relationships in the regression analyses, there was a general trend involving decrease in both ultimate tensile stress and linear stiffness with increase in the mean SEL. In the comparison between the high-SEL group and the low-SEL group, there were significant differences both in the ultimate tensile stress and the linear stiffness.
Based on our results, we assumed that the critical dose of estrogen might be higher than the dose used by Strickland et al. (Citation2003). With a purely physiological dose of estrogen, it might be difficult to discern statistically significant differences within a limited experimental period. We believe that the pathogenesis of ACL rupture is really multifactorial and that high SEL may be one of the pathogenetic factors.
The major limitations in this study were the small number of animals and the short observation period. Thus, longer-term observation with a larger number of animals is under way.
The authors thank Clinical Professor Katsumi Sato at Tohoku Rosai Hospital whose leadership inspired us to perform this study, and Professor N. Yamamoto of Ritsumeikan University for the information concerning the area micrometer. Finally, we thank Mr Katsuyoshi Shoji, Ms Michiko Fukuyama and Ms Masumi Nakamura at Tohoku University for their technical assistance.
No competing interests declared.
Contributions of authors
TK: planned the research concept and design, collected data, preformed the statistical analysis and wrote the first draft of the paper. TS, HS and MH: participated in developing ideas of the study and gave critical review on the manuscript. MW, YY and TK: participated in collecting the data. TM: was involved in data analysis. SK: supervised the study from a biomechanical perspective and presented valuable comments. All authors took part in the interpretation of the results and preparation of the final version of the paper.
- Bjordal J M, Arnly F, Hannestad B, Strand T. Epidemiology of anterior cruciate ligament injuries in soccer. Am J Sports Med 1997; 25(3)341–5
- Challis J R G, Davies I J, Ryan K J. The concentration of progesteron, estron and estradiol-17βin the plasma of pregnant rabbits. Endocrinology 1973; 93: 971–6
- Costill D L, Daniels J, Evans W, Fink W, Krahenbuhl G, Saltin B. Skeletal muscle enzymes and fiber composition in male and female track athletes. J Appl Physiol 1976; 40(2)149–54
- Danto M I, Woo S L. The mechanical properties of skeletally mature rabbit anterior cruciate ligament and patellar tendon over a range of strain rates. J Orthop Res 1993; 11(1)58–67
- Grace T G, Sweetser E R, Nelson M A, Ydens L R, Skipper B J. Isokinetic muscle imbalance and knee-joint injuries. A prospective blind study. J Bone Joint Surg (Am) 1984; 66(5)734–40
- Gwinn D E, Wilckens J H, McDevitt E R, Ross G, Kao T C. The relative incidence of anterior cruciate ligament injury in men and women at the United States Naval Academy. Am J Sports Med. 2000; 28(1)98–102
- Heitz N A, Eisenman P A, Beck C L, Walker J A. Hormonal changes throughout the menstrual cycle and increased anterior cruciate ligament laxity in females. J Athl Train 1999; 34(2)144–9
- Hutchinson M R, Ireland M L. Knee injuries in female athletes. Sports Med. 1995; 19(4)288–302
- LaPrade R F, Burnett Q M. II. Femoral intercondylar notch stenosis and correlation to anterior cruciate ligament injuries. A prospective study. Am J Sports Med 1994; 22(2)198–202
- Lebrun C M. The effect of the phase of the menstrual cycle and the birth control pill on athletic performance. Clin Sports Med 1994; 13(2)419–41
- Levy A S, Wetzler M J, Lewars M, Laughlin W. Knee injuries in women collegiate rugby players. Am J Sports Med 1997; 25(3)360–2
- Liu S H, al-Shaikh R, Panossian V, Yang R S, Nelson S D, Soleiman N, Finerman G A, Lane J M. Primary immunolocalization of estrogen and progesterone target cells in the human anterior cruciate ligament. J Orthop Res 1996; 14(4)526–33
- Liu S H, Al-Shaikh R A, Panossian V, Finerman G A, Lane J M. Estrogen affects the cellular metabolism of the anterior cruciate ligament. A potential explanation for female athletic injury. Am J Sports Med. 1997; 25(5)704–9
- Messina D F, Farney W C, DeLee J C. The incidence of injury in Texas high school basketball. A prospective study among male and female athletes. Am J Sports Med 1999; 27(3)294–9
- Rau M D, Renouf D, Benfield D, Otto D D, Thornton G M, Raso V J, Bagnall K M. Examination of the failure properties of the anterior cruciate ligament during the estrous cycle. Knee 2005; 12(1)37–40
- Roos H, Ornell M, Gärdsell P, Lohmander L S, Lindstrand A. Soccer after anterior crusiate ligament injury - an incompatible combination? A national survey of incidence and risk factors and a 7-year follow-up of 310 players. Acta Orthop Scand 1995; 66(2)107–12
- Sano H, Ishii H, Yeadon A, Backman D S, Brunet J A, Uhthoff H K. Degeneration at the insertion weakens the tensile strength of the supraspinatus tendon: a comparative mechanical and histologic study of the bone-tendon complex. J Orthop Res 1997; 15(5)719–26
- Schantz P, Randall-Fox E, Hutchison W, Tyden A, Astrand P O. Muscle fibre type distribution, muscle cross-sectional area and maximal voluntary strength in humans. Acta Physiol Scand 1983; 117(2)219–26
- Slauterbeck J, Clevenger C, Lundberg W, Burchfield D M. Estrogen level alters the failure load of the rabbit anterior cruciate ligament. J Orthop Res 1999; 17(3)405–8
- Strickland S M, Belknap T W, Turner S A, Wright T M, Hannafin J A. Lack of hormonal influences on mechanical properties of sheep knee ligaments. Am J Sports Med. 2003; 31(2)210–5
- Wojtys E M, Huston L J, Lindenfeld T N, Hewett T E, Greenfield M L. Association between the menstrual cycle and anterior cruciate ligament injuries in female athletes. Am J Sports Med 1998; 26(5)614–9
- Wojtys E M, Huston L J, Boynton M D, Spindler K P, Lindenfeld T N. The effect of the menstrual cycle on anterior cruciate ligament injuries in women as determined by hormone levels. Am J Sports Med 2002; 30(2)182–8
- Woo S L, Newton P O, MacKenna D A, Lyon R M. A comparative evaluation of the mechanical properties of the rabbit medial collateral and anterior cruciate ligaments. J Biomech 1992; 25(4)377–86
- Yamamoto N, Hayashi K. Mechanical properties of rabbit patellar tendon at high strain rate. Biomed Mater Eng 1998; 8(2)83–90
- Yu W D, Liu S H, Hatch J D, Panossian V, Finerman G A. Effect of estrogen on cellular metabolism of the human anterior cruciate ligament. Clin Orthop 1999, 366: 229–38