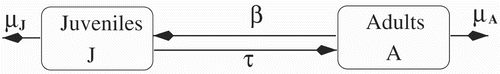Abstract
In this work, we analyse a deterministic epidemic mathematical model motivated by the propagation of a hantavirus (Puumala hantavirus) within a bank vole population (Clethrionomys glareolus). The host population is split into juvenile and adult individuals. A heterogeneous spatial chronological age and infection age structure is considered, and also indirect transmission via the environment. Maturation rates for juvenile individuals are adult density-dependent. For the reaction–diffusion systems with age structures derived, we give global existence, uniqueness and global boundedness results. A model with transmission to humans is also studied here.
AMS Subject Classification :
1. Introduction
We are mainly interested in the mathematical analysis of a deterministic mathematical model describing the propagation of a macroparasite within a single-structured host population. This study is supplemented by a related epidemiological model, wherein a macroparasite is transmitted from a reservoir host population to a second host population. This work is motivated by the specific Puumala hantavirus (PUU) – bank vole (Clethrionomys glareolus) system in Europe. In that particular system, the macroparasite is benign in the reservoir host population and can be transmitted to humans, an epidemiological dead end, with a mild lethal impact (see Citation22–24,Citation31 Citation32 for details).
First, in addition to the age-dependence that is commonly used in population dynamics Citation2,Citation14–16,Citation28, we want to take into account a stage structuration, due to the fact that the sexual maturation of juveniles depends on the density of adults: the higher the density of the adults, the slower the maturation Citation24. We have included that the hantavirus seems not to affect the demography of the bank vole population (but may be lethal for humans) and that there is no vertical transmission of the disease (offspring of infected individuals are healthy at birth). Two modes of horizontal propagation are considered: by direct contacts from infected to healthy individuals, and by contacts of healthy individuals with the environment that can be contaminated by infected individuals Citation24. In addition to time and chronological age, we consider a third structure variable which is the age of the disease in a given animal. This first model was constructed and studied in Wolf Citation30. Numerical simulations based on this model Citation31 lead to dynamics close to those which were observed on fields.
Moreover, bank voles also move in space, but the previous model does not take into account this fact. A spatial-dependent model was constructed and studied in Wolf et al. Citation32. This model highlighted the importance of the spatial structuration in infection evolution, at local and global scales. But the model studied there was simply structured for age and disease status (non-infected and infected juveniles or adults), which is not well adapted to the evolution of the disease Citation30.
The purpose of this work is to suggest and analyse a model combining the whole of these important phenomena into a single system. This will lead to a strongly structured system which is too complex for qualitative studies of the dynamic. Nevertheless, we prove that the solution of this system is unique and bounded and thus this model is a good one in order to understand the epidemic spreading and dynamic.
In Section 2, we construct a disease-free model for a closed population; we derive here a global existence, uniqueness result and then we give a global uniform bound on solutions.
Next in Section 3, we construct the epidemic model and supply global existence, uniqueness and boundedness results.
Then in Section 4, we look at a simplified model which includes transmission of the parasite to a second host population.
Lastly in Section 5, we give the proof of the results stated in the previous sections.
2. The JA disease-free model
In this section, we analyse a disease-free demographic model described in Section 1.
2.1. Modelling
The construction of the model is first based on a disease-free one for the host population. Because of intraspecific competition and different behaviours between juvenile and adult individuals (Citation5
Citation17
Citation22 and references therein), the host population is split into juvenile (J) and adult (A) sub-populations. Let J(t, x, a) and A(t, x, a) be their respective densities at time t, position x∈Ω, , n≥1, and chronological age
for juveniles and
for adults, with a
1>0 Citation16
Citation26
Citation28. The host population reads
. We assume maturation of juveniles depends on the total density of adults and cannot occur prior to age a
1. Let
be the maturation rate at time t of juveniles having age a and position x for a spatial density of adults given by
; we assume τ is non-increasing with respect to the last variable, 𝔸. Let
, be the adult fertility rate depending on the spatial population density given by
. Let
and
be the respective mortality rates for juveniles and adults.
The resulting compartmental model is depicted in ; see Wolf et al. Citation32.
Populations disperse via Fickian law with diffusion rates d J (t, a, x) and d A (t, a, x). The resulting mathematical model is the following:
2.2. Assumptions
We introduce a set of conditions used through out this work.
Definition 2.1
Suppose:
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • |
| ||||
Let
Definition 2.2
For all R>0,
| • | There exists | ||||
| • | There exists K
τ(R)>0 such that for | ||||
| • | For Z=J, A, there exists K
Z
(R)>0 such that for | ||||
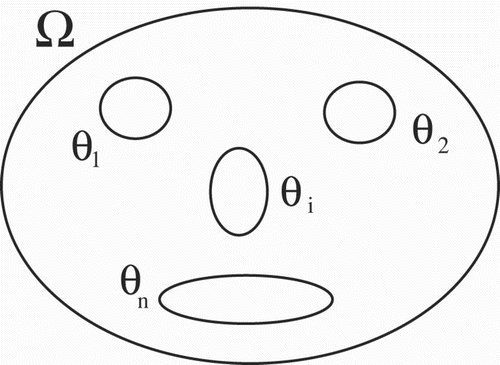
Let Ω be an open-bounded domain in ℝ
n
with a smooth boundary , such that locally Ω lies on one side of its boundary. Let η(x) be a unit normal vector to Ω along
. In order to take into account spatial heterogeneities, we introduce open subsets θ
i
with
,
having the same regularity properties as Ω ().
Let:
Definition 2.3
For Z=J, A, we suppose that
| • |
| ||||
| • |
| ||||
Remark 2.4
Discontinuity in diffusion rates implies that we cannot expect the spatial regularity afforded by classical diffusion processes. Systems with such diffusion rates are, for example, studied in Fitzgibbon et al. Citation8–10,Citation12.
2.3. Main results
We are now interested in the study of system Equation(1) and Equation(2)
. We first establish the existence of weak solutions, for which a definition is given below (see also Citation13
Citation19
Citation21).
Definition 2.5
For
(J, A) is a weak solution of Equations
Equation(1)
and
Equation(2)
in
if
and a similar definition for :
Definition 2.6
For
(J, A) is a weak solution of Equations
Equation(1)
and
Equation(2)
in
if for all
(J, A) is a weak solution of Equations
Equation(1)
and
Equation(2)
in
.
We have the following Theorem:
Theorem 2.7
Suppose that assumption Hypothesis 2.1–2.3 are satisfied and that initial conditions (J
0, A
0) are continuous, non-negative and L
∞
in Q
J, a
and Q
A, a
. Then for all T>0 problems
Equation(1)
and
Equation(2)
have a unique global weak solution (J, A) with non-negative components defined in
.
Remark 2.8
We could also consider continuous μ
J
and μ
A
going to infinity when a is going to a
† when . It follows that densities go to 0 in a
†; see Naulin Citation21. An additional truncation step is then required in the following proofs.
The proof goes through several steps: first we solve two auxiliary problems, then we derive a fixed point method. The proof can be found in Section 5.1.
Under additional assumptions, we establish a global bound L
∞ for solutions of system Equation(1) and Equation(2)
. More precisely, we prove that we can estimate quantities
and
independently on t.
The additional assumption are
Hypothesis 2.9
Diffusion rates d J and d A are not dependent on chronological age a. and, in order to consider death rates of logistic types:
Hypothesis 2.10
For Z=J, A, we have an under bound of the form: there exists such that
Theorem 2.11
Suppose assumption Hypotheses 2.1–2.10 satisfied and
with non-negative components. If (J, A) is a solution of system
Equation(1)
and
Equation(2)
, then there exists a positive constant
independent on t such that
The proof is based on many estimates derive by iterations each depending on the others and is given in Section 5.2.
3. Epidemic model
We are now interested in the analysis of an epidemic model.
3.1. Modelling
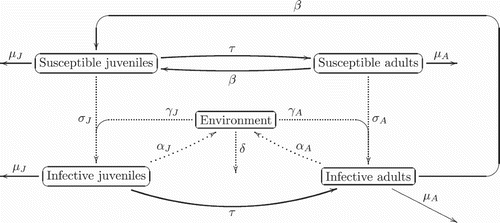
Concerning the epidemic model for a single-host population, we shall consider a basic SI model with susceptible (S) and infective (I) classes. Newly infected individuals highly excrete the virus and are very infectious, but chronically infected individuals excrete very few viruses and are less infectious Citation23. Thus, we will consider a continuous age structure with the age of infection b≤b † where the age of infection is the duration of the disease. Then we have four classes of population
| • | J s (t, a, x) represents susceptible (i.e. not yet infected) juveniles, | ||||
| • |
| ||||
| • | A s (t, a, x) represents susceptible adults, | ||||
| • | and | ||||
We assume the microparasite is benign in the host population: this means there is no additional mortality due to the parasite, fertility and maturation rates, as well as diffusivities of infected individuals being identical to those of susceptibles. We use different incidence functions for direct transmission of the parasite from infected individuals, since different times b to susceptibles: a frequency-dependent rate for the former and a density-dependent one for the latter Citation4 Citation6 Citation7.
In our model, we also consider that indirect transmission of the parasite through the environment is possible. We shall also need an equation to handle the evolution of the proportion (G) of the contaminated environment. The resulting compartmental model is depicted in Citation31.
For direct propagation, newly infected individuals are more infective than chronically infected ones. Then the type of incidence change with the age of the infection (mass action type incidence is dominant for small values of b and proportionate mixing type incidence is dominant for high values of b). The incidence functions are given below (EquationEquation (7)).
Indirect transmission occurs by via the release of the virus from the faeces, vomit, urine and other bodily fluids. Hence, infective individuals will contaminate the environment at a rate (α
J
or α
A
depending on the infected class); while susceptible individuals are infected by the contaminated environment at a rate
for juveniles and
for adults. G(t, x) represents the proportion of the contaminated environment. We consider that the environment eliminates viruses with time at a rate
. Let 𝕀 (t) be the density of infected individuals, and G(t)≥0 be the percentage of contaminated environment; for unstructured population, an equation for G(t) has the form:
Citation3.
The resulting compartmental model is depicted in .
The resulting mathematical model couples the partial differential equations to an ordinary differential equation.
Let , the system corresponding to the epidemic model is
The matrix of diffusion rates D and vectors Φ(U) for demography, Ψ(U) for transmission rates in the host population (direct and indirect), and Υ representing the environment contamination by infected individuals are as follows:
Remark 3.1
Integrating in b the equation for J
i
and adding the result with the equation for J
s
on one hand and those for A
i
and A
s
, on the other hand, one gets system Equation(1) and Equation(2)
.
3.2. Assumptions
We suppose that initial conditions in t=0 ,
,
and
are continuous, non-negative and L
∞. In addition to assumptions Hypotheses 2.1–2.3, concerning demographic and diffusion rates, we make the following two assumptions concerning transmission rates.
Definition 3.2
| • | for Z=J, A, let | ||||
| • | for Z=J, A and z=n, c, let | ||||
| • | and for Z=J, A, Z′=j, a, and z=pm, am let | ||||
Definition 3.3
Let in
and
in
with
.
This last assumption is useful to prove Lemma 5.18 which is used to treat the proportionate mixing part in the following:
In order to simplify the notations, we set the following:
3.3. Mains results
As in the previous section, we prove existence and uniqueness of a global weak solution for system Equation(3)–(6). The notion of weak solution is defined in Definitions 2.5 and 2.6.
We have the Theorem:
Theorem 3.4
Suppose assumptions Hypotheses 2.1–2.3, 3.2 and 3.3 are satisfied, and that initial conditions
are continuous, non-negative and L
∞
in Q
J, a
and Q
A, a
. Then for all T>0 problem
Equation(3)–(6)
has a unique global weak solution
with non-negative components, with
and defined in
.
The proof is similar to those of Theorem 2.7. Details for the points that make it more complicated are given in Section 5.3.
As for the JA demographic model, assuming assumption Hypothesis 2.9 is satisfied, it follows:
Theorem 3.5
Suppose assumptions Hypotheses 2.1–2.10, 3.2 and 3.3 are satisfied. If
is a solution of system
Equation(3)–(6)
with
non-negative and
, then there exists a positive constant
independent on t such that
Proof
We have seen that . Furthermore, it is easy to check that
is invariant by a system Equation(3)–(6). Thus, integrating on b the equation for J
i
and adding the equation of J
s, but also those for A
i
and A
s, one gets EquationEquations (1)
and Equation(2)
. One can conclude using Theorem 2.11. ▪
4. Model with transmission to humans
Let us now consider the situation where the parasite is indirectly transmitted through the environment from the previous host population, a reservoir, to a second host population spatially distributed in a neighbouring spatial domain (Ω
H
) with . Assuming different times scales between these two host populations, neither age structure nor demography are considered in the second one. We use a basic spatially structured SIR epidemic model for the second population with an additional mortality rate.
4.1. Modelling
We extend the previous model by taking into account transmission to humans. We consider that this transmission is only due to the contamination of humans by the infected environment. Humans do not contaminate the environment, and there is no transmission from human to human; see Sauvage Citation22. The model used here is inspired by the works of Sauvage Citation22 and Fitzgibbon et al. Citation12. We consider three classes of human population: H s represents susceptible individuals, H i represents the infected (but not infectious) individuals and H r represents recovered individuals, whom we will consider as immune. Let γH be the contamination rate by the environment, λ be the rate at which infected individuals are recovered and ϵ be the survival rate of the disease (it can be lethal for humans). Considering the smallness of times for bank voles demography, transmission and incubation of the virus, we do not take demography into account for humans.
It is also useless to introduce an age structure for humans; only a space structure is considered in our model. Thus, the system is composed of EquationEquations (3)–(6) with the additional equations for humans: for t>0 and x∈Ω,
Definition 4.1
| • | Let λ>0 and | ||||
| • | Let | ||||
| • | and for x∈Ω, let | ||||
Differences with the previous epidemic model comes only from the additional equations for humans. However, the human population is only influenced by the equation for contaminated environment and does not influence equations for the host population neither those for contaminated environment. Thus, results obtained in the previous section are still true and we can study the system Equation(8)–Equation(10)
with the minimal assumption that
for all T.
We have the following result: see Fitzgibbon et al. Citation12:
Theorem 4.2
Suppose initial conditions
are non-negative and continuous on Ω. Then there exists a unique global classical solution of system
Equation(8)
–Equation(10)
with non-negative components and uniformly bounded on
. Furthermore one has the following:
Proof
Local existence comes from the Banach fixed point Theorem. Global existence is granted by a priori estimates and regularity results in Ladyzhenskaya et al.
Citation18. For the system considered here, this a priori estimate comes from the maximum principle and the fact that applied to the three equations for H
s, H
i
and H
r; it follows inequalities in EquationEquation (8)
; see Fitzgibbon et al.
Citation10–12. ▪
Remark 4.3
Integrating the three EquationEquations (8)–Equation(10)
in space and adding them, one gets
5. Proofs
5.1. Proof of Theorem 2.7
First, we will study two auxiliary problems, which will be useful in the general case.
5.1.1. First auxiliary problem
We consider the following system:
Definition 5.1
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • | d satisfies the assumption Hypothesis 2.3. | ||||
Then we have the proposition:
Proposition 5.2
Suppose assumption Hypothesis 5.1 is satisfied. Then problem
Equation(11)
has a unique solution u in
non-negative and satisfying
Proof
A proof is based on the Galerkin method using a convenient regular basis of H
1(Ω) and tools of Garroni and Langlais Citation13. We can also use the characteristics method and classical results for hyperbolic problems Citation25. We treat here the case , but the case
can be treated in similar ways. We begin by considering 0<t<a. Let
,
, and we set t=c, a=a
0+c and
. Then w is solution of the following linear parabolic problem: for
and x∈Ω,
There exists for parabolic equations a comparison theorem, from which we can get from the previous proof the following corollary:
Corollary 5.3
If
in Q
J
,
in Q
J, a
,
in Q
J
and
in Q
J
then corresponding solutions of system
Equation(11)
satisfies
in Q
J
.
We now establish a boundedness result for the solution of system Equation(11):
Proposition 5.4
Suppose that assumption Hypothesis 5.1 is satisfied for all T>0. Then: If
, for all T>0 there exists a constant M
0(T)>0 depending on
such that u solution of Equation
Equation(11)
satisfies
Proof
We only deal with the case . Yet we know that u≥0 and using Corollary 5.3 it is sufficient to consider the case of
. We use the characteristics method and the results in Alikakos Citation1 and Ladyszenskaya et al.
Citation18. We first consider 0<t<a. We use the notations in the proof of Proposition 5.2 to get w(c, x) solution for
and x∈Ω of
5.1.2. Second auxiliary problem
We are interested in solutions (J*, A*) of the following problem:
Proposition 5.5
For all T>0, for all 0<A<a
†, system
Equation(12)
and
Equation(13)
has a unique solution
with non-negative components. Furthermore, if
the following estimate is satisfied:
Proof
Existence and uniqueness in L ∞ is a consequence of a similar result in a more complicated case; see Wolf Citation30. Furthermore, adding the two systems, one has
5.1.3. End of proof of Theorem 2.7
We only deal with the case , the case
can be treated similarly by truncation.
Let (J*, A*) be the solution of EquationEquations (12) and Equation(13)
.
Let also 𝒦 be the closed convex set defined by
Proposition 5.2 insures the existence of (J, A).
Comparing J to the solution of EquationEquation (11) with μ=0, f=0 and
and using Corollary 5.3, one has
This way is well defined.
It remains to prove that Φ is a strict contraction to have the existence of fixed point, and then to show that this fixed point is a weak solution.
Thus we consider and
. The following lemma holds:
Lemma 5.6
There exist two constants k
1
and k
2
depending on
and
, for Z*=J*, A* such that for t∈(0, T), one has
Proof
k i , i≥3 will be constants with the same property as k 1 and k 2. We begin by estimating the equation corresponding to J 1−J 2, that we multiplied by J 1−J 2. one gets
Lemma 5.7
The mapping Φ is a strict contraction on
with τ* small enough, i.e. there exists
such that
Proof
First, note that if y(t) is solution of the system:
As Φ is a strict contraction on a Banach space, there exists a unique fixed point such that
. Furthermore, one has from EquationEquation (17)
5.2. Proof of Theorem 2.11
First, in order to justify calculations below, we need the following Corollary of Proposition 5.2 for regularity of 𝕁 and 𝔸:
Corollary 5.8
Suppose assumption Hypotheses 2.1–2.3 and 2.9 are satisfied. The the unique non-negative weak solution u in
of problem
Equation(11)
satisfies
Proof
We return to the proof of Proposition 5.2, if the diffusion rate d does not depend on the variable (Assumption 2.9) one can guarantee regularity on
. So as to get this, we work with the approximate solutions given by the Galerkin's method which have sufficient regularity:
. Taking
in the weak formulation, one has
Integrating EquationEquation (1) in a from 0 towards a
†, i.e. using u=1 as a test function, one has
Our goal is to prove the existence of a constant M 0>0, independent on t such that
In order to do this, we adapt a work of Fitzgibbon et al. Citation10. First, we establish the following lemma:
Lemma 5.9
If
are classical non-negative solutions of Equations
Equation(1)
–Equation(2)
in
then noting
Proof
Integrating the inequality in 𝕁 on Ω, one has
Now we give the following result, for regularity:
Lemma 5.10
Suppose initial conditions (J
0, A
0) are non-negative and continuous on Ω¯, and assumptions Hypotheses 2.1–2.10 are satisfied. Then there exists
such that for 0 ≤ l<T:
Proof
From Lemma 5.9, ons has that ℙ and so 𝕁 and 𝔸 are bounded in L
2(Q(0, T)). Multiplying inequality Equation(28) by 𝕁 and integrating on Ω one gets
As 𝕁 and 𝔸 are bounded in L 6(Q(0, T)), each component of
The following lemma completes the estimates given in Lemma 5.9:
Lemma 5.11
If
are non-negative classical solutions of Equations
Equation(1)
–Equation(2)
on
then
Proof
For , one has using the Holder inequality:
We now have the following result:
Proposition 5.12
For fixed l large enough and l*>0, there exists a constant C(6, l*) independent on initial conditions
and
such that for Z=J, A,
Proof
Integrating estimate Equation(33) in time from l towards l+l*, one has
Finally, we can get the global estimates given in Theorem 2.11:
Proof
From Proposition 5.12, one has existence for l large enough (l≥l 0) of C(6, l*) such that
Remark 5.13
A priori estimates allowing estimates in Lemma 5.9 and M(t) in the proof of Proposition 5.10 can be obtained directly from equations of system Equation(1) and Equation(2)
by integrating also equations in age a. However, we do not have results concerning the theory of parabolic equations from, for example, Smoller Citation25 or Ladyzhenskaya et al.
Citation18 to conclude global existence of solutions of the system.
5.3. Proof of Theorem 3.4
In order to prove Theorem 3.4, we will need these two results:
5.3.1. Environment equation
This result is proved by the variation of the constant
Lemma 5.14
The solution of equation
We also need another auxiliary problem:
5.3.2. Third auxiliary problem
We consider the following system:
Definition 5.15
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • | d satisfies the assumption Hypothesis 2.3. | ||||
Then one gets
Proposition 5.16
Suppose Assumption 5.15 is satisfied. Then the problem
Equation(39)
has a unique weak solution u in
non-negative and satisfying
Proof
As in the Proposition 5.2 we prove this result by the characteristics method and classical results for hyperbolic problems. This time there are three different cases: 0<a<t, b, 0<b<t, a and 0<t<a, b. We begin by considering 0<a<t, b. Let u be a solution of EquationEquation (39). For c>0, we set
. w is a solution of the following system:
As for the first auxiliary problem, comparison theorem for parabolic equations implies the following corollary:
Corollary 5.17
If
and
in Q
J
,
in Q
J, a
then the solutions of Equation
Equation(39)
are non-negatives.
5.3.3. Proof of the Theorem 3.4
Proof
Let (J*, A*) be the solution of EquationEquations (12) and Equation(13)
. Let 𝒦 be the closed convex subset of
defined by
First note that we have the following lemma, useful in order to treat the incidence part corresponding to the proportionate mixing term:
Lemma 5.18
Let w
j
(respectively w
a
) the non-negative solution of the linear problem
Equation(11)
with d=d
J
(respectively, d=d
A
),
(respectively,
f=0, β=0 and
(respectively,
. Then there exists a constant m(T)>0 such that
Proof
By the Assumption 3.2:
Let us also define the mapping by
where (U, G) is solution of the linear problem:
Equation for J
s is of the form Equation(11) with
and f=0, so that J
s is non-negative.
Equation for J
i
is of the form Equation(39) with
and f=0, so that J
i
is non-negative.
Equations for A
s and A
i
can be treated in the same way. Furthermore, one then gets for all t, because all rates are non-negative. Using results for linear equations in the previous section and lemma 5.14,
is well defined.
We now have to check that Φ is a strict contraction. We consider and
.
Lemma 5.19
There exist constants k
1
and k
2
depending on
and
for Z=J, A such that for t∈(0, T), one has
Proof
Let k i , i≥3 be constants with the same property as k 1. The main differences with the proof of Lemma 5.6 are that there is also a term for the transmission of infection, the equation of infected individuals are more structured, and there is the equation for G. Let us focus on the equations for J s, J i and G. First multiply the equation corresponding to J s, 1−J s, 2 by J s, 1−J s, 2, and integrating on Ω, it follows:
| • |
| ||||
| • |
| ||||
| • |
| ||||
| • |
| ||||
Similarly to Lemma 5.7, there is
Lemma 5.20
The mapping Φ is a strict contraction on
with τ* small enough, i.e. there exists
such that
The end of the proof of Theorem 3.4 is similar to those of Theorem 2.7.
6. Conclusion
Our objective in this paper was to build and study a deterministic mathematical model describing the propagation of a virus within a structured host population.
In the existing literature on propagation of diseases, various features have been identified, which govern the propagation of a given virus Citation4–7,Citation16 Citation17 Citation22 Citation23 Citation26 Citation28 Citation30 Citation32. Motivated by these previous works, our model has been built so as to take into account three major features which are important in the specific case of the Puumala hantavirus – bank vole system in Europe.
(1) Maturation of juveniles depending on the density of adult individuals. This leads to a stage structure with juvenile and mature individuals, and a chronological age structure on each stage; | |||||
(2) transmission rates depend on the time elapsed since infection. Hence, in addition to the usual stage structure between susceptible and infected, we use a third chronological variable: the age of the disease; | |||||
(3) a spatial structure is considered for the host population. | |||||
Our new model combines all the structures into a single strongly structured system. In this model, we also considered three other assumptions, based on the Puumala hantavirus – bank vole system: Equation(1) the virus is benign in the host population, Equation(2)
virus propagation occurs through direct transmission from infective to susceptible individuals and through indirect contamination of susceptibles via the contaminated environment, and Equation(3)
the dispersion rates are discontinuous. Most of these features may be of some interest for most of the epidemiological systems.
We first analysed a demographic model for a closed population with chronological age and spatial structure and we derived here a mathematical analysis of this model. We get global existence, uniqueness and global boundedness results.
Then we studied an epidemic model with a continuous structure in age of infection and direct and indirect transmission. Global existence, uniqueness and global boundedness results was also performed in this case.
Finally, we looked at a model including the transmission of the virus to human populations with possible lethal consequences, and we also had global existence, uniqueness and global boundedness results.
The next step of this work will be to take into account some others biological assumptions, such as density dependencies for mortality rates or maturation rates (that should be decreasing towards adult density because of adults’ pressure on maturation in the Puumala hantavirus – bank vole system). In the same way, density-dependent diffusion rates may also be of some interest: diffusion is favoured by high-population densities because of territorial reasons.
Hence, we obtained a well-posed model taking into account many significant features. We believe this model can be very useful in diseases propagations studies. Unfortunately, this system seems to be too complex to allow qualitative studies mathematically; but numerical simulations may give lots of information of biological interest and may be compared with data collected in the field. A difficult point in simulating this system is its very strong structured character that leads to a 1+1+1+2 dimension problems, but parameters t, a and b are basically the time, thus numerical simulations can be related to 3D ones. Some parameters are quantifiable with field data, but others will be more difficult to estimate. However, qualitative studies are possible and sensitivity studies can help to determine importance of the parameters that are poorly known; we will focus on this.
Numerical simulations (Citation27 Citation29 and references therein) have studied the hantavirus system, without spatial structure, in the case when the parameters depend periodically with time. These works show that the demography and the propagation of diseases change dramatically when the coefficients differ from their average. This is especially true as far as propagation to a human population is concerned. In this direction, our model investigates the effect of the spatial variations of the coefficients (as opposed to temporal variations). The corresponding numerical study work is in progress.
Additional information
Notes on contributors
S. Madec
Current affiliation: Institut de Mathématiques de Bordeaux, UMR CNRS 5251, Université Victor Segalen, Bordeaux 2, France.C. Wolf
Author Email: [email protected]References
- Alikakos , N. D. 1979 . An application of the invariance principle to reaction-diffusion equations . J. Differ. Equ. , 33 : 201 – 225 .
- Anita , S. 2000 . Analysis and Control of Age-Dependent Population Dynamics , Dordrecht : Kluwer Academic Publishers .
- Berthier , K. , Langlais , M. , Auger , P. and Pontier , D. 2000 . Dynamics of a feline virus with two transmission modes within exponentially growing host populations . Proc. R. Soc. Lond. Ser. B , 267 : 2049 – 2056 .
- Brauer , F. and Castillo Chavez , C. 2001 . Mathematical Models in Population Biology and Epidemiology , Vol. 40 , New York : Springer Verlag . Texts in Applied Mathematics
- Bujalska , G. 1988 . Life history consequences of territoriality in the bank vole, in Evolution of Life Histories: Theories and Patterns from Mammals , Edited by: Boyce , M. S. 75 – 90 . Yale : Yale University Press .
- Busenberg , S. and Cooke , K. 1993 . Vertically Transmitted Diseases , Vol. 23 , New York : Springer Verlag . Biomathematics
- Diekmann , O. and Heesterbeek , J. A.P. 2000 . Mathematical Epidemiology of Infectious Diseases , Chichester : J. John Wiley & Sons . Mathematical and Computational Biology
- Fitzgibbon , W. E. and Morgan , J. J. 1994 . Diffractive diffusion systems with locally defined reactions, in Evolution Equation , Edited by: Goldstein , G. 177 – 186 . New York : M. Dekker .
- Fitzgibbon , W. E. , Hollis , S. and Morgan , J. J. 1999 . Steady-state solutions for balanced reaction diffusion systems on heterogeneous domains . J. Differ. Integral Equ. , 12 : 225 – 241 .
- Fitzgibbon , W. E. , Langlais , M. and Morgan , J. J. 2001 . A mathematical model of the spread of Feline Leukemia Virus (FeLV) through a highly heterogeneous spatial domain . SIAM J. Math. Anal. , 33 : 570 – 588 .
- Fitzgibbon , W. E. , Langlais , M. and Morgan , J. J. 2004 . A reaction-diffusion system modelling direct and indirect transmission of a disease . : 893 – 910 . DCDS-B 4
- Fitzgibbon , W. E. , Langlais , M. and Morgan , J. J. 2007 . A mathematical model for indirectly transmitted disease . Math. Biosci. , 206 : 233 – 248 .
- Garroni , M. G. and Langlais , M. 1982 . Age dependent population diffusion with external constraints . J. Math. Biol. , 14 : 77 – 94 .
- Gurtin , M. E. 1973 . A system of equations for age dependent population diffusion . J. Theoret. Biol. , 40 : 389 – 392 .
- Gurtin , M. E. and Mac Camy , R. C. 1974 . Nonlinear age-dependent population dynamics . Arch. Ration. Mech. Anal. , 54 : 281 – 300 .
- M. Iannelli, Mathematical Theory of Age-Structured Population Dynamics, Applied Mathematics Monographs No. 7, C.N.R. Pisa, 1994
- Kostova , T. , Li , J. and Friedman , M. 1999 . Two models for competition between age classes . Math. Biosci. , 157 : 65 – 89 .
- Ladyzhenskaya , O. A. , Solonnikov , V. A. and Ural'ceva , N. N. 1968 . Linear and quasilinear equations of parabolic type . Transl. Math. Monogr. , 23
- Langlais , M. 1985 . A nonlinear problem in age dependent population diffusion . SIAM J. Math. Anal. , 16 : 510 – 529 .
- Langlais , M. 1988 . Large time behavior in a nonlinear age-dependant population dynamics problem with spatial diffusion . J. Math. Biol. , 26 : 319 – 346 .
- J.M. Naulin, Simulations numériques et analyse mathématique de modèles de viroses dans des populations structurées, Ph.D. thesis, Univ. Bordeaux I, 2001 (in French)
- F. Sauvage, Synergy between the dynamics of the red bank vole populations and the shedding of the hantavirus Puumala: Description of the emergence mechanism of Nephropathia Epidemica, Ph.D. thesis, Univ. C.Bernard Lyon I, 2004 (in French)
- Sauvage , F. , Langlais , M. and Pontier , D. 2007 . Predicting the emergence of human hantavirus disease using a combination of viral dynamics and rodent demographic patterns . Epidemiol. Infect. , 135 : 46 – 56 .
- Sauvage , F. , Langlais , M. , Yoccoz , N. G. and Pontier , D. 2003 . Modelling Hantavirus in cyclic bank voles: The role of indirect transmission on virus persistence . J. Anim. Ecol. , 72 : 1 – 13 .
- Smoller , J. 1983 . Shock Wawes and Reaction Diffusion Equations , New York : Springer Verlag .
- Thieme , H. R. 2003 . Mathematics in Population Biology , Princeton : Princeton University Press .
- Wang , W. and Zhao , X.-Q. 2008 . Threshold dynamics for compartmental epidemic models in periodic environments . J. Dyn. Differ. Equ. , 20 : 699 – 717 .
- Webb , G. F. 1985 . Theory of Nonlinear Age-Dependent Population Dynamics , New York : Marcel Dekker .
- Wesley , C. , Allen , L. and Langlais , M. 2010 . Models for the spread and persistence of Hantavirus infection in rodents with direct and indirect transmission . Math. Biosci. Eng. , 7 : 199 – 215 .
- C. Wolf, A mathematical model for the propagation of a hantavirus in structured populations, DCDS-B 4 (2004), 1065–1089
- C. Wolf, Modelling and mathematical analysis of the propagation of a microparasite in a structured population in heterogeneous environment, Ph.D. thesis, Univ. Bordeaux I, 2005 (in French)
- Wolf , C. , Sauvage , F. , Pontier , D. and Langlais , M. 2006 . Multi-patch epidemic model with periodic demography, direct and indirect transmission and variable maturation rate . Math. Popul. Stud. , 13 : 153 – 177 .