ABSTRACT
Background: Climate warming is amplifying and exacerbating drought stress worldwide. Long-term trends of increasing evaporative demand and decreasing soil moisture availability occur superimposed on severe spells of drought. These rare, extreme droughts have triggered episodes of forest dieback that have led to reduced productivity and rising mortality rates, usually at small scales (dieback hotspots), but affecting biomes worldwide.
Aims: This review summarizes and discusses the drivers, patterns and mechanisms of forest dieback caused by drought.
Methods: I review studies on forest dieback and tree death linked to dry spells with a focus on tools to forecast dieback.
Results: Several mechanisms have been described as physiological drivers of dieback, including hydraulic failure and carbon starvation, however hydraulics-based models have shown little predictive power of dieback and mortality. Field proxies of tree vigour, including changes in canopy defoliation and water content, combined with surrogates of tree functioning (tree-ring growth, wood anatomy, tree-ring δ13C or δ18O composition) may improve predictions of forest dieback or at least render early-warning signals of impending tree death.
Conclusions: Drought-induced dieback and mortality are concerning phenomena which lack forecasting tools with sufficient predictive power. Surrogates of tree vigour, growth and functioning should be used to build more accurate models of tree death in response to extreme climate events linked to drought. Here, I argue for combining and comparing those surrogates to better forecast forest dieback.
Introduction
Rising temperatures are exacerbating drought stress for trees at global scales causing forest dieback (stand level decline and death of trees) and episodes of mortality (Allen et al. Citation2010). Climatic projections indicate that mean global temperatures are expected to rise at least 1.5°C until 2100 in response to anthropogenic emissions (IPCC Citation2018). Such projections of warming indicate the duration and severity of droughts are expected to amplify, probably causing widespread forest dieback and tree mortality by affecting sites or tree species currently not assumed to be prone to drought stress (e.g., tropical or boreal forests) or known to be resistant to drought stress (e.g., Mediterranean shrubs).
Ecologists and natural resource and conservation managers have shown a growing interest in the impacts of climate change and aridification trends on forests, including the study of canopy dieback and tree mortality caused by droughts (Allen et al. Citation2015). Since forests cover 31% of land surface and their woody biomass growth compensates for 15% of annual anthropogenic CO2 emissions (FAO Citation2020), widespread forest dieback would reduce their productivity and diminish their potential to mitigate climate warming. In addition, forests play prominent roles for preserving biodiversity, provide key ecosystem goods and services to mankind, and regulate carbon and water cycles (Hartmann et al. Citation2018).
In the following five sections, I summarise my views on forest dieback as related to drought showing our current limitations to gain predictive power in events linked to rare, extreme dry spells. This Grubb review is focused on field observational studies, forest ecology and, particularly, dendroecology. It is not intended to cover all aspects of the drought–dieback–death conundrum in trees and forests, but aims to reconcile mechanistic, physiological approaches and related models with field-based frameworks considering longer time scales, as those provided by tree-ring data, which may provide early-warning indicators of impending tree death.
I start the review by describing forest dieback and tree mortality related to drought. Then, death models based on hydraulic and functional traits are discussed. The use of tree-ring data as early-warning signals is also discussed with the intention of reconciling process-based and empirical approaches to tree drought mortality. Finally, a research agenda on drought, forest dieback and tree mortality is proposed based on the research gaps commented in previous sections.
Drought and forest dieback
Forest dieback is a complex, lasting phenomenon in which several stressors may result in positive feedbacks that impair the vitality of trees (Manion Citation1981). Many of these acute episodes of dieback () characterised by a sharp loss of tree vigour (e.g., shoot death and leaf shedding, production of epicormic shoots, loss of fine roots), decline in growth and elevated mortality rates have been attributed to rising temperatures (Adams et al. Citation2009; Van Mantgem et al. Citation2009; Williams et al. Citation2013; Sánchez-Salguero et al. Citation2017a, Citation2017b), elevated evaporative demand (high vapour pressure deficit, VPD) and reduced soil moisture, often accompanied or followed by biotic stressors such as insects or fungi (Gaylord et al. Citation2013). Usually, episodes of dieback are preceded by extreme dry spells (); however, there is a lack information on the legacy impacts or cumulative stress that may predispose trees to die in such rare, extreme droughts. Dieback impacts are evident; they cause localised reductions in forest cover and productivity, alter the carbon and water cycles and trigger compositional shifts in the vegetation, often towards assemblages with more drought-tolerant tree and shrub species, or by accelerating successional dynamics (Anderegg et al. Citation2013a, Citation2015a; Batllori et al. Citation2020).
Figure 1. Aerial view of post-dieback stand structure in a Scots pine (Pinus sylvestris) stand severely impacted by the drought in 2012, Corbalán, Aragón, north-eastern Spain. Note that the surviving individuals are of drought-tolerant tree species: Pinus nigra, Juniperus thurifera, Quercus ilex and Quercus faginea. The inset shows a dead Pinus sylvestris with individuals of J. thurifera in the midstorey

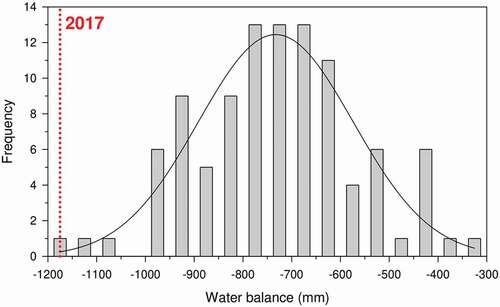
Figure 2. Drought-induced dieback is usually triggered by extreme dry spells. In this example, a very low annual climatic annual water balance in 2017 (dotted red line) characterised a dieback episode that affected a Mediterranean Pinus pinaster stand located near Miedes de Aragón, north-eastern Spain. The year 2017 was characterised by one of the driest growing seasons in the region during the last 100 years
Process-based, bottom-up approaches: hydraulics has low predictive power of dieback and mortality
Predictions of dieback and tree mortality based on physiological approaches are not accurate enough because momentaneous physiological variables do not contain sufficient information about the ecology of trees, which are long-lived, complex organisms and which interact with their environment and other organisms (Trugman et al. Citation2021). An improved mechanistic understanding of drought-induced dieback is imperative. Two non-exclusive mechanisms that may lead to tree death have been widely discussed: hydraulic failure and carbon starvation (McDowell et al. Citation2008, Citation2011; McDowell et al. Citation2016; Choat et al. Citation2018). Hydraulic failure has been widely reported from several tree species in different biomes (cf. Rowland et al. Citation2015; Adams et al. Citation2017). Heat and drought stress may increase the risk of hydraulic failure by increasing water losses through the leaf due to high cuticular permeability and leaf-to-air VPD (Cochard Citation2019). An excessive focus on xylem hydraulics has diverted attention from other, potentially equally relevant parts of the soil-plant-hydraulic continuum. For instance, disruption of the soil-root hydraulic continuum due to reduced soil moisture or fine root mortality may be a proximate cause of tree dehydration and death (Körner Citation2019). Hydraulic safety margins (differences between naturally occurring xylem pressures and pressures that would cause hydraulic dysfunction) and xylem embolism are often poor predictors of tree mortality (Johnson et al. Citation2018; De Kauwe et al. Citation2020; Venturas et al. Citation2020), which reveals the need for more integrative functional measures including data on changes in water pools through measures of relative water content (the difference between fresh and dry weights of a tissue divided by the difference between saturated and dry weights) in stem, shoots, leaves and roots (Martínez-Vilalta et al. Citation2019), and on below-ground loss of hydraulic conductance and root capacity to uptake water from deeper soil sources (Barbeta et al. Citation2015; Ripullone et al. Citation2020). Changes in water status at the tree and landscape levels provided by measures of relative water content and remote-sensing imagery, respectively, constitute relevant tools for monitoring and effectively forecasting dieback (Marusig et al. Citation2020). Remote sensing studies can indicate declines in canopy water content prior to increases in drought-related tree mortality (e.g., Asner et al. Citation2016). Plant water content has been shown to be a reliable predictor of drought-induced mortality in seedlings (Sapes et al. Citation2019), and one needs to apply similar measurements to adult trees in the field to test if rapid drops in wood or leaf relative water content precede tree death. Moreover, most studies have focused on the variability between species in the responses and vulnerability to drought (Choat et al. Citation2012, Citation2018), often from a functional perspective using ‘soft’ traits such as wood density or leaf area (Greenwood et al. Citation2017), which have low predictive power of dieback and mortality. However, for accurate predictions of dieback one should be able to detect which are the most vulnerable trees or populations within stands or species and when a threshold or tipping point of loss in vigour is being surpassed (Camarero et al. Citation2015). In glasshouse experiments for Pinus taeda saplings, this point of no return or lethal threshold has been quantified (e.g., 80% loss of hydraulic conductivity; Hammond et al. Citation2019); however, translating such a value on vascular damage to mature trees in the field is challenging because of ontogenetic changes that affect post-drought recovery of growth and water storage (Anderegg et al. Citation2015a).
Inaccurate predictions of dieback and tree death based on current process-based approaches are explained by the fact that trees may still grow after a high loss of conductivity (e.g., Pinus halepensis trees can recover after losing more than 80% of xylem conductivity; Klein et al. Citation2012), and it is unclear if drought induces enough carbon starvation so as to kill trees (McDowell Citation2011; Sevanto et al. Citation2014; He et al. Citation2020). Mortality thresholds linked to percentages of loss of conductivity defined experimentally appear to be species- and context-dependent, which makes prediction of tree death challenging. Understudied aspects of tree functioning such as shifts in tree water capacitance, i.e., how water is stored and transferred among compartments and tissues, should be better quantified to better parameterise ecophysiological models of tree drought mortality (Blackman et al. Citation2016).
Functional traits as predictors of tree mortality
Functional traits such as wood density or leaf characteristics show patterns that are diverse and related to species‐specific drought‐resistance strategies, drought severity or duration and site features such as soil water holding capacity (McDowell et al. Citation2008). For instance, a higher soil stoniness and rock cover increased soil water availability and reduced the mortality of drought-stressed individuals of P. halepensis (Preisler et al. Citation2019). A global meta‐analysis of drought‐induced forest mortality cases has found that mortality increased with drought severity, but the variation related to functional traits (wood density, specific leaf area) was only 37% (Greenwood et al. Citation2017). These authors concluded that tree species with denser wood and lower specific leaf area suffered less mortality. This pattern agrees with some studies made in temperate forests (Nardini et al. Citation2013). However, the opposite pattern has also been reported showing a positive correlation between tree mortality and wood density (Hoffmann et al. Citation2011). These discrepancies may be due to the different scales of the studies; studies carried out at the local (Hoffmann et al. Citation2011) and global (Greenwood et al. Citation2017) scales are not comparable. Overall, functional traits show idiosyncratic responses and are not reliable predictors of dieback and mortality. Naturally, hydraulic ‘hard’ traits (hydraulic safety margin, xylem vulnerability to embolism) described in the previous section may have more predictive power of patterns in tree mortality from drought than functional ‘soft’ traits; however, the former are measured for less species, which limits our ability to make mechanistic predictions in highly diverse biomes such as tropical forests (Anderegg et al. Citation2016).
Empirical, top-down approaches: growth variability as early-warning indicators of impending dieback
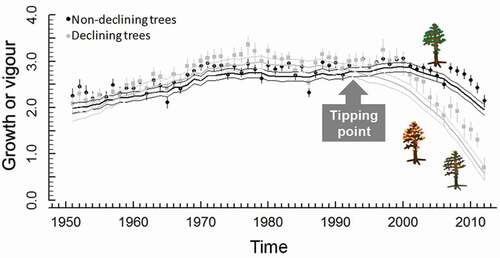
Drought constrains growth before it limits photosynthesis (Körner Citation2003, Citation2019), often not causing severe carbon shortage but driving hydration levels of meristems towards lethal levels (Lempereur et al. Citation2015). This is translated into reduced radial growth rates (narrow tree rings) and partial or complete leaf shedding (crown defoliation) in response to drought (Dobbertin Citation2005), signals which have been identified and characterised by using retrospective approaches (dendroecology) to infer changes in vigour before tree death (Waring Citation1987). Changes in growth rates have been explored for their usefulness as early-warning signals () since declining or dying trees show lower growth rates than non-declining or surviving trees, often after surpassing a tipping point or stress threshold (Pedersen Citation1998; Suarez et al. Citation2004; Sánchez-Salguero et al. Citation2012, Citation2017a; Camarero et al. Citation2015). The decrease in radial growth before death may last from one year to several decades depending on the lifespan of the tree and the intensity of the drought to which the tree is exposed to (Cailleret et al. Citation2017). Another pattern characteristic of dieback processes is the increase in growth variability about 20 years before tree death observed in many gymnosperms (Cailleret et al. Citation2018), but not in all sites and species (Camarero et al. Citation2015). These patterns in growth rates suggest a lower capacity of resilience in stressed/dying trees. In fact, as has been shown by DeSoto et al. (Citation2020), trees that died after drought were less resilient after previous droughts, relative to surviving conspecific individuals. In gymnosperms, the drought-related mortality risk has been associated with a low capacity to recover after drought, whereas in angiosperms it was associated with a low capacity to mitigate the negative impact of drought on tree growth (DeSoto et al. Citation2020).
Figure 3. Early-warning signals vigour of drought-triggered mortality appear to be species specific and reflect how different tree species cope with drought stress. The plot shows the divergence in growth between non-declining and declining Abies alba trees about 20 years before the onset of dieback in 2012. Modified from Camarero et al. (Citation2015)
Growth patterns associated to dieback and mortality are not universal. For instance, in some cases dying, water-spending trees form wide conduits prone to cavitation and present higher growth rates than water-saving surviving trees (Voltas et al. Citation2013). The legacy or carryover effects of drought are important to know because trees may take a few years to recover pre-drought growth levels (Anderegg et al. Citation2013b, Citation2015b). Frequent drought episodes may incur increased growth declines than single drought events and thus may lead to changes in growth–climate sensitivity as a result of multiyear cumulative stress and ‘climatic memory’ (Camarero et al. Citation2018; Peltier et al. Citation2018; Peltier and Ogle Citation2019; Serra-Maluquer et al. Citation2021). Indeed, multiyear legacy effects of more frequent and severe droughts could impair the land carbon sink if the time between droughts became shorter than drought recovery time, i.e., the time an ecosystem requires to revert to a pre-drought state, or in case of compositional changes replacing species were not able to equal carbon stocks of the replaced species in the long-term (Schwalm et al. Citation2017).
Reconciling process-based and empirical approaches
The predictive ability of both process-based (bottom-up) and empirical (top-down) approaches is context-dependent. Therefore, to improve the predictive power of dieback and mortality models one needs to reconcile these two complementary approaches. There are several approaches that can be taken to reconcile process- and pattern-based frameworks on forest dieback and tree mortality; for example, by using tree-ring, multiproxy measures (isotopes, wood anatomy), and by explicitly considering the size-dependence vulnerability to drought damage (Camarero Citation2020).
Tree-ring stable isotopes provide retrospective indirect information about stomatal activity and photosynthesis rates, and may be better long-term, functional indicators of changes in tree vigour than tree ring width (Cherubini et al. Citation2021). Multi-proxy approaches combining tree-ring, carbon (δ13C, i.e., 13C/12C) and oxygen (δ18O, i.e., 18O/16O) isotopic signatures have shown an increase of intrinsic water‐use efficiency in dying conifer trees (Wang et al. Citation2021). During photosynthesis, C3 plants discriminate against the rare naturally occurring stable isotope 13C and this explains why carbon isotope ratios (δ13C) or isotopic signatures of leaf and wood tissues are proxies of water use by C3 plants (Farquhar et al. Citation1982). However, differences in growth rate and isotope discrimination between non-declining or living and declining or dead trees are not consistent across species affected by drought stress as has been shown for Mediterranean Quercus species (Colangelo et al. Citation2017a). Isotope signals may be masked by beetle attacks in some declining conifers (Reed and Hood Citation2021) or may indicate profuse water consumption in fast-growing trees forming wide conduits (perhaps reflecting structural overshoot sensu Jump et al. Citation2017) making them prone to drought-induced dieback (Voltas et al. Citation2013). Favourable climatic conditions that enhance tree growth can lead to structural overshoot of aboveground tree biomass, and such overgrowth can make trees more vulnerable to rapid drought or heat stress due to a temporal mismatch between water demand and availability (Jump et al. Citation2017). Models based on tree-ring width, δ13C or δ18O data are useful aids to better comprehend how dieback occurs after drought (Gessler et al. Citation2018). Nevertheless, there are still few study cases testing the models’ assumptions and predictions (e.g., hydraulic failure would correspond to rapid drops in both stomatal conductance and growth rates prior to death). For instance, in temperate hardwood forests in northern Spain, declining Fagus sylvatica trees showed higher water-use efficiency in contrast to declining Quercus pubescens trees which showed lower growth rates and were less efficient in their water use (Camarero et al. Citation2020), Q. pubescens being in agreement with the conceptual model proposed by Gessler et al. (Citation2018). There is still a large research gap regarding the testing of predictions derived from the isotope model of dieback across tree species, drought types (differing in duration, severity and seasonality) and dieback episodes as has been done with tree-ring width data (Cailleret et al. Citation2017, Citation2018). For instance, it is unclear if dying or vulnerable trees should show a higher water-use efficiency than surviving trees prior to the dieback onset.
Tree ring oxygen isotope ratios (δ18O) can be used to infer water sources accessed by the tress over the years through retrospective analyses stemming from inverse-modelling (Sargeant et al. Citation2019). This is another promising approach because it could allow reconstructing the multi-year changes in the water source trees have used, including groundwater uptake, and improving our predictive capacity of which trees, stands, species or forests will experience dieback due to drought.
Quantitative wood anatomy may be used as an early-warning indicator since long-term reductions of lumen area, indicative of loss in potential hydraulic conductivity, has been shown to have preceded dieback in affected conifer and oak forests (Pellizzari et al. Citation2016; Colangelo et al. Citation2017a). However, the use of this tool is time and labour intensive, and it requires to be used in considering tree height to account for tapering effects (Olson et al. Citation2018; Kiorapostolou et al. Citation2020).
Forward growth models also offer potential tools to define growth vulnerability to extreme droughts and to forecast how projected climate warming could lead to new drought-induced forest dieback (Sánchez-Salguero et al. Citation2017b). In these models, radial growth is simulated as a function of climate variability (Vaganov et al. Citation2006), explicitly considering non-linear relationships, and simulations could allow detecting climatic and growth vulnerability thresholds. I propose combining this type of models with physiological, process-based models to predict tree growth in projected future climate conditions and then use the simulated growth patterns to predict mortality. This combination of models would advance our ability to predict dieback and mortality, but error propagation in combined models should be also quantified. Nevertheless, it must be evaluated how accurate are those growth projections to simulate the effects of rare extreme droughts or the responses to drought (e.g., the existence of an extreme decline in growth, post-drought resilience, legacy effects).
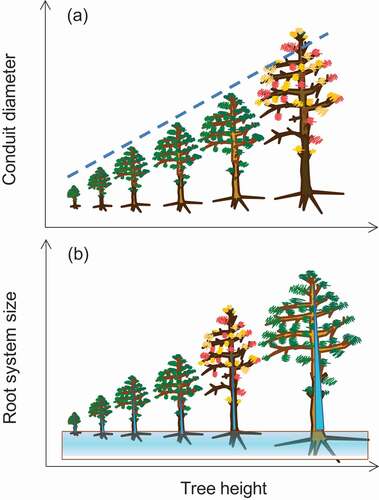
Forest ecologists have sought to identify characteristics of trees such as tree height or functional and hydraulic traits (Waring Citation1987; Anderegg et al. Citation2016) that make trees vulnerable to drought-induced dieback and mortality. Allocation measures vary following allometric relationships, and significant departure from such a size-related scaling can be defined as stress (Anfodillo et al. Citation2016). Based on hydraulic theory (Darcy’s law relating canopy-scale water conductance with leaf area, tree height and VPD), it has been argued that tall trees are at the greatest risk of dying due to drought stress (McDowell and Allen Citation2015) and while some studies have supported this prediction (Bennett et al. Citation2015; Stovall et al. Citation2019) others have contradicted it or failed to confirm it (Camarero et al. Citation2015; Hember et al. Citation2017; Colangelo et al. Citation2017b; Camarero Citation2020). Anthropogenic climate warming may amplify the vulnerability in tall trees by causing higher evaporative demand () and may intensify drought stress in trees showing low hydraulic capacitance or having shallow root systems (Camarero Citation2020; Trugman et al. Citation2021). However, it can also be expected that larger trees may show greater capacity for desiccation avoidance through bigger or more efficient root systems that can exploit deep soil water sources (Ripullone et al. Citation2020) and have a greater hydraulic capacitance (). Trait variability (e.g., conduit size, water storage capacitance, rooting depth, canopy volume) should reflect a balance between accessing and storing water for a tree given its size. These factors may explain why tall trees with large crowns may be less susceptible to drought damage than smaller conspecifics, but different height effects in the comparisons within- vs. between-species should be also considered (Camarero Citation2020). In addition, shifts in composition rather than vulnerability of tall trees may drive the shortening of some forests (Stephenson and Das Citation2020), and beetles can mask size-related vulnerability since they may preferentially attack large trees (Gaylord et al. Citation2013). Ideally, to test if crown size and tree height are coupled in relation to predisposition to drought damage, comparisons should be made between large and small trees of similar vigour or growth rate and along ample environmental gradients. Current evidence indicates that taller trees are not necessarily more vulnerable to drought than shorter conspecifics.
Figure 4. Different views on intra-specific relationships between tree size (total height) and vulnerability to drought-induced dieback and mortality (represented as trees with red, yellow and shedding leaves): (a) according to hydraulic and allometry relationships (dashed line) between tree height and conduit diameter, the tallest trees are those more prone to dying; however (b) large trees can also store more water (higher hydraulic capacitance) and develop large root systems reaching deep soil water than mid-size trees leading to higher damage in in the latter. Modified from Camarero (Citation2020)
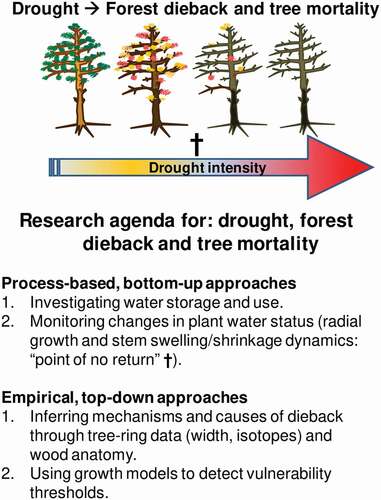
Prospects for the future: a research agenda on drought, forest dieback and tree mortality
Here I outline a research agenda on drought, dieback and tree mortality (see ). The rising consideration of relative water content as a highly reliable measurement of tree water status may be related to the fact that capacitance, water storage and the transfer between internal tree water pools are still understudied aspects of tree functioning. These topics require more attention from the scientific community, and can help reveal mechanisms that trigger drought-induced dieback and determine the recovery of functions after drought. Water stored in the xylem matrix is not only used to prevent vessel embolism during drought (Knipfer et al. Citation2019), and may potentially be used for cambium reactivation after water shortage. Such a resilience mechanism is tightly linked to xylem and phloem transport (Pfautsch et al. Citation2015). The point of no return or no recovery preceding tree mortality could be conditioned by the ability of trees to store, transfer and withdraw water from the xylem matrix. This ability may also determine how water can be transferred towards meristems such as the cambium and, therefore, related to post-drought resilience.
Figure 5. Conceptual figure summarising the research agenda proposed to investigate the causes and mechanisms of forest dieback and tree mortality after drought. The two approaches correspond to sections of the main text that describe process-based and empirical approaches. The cross along the gradient of drought intensity (arrow) indicates the ‘point of no return’ preceding tree mortality, which occurs when stem radial growth ceases and diurnal stem diameter fluctuations decrease due to irreversible stem shrinkage (see Lamacque et al. Citation2020; Preisler et al. Citation2021)
I suggest that patterns and processes be linked through the detection of points of no recovery by combining ecophysiological measures such as changes in stem diameter and relative water content as proxies of tree functioning during dieback and tree mortality. An exciting avenue of research is the combination of dendrometers and sap flow measurements which allow detecting the irreversible slowdown of stem swelling/shrinkage dynamics linked to impending tree death (Lamacque et al. Citation2020; Preisler et al. Citation2021). During dieback and tree death, changes in branch or stem diameter as decreased growth and reduced diurnal stem fluctuations due to irreversible stem shrinkage can provide reliable proxies of plant water use and storage. These integrative measures can be used to detect thresholds of loss of rehydration recovery. This approach could be used for monitoring mortality processes using dendrometers and sap flow sensors placed in sample trees in the field, at the stand level. However, this individualistic framework should be upscaled to landscape and continental scales through monitoring networks including inventories of forest structure and health to provide information at the spatial scales required for planning and managing ecosystem services.
In upscaling from trees to forests it should be considered that competition for soil water among coexisting trees from the same or different species can be a relevant factor in drought-induced mortality, particularly in closed-canopy forests. Indeed, forest structure strongly controls demography, and the effects of drought might be tightly linked to competition for water resources and self-thinning (Vilà-Cabrera et al. Citation2011). Therefore, data from forest inventories could be combined with long-term monitoring plots or networks of forest damage to evaluate the relative importance of drought and competition among neighbouring trees during dieback and tree mortality (e.g., Andrus et al. Citation2021).
Large monitoring networks such as those developed through national forest inventories or international monitoring schemes of canopy defoliation may underestimate dieback impacts. Extreme droughts and dieback episodes are relatively rare events. Therefore, traditional inventory sampling may generate abundant information on normal mortality processes of average-sized trees during standard climatic conditions at large spatial scales, but obviate extreme hydrological conditions defining drought responses which characterise local dieback processes (Hember et al. Citation2017). We need international, purpose-built field networks focusing on long-term monitoring of dieback and mortality hotspots arising from rare, extreme dry spells. For instance, an international network of long-term monitoring plots allowed estimating the impacts of drought and subsequent dieback on carbon fluxes of Amazon rainforests (Phillips et al. Citation2009).
Conclusions
Drought-induced dieback is a widely studied phenomenon, but current available forecasting tools are far of having enough predictive power to envisage which trees, stands or forests may be the most impacted by water shortage associated with weather and climate extremes (heat waves). I argue that reconciling pattern- and process-based field and ecophysiological studies is a first step towards gaining predictive power of dieback and tree mortality. In a second step, stand development and tree-to-tree interactions need to be accounted for because dieback and mortality caused by drought-stress may be also a result of structural overshoot (Jump et al. Citation2017) and competition for water in semi-arid sites (Young et al. Citation2017). Upscaling from tree to stand and forest landscape scales would require considering large-scale datasets including field monitoring networks of tree mortality.
Notes on contributor
J. Julio Camarero is a Research Professor at CSIC. He received his academic degrees at the Universities of Salamanca and Barcelona. He is interested on disentangling the patterns and processes driving tree growth responses to climatic (drought, low temperature) and biotic stressors (pathogens, herbivores). His research fields include forest ecology, dendroecology and global change biology.
Acknowledgements
I am grateful to several reviewers for their constructive comments on previous drafts, and to the editors for inviting me to contribute this Grubb Review. I dedicate this essay to Emilio Cervantes (IRNASA, CSIC) and Emilia Gutiérrez (University of Barcelona) who educated me as a researcher, and to Prof. Ramón Margalef, who passed away in May 2004, and whose unforgettable books attracted me towards ecology. I also thank many colleagues who have helped me in the field and with whom I discussed some of the ideas exposed here.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Correction Statement
This article has been republished with minor changes. These changes do not impact the academic content of the article.
Additional information
Funding
References
- Adams HD, Guardiola‐Claramonte M, Barron‐Gafford GA, Villegas JC, Breshears DD, Zou CB, Troch PA, Huxman TE. 2009. Temperature sensitivity of drought-induced tree mortality portends increased regional die-off under global-change-type drought. Proc Nat Acad Sci USA. 106(17):7063–7066. doi:https://doi.org/10.1073/pnas.0901438106.
- Adams HD, Zeppel MJB, Anderegg WRL, Hartmann H, Landhausser SM, Tissue DT, Huxman TE, Hudson PJ, Franz TE, Allen CD, et al. 2017. A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nat Ecol Evol. 1(9):1285–1291. doi:https://doi.org/10.1038/s41559-017-0248-x
- Allen CD, Breshears DD, McDowell NG. 2015. On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere. 6(8):art129. doi:https://doi.org/10.1890/ES15-00203.1.
- Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EH, et al. 2010. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manage. 259(4):660–684. doi:https://doi.org/10.1016/j.foreco.2009.09.001.
- Anderegg WR, Flint A, Huang C, Flint L, Berry JA, Davis FW, Sperry JS, Field CB. 2015a. Tree mortality predicted from drought-induced vascular damage. Nat Geosci. 8(5):367–371 doi:https://doi.org/10.1038/ngeo2400.
- Anderegg WR, Plavcova L, Anderegg LD, Hacke UG, Berry JA, Field CB. 2013b. Drought’s legacy: multiyear hydraulic deterioration underlies widespread aspen forest die-off and portends increased future risk. Glob Change Biol. 19(4):1188–1196 doi:https://doi.org/10.1111/gcb.12100.
- Anderegg WR, Schwalm C, Biondi F, Camarero JJ, Koch G, Litvak M, Ogle K, Shaw JD, Shevliakova E, Williams AP, et al. 2015b. Pervasive drought legacies in forest ecosystems and their implications for carbon cycle models. Science. 349(6247):528–532 doi:https://doi.org/10.1126/science.aab1833.
- Anderegg WRL, Kane JM, Anderegg LDL. 2013a. Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Ch. 3(1):30–36 doi:https://doi.org/10.1038/nclimate1635.
- Anderegg WRL, Klein T, Bartlett M, Sack L, Pellegrini AFA, Choat B, Jansen S. 2016. Meta-analysis reveals that hydraulic traits explain cross-species patterns of drought-induced tree mortality across the globe. Proc Nat Acad Sci USA. 113(18):5024–5029. doi:https://doi.org/10.1073/pnas.1525678113.
- Andrus RA, Chai RK, Harvey BJ, Rodman KC, Veblen TT. 2021. Increasing rates of subalpine tree mortality linked to warmer and drier summers. J Ecol. 109(5):2203–2218. doi:https://doi.org/10.1111/1365-2745.13634.
- Anfodillo T, Petit G, Sterck F, Lechthaler S, Olson ME. 2016. Allometric trajectories and “stress”: a quantitative approach. Front Plant Sci. 7:1681. doi:https://doi.org/10.3389/fpls.2016.01681.
- Asner GP, Brodrick PG, Anderson CB, Vaughn N, Knapp DE, Martin RE. 2016. Progressive forest canopy water loss during the 2012–2015 California drought. Proc Nat Acad Sci USA. 113(2):E249–E255. doi:https://doi.org/10.1073/pnas.1523397113.
- Barbeta A, Mejía-Chang M, Ogaya R, Voltas J, Dawson TE, Peñuelas J. 2015. The combined effects of a long-term experimental drought and an extreme drought on the use of plant-water sources in a Mediterranean forest. Glob Chang Biol. 21(3):1213–1225. doi:https://doi.org/10.1111/gcb.12785.
- Batllori E, Lloret F, Aakala T, Anderegg WRL, et al. 2020. Forest and woodland replacement patterns following drought-related mortality. Proc Nat Acad Sci USA USA. 117(47):29720–29729. doi:https://doi.org/10.1073/pnas.2002314117.
- Bennett AC, McDowell NG, Allen CD, Anderson-Teixeira KJ. 2015. Larger trees suffer most during drought in forests worldwide. Nat Plants. 1(10):15139. doi:https://doi.org/10.1038/nplants.2015.139.
- Blackman CJ, Pfautsch S, Choat B, Delzon S, Gleanson SM, Duursma RA. 2016. Toward an index of desiccation time to tree mortality under drought. Plant Cell Env. 39(10):2342–2345. doi:https://doi.org/10.1111/pce.12758.
- Cailleret M, Dakos V, Jansen S, Robert EMR, Aakala T, Amoroso MM, Antos JA, Bigler C, Bugmann H, Caccianaga M, et al. 2018. Early-warning signals of individual tree mortality based on annual radial growth. Front Plant Sci. 9:e1964. doi:https://doi.org/10.3389/fpls.2018.01964.
- Cailleret M, Jansen S, Robert EM, Desoto L, Aakala T, Antos JA, Beikircher B, Bigler C, Bugmann H, Caccianiga M, et al. 2017. A synthesis of radial growth patterns preceding tree mortality. Glob Change Biol. 23(4):1675–1690. doi:https://doi.org/10.1111/gcb.13535.
- Camarero JJ, Colangelo M, Gazol A, Azorín-Molina C. 2020. Drought and cold spells trigger dieback in temperate forests. Dendrochronologia. 66:125802.
- Camarero JJ, Gazol A, Sangüesa-Barreda G, Cantero A, Sánchez-Salguero R, Sánchez-Miranda A, Granda E, Serra-Maluquer X, Ibañez R. 2018. Forest growth responses to drought at short- and long-term scales in Spain: squeezing the stress memory from tree rings. Front Ecol Evol. 6:9. doi:https://doi.org/10.3389/fevo.2018.00009.
- Camarero JJ, Gazol A, Sangüesa‐Barreda G, Oliva J, Vicente‐Serrano SM, Gibson D. 2015. To die or not to die: early warnings of tree dieback in response to a severe drought. J Ecol. 103(1):44–57. doi:https://doi.org/10.1111/1365-2745.12295.
- Camarero JJ. 2020. Within- versus between-species size effects on drought-induced dieback and mortality. Tree Physiol. 41(5):679–682. doi:https://doi.org/10.1093/treephys/tpaa167.
- Cherubini P, Battipaglia G, Innes JL. 2021. Tree vitality and forest health: can tree-ring stable isotopes be used as indicators? Curr For Rep. doi:https://doi.org/10.1007/s40725-021-00137-8
- Choat B, Brodribb TJ, Brodersen CR, Duursma RA, Lopez R, Medlyn BE. 2018. Triggers of tree mortality under drought. Nature. 558(7711):531–539. doi:https://doi.org/10.1038/s41586-018-0240-x.
- Choat B, Jansen S, Brodribb TJ, Cochard H, Delzon S, Bhaskar R, Bucci SJ, Feild TS, Gleason SM, Hacke UG, et al. 2012. Global convergence in the vulnerability of forests to drought. Nature. 491(7426):752–755. doi:https://doi.org/10.1038/nature11688
- Cochard H. 2019. A new mechanism for tree mortality due to drought and heatwaves. In: bioRxiv. p. 531632.
- Colangelo M, Camarero JJ, Battipaglia G, Borghetti M, De Micco V, Gentilesca T, Ripullone F. 2017a. A multi-proxy assessment of dieback causes in a Mediterranean oak species. Tree Physiol. 37(5):617–631. doi:https://doi.org/10.1093/treephys/tpx002.
- Colangelo M, Camarero JJ, Borghetti M, Gazol A, Ripullone F, Ripullone F. 2017b. Size matters a lot: drought-affected Italian oaks are smaller and show lower growth prior to tree death. Front Plant Sci. 8:135. doi:https://doi.org/10.3389/fpls.2017.00135.
- De Kauwe MG, Medlyn BE, Ukkola AM, Mu M, Sabot ME, Pitman AJ, Meir P, Cernusak L, Rifai SW, Choat B. 2020. Identifying areas at risk of drought-induced tree mortality across South-Eastern Australia. Global Change Biol. 26(10):5716–5733. doi:https://doi.org/10.1111/gcb.15215.
- DeSoto L, Cailleret M, Sterck F, Jansen S, Kramer K, Robert EMR, Aakala T, Amoroso MM, Bigler C, Camarero JJ, et al. 2020. Low growth resilience to drought is related to future mortality risk in trees. Nat Comm. 11(1):545. doi:https://doi.org/10.1038/s41467-020-14300-5
- Dobbertin M. 2005. Tree growth as indicator of tree vitality and of tree reaction to environmental stress: a review. Eur J For Res. 124(4):319–333. doi:https://doi.org/10.1007/s10342-005-0085-3.
- FAO. 2020. The State of the World’s Forests 2020. In: Forests, biodiversity and people. Rome: FAO.
- Farquhar GD, O’Leary MH, Berry JA. 1982. On the relationship between carbon isotope discrimination and the intercellular carbon-dioxide concentration in leaves. Australian J Plant Physiol. 9:121–137.
- Gaylord ML, Kolb TE, Pockman WT, Plaut JA, Yepez EA, Macalady AK, Pangle RE, McDowell NG. 2013. Drought predisposes piñon–juniper woodlands to insect attacks and mortality. New Phytol. 198(2):567–578. doi:https://doi.org/10.1111/nph.12174.
- Gessler A, Cailleret M, Joseph J, Schönbeck L, Schaub M, Lehmann M, Treydte K, Rigling A, Timofeeva G, Saurer M, et al. 2018. Drought induced tree mortality - a tree-ring isotope based conceptual model to assess mechanisms and predispositions. New Phytol. 219(2):485–490. doi:https://doi.org/10.1111/nph.15154
- Greenwood S, Ruiz‐Benito P, Martínez‐Vilalta J, Lloret F, Kitzberger T, Allen CD, Fensham R, Laughlin DC, Kattge J, Bönisch G, et al. 2017. Tree mortality across biomes is promoted by drought intensity, lower wood density and higher specific leaf area. Ecol Lett. 20(4):539–553. doi:https://doi.org/10.1111/ele.12748
- Hammond WM, Yu K, Wilson LA, Will RE, Anderegg WRL, Adams HD. 2019. Dead or dying? Quantifying the point of no return from hydraulic failure in drought-induced tree mortality. New Phytol. 223(4):1834–1843. doi:https://doi.org/10.1111/nph.15922.
- Hartmann H, Moura CF, Anderegg WR, Ruehr NK, Salmon Y, Allen CD, Arndt SK, Breshears DD, Davi H, Galbraith D. 2018. Research frontiers for improving our understanding of drought-induced tree and forest mortality. New Phytol. 218(1):15–28. doi:https://doi.org/10.1111/nph.15048.
- He W, Liu H, Qi Y, Liu F, Zhu X. 2020. Patterns in nonstructural carbohydrate contents at the tree organ level in response to drought duration. Global Change Biol. 26(6):3627–3638. doi:https://doi.org/10.1111/gcb.15078.
- Hember REA, Kurz WA, Coops NC. 2017. Relationships between individual-tree mortality and water-balance variables indicate positive trends in water stress-induced tree mortality across North America. Global Change Biol. 23(4):1691–1710. doi:https://doi.org/10.1111/gcb.13428.
- Hoffmann WA, Marchin RM, Abit P, Lau OL. 2011. Hydraulic failure and tree dieback are associated with high wood density in a temperate forest under extreme drought. Global Change Biol. 17(8):2731–2742. doi:https://doi.org/10.1111/j.1365-2486.2011.02401.x.
- IPCC. 2018. Global warming of 1.5°C. An IPCC special report on the impacts of global warming of 1.5°C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change, sustainable development, and efforts to eradicate poverty. Geneva: World Meteorological Organization.
- Johnson DM, Domec J-C, Berry ZC, Schwantes AM, McCulloh KA, Woodruff DR, Wayne Polley H, Wortemann R, Swenson JJ, Scott Mackay D, et al. 2018. Co-occurring woody species have diverse hydraulic strategies and mortality rates during an extreme drought. Plant Cell Env. 41(3):576–588. doi:https://doi.org/10.1111/pce.13121
- Jump AS, Ruiz-Benito P, Greenwood S, Allen CD, Kitzberger T, Fensham R, Martínez-Vilalta J, Lloret F. 2017. Structural overshoot of tree growth with climate variability and the global spectrum of drought-induced forest dieback. Global Change Biol. 23(9):3742–3757. doi:https://doi.org/10.1111/gcb.13636.
- Kiorapostolou N, Camarero JJ, Carrer M, Sterck F, Brigita B, Sangüesa-Barreda G, Petit G. 2020. Scots pine trees react to drought by increasing xylem and phloem conductivities. Tree Physiol. 40(6):774–781. doi:https://doi.org/10.1093/treephys/tpaa033.
- Klein T, Di Matteo G, Rotenberg E, Cohen S, Yakir D. 2012. Differential ecophysiological response of a major Mediterranean pine species across a climatic gradient. Tree Physiol. 33(1):26–36. doi:https://doi.org/10.1093/treephys/tps116.
- Knipfer T, Reyes C, Earles JM, Berry ZC, Johnson DM, Brodersen CR, McElrone AJ. 2019. Spatiotemporal coupling of vessel cavitation and discharge of stored xylem water in a tree sapling. Plant Physiol. 179(4):1658–1668. doi:https://doi.org/10.1104/pp.18.01303.
- Körner C. 2003. Carbon limitation in trees. J Ecol. 91(1):4–17. doi:https://doi.org/10.1046/j.1365-2745.2003.00742.x.
- Körner C. 2019. No need for pipes when the well is dry—a comment on hydraulic failure in trees. Tree Physiol. 39(5):695–700. doi:https://doi.org/10.1093/treephys/tpz030.
- Lamacque L, Charrier G, dos Santos Farnese F, Lemaire B, Ameglio T, Herbette S. 2020. Drought-induced mortality: branch diameter variation reveals a point of no recovery in lavender species. Plant Phisiol. 183(4):1638–1649. doi:https://doi.org/10.1104/pp.20.00165.
- Lempereur M, Martin-StPaul NK, Damesin C, Joffre R, Ourcival JM, Rocheteau A, Rambal S. 2015. Growth duration is a better predictor of stem increment than carbon supply in a M editerranean oak forest: implications for assessing forest productivity under climate change. New Phytol. 207(3):579–590. doi:https://doi.org/10.1111/nph.13400.
- Manion PD. 1981. Tree disease concepts. Upper Saddle River: Prentice Hall.
- Martínez-Vilalta J, Anderegg WRL, Sapes G, Sala A. 2019. Greater focus on water pools may improve our ability to understand and anticipate drought-induced mortality in plants. New Phytol. 223(1):22–32. doi:https://doi.org/10.1111/nph.15644.
- Marusig D, Petruzzellis F, Tomasella M, Napolitano R, Altobelli A, Nardini A. 2020. Correlation of field-measured and remotely sensed plant water status as a tool to monitor the risk of drought-induced forest decline. Forests. 11(1):77. doi:https://doi.org/10.3390/f11010077.
- McDowell NG. 2011. Mechanisms linking drought, hydraulics, carbon metabolism, and vegetation mortality. Plant Physiol. 155(3):1051–1059. doi:https://doi.org/10.1104/pp.110.170704.
- McDowell NG, Allen CD. 2015. Darcy’s law predicts widespread forest mortality under climate warming. Nat Clim Chang. 5(7):669–672. doi:https://doi.org/10.1038/nclimate2641.
- McDowell NG, Beerling DJ, Breshears DD, Fisher RA, Raffa KF, Stitt M. 2011. The interdependence of mechanisms underlying climate-driven vegetation mortality. Trends Ecol Evol. 26(10):523–532. doi:https://doi.org/10.1016/j.tree.2011.06.003.
- McDowell NG, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, et al. 2008. Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol. 178(4):719–739. doi:https://doi.org/10.1111/j.1469-8137.2008.02436.x
- McDowell NG, Williams AP, Xu C, Pockman WT, Dickman LT, Sevanto S, Pangle R, Limousin J, Plaut J, Mackay DS, et al. 2016. Multi-scale predictions of massive conifer mortality due to chronic temperature rise. Nat Clim Ch. 6(3):295–300. doi:https://doi.org/10.1038/nclimate2873
- Nardini A, Battistuzzo M, Savi T. 2013. Shoot desiccation and hydraulic failure in temperate woody angiosperms during an extreme summer drought. New Phytol. 200(2):322–329. doi:https://doi.org/10.1111/nph.12288.
- Olson ME, Soriano D, Rosell JA, Anfodillo T, Donoghue MJ, Edwards EJ, León-Gómez C, Dawson T, Camarero Martínez JJ, Castorena M, et al. 2018. Plant height and hydraulic vulnerability to drought and cold. Proc Natl Acad Sci. 115(29):7551–7556. doi:https://doi.org/10.1073/pnas.1721728115
- Pedersen BS. 1998. The role of stress in the mortality of Midwestern oaks as indicated by growth prior to death. Ecology. 79(1):79–93. doi:https://doi.org/10.1890/0012-9658(1998)079[0079:TROSIT]2.0.CO;2.
- Pellizzari E, Camarero JJ, Gazol A, Sangüesa-Barreda G, Carrer M. 2016. Wood anatomy and carbon-isotope discrimination support long-term hydraulic deterioration as a major cause of drought-induced dieback. Global Change Biol. 22(6):2125–2137. doi:https://doi.org/10.1111/gcb.13227.
- Peltier DMP, Barber JJ, Ogle K, McGlone M. 2018. Quantifying antecedent climatic drivers of tree growth in the Southwestern US. J Ecol. 106(2):613–624. doi:https://doi.org/10.1111/1365-2745.12878.
- Peltier DMP, Ogle K. 2019. Legacies of more frequent drought in ponderosa pine across the western United States. Global Change Biol. 25(11):3803–3816. doi:https://doi.org/10.1111/gcb.14720.
- Pfautsch S, Renard J, Tjoelker MG, Salih A. 2015. Phloem as capacitor: radial transfer of water into xylem of tree stems occurs via symplastic transport in ray parenchyma. Plant Physiol. 167(3):963–971. doi:https://doi.org/10.1104/pp.114.254581.
- Phillips OL, Aragao LEOC, Lewis SL, Fisher JB, Lloyd, Lloyd J, Lopez-Gonzalez G, Malhi Y, Monteagudo A, Peacock J, et al. 2009. Drought sensitivity of the Amazon rainforest. Science. 323(5919):1344–1347. doi:https://doi.org/10.1126/science.1164033
- Preisler Y, Tatanirov F, Grünzweig JM, Yakir D. 2021. Seeking the “point of no return” in the sequence of events leading to mortality of mature trees. Plant Cell Env. 44(5):1315–1328. doi:https://doi.org/10.1111/pce.13942.
- Preisler Y, Tatarinov F, Grünzweig JM, Bert D, Ogée J, Wingate L, Rotenberg E, Rohatyn S, Her N, Moshe I, et al. 2019. Mortality versus survival in drought-affected Aleppo pine forest depends on the extent of rock cover and soil stoniness. Funct Ecol. 33(5):901–912. doi:https://doi.org/10.1111/1365-2435.13302.
- Reed CC, Hood SM. 2021. Few generalizable patterns of tree-level mortality during extreme drought and concurrent bark beetle outbreaks. Sci Tot Env. 750:141306. doi:https://doi.org/10.1016/j.scitotenv.2020.141306.
- Ripullone F, Camarero JJ, Colangelo M, Voltas J. 2020. Variation in the access to deep soil water pools explains tree-to-tree differences in drought-triggered dieback of Mediterranean oaks. Tree Physiol. 40(5):591–604. doi:https://doi.org/10.1093/treephys/tpaa026.
- Rowland L, da Costa AC, Galbraith DR, Oliveira RS, Binks OJ, Oliveira AA, Pullen AM, Doughty CE, Metcalfe DB, Vasconcelos SS, et al. 2015. Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature. 528(7580):119–122. doi:https://doi.org/10.1038/nature15539
- Sánchez-Salguero R, Camarero JJ, Grau JM, De la Cruz A, Gil PM, Minaya M, Fernández-Cancio A. 2017a. Analysing atmospheric processes and climatic drivers of tree defoliation to determine forest vulnerability to climate warming. Forests. 8(1):13. doi:https://doi.org/10.3390/f8010013.
- Sánchez-Salguero R, Camarero JJ, Gutiérrez E, González Rouco F, Gazol A, Sangüesa-Barreda G, Andreu-Hayles L, Linares JC, Seftigen K. 2017b. Assessing forest vulnerability to climate warming using a process-based model of tree growth: bad prospects for rear-edges. Global Change Biol. 23(7):2705–2719. doi:https://doi.org/10.1111/gcb.13541.
- Sánchez-Salguero R, Navarro-Cerrillo RM, Camarero JJ, Fernández-Cancio A. 2012. Selective drought-induced decline of pine species in southeastern Spain. Clim Ch. 113(3–4):767–785. doi:https://doi.org/10.1007/s10584-011-0372-6.
- Sapes G, Roskilly B, Dobrowski S, Maneta M, Anderegg WRL, Martinez-Vilalta J, Sala A. 2019. Plant water content integrates hydraulics and carbon depletion to predict drought-induced seedling mortality. Tree Physiol. 39(8):1300–1312. doi:https://doi.org/10.1093/treephys/tpz062.
- Sargeant CI, Singer MB, Vallet-Coulomb C. 2019. Identification of source-water oxygen isotopes in trees toolkit (ISO-Tool) for deciphering historical water use by forest trees. Water Resour Res. 55(12):10954–10975. doi:https://doi.org/10.1029/2018WR024519.
- Schwalm CR, Anderegg WR, Michalak AM, Fisher JB, Biondi F, Koch G, Litvak M, Ogle K, Shaw JD, Wolf A, et al. 2017. Global patterns of drought recovery. Nature. 548(7666):202–205. doi:https://doi.org/10.1038/nature23021
- Serra‐Maluquer X, Granda E, Camarero JJ, Vilà‐Cabrera A, Jump AS, Sánchez‐Salguero R, Sangüesa‐Barreda G, Imbert JB, Gazol A, Jucker T. 2021. Impacts of recurrent dry and wet years alter long-term tree growth trajectories. J Ecol. 109(3):1561–1574. doi:https://doi.org/10.1111/1365-2745.13579.
- Sevanto S, McDowell NG, Dickman LT, Pangle R, Pockman WT. 2014. How do trees die? A test of the hydraulic failure and carbon starvation hypotheses. Plant Cell Env. 37(1):153–161. doi:https://doi.org/10.1111/pce.12141.
- Stephenson NL, Das AJ. 2020. Height-related changes in forest composition explain increasing tree mortality with height during an extreme drought. Nat Comm. 11(1):3402. doi:https://doi.org/10.1038/s41467-020-17213-5.
- Stovall AEL, Shugart H, Yang X. 2019. Tree height explains mortality risk during an intense drought. Nat Comm. 10(1):4385. doi:https://doi.org/10.1038/s41467-019-12380-6.
- Suarez ML, Ghermandi L, Kitzberger T. 2004. Factors predisposing episodic drought-induced tree mortality in Nothofagus - site, climatic sensitivity and growth trends. J Ecol. 92(6):954–966. doi:https://doi.org/10.1111/j.1365-2745.2004.00941.x.
- Trugman A, Anderegg LDL, Anderegg WRL, Das AJ, Stephenson NL. 2021. Why is tree drought mortality so hard to predict? Trends Ecol Evol. 36(6):520–532. doi:https://doi.org/10.1016/j.tree.2021.02.001.
- Vaganov EA, Hughes MK, Shashkin AV. 2006. Growth dynamics of conifer tree rings: images of past and future environments. Springer.
- Van Mantgem PJ, Stephenson NL, Byrne JC, Daniels LD, Franklin JF, Fulé PZ, Harmon ME, Larson AJ, Smith JM, Taylor AH. 2009. Widespread increase of tree mortality rates in the western United States. Science. 323(5913):521–524. doi:https://doi.org/10.1126/science.1165000.
- Venturas MD, Todd HN, Trugman AT, Anderegg WRL. 2020. Understanding and predicting forest mortality in the western United States using long-term forest inventory data and modeled hydraulic damage. New Phytol. 230(5):1896–1910. doi:https://doi.org/10.1111/nph.17043.
- Vilà-Cabrera A, Martínez-Vilalta J, Vayreda J, Retana J. 2011. Structural and climatic determinants of demographic rates of Scots pine forests across the Iberian Peninsula. Ecol Appl. 21(4):1162–1172. doi:https://doi.org/10.1890/10-0647.1.
- Voltas J, Camarero JJ, Carulla D, Aguilera M, Ortiz A, Ferrio JP. 2013. A retrospective, dual-isotope approach reveals individual predispositions to winter-drought induced tree dieback in the southernmost distribution limit of Scots pine. Plant, Cell Env. 36(8):1435–1448. doi:https://doi.org/10.1111/pce.12072.
- Wang W, English NB, Grossiord C, Gessler A, Das AJ, Stephenson NL, Baisan CH, Allen CD, McDowell NG. 2021. Mortality predispositions of conifers across western USA. New Phytol. 229(2):831–844. doi:https://doi.org/10.1111/nph.16864.
- Waring RH. 1987. Characteristics of trees predisposed to die. BioScience. 37(8):569–574. doi:https://doi.org/10.2307/1310667.
- Williams AP, Allen CD, Macalady AK, Griffin D, Woodhouse CA, Meko DM, Swetnam TW, Rauscher SA, Seager R, Grissino‐Mayer HD, et al. 2013. Temperature as a potent driver of regional forest drought stress and tree mortality. Nat Clim Ch. 3(3):292–297. doi:https://doi.org/10.1038/nclimate1693
- Young DJ, Stevens JT, Earles JM, Moore J, Ellis A, Jirka AL, Latimer AM, Lloret F. 2017. Long-term climate and competition explain forest mortality patterns under extreme drought. Ecol Lett. 20(1):78–86. doi:https://doi.org/10.1111/ele.12711.
