ABSTRACT
Annually, approximately 2 million assisted reproductive technology (ART) procedures are performed worldwide, of which, only ~25% lead to successful delivery. There are two major factors contributing to successful implantation: embryo quality and endometrial receptivity (ER). Although embryo quality might be assessed through morphological and genetic testing, no clinically approved techniques are available to evaluate ER. Mucus in different parts of the female reproductive tract contains many cytokines, chemokines, growth factors, and nucleic acids, which influence and reflect various implantation-related processes. Therefore, the aim of the present review was to summarize available data regarding noninvasively obtained mucosal biomarkers for ER and to investigate their ability to predict the outcome of ART procedures. A broad literature search was performed to define studies related to noninvasive ER assessments. More than 50 biomarkers detectable in endometrial fluid, embryo transfer cannula leftover cells and mucus, menstrual blood, cervicovaginal washings are discussed herein. The remarkable methodological heterogeneity of the reviewed studies complicates the comparison of their results. Nevertheless, certain promising analytical targets may already be identified, such as urocortin, activin A, IL-1β, TNF-α, IP-10, MCP-1, and several oxidative stress biomarkers. The present review contains a collection of currently available mucosal biomarker-related data, which may provide insights for future studies.
Abbreviations: ART: assisted reproductive technology; ER: endometrial receptivity; IVF: in vitro fertilization; ICSI: intracytoplasmic sperm injection; IUI: intrauterine insemination; MeSH: Medical Subject Headings; hDP 200: human decidua-associated protein 200; ET: embryo transfer; IL-18: Interleukin-18; LRG: leucine-rich α2-glycoprotein; ROC: receiver operating characteristic; AUC: area under the ROC-curve; LH: luteinizing hormone; LIF: leukemia inhibitory factor; TNF-α: tumor necrosis factor alpha; IFN-γ: interferon γ; MCP-1: monocyte chemoattractant protein-1; VEGF: vascular endothelial growth factor; SOD: superoxide dismutase; CAT: catalase; LPO: lipid peroxidation; TTG: total thiol groups; TAP: total antioxidant power; CE: chronic endometritis
Introduction
Infertility is defined as the inability to conceive within one year of regular unprotected intercourse (or 6 months if a woman is ≥35 years old) (Practice Committee of the American Society for Reproductive Medicine Citation2013). Globally, this problem affects 15% of couples of reproductive age, with the male factor serving as a primary or contributing factor in approximately half of cases (Sun et al. Citation2019). Infertility is defined as a public health priority in many regions of the world (Duffy et al. Citation2020). Infertility and its treatment inevitably impact patients’ quality of life, causing economic strain, depression, social disruption, and family discord (Gdanska et al. Citation2017; Massarotti et al. Citation2019).
Currently, assisted reproductive technologies (ART), such as in vitro fertilization (IVF), intracytoplasmic sperm injection (ICSI), and intrauterine insemination (IUI), are available to treat infertility. Annually, approximately 2 million ART cycles are performed worldwide, of which, only ~25% lead to successful delivery (De Mouzon et al. Citation2020). Considering the high cost of ART procedures as well as possible adverse effects of an increased hormonal load during cycles, the development of new approaches to predict the outcome of ART treatment is of great value.
Embryo quality is known to be responsible for approximately 30% of the implantation failures (Oron et al. Citation2014; Zhu et al. Citation2014). Although the implementation of morphological grading and preimplantation genetic testing helped to moderately increase the likelihood of successful IVF and ICSI outcomes, the implantation rate is still suboptimal, thereby it is important to address the other major cause of implantation failures – impaired endometrial receptivity (ER) (Baltaci et al. Citation2006).
ER is defined as the ability of the endometrium to accept the embryo, enabling its development and securing optimal trophic and microenvironmental status (Lessey and Young Citation2019). Currently, there is no widely accepted approach for the quantitative evaluation of this parameter. A plethora of studies have been conducted to evaluate ER based on endometrial biopsy analysis, hysteroscopy, and ultrasound findings (Sakumoto et al. Citation1992; Jinno et al. Citation2001; Santi et al. Citation2012; Voa et al. Citation2015; Jin et al. Citation2017). Nevertheless, in a recent meta-analysis, it was concluded that these approaches are characterized by a poor ability to predict clinical pregnancy (Craciunas et al. Citation2019).
Despite the above-mentioned advances in endometrial biopsy analysis, the modern trends in precision medicine are gravitating toward noninvasive approaches. Fortunately, there are certain options to obtain biomaterial from the female reproductive tract without any traumatizing interventions, such as cervicovaginal/endometrial washing, direct endometrial fluid aspiration, and menstrual blood collection. ER studies involving these biological substrates are scarce and heterogeneous. Therefore, they are either completely ignored or simply mentioned in the currently published reviews and meta-analyses. The aim of the present review was to summarize the available knowledge regarding noninvasively obtained mucosal biomarkers for ER and to investigate their ability to predict the outcome of ART procedures.
Search strategy
A literature search was performed using PubMed resources: MEDLINE, PubMed Central, and Bookshelf. To gain insight into the historical evolution of ER studies, no restrictions regarding publication date were applied. The search input consisted of a broad spectrum of terms and keywords related to the topic and Medical Subject Headings (MeSH), such as endometrium, uterus, ER, assisted reproductive technologies, embryo implantation, pregnancy, biomarkers, and endometrial fluid. The PubMed function ‘Similar articles’ were used as an auxiliary tool to reveal articles beyond the scope of the implemented search input.
Single endometrial fluid biomarker studies
A summary of studies that sought to analyze a single molecular biomarker in endometrial fluid is presented in . In all cases, endometrial fluid samples were obtained by flushing the uterine cavity with a physiological solution. The timing of sample collection varied significantly from study to study.
Table 1. Summary of studies that sought to analyze a single endometrial fluid biomarker.
Human decidua-associated protein 200
The first attempt to evaluate the relationship between endometrial fluid biomarkers and IVF outcomes was conducted by Halperin et al. (Citation1995). Human decidua-associated protein 200 (hDP 200), which was previously described and detected in endometrial secretion by the same research group, was selected as a biomarker (Halperin et al. Citation1990; Golan et al. Citation1994). The secretory glycoprotein, hDP 200, was hypothesized to be involved in the functional development of the endometrial tissue. hDP 200 was evaluated in endometrial washings obtained on the day of embryo transfer (ET). A significant positive correlation (p < 0.05) was found between the hDP 200 level and pregnancy rate. However, analytical parameters such as sensitivity and specificity were suboptimal (57.14% and 69.47%, respectively). No further attempts to evaluate its clinical value regarding ART procedures outcomes were performed. Three years later, this research group attempted to investigate the ability of hDP 200 to detect ectopic pregnancy; however, no statistical significance was found (Halperin et al. Citation1998).
Interleukin-18
Interleukin-18 (IL-18) plays an important role in the regulation of the uterine cytokine microenvironment (Ida et al. Citation2000; Yoshino et al. Citation2001; Yasuda et al. Citation2019). IL-18 modulates innate and acquired immune responses, controls the Th1/Th2 balance, promotes the expansion of natural killer cells and changes in their phenotype (Smeltz et al. Citation2001; Senju et al. Citation2018). Due to its ability to possibly reflect the whole status of cytokine balance in the uterus, Lédée-Bataille et al. (Citation2004) selected IL-18 as a biomarker for ER. In their study (n = 133), IL-18 was analyzed in endometrial fluid collected prior to oocyte retrieval in patients undergoing IVF. In a group of patients with undetectable levels of IL-18, the rate of pregnancy was higher than that found in the group with detectable IL-18 levels (37.9% vs. 15%, respectively, p < 0.05). Despite the statistical significance of raw group comparisons, the prognostic value of IL-18 as a sole biomarker for ER remains low. Nonetheless, in contrast to hDP 200, IL-18 has been studied in future multiple biomarker studies, which will be discussed later.
Leucine-rich α2-glycoprotein
Leucine-rich α2-glycoprotein (LRG) plays a role in the regulation of immune cell infiltration, re-epithelialization, and angiogenesis (Liu et al. Citation2020). Knowledge regarding the role of LRG in ER is limited, although there is indirect evidence of its participation in implantation and decidualization (Tabibzadeh and Babaknia Citation1995). Such findings served as the basis of a small-scale study conducted by Gillott et al. (Citation2008). The study included 31 patients who underwent unspecified infertility treatment in an ART clinic. A statistically significant abundance of LRG isoforms III and IV (p < 0.01) was observed in women with treatment-independent pregnancies (n = 3) compared to non-pregnant (n = 25) and pregnant women (n = 3). These results did not promote any future studies on ER using LRG as a biomarker.
Urocortin
Urocortin is a peptide produced by the luminal and glandular epitheliocytes of the human endometrium (Florio et al. Citation2002). This peptide is closely related to corticotropin-releasing hormone in terms of structure, and partially shares receptors with this hormone (Reyes et al. Citation2001). There is evidence that urocortin might play a role in decidualization and endometrial immune response, inducing the expression of various cytokines (Torricelli et al. Citation2007; Novembri et al. Citation2011). Florio et al. (Citation2008) conducted a prospective controlled study (n = 71) to evaluate urocortin as a biomarker of successful IUI outcomes. Endometrial wash fluid was obtained prior to the IUI in the periovulatory phase. Urocortin levels were significantly higher in pregnant than non-pregnant study participants (0.38 vs. 0.13 ng/mL, respectively, p < 0.001). Based on receiver operating characteristic (ROC) analysis, the researchers revealed that at a cutoff value of 0.321 ng/mL, this biomarker achieved sensitivity of 60.7% and specificity of 97.7%, with an area under the ROC-curve (AUC) as high as 0.88. Surprisingly, despite the great results of this study, subsequent biomarker studies did not include urocortin.
Activin A
In contrast to previously discussed biomarkers, the involvement of activin A in essential endometrial processes has been supported by numerous in vitro and in vivo studies (Jones et al. Citation2000; Popovici et al. Citation2000; Florio et al. Citation2003). This glycoprotein belongs to the transformation factor β superfamily and is mainly expressed in reproductive organs. Its functions include stimulation of decidualization, trophoblast invasion, embryonic differentiation, and fetal brain protection under hypoxic conditions (Bloise et al. Citation2019). Due to its prominent role in the implantation process, Florio et al. (Citation2010) examined the ability of activin A to reflect the ER. Endometrial washing samples were collected during the periovulatory phase on the day of human chorionic gonadotropin prescription. The levels of activin A in the pregnant group were statistically significantly increased relative to levels in the non-pregnant group (0.800 vs. 0.022 ng/mL, respectively, p < 0.001). According to the results of ROC analysis, activin A levels were characterized by a sensitivity of 76%, specificity of 100%, and AUC of 0.926 (at a cutoff value of 0.04). Thus, activin A demonstrated excellent predictive power for successful implantation in patients undergoing IUI. Nevertheless, there have been no attempts to investigate its predictive power in other cohorts of patients receiving ART treatment, which already appears to be a common trend in the field of endometrial fluid biomarker research.
Glycodelin
Glycodelin is a secretory glycoprotein, which is primarily expressed in different parts of the male and female reproductive system (Uchida et al. Citation2013). Glycodelin is frequently referred to as placental protein 14 because of its initial discovery in amniotic fluid (Krivonosov et al. Citation1979). In humans, glycodelin is present in four glycoforms (glycodelin-A, -S, -F, -C), which are characterized by different functions, including various interactions with spermatozoa, endometrial epithelial cell adhesion toward embryos, and suppression of natural killer cytotoxicity (Uchida et al. Citation2013). The expression of glycodelin in endometrial tissue was found to be significantly upregulated during the peak estradiol and progesterone levels timed to luteinizing hormone (LH) surge – this exact timeframe is considered to be ‘the window of implantation’ by many researchers (Kao et al. Citation2002a).
Based on this evidence, Bentin-Ley et al. (Citation2011) conducted a study (n = 96) to evaluate glycodelin as a biomarker for ER. Endometrial washing samples were obtained on days 1 and 7 after the LH surge (LH+1 and LH+7). At LH+7, no statistically significant difference was observed between the fertile and infertile groups, in contrast to LH+1, where glycodelin levels were found to be decreased in fertile patients. ROC analysis revealed poor accuracy of this biomarker at LH+1 to predict successful IVF outcomes (AUC of 0.58).
Multiple endometrial fluid biomarkers studies
Studies that sought to analyze multiple biomarkers for ER in endometrial secretion are summarized in . In these studies, uterine flushing was not the only option to acquire biomaterial owing to the emergence of a new approach – the direct aspiration of small volumes of endometrial fluid. All researchers in this section collected samples on the day of ET prior to the procedure. The development of bead-based multiplex immunoassays enabled the simultaneous quantification of up to 100 analytes in a single 50 µL sample (Houser Citation2012). This technology was expected to be prevalent in multiple endometrial fluid biomarkers studies, but was only implemented once (Boomsma et al. Citation2009).
Table 2. Summary of studies that sought to analyze multiple endometrial fluid biomarkers.
Cytokines
In the past 20 years, a plethora of global gene expression studies have been conducted to describe the variability of the transcriptome at different points of the menstrual cycle (Kao et al. Citation2002a; Laird et al. Citation2006; Wang et al. Citation2020). For all the discussed cytokines, there is growing evidence of up-/downregulation either at the implantation window or in fertile/infertile patients. It is well known that cytokines guide all steps of implantation and embryo-endometrial crosstalk (Massimiani et al. Citation2020). Therefore, the reasons for the selection of various cytokines as ER biomarkers are clear.
The first study that evaluated the ability of more than one biomarker to reflect the ER was conducted by Lédée-Bataille et al. (Citation2002) (n = 33). Leukemia inhibitory factor (LIF) and tumor necrosis factor alpha (TNF-α) were the analytical targets of choice. Their levels were evaluated in endometrial washings of patients undergoing either IVF or IUI. Statistically significant differences were only found for LIF concentrations, which differed between pregnant (0 [range: 0–177] pg/mL) and non-pregnant (203 [range: 0–1620] pg/mL) patients (p < 0.01). These results were surprising as the researchers initially expected contrary outcomes due to the results obtained by Laird et al. (Citation1997), who demonstrated that LIF levels were higher in fertile patients than in infertile patients. Unfortunately, no data regarding the prognostic value of LIF as a biomarker of successful IVF or IUI outcomes are available.
The next study, carried out by Boomsma Carolien et al. (Citation2009) (n = 210), is remarkable because it is the only study that utilized the above-mentioned bead-based multiplex immunoassay, which allowed the simultaneous determination of 17 cytokines. Endometrial fluid was directly aspirated in a volume of 1–4 µL from patients receiving IVF/ISCI treatment. The multiplex immunoassay included: IL-1β, IL-5 (excluded from the analysis due to low detection rate), IL-6, IL-10, IL-12, IL-15, IL-17, IL-18, TNF-α, interferon γ (IFN-γ, was undetectable in all samples), macrophage migration inhibitory factor, eotaxin, IFN-γ-inducible 10 kDa protein (IP-10), monocyte chemoattractant protein-1 (MCP-1), Dickkopf homolog 1, heparin-binding epidermal growth factor (excluded from the analysis due to low detection rate), and vascular endothelial growth factor (VEGF). The results of multivariable logistic regression revealed statistically significant associations between MCP-1 (p < 0.01) and IP-10 (p < 0.05) with implantation, and associations between IL-1β (p < 0.05) and TNF-α (p < 0.05) with clinical pregnancy. The AUC for IL-1β and TNF-α as biomarkers for clinical pregnancy was suboptimal (0.61), and inclusion of embryo quality slightly improved the model (AUC of 0.67).
Two small-scale studies that aimed to prove the results obtained by Boomsma Carolien et al. (Citation2009) were conducted by Rahiminejad et al. (Citation2015) and Khadem et al. (Citation2019). Both studies included patients undergoing IVF, although Khadem et al. (Citation2019) (n = 76) analyzed IL-1β and TNF-α in endometrial washings, while Rahiminejad et al. (Citation2015) (n = 50) used the direct aspiration method to assess the levels of IL-1β, TNF-α, IP-10, and MCP-1. In contrast to Boomsma Carolien et al. (Citation2009), who used a multivariable approach, in both studies, the predictive power of the biomarkers was evaluated individually in a direct comparison of the pregnant and non-pregnant groups. Thus, for further evaluation of the data, it is important to mention that Boomsma Carolien et al. (Citation2009) did not observe any statistically significant differences for individual biomarkers, but found statistically significant differences for certain combinations.
Rahiminejad et al. (Citation2015) demonstrated that TNF-α levels were decreased in the pregnancy group (p < 0.01), which contradicts the results of Khadem et al. (Citation2019). Neither study demonstrated the ability of IL-1β in endometrial fluid to predict clinical pregnancy. Finally, IP-10 and MCP-1, which were the subjects of only one study, were indicative of clinical pregnancy (p < 0.01).
This discrepancy in the results of the three multiple biomarkers studies originates from methodological heterogeneity. Currently, it is difficult to evaluate the real prognostic value of TNF-α, IL-1β, IP-10, and MCP-1 in endometrial fluid, and an additional investigation is needed.
Biomarkers of oxidative stress
Oxidative stress is defined as an imbalance between free radicals and antioxidants in a certain environment (Jones Citation2008). The role of oxidative stress in various pathological conditions is mostly clear, but is still extensively investigated (Sies Citation2015). Endometrial cells are also subject to reactive oxygen species-mediated damage, which may be part of the ER reduction process (Wojsiat et al. Citation2017). Rahiminejad et al. (Citation2016) (n = 100) conducted a study to evaluate common oxidative stress biomarkers in endometrial fluid by means of direct aspiration on the day of ET in patients undergoing IVF/ICSI. The activities of superoxide dismutase (SOD), catalase (CAT), lipid peroxidation (LPO), total thiol groups (TTG), and total antioxidant power (TAP) were analyzed. Statistically significant differences were found in the levels of SOD, LPO, CAT, and TAP in pregnant and non-pregnant patients (p < 0.01). ROC analysis revealed descent predictive power for CAT, TAP, and SOD individual levels (AUC of 0.766, 0.788, and 0.711, respectively). Unfortunately, the data are limited, and a multifactor model simultaneously including CAT, TAP, and SOD levels was not developed. Nevertheless, this study clearly demonstrated that oxidative stress biomarkers have great potential for the prediction of clinical pregnancy. Their inclusion in future multiple biomarkers studies is desirable.
Endometrial fluid sampling
According to the methodologies of the discussed studies, there are two main approaches to endometrial fluid collection: flushing and direct aspiration. Cheong et al. (Citation2013) argued that washings are inferior to direct aspirates due to increased dilution and leakage of flushing solution through fallopian tubes, which makes it difficult to compare the extent of dilution in different samples. The statement regarding increased sample dilution is undeniable, as the mean volume of saline water, which was used to obtain endometrial washings in the previously discussed articles, was 2 mL. However, for direct aspiration, the samples were diluted with only 70 µL of physiological solution. Nevertheless, the extent of dilution is difficult to compare in both sampling methods, as the direct aspirates, according to Boomsma Carolien et al. (Citation2009), are characterized by inconsistent volumes of 1–4 µL. Thus, samples required normalization by the total protein level, which was successfully implemented by the researchers. Of note, normalization was not used in any of the studies that utilized endometrial flushing.
Indeed, extensive dilution may force the researchers to work with low target concentrations, neighboring the immunoassay limit of detection, which was the case in all the discussed studies. Information about the linearity of the immunoassays used in the lowest concentration range was questionable and has not been presented in most studies.
Thereby, it is interesting to compare the results of IL-18 quantification in endometrial washings and direct aspirates. Lédée-Bataille et al. (Citation2004) reported that only 38/133 patients had detectable levels of this cytokine in endometrial washings and that the difference in pregnancy rates between the IL-18 positive and negative groups was statistically significant (p < 0.05). In contrast, Boomsma Carolien et al. (Citation2009) demonstrated the absence of statistical significance, while IL-18 was detectable in all 210 patients. However, in the latter, an analysis technique with a 10-fold lower limit of detection was used; this discrepancy was, by a large margin, a result of an excessive dilution, which was more than 10-fold higher in cases of uterine flushing.
Other sources of mucosal ER biomarkers
Endometrial fluid is the most obvious source of ER biomarkers; however, it has certain drawbacks, such as low sampling volume and relative invasiveness, as access to the uterine cavity is required. Secretion in other parts of the female reproductive tract may also reflect endometrial status. The available sources of mucosal ER biomarkers are summarized in . Although follicular fluid has been extensively studied regarding ART procedures outcomes, it was not considered to be the subject of the present review. This is because follicular fluid biomarkers predict the outcome of ART procedures via reflection of certain oocyte related characteristics, such as oocyte quantity and quality, fertilization rate, and embryo fragmentation rate, rather than ER (Ciepiela et al. Citation2018; Nagy et al. Citation2019; Yang et al. Citation2020).
Figure 1. Sources of mucosal biomarkers for endometrial receptivity. ET: embryo transfer, IVF: in vitro fertilization.
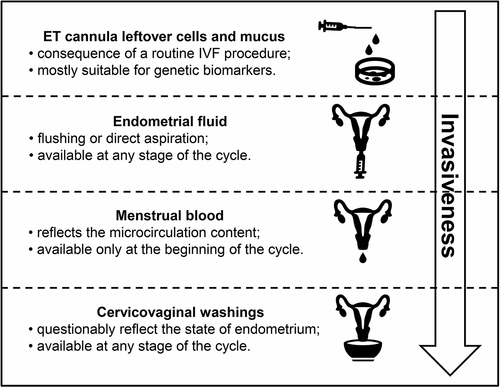
Cervicovaginal washings
Cervicovaginal secretion has a clear advantage over endometrial fluid because its sampling is incomparably easier and less invasive. Notably, cervical secretion has a distinctive cytokine profile compared to endometrial fluid, as shown by Boomsma Carolien et al. (Citation2009) in a within-patient comparison (n = 22). However, these data do not exactly mean that cytokine levels in cervicovaginal washings are not suitable for the analysis of ER. In fact, Zanotta et al. (Citation2019) proved the opposite in a study with 155 women undergoing IVF cycles. A panel of 48 cytokines, chemokines, and growth factors was analyzed using a multiplex bead-based immunoassay in cervicovaginal washings obtained prior to oocyte retrieval. Logistic regression revealed a pattern of four pro-inflammatory immune factors that were significantly increased in patients who achieved clinical pregnancy (IL-12p40, IFN-α, macrophage migration inhibitory factor, and MCP-3, p < 0.001). Data regarding predictive power for successful IVF outcomes of these biomarkers are not available. Nevertheless, these results are of great value as they clearly demonstrate the potential of cervicovaginal secretion as a source of biomarkers for ER. Further large-scale studies of this biomaterial with the inclusion of additional biomarkers are needed.
Embryo transfer cannula leftover cells and mucus
Endometrial epithelial cells at the time of ET might serve as the best sources of information regarding ER in the current IVF cycle. However, it is difficult to imagine an ethics committee that allows traumatizing endometrial biopsy immediately after (or prior to) ET. A sophisticated approach to solve this issue was proposed by Camargo-Díaz et al. (Citation2017) (n = 76), who used the ET cannula leftover cells and mucus as a biomaterial for ER assessment. Hematoxylin-eosin staining with microscopic evaluation confirmed that cells that were attached to the ET cannula were mostly of endometrial origin. Total RNA was isolated from a mixture of endometrial epithelial cells and mucus. The levels of mucin 1 (MUC-1), homeobox A10 (HOXA-10), LIF, and colony stimulating factor-1 (CSF-1) RNA molecules were selected as ER biomarkers because of their altered expression profiles in infertile patients (Othman et al. Citation2012). LIF and CSF-1 were significantly upregulated in pregnant patients (p < 0.05). The increased LIF expression contradicts the previously discussed results of Lédée-Bataille et al. (Citation2002), who observed a significant decrease in LIF levels in the endometrial fluid of pregnant patients and confirmed the results of Laird et al. (Citation1997), who compared fertile and infertile women. Thus, the ability of LIF to reflect the ER remains unclear and requires further investigation.
Menstrual blood
Menstrual blood has been extensively studied as a source of stem cells, which has been applied in regenerative medicine, although the use of this biological fluid as a substrate for diagnostic manipulations is limited (Bozorgmehr et al. Citation2020). Menstrual blood has certain advantages over endometrial fluid. This biomaterial can be easily and painlessly collected directly from the uterine cavity as the cervix is slightly dilated during menstruation. Its main drawback is obviously the fact that it can be sampled only in a certain short window of the menstrual cycle, which is quite delayed from the day of implantation.
Currently, menstrual blood is not used for ER evaluation; however, Tortorella et al. (Citation2014) successfully attempted to detect three common pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) in this biomaterial of 64 infertile patients to develop a new noninvasive approach to chronic endometritis (CE) diagnosis (Tortorella et al. Citation2014). It is known that CE is prevalent in patients with infertility, repeated implantation failure, and recurrent miscarriage, although the clinical significance of this condition is still debated (Cicinelli et al. Citation2015). A recent meta-analysis evaluating the impact of antibiotic treatment of CE on IVF outcomes concluded that histological confirmation of CE cure leads to increased pregnancy and live-birth rates compared to cases of persistent CE (Vitagliano et al. Citation2018).
Tortorella et al. (Citation2014) demonstrated that all three pro-inflammatory cytokines were significantly increased in women with histologically proven CE (p < 0.001). ROC analysis revealed that the IL-6/TNF-α-based model is an efficient predictor of CE with a sensitivity of 91.7% and specificity of 96.4% (AUC of 0.989). According to these results, menstrual blood is a promising biological substrate for the analysis of various biomarkers, although its suitability for the evaluation of ER is a subject for future studies.
Safety of endometrial sampling
Mainly among clinicians, the endometrium of women undergoing the IVF cycle is considered an extremely fragile system; thus, any attempt to obtain its biomaterial (especially close to the day of implantation) will be declared undesirable. Although the previously discussed endometrial fluid aspiration is hardly an invasive procedure, its non-disruptiveness for implantation must be demonstrated. van der Gaast et al. (Citation2003) and Boomsma Carolien et al. (Citation2009) conducted studies to prove the safety of direct endometrial fluid aspiration prior to ET using the same method. They compared 276 women undergoing the above-mentioned sampling with 276 matching controls; there was no statistically significant difference in pregnancy rates between the groups. Therefore, endometrial fluid has certain advantages over endometrial biopsy, namely it is less invasive and safe to collect even on the day of ET. However, it is clearly less suitable for genetic and histological evaluation because of the low content of the desired targets.
Evidently, no studies that performed the risk assessment of endometrial biopsy on the day of ET are available; however, certain studies have evaluated the safety of this procedure in the preceding ovarian stimulation cycle and found that endometrial injury, as a result of biopsy, might be beneficial for further implantation (Barash et al. Citation2003). Further small-scale studies and reviews indicated that endometrial scratching indeed improves the outcomes of ART procedures, and clinicians began to implement this intervention with quite limited scientific data on the underlying mechanism and real clinical benefit, which might be obtained only as a result of large randomized controlled trials (Lensen et al. Citation2016). The biological mechanism remained purely hypothetical, and it was thought that endometrial injury induces certain regenerative processes that lead to positive changes in cytokine balance and alterations in tissue thickness (Li and Hao Citation2009).
Following the completion of proper randomized controlled trials and respective meta-analyses years later, it was concluded that overall endometrial scratching had either no effect on clinical pregnancy and live-birth rates, or a slightly positive effect in the case of a certain subgroup analysis; thus, it is not advised for practical use until further evidence is available (van Hoogenhuijze et al. Citation2019; Sar-Shalom Nahshon et al. Citation2019). For ER biomarkers studies, these data indicate that endometrial biopsy in the cycle preceding IVF is a rather safe intervention. However, the main disadvantage is that this process is spaced in time from the moment of actual implantation.
Discussion
Healthy embryo-endometrial crosstalk is vital for a successful pregnancy. It is mediated by various complex molecular interactions that occur throughout gestation. However, this begins with implantation, and the goal of ART specialists is to guide a patient to this stage. All other steps of embryo-endometrial crosstalk are left to further echelons of obstetric care. Successful implantation requires proper ER and endometrial selectivity. Endometrial selectivity is the ability of the endometrium to distinguish low quality embryos (Macklon and Brosens Citation2014). In clinical practice, certain tools are available to filter embryos and meet the criteria raised by the endometrium, such as the previously mentioned morphological and genetic preimplantation testing. Currently, there is no option not only to reliably increase ER, but also to measure it.
Thus, the main issue in the field of ER research is the lack of a gold standard for its evaluation. Currently, the clinical pregnancy rate is the only suitable option to confirm the presence of receptive endometrium. Unfortunately, the outcome of implantation is influenced by several other factors, including embryo quality, uterine structural defects, maternal systemic conditions, and various environmental factors. Therefore, the use of clinical pregnancy as a reference to the presence of a receptive endometrium will never allow mucosal biomarker-based tests to achieve a sensitivity of 100%. Although it is theoretically possible to build a comprehensive prognostic model that includes not only biomarker-related data, but all possible embryo characteristics, results of hysteroscopy, and demographic/health-related data, it is still impossible to account for all other factors. The cumulative pregnancy rate calculated in a series of ART cycles might reduce their influence. However, this approach complicates the study design, and patient adherence will become a major concern.
Although a great variety of mucosal biomarkers for ER has been studied, data regarding their clinical potential are limited. Owing to remarkable heterogeneity in the methodology and the selected biomarkers, it is impossible to conduct a meta-analysis.
The levels of various molecular agents and oxidative stress influence the ER; however, their interactions are complex. Owing to the simplicity of statistical analysis in most discussed studies, certain promising biomarkers were compared individually among the studied groups and no attempts were made to develop a complex multifactor model.
A multivariable approach, which might reflect the relationship between certain analytical targets, rather than individual comparisons, is the ideal path for ER biomarkers studies. Modern laboratory analysis markets provide various options for biomarker screening. In addition, premade 48-target multiplex immunoassays are commercially available. However, these immunoassays were underused in the discussed studies.
Among the mentioned molecular biomarkers, urocortin and activin A demonstrated the best prognostic value, with an overall accuracy of 79%–88% (Florio et al. Citation2008, Citation2010). Interestingly, both molecules were completely abandoned in subsequent studies. It is advisable to incorporate these molecules into future multiple biomarkers panels to further validate the promising results obtained previously.
Therefore, the design of a future ideal ER study must include simultaneous analysis of multiple biomarkers, preferably with an assessment of oxidative stress level (which also seems promising), multivariable approach to the statistical analysis, and an attempt to create a complex prognostic model. A topic beyond the scope of the present review is the influence of endometrial microbiota and immunological balance on ER. There is growing evidence that uterine bacteria play an important role in the modulation of immune cell subsets in the endometrium (Benner et al. Citation2018). Recently, it was reported that endometrium and ovulatory cervical mucus of patients with recurrent pregnancy loss are characterized by an increased density of natural killer cells CD56+ and CD16+, indicating a possible autoimmune component of infertility (Ulčová-Gallová et al. Citation2019). Menstrual blood might be also used as a source of endometrial lymphocytes, as it is known to have a clearly distinct immune microenvironment compared to peripheral blood (van der Molen et al. Citation2014). Thus, the results of microbiological and immunological assessment of endometrium-related biomaterial may reveal potential prognostic factors.
Various sample collection timings were employed in these studies. Intuitively, the patient’s biomaterial was the most predictive of ART treatment outcome on the day of ET. Accordingly, this day was the sampling timing of choice in most cases. However, from a purely practical point of view, this timing is useless for predicting the implantation outcome, as all clinical manipulations would have been carried out by the time the results arrive from the laboratory. Currently, there are insufficient data regarding the consistency of ER biomarkers levels in the following menstrual cycles. From a clinical perspective, the optimal implantation outcome prediction timing should be either at the beginning of the current cycle or in the previous cycle, assuming it is proven that a certain biomarker is consistent across menstrual cycles. Therefore, studies evaluating the consistency of ER biomarkers levels across different days of the cycle as well as in consecutive cycles are needed.
Conclusions
A receptive endometrium is crucial for successful implantation. The discovery of a reliable approach to measure ER may substantially promote an investigation of the influence of various internal and external factors on the likelihood of pregnancy, in addition to its prognostic value in ART procedures. Currently, our knowledge about the molecular mechanisms underlying impaired ER is limited. The studies reviewed had methodological heterogeneity and inconsistent results. Nevertheless, based on the data discussed, mucosal biomarkers can reflect the state of the endometrium and the noninvasiveness of their analysis makes them a remarkable alternative to endometrial biopsy. The challenge of studying ER is that it is influenced by various cross-disciplinary factors. Comprehensive research requires cooperation from specialists in reproductive medicine, endocrinology, immunology, and possibly microbiology. Therefore, future large-scale studies of mucosal ER biomarkers in endometrial fluid, menstrual blood, ET cannula leftover cells, and cervicovaginal washings are anticipated.
Ethics approval
Not applicable.
Authors’ contributions
Conceptualization: MJ, OP; methodology: EM, MJ; software: MJ; validation: OP; formal analysis: OP; investigation: EM, MJ; resources: LS; data curation: LS, OP; writing-original draft preparation: MJ; writing-review and editing: EM, OP; visualization: MJ; supervision: LS; project administration: LS; funding acquisition: LS. All authors have read and agreed to the published version of the manuscript.
Disclosure statement
The authors report no conflict of interest.
Additional information
Funding
References
- Baltaci V, Satiroglu H, Kabukçu C, Ünsal E, Aydinuraz B, Ö Ü, Aktas Y, Çetinkaya E, Turhan F, Aktan A. 2006. Relationship between embryo quality and aneuploidies. Reprod Biomed Online. 12(1):77–82. doi:https://doi.org/10.1016/S1472-6483(10)60984-4.
- Barash A, Dekel N, Fieldust S, Segal I, Schechtman E, Granot I. 2003. Local injury to the endometrium doubles the incidence of successful pregnancies in patients undergoing in vitro fertilization. Fertil Steril. 79(6):1317–1322. doi:https://doi.org/10.1016/S0015-0282(03)00345-5.
- Benner M, Ferwerda G, Joosten I, van der Molen RG. 2018. How uterine microbiota might be responsible for a receptive, fertile endometrium. Hum Reprod Update. 24(4):393–415. doi:https://doi.org/10.1093/humupd/dmy012.
- Bentin-Ley U, Lindhard A, Ravn V, Islin H, Sørensen S. 2011. Glycodelin in endometrial flushing fluid and endometrial biopsies from infertile and fertile women. Eur J Obstet Gynecol Reprod Biol. 156(1):60–66. doi:https://doi.org/10.1016/j.ejogrb.2010.12.040.
- Bloise E, Ciarmela P, Dela Cruz C, Luisi S, Petraglia F, Reis FM. 2019. Activin A in mammalian physiology. Physiol Rev. 99(1):739–780. doi:https://doi.org/10.1152/physrev.00002.2018.
- Boomsma Carolien M, Kavelaars A, Eijkemans MJC, Amarouchi K, Teklenburg G, Gutknecht D, Fauser BJCM, Heijnen CJ, Macklon NS. 2009. Cytokine profiling in endometrial secretions: a non-invasive window on endometrial receptivity. Reprod Biomed Online. 18(1):85–94. doi:https://doi.org/10.1016/S1472-6483(10)60429-4.
- Boomsma CM, Kavelaars A, Eijkemans MJC, Lentjes EG, Fauser BCJM, Heijnen CJ, MacKlon NS. 2009. Endometrial secretion analysis identifies a cytokine profile predictive of pregnancy in IVF. Hum Reprod. 24(6):1427–1435. doi:https://doi.org/10.1093/humrep/dep011.
- Bozorgmehr M, Gurung S, Darzi S, Nikoo S, Kazemnejad S, Zarnani AH, Gargett CE. 2020. Endometrial and Menstrual Blood Mesenchymal Stem/Stromal Cells: biological Properties and Clinical Application. Front Cell Dev Biol. 8. https://pubmed.ncbi.nlm.nih.gov/32742977/.
- Camargo-Díaz F, García V, Ocampo-Bárcenas A, González-Marquez H, López-Bayghen E. 2017. Colony stimulating factor-1 and leukemia inhibitor factor expression from current-cycle cannula isolated endometrial cells are associated with increased endometrial receptivity and pregnancy. BMC Womens Health. 17(1). doi:https://doi.org/10.1186/s12905-017-0418-7.
- Cheong Y, Boomsma C, Heijnen C, MacKlon N. 2013. Uterine secretomics: a window on the maternal-embryo interface. Fertil Steril. 99(4):1093–1099. doi:https://doi.org/10.1016/j.fertnstert.2013.01.144.
- Cicinelli E, Matteo M, Tinelli R, Lepera A, Alfonso R, Indraccolo U, Marrocchella S, Greco P, Resta L. 2015. Prevalence of chronic endometritis in repeated unexplained implantation failure and the IVF success rate after antibiotic therapy. Hum Reprod. 30(2):323–330. doi:https://doi.org/10.1093/humrep/deu292.
- Ciepiela P, Dulęba AJ, Kowaleczko E, Chełstowski K, Kurzawa R. 2018. Vitamin D as a follicular marker of human oocyte quality and a serum marker of in vitro fertilization outcome. J Assist Reprod Genet. 35(7):1265–1276. doi:https://doi.org/10.1007/s10815-018-1179-4.
- Craciunas L, Gallos I, Chu J, Bourne T, Quenby S, Brosens JJ, Coomarasamy A. 2019. Conventional and modern markers of endometrial receptivity: a systematic review and meta-analysis. Hum Reprod Update. 25(2):202–223. doi:https://doi.org/10.1093/humupd/dmy044.
- De Mouzon J, Chambers GM, Zegers-Hochschild F, Mansour R, Ishihara O, Banker M, Dyer S, Kupka M, David Adamson G. 2020. International committee for monitoring assisted reproductive technologies world report: assisted reproductive technology 2012. Hum Reprod. 35(8):1900–1913. doi:https://doi.org/10.1093/humrep/deaa090.
- Duffy JMN, Bhattacharya S, Bofill M, Collura B, Curtis C, Evers JLH, Giudice LC, Farquharson RG, Franik S, Hickey M, et al. 2020. Standardizing definitions and reporting guidelines for the infertility core outcome set: an international consensus development study. Hum Reprod. 35(12):2735–2745. doi:https://doi.org/10.1093/humrep/deaa243
- Florio P, Arcuri F, Ciarmela P, Runci Y, Romagnoli R, Cintorino M, Di Blasio AM, Petraglia F. 2002. Identification of urocortin mRNA and peptide in the human endometrium. J Endocrinol. 173(2). doi:https://doi.org/10.1677/joe.0.173r009.
- Florio P, Bruni L, De Falco C, Filardi G, Torricelli M, Reis FM, Galleri L, Voltolini C, Bocchi C, De Leo V, et al. 2008. Evaluation of endometrial urocortin secretion for prediction of pregnancy after intrauterine insemination. Clin Chem. 54(2):350–355. doi:https://doi.org/10.1373/clinchem.2007.094987.
- Florio P, Bruni L, Galleri L, Reis FM, Borges LE, Bocchi C, Litta P, De Leo V, Petraglia F. 2010. Evaluation of endometrial activin A secretion for prediction of pregnancy after intrauterine insemination. Fertil Steril. 93(7):2316–2320. doi:https://doi.org/10.1016/j.fertnstert.2008.12.125.
- Florio P, Severi FM, Luisi S, Ciarmela P, Calonaci G, Cobellis L, Petraglia F. 2003. Endometrial expression and secretion of activin A, but not follistatin, increase in the secretory phase of the menstrual cycle. J Soc Gynecol Investig. 10(4):237–243. doi:https://doi.org/10.1016/S1071-5576(03)00045-5.
- Gdanska P, Drozdowicz-Jastrzebska E, Grzechocinska B, Radziwon-Zaleska M, Wegrzyn P, Wielgos M. 2017. Anxiety and depression in women undergoing infertility treatment. Ginekol Pol. 88(2):109–112. doi:https://doi.org/10.5603/GP.a2017.0019.
- Gillott DJ, Al-Rumaih HM, Leung KY, Eldib A, Grudzinskas JG. 2008. Specific isoforms of leucine-rich α2-glycoprotein detected in the proliferative endometrium of women undergoing assisted reproduction are associated with spontaneous pregnancy. Fertil Steril. 90(3):761–768. doi:https://doi.org/10.1016/j.fertnstert.2007.01.094.
- Golan A, Halperin R, Herman A, Hadas E, Soffer Y, Bukovsky I, Caspi E, Ron-El R. 1994. Human decidua-associated protein 200 levels in uterine fluid at hysteroscopy. Gynecol Obstet Invest. 38(4):217–219. doi:https://doi.org/10.1159/000292484.
- Halperin R, Hadas E, Bukovsky I, Schneider D. 1998. Measurement of intrauterine human decidua-associated protein 200 and diagnosis of ectopic pregnancy. Gynecol Obstet Invest. 45(4):225–228. doi:https://doi.org/10.1159/000009972.
- Halperin R, Hadas E, Fleminger G, Ovadia Y, Kraicer PF. 1990. Identification, immunoaffinity purification and partial characterization of a human decidua-associated protein. J Reprod Fertil. 88(1):159–165. doi:https://doi.org/10.1530/jrf.0.0880159.
- Halperin R, Ron-el R, Golan A, Hadas E, Schneider D, Bukovsky I, Herman A. 1995. Implantation: uterine fluid human decidua-associated protein 200 and implantation after embryo transfer. Hum Reprod. 10(4):907–910. doi:https://doi.org/10.1093/oxfordjournals.humrep.a136059.
- Houser B. 2012. Bio-rad’s Bio-Plex® suspension array system, xMAP technology overview. Arch Physiol Biochem. 118(4):192–196. doi:https://doi.org/10.3109/13813455.2012.705301.
- Ida A, Tsuji Y, Muranaka J, Kanazawa R, Nakata Y, Adachi S, Okamura H, Koyama K. 2000. IL-18 in pregnancy; the elevation of IL-18 in maternal peripheral blood during labour and complicated pregnancies. J Reprod Immunol. 47(1):65–74. doi:https://doi.org/10.1016/S0165-0378(00)00058-9.
- Jin XY, Zhao LJ, Luo DH, Liu L, Dai YD, Hu XX, Wang YY, Lin X, Hong F, Li TC, et al. 2017. Pinopode score around the time of implantation is predictive of successful implantation following frozen embryo transfer in hormone replacement cycles. Hum Reprod. 32(12):2394–2403. doi:https://doi.org/10.1093/humrep/dex312.
- Jinno M, Ozaki T, Iwashita M, Nakamura Y, Kudo A, Hirano H. 2001. Measurement of endometrial tissue blood flow: a novel way to assess uterine receptivity for implantation. Fertil Steril. 76(6):1168–1174. doi:https://doi.org/10.1016/S0015-0282(01)02897-7.
- Jones DP. 2008. Radical-free biology of oxidative stress. Am J Physiol - Cell Physiol. 295(4). doi:https://doi.org/10.1152/ajpcell.00283.2008.
- Jones RL, Salamonsen LA, Critchley HOD, Rogers PAW, Affandi B, Findlay JK. 2000. Inhibin and activin subunits are differentially expressed in endometrial cells and leukocytes during the menstrual cycle, in early pregnancy and in women using progestin-only contraception. Mol Hum Reprod. 6(12):1107–1117. doi:https://doi.org/10.1093/molehr/6.12.1107.
- Kao LC, Tulac S, Lobo S, Imani B, Yang JP, Germeyer A, Osteen K, Taylor RN, Lessey BA, Giudice LC. 2002a. Global gene profiling in human endometrium during the window of implantation. Endocrinology. 143(6):2119–2138.
- Khadem N, Mansoori M, Attaran M, Attaranzadeh A, Zohdi E. 2019. Association of il-1 and tnf-α levels in endometrial secretion and success of embryo transfer in ivf/icsi cycles. Int J Fertil Steril. 13(3):236–239.
- Krivonosov SK, Mikhailov AT, Tatarinov IS. 1979. Comparative immunochemical analysis of specific beta globulins of the “pregnancy zone” of the humans and other mammals. Byulleten Eksp Biol I Meditsiny. 88(7):51–53.
- Laird SM, Tuckerman EM, Dalton CF, Dunphy BC, Li TC, Zhang X. 1997. The production of leukaemia inhibitory factor by human endometrium: presence in uterine flushings and production by cells in culture. Hum Reprod. 12(3):569–574. doi:https://doi.org/10.1093/humrep/12.3.569.
- Laird SM, Tuckerman EM, Li TC. 2006. Cytokine expression in the endometrium of women with implantation failure and recurrent miscarriage. Reprod Biomed Online. 13(1):13–23. doi:https://doi.org/10.1016/S1472-6483(10)62011-1.
- Lédée-Bataille N, Laprée-Delage G, Taupin JL, Dubanchet S, Frydman R, Chaouat G. 2002. Concentration of leukaemia inhibitory factor (LIF) in uterine flushing fluid is highly predictive of embryo implantation. Hum Reprod. 17(1):213–218. doi:https://doi.org/10.1093/humrep/17.1.213.
- Lédée-Bataille N, Olivennes F, Kadoch J, Dubanchet S, Frydman N, Chaouat G, Frydman R. 2004. Detectable levels of interleukin-18 in uterine luminal secretions at oocyte retrieval predict failure of the embryo transfer. Hum Reprod. 19(9):1968–1973. doi:https://doi.org/10.1093/humrep/deh356.
- Lensen S, Sadler L, Farquhar C. 2016. Endometrial scratching for subfertility: everyone’s doing it. Hum Reprod. 31(6):1241–1244. doi:https://doi.org/10.1093/humrep/dew053.
- Lessey BA, Young SL. 2019. What exactly is endometrial receptivity? Fertil Steril [Internet]. 111(4):611–617. doi:https://doi.org/10.1016/j.fertnstert.2019.02.009.
- Li R, Hao G. 2009. Local injury to the endometrium: its effect on implantation. Curr Opin Obstet Gynecol. 21(3):236–239. doi:https://doi.org/10.1097/GCO.0b013e32832a0654.
- Liu C, Teo MHY, Pek SLT, Wu X, Leong ML, Tay HM, Hou HW, Ruedl C, Moss SE, Greenwood J, et al. 2020. A multifunctional role of leucine-rich a-2-glycoprotein 1 in cutaneous wound healing under normal and diabetic conditions. Diabetes. 69(11):2467–2480. doi:https://doi.org/10.2337/db20-0585.
- Macklon NS, Brosens JJ. 2014. The human endometrium as a sensor of embryo quality. Biol Reprod. 91(4):98. doi:https://doi.org/10.1095/biolreprod.114.122846.
- Massarotti C, Gentile G, Ferreccio C, Scaruffi P, Remorgida V, Anserini P. 2019. Impact of infertility and infertility treatments on quality of life and levels of anxiety and depression in women undergoing in vitro fertilization. Gynecol Endocrinol. 35(6):485–489. doi:https://doi.org/10.1080/09513590.2018.1540575.
- Massimiani M, Lacconi V, La Civita F, Ticconi C, Rago R, Campagnolo L. 2020. Molecular signaling regulating endometrium–blastocyst crosstalk. Int J Mol Sci. 21:1.
- Nagy B, Poto L, Farkas N, Koppan M, Varnagy A, Kovacs K, Papp S, Bohonyi N, Bodis J. 2019. Follicular fluid progesterone concentration is associated with fertilization outcome after IVF: a systematic review and meta-analysis. Reprod Biomed Online. 38(6):871–882. doi:https://doi.org/10.1016/j.rbmo.2018.12.045.
- Novembri R, Carrarelli P, Toti P, Rocha ALL, Borges LE, Reis FM, Piomboni P, Florio P, Petraglia F. 2011. Urocortin 2 and urocortin 3 inendometriosis: evidence for a possible role in inflammatory response. Mol Hum Reprod. 17(9):587–593. doi:https://doi.org/10.1093/molehr/gar020.
- Oron G, Son WY, Buckett W, Tulandi T, Holzer H. 2014. The association between embryo quality and perinatal outcome of singletons born after single embryo transfers: a pilot study. Hum Reprod. 29(7):1444–1451. doi:https://doi.org/10.1093/humrep/deu079.
- Othman R, Omar MH, Shan LP, Shafiee MN, Jamal R, Mokhtar NM. 2012. Microarray profiling of secretoryphase endometrium from patients with recurrent miscarriage. Reprod Biol. 12(2):183–199. doi:https://doi.org/10.1016/S1642-431X(12)60085-0.
- Popovici RM, Kao L-C, Giudice LC. 2000. Discovery of New Inducible Genes in in vitro Decidualized Human Endometrial Stromal Cells Using Microarray Technology. Endocrinology. 141(9):3510–3515. doi:https://doi.org/10.1210/endo.141.9.7789.
- Practice Committee of the American Society for Reproductive Medicine. 2013. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril. 99(1):63. doi:https://doi.org/10.1016/j.fertnstert.2012.09.023.
- Rahiminejad ME, Moaddab A, Ebrahimi M, Rabiee S, Zamani A, Ezzati M, Shamshirsaz AA. 2015. The relationship between some endometrial secretion cytokines and in vitro fertilization. Iran J Reprod Med. 13(9):557–562.
- Rahiminejad ME, Moaddab A, Ganji M, Eskandari N, Yepez M, Rabiee S, Wise M, Ruano R, Ranjbar A. 2016. Oxidative stress biomarkers in endometrial secretions: a comparison between successful and unsuccessful in vitro fertilization cycles. J Reprod Immunol. 116:70–75. doi:https://doi.org/10.1016/j.jri.2016.05.003.
- Reyes TM, Lewis K, Perrin MH, Kunitake KS, Vaughan J, Arias CA, Hogenesch JB, Gulyas J, Rivier J, Vale WW, et al. 2001. Urocortin II: a member of the corticotropin-releasing factor (CRF) neuropeptide family that is selectively bound by type 2 CRF receptors. Proc Natl Acad Sci U S A. 98(5):2843–2848. doi:https://doi.org/10.1073/pnas.051626398.
- Sakumoto T, Inafuku K, Miyara M, Takamiyagi N, Miyake A, Shinkawa T, Nakayama M. 1992. Hysteroscopic assessment of midsecretory-phase endometrium, with special reference to the luteal-phase defect. Horm Res Paediatr. 37:48–52. doi:https://doi.org/10.1159/000182349
- Santi A, Felser R, Bersinger NA, Wunder DM, McKinnon B, Mueller MD. 2012. The hysteroscopic view of infertility: the mid-secretory endometrium and treatment success towards pregnancy. Gynecol Surg. 9(2):147–150. doi:https://doi.org/10.1007/s10397-011-0687-3.
- Sar-Shalom Nahshon C, Sagi-Dain L, Wiener-Megnazi Z, Dirnfeld M. 2019. The impact of intentional endometrial injury on reproductive outcomes: a systematic review and meta-analysis. Hum Reprod Update. 25(1):95–113. doi:https://doi.org/10.1093/humupd/dmy034.
- Senju H, Kumagai A, Nakamura Y, Yamaguchi H, Nakatomi K, Fukami S, Shiraishi K, Harada Y, Nakamura M, Okamura H, et al. 2018. Effect of IL-18 on the expansion and phenotype of human natural killer cells: application to cancer immunotherapy. Int J Biol Sci. 14(3):331–340. doi:https://doi.org/10.7150/ijbs.22809
- Sies H. 2015. Oxidative stress: a concept in redox biology and medicine. Redox Biol. 4:180–183. doi:https://doi.org/10.1016/j.redox.2015.01.002.
- Smeltz RB, Chen J, Hu-Li J, Shevach EM. 2001. Regulation of interleukin (IL)-18 receptor α chain expression on CD4+ T cells during T helper (Th)1/Th2 differentiation: critical downregulatory role of IL-4. J Exp Med. 194(2):143–153. doi:https://doi.org/10.1084/jem.194.2.143.
- Sun H, Gong TT, Jiang YT, Zhang S, Zhao YH, Wu QJ. 2019. Global, Regional, and National Prevalence and Disability-Adjusted Life-Years for Infertility in 195 Countries and Territories, 1990–2017: results from a Global Burden of Disease Study, 2017. Aging (Albany NY). doi:https://doi.org/10.18632/aging.102497
- Tabibzadeh S, Babaknia A. 1995. Molecular aspects of implantation: the signals and molecular pathways involved in implantation, a symbiotic interaction between blastocyst and endometrium involving adhesion and tissue invasion. Hum Reprod. 10(6):1579–1602. doi:https://doi.org/10.1093/HUMREP/10.6.1579.
- Torricelli M, De Falco G, Florio P, Rossi M, Leucci E, Viganò P, Leoncini L, Petraglia F. 2007. Secretory endometrium highly expresses urocortin messenger RNA and peptide: possible role in the decidualization process. Hum Reprod. 22(1):92–96. doi:https://doi.org/10.1093/humrep/del331.
- Tortorella C, Piazzolla G, Matteo M, Pinto V, Tinelli R, Sabbà C, Fanelli M, Cicinelli E. 2014. Interleukin-6, interleukin-1β, and tumor necrosis factor α in menstrual effluents as biomarkers of chronic endometritis. Fertil Steril. 101(1):242–247. doi:https://doi.org/10.1016/j.fertnstert.2013.09.041.
- Uchida H, Maruyama T, Nishikawa-Uchida S, Miyazaki K, Masuda H, Yoshimura Y. 2013. Glycodelin in reproduction. Reprod Med Biol. 12(3):79–84. doi:https://doi.org/10.1007/s12522-013-0144-2.
- Ulčová-Gallová Z, Mukenšnabl P, Haschová M, Pešek M, Chaloupka P, Lošan P, Bibková K, Mičanová Z, Cibulka J, Švecová M. 2019. NK cells not only in endometrium but also in ovulatory cervical mucus in patients with decreased fertility. Ces Gynekol. 84(3):184–189.
- van der Gaast M, Beier-Hellwig K, Fauser B, Beier H, Macklon N. 2003. Endometrial secretion aspiration prior to embryo transfer does not reduce implantation rates. Reprod Biomed Online. 7:105–109. doi:https://doi.org/10.1016/S1472-6483(10)61737-3
- van der Molen RG, Schutten JHF, van Cranenbroek B, ter Meer M, Donckers J, Scholten RR, van der Heijden OWH, Spaanderman MEA, Joosten I. 2014. Menstrual blood closely resembles the uterine immune micro-environment and is clearly distinct from peripheral blood. Hum Reprod. 29(2):303–314. doi:https://doi.org/10.1093/humrep/det398.
- van Hoogenhuijze NE, Kasius JC, Broekmans FJM, Bosteels J, Torrance HL. 2019. Endometrial scratching prior to IVF; does it help and for whom? A systematic review and meta-analysis. Hum Reprod Open. 2019(1). doi:https://doi.org/10.1093/hropen/hoy025.
- Vitagliano A, Saccardi C, Noventa M, Di Spiezio Sardo A, Saccone G, Cicinelli E, Pizzi S, Andrisani A, Ps L. 2018. Effects of chronic endometritis therapy on in vitro fertilization outcome in women with repeated implantation failure: a systematic review and meta-analysis. Fertil Steril. 110(1):103–112.e1. doi:https://doi.org/10.1016/j.fertnstert.2018.03.017.
- Voa M-F, Rocha AM, Ferreira FP, Bonetti TCS, Serafini P, Motta ELA. 2015. Matrix metalloproteinases 2 and 9 and E-cadherin expression in the endometrium during the implantation window of infertile women before in vitro fertilization treatment. Reprod Sci. 22(4):416–422. doi:https://doi.org/10.1177/1933719114529373.
- Wang W, Vilella F, Alama P, Moreno I, Mignardi M, Isakova A, Pan W, Simon C, Quake SR. 2020. Single-cell transcriptomic atlas of the human endometrium during the menstrual cycle. Nat Med. 26(10):1644–1653. doi:https://doi.org/10.1038/s41591-020-1040-z.
- Wojsiat J, Korczyński J, Borowiecka M, Żbikowska HM. 2017. The role of oxidative stress in female infertility and in vitro fertilization. Postepy Hig Med Dosw. 71:359–366. doi:https://doi.org/10.5604/01.3001.0010.3820.
- Yang J, Yang X, Yang H, Bai Y, Zha H, Jiang F, Meng Y. 2020. Interleukin 6 in follicular fluid reduces embryo fragmentation and improves the clinical pregnancy rate. J Assist Reprod Genet. 37(5):1171–1176. doi:https://doi.org/10.1007/s10815-020-01737-2.
- Yasuda K, Nakanishi K, Tsutsui H. 2019. Interleukin-18 in health and disease. Int J Mol Sci. 20:3. doi:https://doi.org/10.3390/ijms20030649.
- Yoshino O, Osuga Y, Koga K, Tsutsumi O, Yano T, Fujii T, Kugu K, Momoeda M, Fujiwara T, Tomita K, et al. 2001. Evidence for the expression of interleukin (IL)-18, IL-18 receptor and IL-18 binding protein in the human endometrium. Mol Hum Reprod. 7(7):649–654. doi:https://doi.org/10.1093/molehr/7.7.649.
- Zanotta N, Monasta L, Skerk K, Luppi S, Martinelli M, Ricci G, Comar M. 2019. Cervico-vaginal secretion cytokine profile: a non-invasive approach to study the endometrial receptivity in IVF cycles. Am J Reprod Immunol. 81(1). doi:https://doi.org/10.1111/aji.13064.
- Zhu J, Lian Y, Li M, Chen L, Liu P, Qiao J. 2014. Does IVF cleavage stage embryo quality affect pregnancy complications and neonatal outcomes in singleton gestations after double embryo transfers? J Assist Reprod Genet. 31(12):1635–1641. doi:https://doi.org/10.1007/s10815-014-0351-8.
