ABSTRACT
Coronavirus Disease 2019, caused by the virus, Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2), is a pandemic first discovered in Wuhan, China which has claimed over 1.7 million lives to date across the globe as of 24 December 2020. As the virus spreads across the world affecting millions of patients, there has been a massive movement to discover readily available and effective treatment options including vaccines. One of the limiting factors in treating the disease is its varied presentation and effect in patients, ranging from asymptomatic patients to those left in intensive care units, intubated and fighting for their lives. There are numerous clinical trials and small-scale studies underway to investigate potential treatment options. However, very few studies and drugs demonstrated efficacy while many more are under investigation, leaving care teams dependent on supportive care coupled with experimental treatment options. In this review, we summarize the various treatment options explored to treat COVID-19, discussing possible the mechanisms of fighting the virus.
Introduction
Of the seven coronaviruses known to infect humans, majority of them cause a mild upper respiratory disease [Citation1]. Three exceptions to these are severe acute respiratory syndrome coronavirus (SARS-CoV) in 2003, middle east respiratory syndrome coronavirus (MERS) in 2012 and the current SARS CoV-2 virus in 2020. Both the SARS-CoV and MERS outbreak were treated with supportive therapy as the main stay treatment since no clear drug or vaccine emerged as a winner [Citation2,(Citation3]. The novel coronavirus, known as SARS-CoV-2 is very closely related to SARS-CoV with a 79.6% sequence similarity [Citation4]. Because of the sequence similarity, many of the drugs which were investigated during the prior pandemics, re-surfaced as potential treatment options. With the gravity of the outbreak, the R0 being 2.24–3.58 [Citation5,(Citation6], and disease claiming more lives when compared to the prior outbreaks, there has been tremendous push towards developing effective treatment options. It is known that SARS-CoV-2 uses the receptor angiotensin-converting enzyme-2 (ACE2) for entry and employs the serine protease TMPRSS2 for S protein priming [Citation7] (). COVID-19 patients, in their early stages of contracting the illness, present with a decrease in both CD4+ and CD8+ T-cells subsets leading to impairment in the host’s immunity [Citation8]. Thus, the use of anti-viral agents early in the disease course has shown to be beneficial in controlling the severity of COVID-19 [Citation9].
Figure 1. Diaphragm representing various experimental drugs used in the treatment of COVID-19 and their mechanisms of action
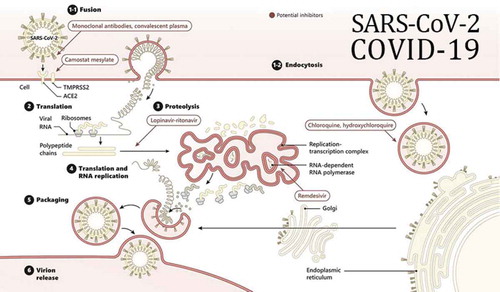
The current management focuses on supportive care in the mild-moderate setting and fluid/ventilation management in the acute respiratory distress phase (ARDS) and extracorporeal membrane oxygenation (ECMO) phases [Citation10,(Citation11]. The role of steroids in this disease has been debated as well. We chose to review many potential medications that have been studied in this regard ().
Treatment options
One of the earlier drugs that were studied during the evolution of this pandemic was Chloroquine/hydroxychloroquine. In this review, we discuss major potential treatments studied until December 2020.
Chloroquine, hydroxychloroquine
The mechanism of action of chloroquine is via inhibition of viral cell entry and cell fusion by increasing endosomal pH. It also impairs the glycosylation of ACE2 receptors which the SARS viruses use for cellular entry [Citation12,(Citation13]. It has shown efficacy in in-vitro studies against SARS-CoV-2, however, failed to demonstrate efficacy as more information was gained during the course of the pandemic [Citation14].
According to Gautret et al. azithromycin was added to hydroxychloroquine for treating 20 patients with severe COVID-19 [Citation15]. The combination therapy showed significant viral load reduction/disappearance [Citation15]. This was followed by Chen et al. where a dose of 400 mg hydroxychloroquine did not lead to nasal clearance in 30 patients [Citation16]. Both of these studies were randomized control trials. However, another study by Molina et al. showed that the combination therapy did not result in viral clearance at 5 or 6 days of treatment [Citation17]. A recent observational study showed that hydroxychloroquine administration was not associated with either a lower or an increased risk for intubation or death [Citation18]. Hydroxychloroquine was also studied for post-exposure prophylaxis and it did not prevent illness after being exposed to confirmed COVID-19 infection when given after 4 days of exposure [Citation19].
Based on a myriad of data proving that these medications did not fasten viral clearance or symptom improval, there has been a change of practice to not include them for routine COVID-19 treatment.
Remdesivir (RDV)
Remdesivir has shown some promise in the treatment of COVID-19. This drug targets viral RNA-dependent RNA polymerase leading to premature termination of viral RNA transcription [Citation20,(Citation21]. Remdesivir has been shown to inhibit the activity of Orthocoronavirinae (such as pathogenic SARS-CoV and MERS) and has shown some efficacy in treating SARS-CoV-2 in vitro [Citation13,Citation22,Citation23]. Since there is sequence of similarities between SARS-CoV and SARS-CoV-2, this was trialled in patients with COVID-19.
This drug was first utilized in the USA in January 2020 in the state of Washington, where the very first COVID-19 patient was treated with it on day 7 for pneumonia [Citation24]. The patient cleared the virus from the respiratory tract by day 12. In a study containing 237 patients by Wang et al. in April 2020, patients admitted to hospital for severe COVID-19 who were treated with remdesivir did not have statistically significant clinical benefits when compared to placebo [Citation25]. The primary end-point in this study was time to recovery within 28 days. However, it was important to note that the patients who received remdesivir showed faster time to clinical improvement. Remdesivir did not show any statistically significant improvement whether treated for 5 or 10 days duration in patients with severe COVID-19 who were not mechanically ventilated [Citation26].
Compassionate use of remdesivir in severe COVID-19 patients led to clinical improvement in 68% patients which led to the utilization in multiple health care facilities all across the world [Citation27]. For the first time, a randomized-controlled trial in 1063 patients revealed positive results in terms of shortening time to recovery in adult patients hospitalized with COVID-19 [Citation28]. Based on this data, the Food and Drug Administration has approved remdesivir for use in hospitalized patients with COVID-19 and the most benefit may be observed in time to recovery, with remdesivir shortening the disease course.
More recently, remdesivir was one of the medications studied among three other to look at in-hospital mortality in a multi-country randomized controlled trial (Solidarity trial). In that study, remdesivir did not show any in-hospital mortality benefit [Citation29]. This study led to changing recommendations from World Health Organization (WHO) recommending against the use of remdesivir in hospitalized patients.
Favipiravir
Favipiravir inhibits the RNA polymerase activity of the viral genome of RNA viruses leading to chain termination and chain elongation [Citation30]. Favipiravir has shown in-vitro viral inhibition activity against SARS-CoV-2 which resulted in it being a potential treatment option [Citation13].
Favipiravir was compared with lopinavir/ritonavir (LPV/RTV) for treatment and in comparison to LPV/RTV, favipiravir led to faster viral clearance times and significant improvement rate in chest imaging [Citation31]. A prospective, multicenter, open-label, randomized trial in China comparing favipiravir for COVID-19 with umifenovir (Arbidol) which is used against influenza viruses in 240 patients, was recently reported. Preliminary results have shown that although favipivar improved time to relief for fever and cough, it did not improve overall clinical recovery rate when compared to Arbidiol [Citation32]. The overall consensus is that there are no definite results to use this drug in routine care of COVID-19 patients.
Additionally, favipiravir is being studied in clinical trials in combination with interferon-α (ChiCTR2000029600) [Citation33] or with baloxavir marboxil (ChiCTR2000029544) [Citation34] in China. Phase 3 clinical trial is currently underway in India.
Interferons
Interferon β (IFNb)-1b was a treatment option during the MERS and SARS-CoV outbreaks [Citation35,(Citation36]. This drug displayed in-vitro susceptibility and SARS-CoV-2 was found to be more sensitive to IFN-I treatment when compared to the SARS-CoV, making this potential option [Citation37]. ACTT-3 is a phase 3 randomized-controlled clinical trial looking at outcomes with combination of interferon beta-1a and remdesivir compared to remdesivir alone in 1038 patients which is ongoing [Citation38]. Although not peer-reviewed yet, IFNb was studied to increase discharge rate at 14 days and decreased mortality rate at 28 days in a study from Iran (IRCT20100228003449N28) [Citation39]. Interferon was also studied as a part of Solidarity trial, it did not show any mortality benefit or decrease the duration of hospital stay [Citation29]
Lopinavir/ritonavir
Protease inhibitors (PIs) are drugs which are widely used in chronic human immunodeficiency virus (HIV) infection. In-vitro, SARS-CoV was susceptible to lopinavir/ritonavir (LPV/RTV) combination [Citation40]. Therefore, it was tested and was deemed susceptible to SARS-CoV-2 in vitro.
LPV/RTV treatment alone failed to improve the primary outcome of clinical improvement in severe COVID-19 neither did it provide a mortality benefit [Citation40]. Per Baden et al. reported that the benefit of this drug may not have been observed since the patients were treated with these drugs late in the infection and the concentration of drug necessary to inhibit the virus in the pulmonary circulation may be higher than the serum level [Citation41]. When tested as a part of the solidarity trial, it too did not show any in-hospital mortality benefit [Citation29].
Tocilizumab and other interleukin inhibitors
Tocilizumab is a monoclonal antibody which is an interleukin-6 receptor inhibitor which reduces the severity of illness by dampening the cytokine release syndrome [Citation42]. COVID-19 patients who are severely ill present with a cytokine storm which led to investigators looking at agents that neutralize the inflammatory factors. Tocilizumab given either intravenously or subcutaneously reduced the risk of invasive mechanical ventilation and death in a retrospective observational study done in 1351 patients [Citation43]. Another observational study in 764 patients showed reduced mortality in patients who received Tocilizumab [Citation44]. However, these are retrospective or observational studies. More recently, in a randomized double-blinded controlled trial, Tocilizumab did not prevent intubation or death in moderately hospitalized patients warning clinicians against its use in moderately-ill COVID-19 patients [Citation45]
Interleukin-10 was also hypothesized in reducing COVID-19 mortality by blocking the pro-inflammatory function, however, studies are warranted to prove this finding [Citation46].
Convalescent plasma
Another investigational method is the administration of convalescent plasma made from recovered patients in China. It was studied in prior pandemics – SARS, H1N1, and severe Ebola virus infections [Citation47]. For the first time for COVID-19, Shen et al. reported this experimental treatment where neutralizing IgG antibodies were transfused to 5 patients who were in severe ARDS. They theorized that adding antibodies from the convalescent plasma helped clear the virus leading to improvement in symptoms, however, very small proportion of patients were tested in this study [Citation48]. However, a recently published randomized controlled trial in 103 patients who were severely infected with COVID-19, plasma therapy did not result in a significant clinical improvement within 28 days [Citation49]. Literature such as this, has discouraged clinicians from using plasma as a potential treatment of COVID-19
Corticosteroids
The use of steroids in COVID-19 have been controversial as well. A study by Wu et al. reported that methylprednisolone may be beneficial, leading to decreased risk of death in patients with ARDS [Citation50]. Some studies have shown a beneficial effect with low dose prednisone in cancer patients with COVID-19 [Citation51,(Citation52]. A metanalysis of one randomized-controlled trial and 22 cohort studies showed that glucocorticoid therapy reduced the duration of fever but did not affect the mortality, duration of hospitalization or lung inflammation absorption [Citation53]. A recently published open-labelled trial which studied dexamethasone vs usual care showed 28-day mortality benefit in those patients receiving invasive mechanical ventilation or oxygen with dexamethasone (Recovery trial). However, the positive results only applied to patients receiving respiratory oxygen support [Citation54].
Other Experimental Modalities
Monoclonal or polyclonal antibodies were used to treat infections such as SARS-CoV, MERS [Citation55]. LY-CoV555 (bamlanivimab) was studied in patients with mild to moderate COVID-19 studies as a phase 2 clinical trial, it led to fewer hospitalization and lower symptom burden leading to FDA approval among outpatients [Citation56]. REGN-COV2 is a cocktail containing two neutralizing antibodies studied among 275 outpatients in a 1–3 phase trial, it showed that it reduced viral load with greater efficacy among those with a high baseline viral loads or those who have not yet developed immunity [Citation57]. It is also now FDA approved for non-hospitalized COVID-19 patients.
Baricitinib, a janus kinase inhibitor, has been studied as a potential treatment for COVID-19 by preventing virus infectivity via inhibition of clathrin-mediated endocytosis [Citation58]. A randomized-controlled trial was done comparing remdesivir plus baricitinib vs usual care and the combination with baricitinib reduced time to recovery in patients with high-flow oxygen and non-invasive ventilation [Citation59].
Type II transmembrane serine protease (TMPRSS2) is a host protease that activates the SARS-CoV protein in cell cultures which makes it an exciting treatment option [Citation7]. The protease inhibitor camostat plus EST [[Citation23,Citation25]trans-epoxysuccinyl-l-leucylamindo-3-methylbutane ethyl ester], a cathepsin inhibitor effectively stopped the SARS-CoV entry into host cells [Citation60]. A randomized-controlled trial is underway to test the efficacy of camostat for COVID-19 (NCT04321096) [Citation61].
Mesenchymal stem cells have improved the pulmonary functions in a case report described in China [Citation62]. On a large scale, stem cell therapy for COVID-19 is being investigated in China currently (NCT04288102) [Citation63].
Medications for concomitant conditions
The most common comorbidities of COVID-19 are hypertension and diabetes [Citation64]. Many of these patients are on ACE inhibitors or angiotensin receptor blockers (ARB) and SARS-CoV-2 uses the ACE2 receptors to enter a host cell [Citation64]. The theory is that these medications cause upregulation of ACE2 receptor which facilitates the viral entry. This is a double-edged sword since the use of these medications may prevent cardiovascular morbidity with could potentially be caused by the virus. A randomized-controlled trial evaluating the safety of continuing vs discontinuing ACE/ARBs is underway [Citation65].
Non-steroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen, were hypothesized to increase ACE2 receptors thereby facilitating the viral entry [Citation64]. However, FDA released a statement that there is no scientific data suggesting that NSAIDs would worsen COVID-19 symptoms [Citation51,(Citation66].
Vaccines
Currently, as of 22nd December, per WHO’s draft evaluation, there are 61 vaccines under clinical investigation and 172 under pre-clinical investigation [Citation67]. Two vaccines have been approved for emergency use authorization by FDA.
Conclusions
Till date, very few vaccines or specific anti-SARS-CoV-2 drug regimens have been identified to treat or prevent COVID-19. None of the drugs that have been studied has emerged as a clear winner. Remdesivir appears to be the most promising agent, however, results have been conflicting and it’s use has shown to have no effect on mortality. The most benefit from remdesivir is that it hastens time to recovery. Dexamethasone may be beneficial in severe COVID-19 cases, including patients on high-flow oxygen and invasive ventilation. The treatment atleast for now mainly focuses on supportive care and preventive measures including social distancing, wearing masks and avoiding large group gatherings.
Abbreviations
SARS-CoV2- severe acute respiratory syndrome coronavirus-2
SARS- CoV- severe acute respiratory syndrome coronavirus
MERS- Middle East Respiratory Syndrome Coronavirus
ACE2- angiotensin converting enzyme 2
ARDS- acute respiratory distress phase
ECMO- extracorporeal membrane oxygenation
RSV- respiratory syncytial virus
WHO- world health organization
IFN- interferon
HIV- human immunodeficiency virus
LPV/RTV- lopinavir/ritonavir
RA- rheumatoid arthritis
SOFA- sequential organ failure assessment
ARB- angiotensin II receptor blockers
NSAIDs- non-steroidal anti-inflammatory drugs
SIC- sepsis-induced coagulopathy
FDA- food and drug administration
Sources of Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author Contributions
RG, NK, NS assisted in acquisition of data, drafting of the manuscript. TD, CK, RJ assisted in article concept and design, interpretation of data, revision of the manuscript and final approval. RJ, NS, RJ further assisted in revisions of the final manuscript.
Disclosure statement
The authors do not have financial relationships with any commercial entity that has an interest in the subject of the presented manuscript or other conflicts of interest to disclose.
References
- Andersen KG, Rambaut A, Lipkin WI, et al. The proximal origin of sars-cov-2 Nat Med. 2020Apr.264:450–452 Available fromhttps://www.nature.com/articles/s41591-020-0820–910.1038/s41591-020-0820-9
- Cheng VCC, Chan JFW, To KKW, et al. Clinical management and infection control of sars: lessons learned. Antiviral Res. Nov 1 2013;100:407–419. Available from http://www.sciencedirect.com/science/article/pii/S016635421300224610.1016/j.antiviral.2013.08.016 2
- Mustafa S, Balkhy H, Gabere MN. Current treatment options and the role of peptides as potential therapeutic components for middle east respiratory syndrome (mers): a review. J Infect Public Health. 2018;11(1):9–17. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7102797/10.1016/j.jiph.2017.08.009
- Zhou P, Yang X-L, Wang X-G, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273. Available from. .
- Wu JT, Leung K, Leung GM. Nowcasting and forecasting the potential domestic and international spread of the 2019-ncov outbreak originating in wuhan, china: a modelling study. Lancet Lond Engl. 202029;395(10225):689–697.
- Lai -C-C, Shih T-P, Ko W-C, et al. Severe acute respiratory syndrome coronavirus 2 (sars-cov-2) and coronavirus disease-2019 (covid-19): the epidemic and the challenges. Int J Antimicrob Agents. 2020 Mar; 55(3):105924.
- Hoffmann M, Kleine-Weber H, Schroeder S, et al. Sars-cov-2 cell entry depends on ace2 and tmprss2 and is blocked by a clinically proven protease inhibitor. Cell. 2020Mar 4;10.1016/j.cell.2020.02.052
- Diao B, Wang C, Tan Y, et al. Reduction and functional exhaustion of t cells in patients with coronavirus disease 2019 (covid-19). medrxiv. 2020Feb 20;2020.02.18.20024364. Available from: https://www.medrxiv.org/content/10.1101/2020.02.18.20024364v1
- To KK-W, Tsang OT-Y, Leung W-S et al. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by sars-cov-2: an observational cohort study. Lancet Infect Dis. 2020Mar23 Available fromhttps://www.thelancet.com/journals/laninf/article/PIIS1473-3099(20)30196-1/abstract
- Cunningham AC, Goh HP, Koh D. Treatment of COVID-19: old tricks for new challenges. Crit Care Lond Engl. 202016;24(1):91.
- Liu Y, Li J, Feng Y. Critical care response to a hospital outbreak of the 2019-ncov infection in shenzhen, china Crit Care. 2020 Feb19.241:56 Available from10.1186/s13054-020-2786-x10.1186/s13054-020-2786-x
- Vincent MJ, Bergeron E, Benjannet S et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005Aug22 21 69 Available from10.1186/1743-422X-2-6910.1186/1743-422X-2-69
- Wang M, Cao R, Zhang L et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020 Mar 30(3) 269–271 Available fromhttps://www.nature.com/articles/s41422-020-0282–010.1038/s41422-020-0282-0
- Cortegiani A, Ingoglia G, Ippolito M, et al. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020Mar10 Available from http://www.sciencedirect.com/science/article/pii/S0883944120303907
- Gautret P, Lagier J-C, Parola P et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020 Mar20 Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7102549/1 10.1016/j.ijantimicag.2020.105949
- Chen J, Liu D, Liu L et al. [A pilot study of hydroxychloroquine in treatment of patients with moderate COVID-19]. Zhejiang Xue Xue Bao Yi Xue Ban J Zhejiang Univ Med Sci 2020 May 25;492:215–219
- Molina JM, Delaugerre C, Le goff J et al. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Médecine Mal Infect. 2020Mar30 Available fromhttp://www.sciencedirect.com/science/article/pii/S0399077X2030085810.1016/j.medmal.2020.03.0064
- Geleris J, Sun Y, Platt J, et al. Observational study of hydroxychloroquine in hospitalized patients with covid-19. N Engl J Med [ null. Available from]. 2020 May7. : 10.1056/NEJMoa2012410
- Boulware DR, Pullen MF, Bangdiwala AS, et al. A randomized trial of hydroxychloroquine as postexposure prophylaxis for covid-19. N Engl J Med. 2020Aug6 383(6):517–525. Available from
- Agostini ML, Andres EL, Sims AC et al. Coronavirus susceptibility to the antiviral remdesivir (gs-5734) is mediated by the viral polymerase and the proofreading exoribonuclease. mBio. 2018Mar6 92 Available fromhttps://www.ncbi.nlm.nih.gov/pmc/articles/PMC5844999/10.1128/mBio.00221-18
- Brown AJ, Won JJ, Graham RL, et al. Broad spectrum antiviral remdesivir inhibits human endemic and zoonotic deltacoronaviruses with a highly divergent RNA dependent RNA polymerase. Antiviral Res. 2019;169:104541.
- Sheahan TP, Sims AC, Graham RL, et al. Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci Transl Med. 201728;9(396):396. .
- Wit E D, Feldmann F, Cronin J et al. Prophylactic and therapeutic remdesivir (GS-5734) treatment in the rhesus macaque model of MERS-CoV infection. Proc Natl Acad Sci. 2020Mar24 11712 6771–6776 Available fromhttps://www.pnas.org/content/117/12/677110.1073/pnas.1922083117
- Holshue ML, DeBolt C, Lindquist S et al. First case of 2019 novel coronavirus in the usa. N Engl J Med. 2020Jan 31 Available fromhttps://www.nejm.org/doi/10.1056/NEJMoa200119110.1056/NEJMoa200119110
- Wang Y, Zhang D, Du G et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020Apr29 Available fromhttps://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31022-9/abstract
- Goldman JD, Lye DCB, Hui DS, et al. Remdesivir for 5 or 10 days in patients with severe covid-19. N Engl J Med. 2020Nov5 383(19):1827–1837. Available from
- Grein J, Ohmagari N, Shin D, et al. Compassionate use of remdesivir for patients with severe covid-19. N Engl J Med. 2020 Jun11 382(24):2327–2336. Available from
- Beigel JH, Tomashek KM, Dodd LE et al. Remdesivir for the treatment of covid-19 — preliminary report. N Engl J Med. 2020 May22 null. Available from 10.1056/NEJMoa2007764
- Repurposed antiviral drugs for covid-19 — interim who solidarity trial results. N Engl J Med. 2020Dec2 null. Available from 10.1056/NEJMoa2023184
- Furuta Y, Takahashi K, Fukuda Y, et al. In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob Agents Chemother. 2002 Apr 46;(4)977–981.
- Cai Q, Yang M, Liu D et al. Experimental treatment with favipiravir for covid-19: an open-label control study. Eng Beijing China. 2020Mar18; Available from. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7185795/
- Chen C, Huang J, Yin P et al. Favipiravir versus arbidol for covid-19: a randomized clinical trial. medRxiv. 2020Apr8 2020.03.17.20037432Available fromhttps://www.medrxiv.org/content/10.1101/2020.03.17.20037432v3
- Chinese clinical trial register (chictr) - the world health organization international clinical trials registered organization registered platform. Available from: http://www.chictr.org.cn/showprojen.aspx?proj=49042
- Chinese clinical trial register (chictr) - the world health organization international clinical trials registered organization registered platform. Available from: http://www.chictr.org.cn/showprojen.aspx?proj=49013
- Hensley LE, Fritz LE, Jahrling PB, et al. Interferon-beta 1a and SARS coronavirus replication. Emerg Infect Dis. 2004 Feb; 10(2):317–319.
- Chan JFW, Chan K-H, Kao RYT, et al. Broad-spectrum antivirals for the emerging middle east respiratory syndrome coronavirus. J Infect. 2013 Dec 67;(6)606–616.
- Lokugamage KG, Hage A, Schindewolf C, et al. Sars-cov-2 is sensitive to type i interferon pretreatment. bioRxiv. 2020Mar18 2020.03.07.982264Available fromhttps://www.biorxiv.org/content/10.1101/2020.03.07.982264v2
- National Institute of Allergy and Infectious Diseases (NIAID). A Multicenter, Adaptive, Randomized Blinded Controlled Trial of the Safety and Efficacy of Investigational Therapeutics for the Treatment of COVID-19 in Hospitalized Adults (ACTT-3). ; 2020 Sep. Report No.: NCT04492475. Available from: https://clinicaltrials.gov/ct2/show/NCT04492475 clinicaltrials.gov
- Davoudi-monfared E, Rahmani H, Khalili H et al. Efficacy and safety of interferon beta-1a in treatment of severe COVID-19: A randomized clinical trial. medRxiv. 2020May30 2020.05.28.20116467Available fromhttps://www.medrxiv.org/content/10.1101/2020.05.28.20116467v1
- Cao B, Wang Y, Wen D et al. A trial of lopinavir–ritonavir in adults hospitalized with severe covid-19. N Engl J Med. 2020Mar18 null. Available from 10.1056/NEJMoa2001282
- Baden LR, Rubin EJ. Covid-19 — the search for effective therapy. N Engl J Med. 2020Mar18 Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7121446/19 10.1056/NEJMe2005477
- Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression Lancet. 2020Mar28.39510229:1033–1034 Available fromhttps://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30628-0/abstract10.1016/S0140-6736(20)30628-0
- Guaraldi G, Meschiari M, Cozzi-lepri A et al. Tocilizumab in patients with severe COVID-19: a retrospective cohort study. Lancet Rheumatol. 2020Jun24 Available fromhttps://www.thelancet.com/journals/lanrhe/article/PIIS2665-9913(20)30173-9/abstract
- Biran N, Ip A, Ahn J et al. Tocilizumab among patients with COVID-19 in the intensive care unit: a multicentre observational study. Lancet Rheumatol. 2020Oct1 210 e603–12 Available fromhttps://www.thelancet.com/journals/lanrhe/article/PIIS2665-9913(20)30277-0/abstract10.1016/S2665-9913(20)30277-0
- Stone JH, Frigault MJ, Serling-boyd NJ, et al. Efficacy of tocilizumab in patients hospitalized with covid-19. N Engl J Med. 2020Dec10 383(24):2333–2344. Available from
- Lu L, Zhang H, Dauphars DJ, et al. A potential role of interleukin 10 in covid-19 pathogenesis. Trends Immunol. [2020Oct31]. Available from: https://www.cell.com/trends/immunology/abstract/S1471-4906(20)30256–8
- Chan JF-W, Yao Y, Yeung M-L, et al. Treatment with lopinavir/ritonavir or interferon-β1b improves outcome of mers-cov infection in a nonhuman primate model of common marmoset. J Infect Dis. 2015 Dec15 212;(12)1904–1913.
- Shen C, Wang Z, Zhao F et al. Treatment of 5 critically ill patients with covid-19 with convalescent plasma. JAMA. 2020Mar27 Available fromhttps://jamanetwork.com/journals/jama/fullarticle/276398310.1001/jama.2020.478316
- Li L, Zhang W, Hu Y, et al. Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life-threatening covid-19: a randomized clinical trial. JAMA. Available from: https://jamanetwork.com/journals/jama/fullarticle/2766943 2020 Jun3.
- Wu C, Chen X, Cai Y et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in wuhan, china. JAMA Intern Med. 2020 Mar 13 Available fromhttps://jamanetwork.com/journals/jamainternalmedicine/fullarticle/276318410.1001/jamainternmed.2020.09947
- Russell B, Moss C, Rigg A, et al. covid-19 and treatment with nsaids and corticosteroids: should we be limiting their use in the clinical setting? ecancermedicalscience. 14. [2020Mar 30]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7105332/
- Russell B, Moss C, George G et al. Associations between immune-suppressive and stimulating drugs and novel COVID-19—a systematic review of current evidence. ecancermedicalscience. 2020Mar 27 14 Available fromhttps://www.ncbi.nlm.nih.gov/pmc/articles/PMC7105343/10.3332/ecancer.2020.1022
- Lu S, Zhou Q, Huang L et al. Effectiveness and safety of glucocorticoids to treat COVID-19: a rapid review and meta-analysis. Ann Transl Med. 2020 May 810 Available fromhttps://www.ncbi.nlm.nih.gov/pmc/articles/PMC7290628/10.21037/atm-20-3307
- Horby P, Lim WS, Emberson J, et al. Effect of dexamethasone in hospitalized patients with covid-19: preliminary report. medRxiv. 2020Jun 22;2020.06.22.20137273. Available from: https://www.medrxiv.org/content/10.1101/2020.06.22.20137273v1
- Beigel JH, Nam HH, Adams PL et al. Advances in respiratory virus therapeutics – a meeting report from the 6th isirv antiviral group conference. Antiviral Res. 2019 Jul 167 45–67 Available fromhttps://www.ncbi.nlm.nih.gov/pmc/articles/PMC7132446/10.1016/j.antiviral.2019.04.006
- Sars-cov-2 neutralizing antibody ly-cov555 in outpatients with covid-19 | NEJM. Available from:https://www.nejm.org/doi/full/10.1056/NEJMoa2029849
- Weinreich DM, Sivapalasingam S, Norton T, et al. Regn-cov2, a neutralizing antibody cocktail, in outpatients with covid-19. N Engl J Med. [2020Dec17]. Available from. 10.1056/NEJMoa2035002
- Richardson PJ, Corbellino M, Stebbing J. Baricitinib for COVID-19: a suitable treatment? – authors’ reply. Lancet Infect Dis. [2020Apr3]. Available from: https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(20)30270-X/abstract
- Kalil AC, Patterson TF, Mehta AK, et al. Baricitinib plus remdesivir for hospitalized adults with covid-19. N Engl J Med. 2020 Dec11: 10.1056/NEJMoa2031994 Available from.
- Kawase M, Shirato K, van der hoek L, et al. Simultaneous treatment of human bronchial epithelial cells with serine and cysteine protease inhibitors prevents severe acute respiratory syndrome coronavirus entry J Virol. 2012 Jun.8612:6537–6545 Available fromhttps://www.ncbi.nlm.nih.gov/pmc/articles/PMC3393535/10.1128/JVI.00094-12
- The impact of camostat mesilate on covid-19 infection - full text view - clinicaltrials.gov. Available from: https://clinicaltrials.gov/ct2/show/NCT04321096
- Zhang Y, Ding J, Ren S, et al. Intravenous infusion of human umbilical cord wharton’s jelly-derived mesenchymal stem cells as a potential treatment for patients with covid-19 pneumonia. Stem Cell Res Ther. 202027;11(1):207. .
- Treatment with mesenchymal stem cells for severe corona virus disease 2019(covid-19) - full text view - clinicaltrials.gov. Available from: https://clinicaltrials.gov/ct2/show/NCT04288102
- Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020Apr1.84:e21 Available fromhttps://www.thelancet.com/journals/lanres/article/PIIS2213-2600(20)30116-8/abstract10.1016/S2213-2600(20)30116-8
- Elimination or prolongation of ace inhibitors and arb in coronavirus disease 2019 - full text view - clinicaltrials.gov. Available from: https://clinicaltrials.gov/ct2/show/NCT04338009
- Research C for DE and. FDA advises patients on use of non-steroidal anti-inflammatory drugs (nsaids) for COVID-19. FDA. 2020Mar19; Available from:https://www.fda.gov/drugs/drug-safety-and-availability/fda-advises-patients-use-non-steroidal-anti-inflammatory-drugs-nsaids-covid–19
- Draft landscape of COVID-19 candidate vaccines. Available from: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines
