A major focus of the recently updated guidelines of the International Society for Extracellular Vesicles (ISEV) on minimal information for studies of extracellular vesicles (MISEV) is the need for appropriate (that is, informative and effective) nomenclature in reporting extracellular vesicle (EV) research [Citation1]. The ISEV consensus recommendation on nomenclature is to use “extracellular vesicle” as the “generic term for particles naturally released from the cell that are delimited by a lipid bilayer and cannot replicate” and to modify “EV” based on clear, measurable characteristics such as cell of origin, molecular markers, size, density, function, etc. [Citation1]. Much like the biological entity it describes, the term “EV” is a scaffold on which to place further defining characteristics. MISEV does not prohibit other terms but rather specifies that they be defined carefully at first use in a manuscript. Three hundred and eighty-two authors contributed to the MISEV guidelines through extensive surveys, and 94% endorsed the nomenclature recommendation. The other 6% were evenly split between disagreeing with the recommendation (i.e. the recommendation should be stricter, less strict, or use terms other than EV) and preferring no nomenclature recommendation.
As the corresponding authors of the latest iteration of MISEV, we recognize that although disagreement with the MISEV recommendation was voiced by only a very small minority of MISEV authors, the MISEV contributors represent a core of highly active EV researchers who tend to be very collaborative and work across national and regional boundaries. Not all EV researchers engage with initiatives like MISEV or indeed with ISEV at all. It is possible that other groups of researchers would have a different distribution of opinion on various MISEV topics including nomenclature. After publication of the latest iteration of MISEV, we have received mostly positive remarks, but also some criticism of the guidelines from a few scientists who are not authors. The criticism we have received is overwhelmingly centred on one of two points. The first is the issue of characterization markers (such as specific proteins) that should be depleted in a pure EV preparation compared with the cell of origin. Here, the suggestion of our correspondents is that MISEV is too strict, since some proposed “negative” markers like GM130 or calnexin can be associated with some EVs and may be detectable in an EV preparation. This criticism is easily addressed, since it is based on a misreading or misunderstanding. In fact, MISEV2018 relaxed the recommendations of MISEV2014 [Citation2] in this regard, such that, while authors should give careful thought to depletion of contaminants, there is no recommendation for a universal negative marker. That is, for any given EV source, markers should be chosen to reveal the level of contaminants that might be expected in that specific material. One size does not fit all!
The second major criticism is the topic of this editorial: nomenclature. Several scientists we have corresponded with very much prefer the term “exosome” as a generic alternative to “extracellular vesicle.” And, yes, the nomenclature disagreement is almost always about exosomes, not microvesicles, ectosomes, microparticles, or other terms. In some cases, we perceive an almost emotional attachment to the term exosome, as in the example of one person who told us several times that they could not support MISEV as a matter of “conscience.” While we cannot argue about feelings or understand why they should enter into scientific discourse, we would like to address some other reasons that are put forward to support the use of exosome or EV: chiefly, these are arguments about primacy, precision, and popularity.
Primacy: the state of being first or most important
Was “exosome” first used to describe EVs?
No. “Exosome” is first found in the biomedical literature in four articles that were published in PNAS from 1970 to 1973 [Citation3–Citation5]. The term described transforming DNA fragments that transferred between Drosophila or Neurospora cells. In Neurospora, for example, “exosomes” could induce inositol independence in inositol-requiring Neurospora mutants [Citation6]. These DNA exosomes were thought not to be integrated into the genome and to be eliminated during meiosis. Although the presumed function might remind us of EVs that carry nucleic acids, association of the DNA with lipid bilayers was not described in these publications, so they cannot easily be construed as early descriptions of extracellular vesicles. However, by a strict application of temporal primacy (and we would disagree with such an application), mobile DNA fragments should be the only use for “exosome” today.
Was “exosome” or “extracellular vesicle” first used to describe EVs?
To our knowledge, the first use of “extracellular vesicle” in the title of a scientific publication was in 1971 in the Journal of Ultrastructure Research, “Ultrastructure of intracellular and extracellular vesicles, membranes, and myelin figures produced by Ochromonas danica.” This paper showed electron microscopy evidence for EV biogenesis from O. danica, a flagellated alga [Citation7]. Importantly, Aaronson, et al., clearly recognized EV biogenesis as a biological phenomenon. EVs were not simply a glutaraldehyde fixation artefact: they could be observed without glutaraldehyde fixation, and intact vesicles could be recovered by ultracentrifugation [Citation7]. One can also glean from this paper that the EV was not an entirely new concept in the microscopy community in 1971. Today, EV researchers often cite the coagulation and platelet studies of Chargaff and West [Citation8] and Wolff [Citation9], or the bone matrix vesicle studies of Anderson and Bonucci [Citation10–Citation12] as early evidence for the existence of EVs. In 1971, Aaronson, et al., referred to several more studies, going back to 1960, remarking that, “(m)embranous structures are also secreted extracellularly by mammalian cells (referring to [Citation13]) and [by non-mammalian] phagocytic cells (referring to [Citation14,Citation15]).” Additional publications in the 1970s referred to extracellular vesicles that were observed in vitro and in vivo. To give a few example, these include findings of EVs released from bat thyroid follicular cells during arousal from hibernation (presaging involvement of EVs in endocrine processes) [Citation16]; EVs in cartilage [Citation17,Citation18]; and what are described as EVs released by various non-mammalian organisms such as Candida [Citation19], Corynebacterium [Citation20], and Acinetobacter [Citation21].
To be sure, EVs were referred to by several names around this time by groups working in different fields, and it was often unclear exactly what these particles were. The multivesicular body was first described in the 1950s [Citation22]. Small, apparent EVs consistent with the size of MVB particles but also with particles released from the cell surface [Citation23] were observed in various animal sera [Citation24,Citation25] and in tissue and milk of breast cancer patients [Citation26], and were called by names from “pequenas particulas” (little particles) to “extracellular microvesicles” to “virus-like particles.” By the mid-1970s at the latest, interest thus emerged in the possible relationship of several observations: MVB intraluminal particles, microvesicles budding from the cell surface, and enveloped viruses [Citation27]. Van Blitterswijk et al. elaborated in a 1979 paper about EVs of ascites fluid in murine leukaemia: “An analogy between this shedding of vesicles, the formation of endocytotic vesicles and the budding of viruses is noted; all these processes select or assemble rigid lipid domains of the cell membrane” [Citation28].
The term “exosome” was not used to refer to EVs until 1981 when Trams et al. reported on “Exfoliation of membrane ecto-enzymes in the form of micro-vesicles” [Citation29]. The term “micro-vesicle”/”microvesicle” was used in the title and text, but the authors also proposed, as the last word of the abstract and discussion, that microvesicles could be referred to as “exosomes.” Although the Trams, et al. publication was cited on average several times per year during the 1980s, including by numerous studies that involved extracellular vesicles, the term “exosome” was not used again in this manner until 1986 [Citation30], and again in 1987 by Rose Johnstone and colleagues [Citation31]. From the latter publication on, consensus quickly developed around the use of exosome to describe MVB-origin EVs. We question the notion that the first-used term must necessarily predominate. Nevertheless, the suggestion of Trams, et al. in 1981 does not establish “primacy” for the term “exosome” as a generic term for all EVs.
Is “exosome” the most important term (i.e. if primacy = most important)?
Answering the question of “primacy” in the sense of “most important” is difficult and depends on the value we assign to various factors. We will thus come back to this question in the “popularity” section, below.
Precision: exactness, accuracy
What would the terms “exosome” and “extracellular vesicle” mean to a non-specialist? And do the terms have clear definitions for the specialist?
The word “exosome” is formed from the Greek “exo” (“outside”) and “soma” (“body”). What this term means would not be clear to a non-specialist (e.g. a biologist who does not study EVs) without further explanation, as exact meaning cannot be discerned immediately from the word’s constituent parts. “Outside body” could conceivably signify numerous entities in biology. Indeed, as we have seen, “exosome” was first used to denote mobile DNA elements. The term has also been employed in yet another way, by Mitchell and colleagues, to name an RNA processing body in the cell: a “soma” or particle that includes “exo”nucleases and processes/degrades RNA [Citation32]. This use of “exosome” was at least as common as the EV usage for some years beginning in the late 1990s. There have thus been at least three uses of “exosome” in recent biological science.
Even for the specialist, that is, the EV scientist, the term “exosome” has a range of inconsistent usages. Exosome has been used canonically, most consistently, and in many cases quite rigorously since the late 1980s to mean an EV originating from the endosomal system of the cell [Citation31,Citation33–Citation37], following a pathway initially described by the groups of Philip Stahl and Rose Johnstone [Citation38–Citation40]. To us, there is nothing ambiguous or controversial about this usage to denote a distinct biogenesis compartment, although the field has recognized increasingly that an exosome is often difficult to recognize as such after it has left the cell (and hence the recommendations of MISEV) [Citation1]. Even so, the clarity of the biogenesis-based usage of “exosome” has increasingly been compromised in the literature in favour of various usages: as a term for small EVs, all EVs, or even a general cell releasate, where EVs are not at all rigorously characterized. Exosomes are often said to be EVs within a particular (often arbitrary) size range, separated using a particular method, or bearing specific cargo, but frequently without demonstration of this presumed exclusivity. However they are defined, exosomes are also commonly ascribed specific functions or potency, albeit frequently without appropriate comparison with other types of EVs or fractions of cell releasates. As such, even the specialist does not know immediately how the term is being used in any given context.
This potentially confusing variety is not limited to “exosome,” though. Other terms for specific types of EVs are also burdened by other usages. “Ectosome” may refer to outer structures of sponges [Citation41], “microvesicle” can refer to structures inside and outside the cell [Citation42], as well as to synthetic particles and features of tissue damage [Citation43], and “microparticle” has a very wide range of uses for both biological and synthetic particles.
In contrast, the term “extracellular vesicle” should have a very clear definition to specialist and non-specialist alike and, as mentioned, can be used as a “scaffold” for progressively more detailed designation. As this has been covered by the consensus MISEV2018 [Citation1], we will not dwell on the perceived advantages or usages of EV. We conclude that EV is an exact and accurate term (if rather broad when used alone), while exosome, at least as commonly used, is not.
Popularity: being liked by many; fashionableness
Is exosome or EV better “liked”?
In our view, the continuing, diffuse usages of “exosome” cannot be defended by arguments of primacy (at least in the definition of “being first”) or precision. Rather, it would seem that exosome is simply a popular term. We have been informed (informally!) by some colleagues that the term “exosome” has been focus group-tested by industry and found to be the most desirable of a certain list of terms. Although we personally have not seen these results, and do not know what terms were compared, it would stand to reason that industry – who may wish to sell EV-based diagnostics or therapeutics – would want to use a word that tests well amongst potential consumers. However, even if evidence for this preference was presented publicly (and we encourage presentation of any available data), it is not at the moment a compelling argument for us as academic scientists to change our discourse.
Which term is most used in the literature?
Perhaps more convincing than how the term rolls off the tongue or what consumers think of it, the number of scientific articles that use a term could be the basis for establishing the second definition of “primacy” that thus unites with “popularity”: importance. Indisputably, the term exosome is the most popular/used in the recent biomedical literature. We observed above, though, that exosome often does not refer to the canonical exosome of MVB origin and is thus diluted by many definitions (and in papers that in many cases do not demonstrably involve EVs). As van Deun et al. recorded in their 2017 analysis of the biomedical literature, most manuscripts on EVs do not include proper characterization of EVs generally, much less demonstration of exosomes versus other types of EVs [Citation44]. This lack of rigour as the field rapidly expanded in the early 2000s formed a major part of the impetus for the International Society for Extracellular Vesicles and its standardization initiatives.
Experts … and expert consensus
Beyond overall popularity, scientific consensus is a kind of “popularity” or “agreement” among established experts. Can we answer the question of whether the established expert opinion has imbued “exosome” or “extracellular vesicle” with legitimacy as the best term for EVs in general? That is a difficult question. Without doubt, MISEV2018 represents the consensus of by far the largest group of EV experts assembled to date as an author team, and in this sense, “extracellular vesicle” is the expert consensus for the general term. Of course, this undisputed fact must be balanced against the many authors who use different definitions of “exosome” individually.
Trajectory
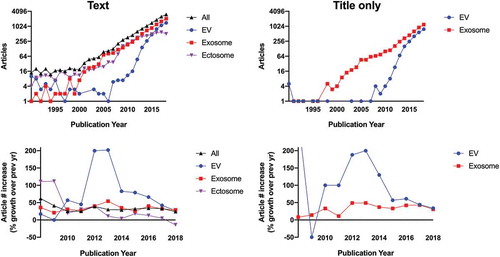
Analyzing trajectory is another way to assess the use of specific terms for EVs. Beginning around 2011, a return to the term “extracellular vesicle” began, driven by needs perceived by leading experts in the field. It is admittedly unclear if the historical use was recognized or contributed to this change. The shift around 2011 reflected a growing recognition of both the diversity of vesicles outside the cell, including the diversity of organisms that release them, and the difficulty of tracing vesicles back to a particular biogenesis pathway after release. We credit this shift in large part to an influential, highly cited review by György, et al. (Edit Buzás’s group) in 2011 [Citation45]. The authors of this review noted the “large number (diversity) of mobile membrane-limited vesicles” for which they stated explicitly in their introduction, “we suggest the term ‘extracellular vesicles’ (EVs).” The suggestion was mirrored by growing efforts in the international community. In the same year as the publication of the review, a meeting held in Paris, France, the “International Workshop on Exosomes” (IWE), initiated the idea of an international scientific society dedicated to this topic. A survey held later that year among the IWE participants, on the name to use for such a society, resulted in a majority of votes for a generic term encompassing all possible current (and future!) subtypes of released, membrane-enclosed vesicles. Hence, the chosen name was International Society for Extracellular Vesicles (ISEV). The first annual meeting of ISEV was held in 2012, followed several months later by a scientific workshop and, in early 2013, the first two position papers of the society, both using the term “extracellular vesicle” in the title [Citation46,Citation47]. From 2011 to 2013, uses of “extracellular vesicle” in the biomedical literature tripled year-over-year and then continued to increase at a healthy rate each year, reflecting but somewhat outpacing the increase in EV research in general. The rate of increase for “EV,” at least for several years, was even steeper than that for “exosome,” both in the text and the title of publications (). However, by 2018, articles mentioning EVs, exosomes, or any of the leading terms settled into a roughly 25–30% rate of increase. (For a general overview of growth in the field, we refer the reader to Roy, et al. [Citation48].) It remains unclear if EV, no matter how preferred as a generic term by expert consensus of the collaborative international EV community, will reach the overall popularity of “exosome.”
Figure 1. Usage and rate of increase of common EV terms in PubMed-indexed articles. Top left: number of published articles from 1990 to 2018 including each term as a text word (singular or plural), “Ectosome” here also encompasses terms such as microvesicle and microparticle. Searches were designed with Boolean operators in an attempt to eliminate articles unrelated to EVs, such as those including alternative definitions as alluded to in the text (e.g. the intracellular “exosome” complex, the sponge “ectosome”, synthetic microparticles, and intracellular microvesicles). Nevertheless, entries from 1990 to 2007 were curated manually to remove non-EV articles. By 2008, non-EV usages fell to <10% of the total, and manual curation was not done for subsequent years. Since some articles use more than one term, the total (“All”) may be less than the sum of the individual terms. Note that false negatives and positives from the searches are possible, so all numbers should be taken as approximate. Bottom left: year-to-year change in usage of each term, starting with 2008 and expressed as per cent change (0 = no change). Top right: number of articles with titles containing “exosome” (or “exosomal”) or “extracellular vesicle”. Bottom right: year-to-year change in article title usage.
Conclusion
The term “extracellular vesicle” or “EV” has now been agreed on by the international community as the consensus generic term for lipid bilayer-delimited particles released from the cell [Citation1]. Here, we have highlighted some of the historical work on EVs that established the primacy of this term before “exosome” was first used or reached its current consensus definition as an EV from the endosomal system. Nevertheless, a perceived popularity of the term “exosome” has resulted in its application as a generic descriptor of EVs. In the end, precision and consensus terminology are ideal, but authors will inevitably use the terms they prefer. When authors choose not to follow the consensus, we can only recommend, in the spirit of a previous letter to the editor [Citation49], that terms other than EV are clearly defined at each use.
Disclosure statement
No potential conflict of interest was reported by the authors.
Additional information
Funding
References
- Théry C, Witwer KW, Aikawa E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. Available from: https://www.tandfonline.com/doi/full/10.1080/20013078.2018.1535750
- Lötvall J, Hill AF, Hochberg F, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles. 2014;3(1):26913.
- Fox AS, Duggleby WF, Gelbart WM, et al. DNA-induced transformation in Drosophila: evidence for transmission without integration. Proc Natl Acad Sci U S A. 1970;67(4):1834–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/5275381
- Fox AS, Yoon SB. DNA-induced transformation in Drosophila: locus-specificity and the establishment of transformed stocks. Proc Natl Acad Sci U S A. 1970;67(3):1608–1615. Available from: http://www.ncbi.nlm.nih.gov/pubmed/5274483
- Fox AS, Yoon SB, Gelbart WM. DNA-induced transformation in Drosophila: genetic analysis of transformed stocks. Proc Natl Acad Sci U S A. 1971;68(2):342–346. Available from: http://www.ncbi.nlm.nih.gov/pubmed/5277082
- Mishra NC, Tatum EL. Non-Mendelian inheritance of DNA-induced inositol independence in Neurospora. Proc Natl Acad Sci U S A. 1973;70(12):3875–3879. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4521213
- Aaronson S, Behrens U, Orner R, et al. Ultrastructure of intracellular and extracellular vesicles, membranes, and myelin figures produced by Ochromonas danica. J Ultrastruct Res. 1971;35(5):418–430. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4111037
- Chargaff E, West R. The biological significance of the thromboplastic protein of blood. J Biol Chem. 1946 Nov 1;166(1):189–197. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20273687
- Wolf P. The nature and significance of platelet products in human plasma. Br J Haematol. 1967 May 1;13(3):269–288. Available from: http://www.ncbi.nlm.nih.gov/pubmed/6025241
- Anderson HC. Vesicles associated with calcification in the matrix of epiphyseal cartilage. J Cell Biol. 1969 Apr 1;41(1):59–72. Available from: http://www.ncbi.nlm.nih.gov/pubmed/5775794
- Anderson HC. Electron microscopic studies of induced cartilage development and calcification. J Cell Biol. 1967;35(1):81–101. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2107116&tool=pmcentrez&rendertype=abstract
- Bonucci E. Fine structure of early cartilage calcification. J Ultrastruct Res. 1967;20(1):33–50. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4195919
- Sun CN. Lattice structures and osmiophilic bodies in the developing respiratory tissue of rats. J Ultrastruct Res. 1966;15(3):380–388. Available from: http://www.ncbi.nlm.nih.gov/pubmed/5943006
- Mercer EH, Shaffer BM. Electron microscopy of solitary and aggregated slime mould cells. J Biophys Biochem Cytol. 1960;7(2):353–356. Available from: http://www.jcb.org/cgi/doi/10.1083/jcb.7.2.353
- VICKERMAN K. Patterns of cellular organisation in Limax amoebae. An electron microscope study. Exp Cell Res. 1962;26:497–519. Available from: http://www.ncbi.nlm.nih.gov/pubmed/13925773
- Nunez EA, Wallis J, Gershon MD. Secretory processes in follicular cells of the bat thyroid. III. The occurrence of extracellular vesicles and colloid droplets during arousal from hibernation. Am J Anat. 1974;141(2):179–201. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4415703
- Kahn SE, Jafri AM, Lewis NJ, et al. Purification of alkaline phosphatase from extracellular vesicles of fracture callus cartilage. Calcif Tissue Res. 1978;25(1):85–92. Available from: http://www.ncbi.nlm.nih.gov/pubmed/647441
- Ketenjian AY, Arsenis C. Morphological and biochemical studies during differentiation and calcification of fracture callus cartilage. Clin Orthop Relat Res. 1975;107(107):266–273. Available from: http://www.ncbi.nlm.nih.gov/pubmed/48443
- Chigaleĭchik AG, Belova LA, Grishchenko VM, et al. Several properties of the extracellular vesicles of Candida tropicalis yeasts grown on n-alkanes. Mikrobiologiia. 1977;46(3):467–471. Available from: http://www.ncbi.nlm.nih.gov/pubmed/895555
- Vysotskiĭ VV, Mazurova IK, Shmeleva EA. Extracellular material of some representatives of the genus Corynebacterium (the electron microscopic aspect). Zh Mikrobiol Epidemiol Immunobiol. 1977;(8):90–95. Available from: http://www.ncbi.nlm.nih.gov/pubmed/411293
- Käppeli O, Finnerty WR. Partition of alkane by an extracellular vesicle derived from hexadecane-grown Acinetobacter. J Bacteriol. 1979;140(2):707–712. Available from: http://www.ncbi.nlm.nih.gov/pubmed/500568
- Palay SL, Palade GE. The fine structure of neurons. J Cell Biol. 1955;1(1):69–88. Available from: http://www.ncbi.nlm.nih.gov/pubmed/,14381429
- Haguenau F. “Viruslike” particles as observed with the electron microscope. In: Dalton A, Hauguenau F, editors. Ultrastructure of animal viruses and bacteriophages an Atlas. New York: Academic Press; 1973. p. 391–397.
- De Tkaczevski LZ. “Pequenas particulas” observadas en algunos seuros animales. Rev Soc Argent Biol. 1968;44(1):19–27. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4902747
- Benz EW, Moses HL. Brief communication: small, virus-like particles detected in bovine sera by electron microscopy2. J Natl Cancer Inst. 1974;52(6):1931–1934. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4834422
- Feller WF, Chopra HC. A small virus-like particle observed in human breast cancer by means of electron microscopy. J Natl Cancer Inst. 1968;40(6):1359–1373. Available from: https://academic.oup.com/jnci/article-lookup/doi/10.1093/jnci/40.6.1359
- Dalton AJ. Microvesicles and vesicles of multivesicular bodies versus “virus-like” particles. J Natl Cancer Inst. 1975;54(5):1137–1148. Available from: http://www.ncbi.nlm.nih.gov/pubmed/165305
- van Blitterswijk WJ, Emmelot P, Hilkmann HA, et al. Rigid plasma-membrane-derived vesicles, enriched in tumour-associated surface antigens (MLr), occurring in the ascites fluid of a murine leukaemia (GRSL). Int J Cancer. 1979;23(1):62–70. Available from: http://www.ncbi.nlm.nih.gov/pubmed/83306
- Trams EG, Lauter CJ, Salem N, et al. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim Biophys Acta. 1981;645(1):63–70. Available from: http://www.ncbi.nlm.nih.gov/pubmed/6266476
- Kassis S, Lauter CJ, Stojanov M, et al. Exfoliation of the beta-adrenergic receptor and the regulatory components of adenylate cyclase by cultured rat glioma C6 cells. Biochim Biophys Acta. 1986;886(3):474–482. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2871868
- Johnstone RM, Adam M, Hammond JR, et al. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19):9412–9420. Available from: http://www.ncbi.nlm.nih.gov/pubmed/3597417
- Mitchell P, Petfalski E, Shevchenko A, et al. The exosome: a conserved eukaryotic RNA processing complex containing multiple 3ʹ–>5ʹ exoribonucleases. Cell. 1997;91(4):457–466. Available from: http://www.ncbi.nlm.nih.gov/pubmed/9390555
- Johnstone RM, Bianchini A, Teng K. Reticulocyte maturation and exosome release: transferrin receptor containing exosomes shows multiple plasma membrane functions. Blood. 1989;74(5):1844–1851. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2790208
- Vidal M, Sainte-Marie J, Philippot JR, et al. Asymmetric distribution of phospholipids in the membrane of vesicles released during in vitro maturation of guinea pig reticulocytes: evidence precluding a role for “aminophospholipid translocase”. J Cell Physiol. 1989;140(3):455–462. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2777884
- Raposo G, Nijman HW, Stoorvogel W, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996 Mar 1;183(3):1161–1172. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8642258
- Vidal MJ, Stahl PD. The small GTP-binding proteins Rab4 and ARF are associated with released exosomes during reticulocyte maturation. Eur J Cell Biol. 1993;60(2):261–267. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8330623
- Johnstone RM. The Jeanne Manery-fisher memorial lecture 1991. Maturation of reticulocytes: formation of exosomes as a mechanism for shedding membrane proteins. Biochem Cell Biol. 1992;70(3–4):179–190. Available from: http://www.ncbi.nlm.nih.gov/pubmed/1515120
- Harding C, Heuser J, Stahl P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329–339. Available from: http://www.ncbi.nlm.nih.gov/pubmed/6309857
- Harding C, Heuser J, Stahl P. Endocytosis and intracellular processing of transferrin and colloidal gold-transferrin in rat reticulocytes: demonstration of a pathway for receptor shedding. Eur J Cell Biol. 1984;35(2):256–263. Available from: http://www.ncbi.nlm.nih.gov/pubmed/6151502
- Pan BT, Teng K, Wu C, et al. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J Cell Biol. 1985;101(3):942–948. Available from: http://www.jcb.org/cgi/doi/10.1083/jcb.101.3.942
- Bretting H, Königsmann K. Investigations on the lectin-producing cells in the sponge Axinella polypoides (Schmidt). Cell Tissue Res. 1979;201(3):487–497. Available from: http://www.ncbi.nlm.nih.gov/pubmed/509491
- Roth LM, LUSE SA. Fine structure of the neurohypophysis of the opossum (Didelphis virginiana). J Cell Biol. 1964;20(3):459–472. Available from: http://www.ncbi.nlm.nih.gov/pubmed/14128048
- Astner S, Cheung AC, Rius-Díaz F, et al. Non-invasive evaluation of the kinetics of allergic and irritant contact dermatitis. J Invest Dermatol. 2005;124(2):351–359. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15675954
- Van Deun J, Mestdagh P, Agostinis P, et al. EV-TRACK: transparent reporting and centralizing knowledge in extracellular vesicle research. Nat Methods. 2017;14(3):228–232. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28245209
- György B, Szabó TG, Pásztói M, et al. Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell Mol Life Sci. 2011;68(16):2667–2688.
- Witwer KW, Buzás EI, Bemis LT, et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. J Extracell Vesicles. 2013;2:1–25.
- Hill AF, Pegtel DM, Lambertz U, et al. ISEV position paper: extracellular vesicle RNA analysis and bioinformatics. J Extracell Vesicles. 2013;2. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24376909
- Roy S, Hochberg FH, Jones PS. Extracellular vesicles: the growth as diagnostics and therapeutics; a survey. J Extracell Vesicles. 2018;7(1):1438720. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29511461
- Gould SJ, Raposo G As we wait: coping with an imperfect nomenclature for extracellular vesicles. J Extracell Vesicles. 2013 Sep 7;2. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24009890
