Abstract
Objective: To describe catch-up growth in children with moderate acute malnutrition (MAM) on targeted supplementation using Ready-to-Use Supplementary Food (RUSF).
Methods: An impact study was done to determine anthropometric changes in children aged 12–60 months who received RUSF (175 kcal/kg/day) for six weeks, managed as outpatients and followed up for 12 weeks until a final assessment.
Results: Default rates were high, with 30% of children returning to the primary healthcare facility for follow-up only once or twice. Despite significant improvement in height-for-age Z-score (HAZ), weight-for-age Z-score (WAZ), weight-for-height Z-score (WHZ) and mid-upper arm circumference (MUAC), 70.5% of the sample remained in the same malnutrition classification and only 26% recovered. The growth velocity of children with a lower initial WHZ was significantly higher (r = –0.15, p < 0.05) than those with less wasting, but only 20% grew at a rate to achieve catch-up growth. The mean growth velocity decreased as the intervention period continued.
Conclusion: All median anthropometric indicators improved with RUSF supplementation. However, catch-up growth or recovery occurred in only 20–25% of children included in the study. These findings create questions about the value of supplementation in the absence of blanket food distribution or other interventions to address food security.
Introduction
Malnutrition in children below the age of five years is associated with inadequate child development and poor survival.Citation1 Recent studies showed that poor growth in the first 1 000-day period, including pregnancy and the first two years of life, may result in irreversible damage during infancy and early childhood, which may impact on children’s cognitive abilities at the age of school enrolment.Citation2 Grantham-McGregor et al.Citation3 estimated that more than 200 million children in developing countries fail to reach their full development potential as a result of poverty, poor nutrition and inadequate care.
Under-nutrition and growth faltering can both result in an increased prevalence of moderate acute malnutrition (MAM). Data from 2008 showed that approximately 8.7% of children in South Africa below the age of five years suffer from moderate wasting and 23.9% from moderate stunting.Citation4 Early identification of these children can be used as an early warning system to intervene in a timely manner and prevent the development of severe acute malnutrition (SAM). The incidence of SAM is increasing in South AfricaCitation5 and these children are at a higher risk of associated morbidity and death.
Current intervention strategies for malnourished children treated in the public health sectorCitation6 allow for targeted supplementation with enriched maize blends, Ready-to-Use Therapeutic Foods (RUTF), enriched energy drinks, or a combination thereof. These supplements are provided in cases where more conventional approaches, such as counselling to promote breastfeeding and appropriate complementary feeding, regular deworming and routine vitamin A supplementation and zinc supplementation for diarrhoea, fail to result in sufficient catch-up growth. It is, however, recognised that these interventions are only sustainable in environments with a functional district health system with strong links between the facility and community levels, as well as sufficient human resources including nutrition staff.Citation7
RUTF and Ready-to-Use Supplementary Food (RUSF) have been developed to provide a nutrient-dense alternative to more conventional supplements. As these supplements have a low water activity, they are safer to use in developing countries where clean water and optimal storage conditions are often problematic. RUTF generally consists of a peanut butter base in vegetable oil with added protein, vitamins and minerals. RUTF has been used in the international setting for the treatment of SAM as an improved alternative to F-100 for a number of years.Citation8
Recently, calls have gone out to use these energy dense supplements in the treatment of moderate malnutrition as well.Citation9 This practice was supported by the World Health Organization (WHO), which also favours short, aggressive supplementation periods in children with malnutrition that allows for rehabilitation in 14 to 40 days.Citation10 Although there is strong evidence for the value of nutrition interventions internationally,Citation11 limited evidence exists regarding the impact of such interventions, especially in relation to growth in South Africa.
This study attempted to address this need by determining the impact of aggressive supplementation with RUSF on catch-up growth in children with MAM, 12–60 months of age, managed in a functional health system in South Africa with sufficient human resources.
Methods
Setting and design
A non-experimental impact design was used, as this intervention supported the nutrition supplementation programme in South Africa, and ethically it would unacceptable to withhold treatment from a comparison group. Between September 2012 and August 2013, the legal guardians of children between 12 and 60 months of age, in three geographical areas, screened with a gender specific Moyo chartCitation12 at primary health care facilities as being moderately malnourished (weight-for-height Z-score of between –2 and –3 standard deviations [SD] of the WHO reference population, or height-for-age less than –2 SD of the WHO reference population), were invited to allow their children to participate.
Children who presented without severe illness were purposefully selected by trained, registered dietitians for inclusion in the study, after written informed consent was obtained from the legal guardian. Ethical approval for the study was obtained from the Research Ethics Committee (Human), Nelson Mandela Metropolitan University (H12-RTI-HIV-003).
Intervention
Children included in the study received RUSF in a quantity sufficient to meet their nutrient requirements for full catch-up growth (175 kcal/kg/day), for a period of six weeks. The appetite of all children were tested prior to inclusion, and children who passed the test, being able to consume 75% of prescribed RUSF, qualified to participate.Citation13 The RUSF quantity was rounded in terms of sachet volumes and children received one to three 92 g sachets per day. The required amounts were calculated for a period of two weeks and the mother or caretaker had to return to the facility every two weeks to collect the required number of sachets, and also to allow follow-up anthropometry data collection.
Measures
The following data were collected: demographic data; underlying illness including HIV and TB from the medical history; baseline weight, height, mid-upper arm circumference (MUAC); number of sachets provided and returned; supplement sharing and nutritional problems or illness that might have interfered with intake. All data were collected during follow-up visits, scheduled with two-week intervals.
Children were weighed using a calibrated Seca™ Paediatric scale (Seca, Hamburg, Germany) accurate to the nearest 10 g. All children were weighed without a nappy, with minimal clothing (underwear only) and without shoes. The scale was placed on a hard surface; the child was placed in the middle and kept still until the measurement was completed.Citation14
A measuring mat accurate to 0.1 cm was used to measure the height of children below 24 months of age. The measuring mat was placed on a flat surface in order to obtain accurate measurements. The researcher and a trained nurse worked together in taking and recording accurate height measurements every two weeks. The measurement was taken when the head was level with the headboard, and the end of the measuring mat was against a flexed heel. In children older than 24 months the standing height was taken, without shoes and hair gear, to the nearest 0.1 cm. In both cases, the measurements were taken at eye level.Citation14 Three height measures were taken, and the mean was calculated and recorded.
Mid-upper arm circumference was measured by the researcher using a MUAC Child 11.5 Red measuring tape (Teaching-aids At Low Cost, UK) with accuracy to the nearest 1 mm. The MUAC was taken during each visit. MUAC was measured on the left upper arm, with the arm in a relaxed position along the side of the body. The measurement was taken at the midpoint between the acromion and the olecranon processes, with the measuring tape fitting snuggly, but without making a dent in the upper arm.Citation14
The rate of weight gain or growth velocity was calculated using the following formula, where W1 is the initial weight in kg and W2 is the weight in kg on the last day of measurement:Citation15
The measurements taken during the last visit were used to calculate growth velocity and determine recovery and overall impact of the RUSF. For the purpose of this study, ‘catch-up growth’ refers to weight gain of 2–5 g/kg/day, and ‘moderate growth velocity’ more than 5 g/kg/day.Citation10 The fieldworkers made an effort to contact defaulters and return them to the programme after one missed visit. It was further attempted to follow-up all participants after four weeks off the programme in order to determine relapse and to readmit children for nutrition support if necessary.
Data analysis
Data were analysed using PASW (Predictive Analytics SoftWare) by SPSS (Version 22; SPSS Inc., Armonk, NY, USA) and SAS/STAT software version 9.3 of the SAS system for Windows (SAS Institute, Cary, NC, USA). Frequencies and percentages were used to describe categorical data. Comparisons of means were performed using t-tests. Chi-square tests and two-tailed Pearson correlations were used to describe and test associations and correlations between variables. A p-value of ≤ 0.05 was considered statistically significant.
During analysis, it became evident that 12 children in the sample with clinically stable SAM were incorrectly classified as MAM during screening and were also managed according to the proposed intervention strategy. Similarly, 17 children with low normal HAZ and WHZ near to the MAM cut-off values used were included according to the Moyo chart. Results were therefore presented to include all three classifications.
Results
The children in the sample (n = 226) had a mean age of 29.3 months (SD = 13.5), with 50.2% of them being male (n = 113), and lived in households with a mean of 5.65 (SD = 2.2) people. One child from the sample was excluded with missing values for initial Z-score measurements in some of the calculations. 7.2% of children were affected by either a father or mother who passed away. Although no HIV or tuberculosis (TB) testing formed part of this study, about 7% of children were reported to be HIV-infected (Table ).
Table 1: Demographic and clinical details of children receiving Ready-to-Use Supplementary Food (RUSF)
Table summarises the anthropometric changes that occurred after the six-week intervention with RUSF. The baseline sample included 196 children with MAM (87.1%), 12 with SAM (5.3%) and 17 (7.6%) with low normal limits based on HAZ and WHZ.
Table 2: Anthropometric changes after six weeks of intervention with RUSF
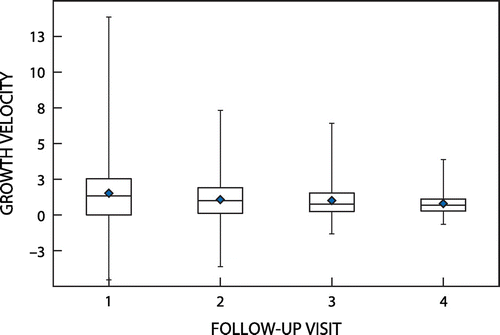
After supplementation for two weeks, at the first follow-up visit children (n = 186) had a mean growth velocity of 1.52 g/kg/day (SD = 2.43), with 7% (n = 13) achieving moderate growth velocity and 29% (n = 54) catch-up growth. At the four-week follow-up among 156 children, a mean growth velocity of 1.07 g/kg/day (SD = 1.51) was recorded, with three children (2%) achieving a moderate growth velocity and only 31 (20%) achieving catch-up growth. At six weeks, the results further deteriorated with only 139 children remaining in the sample, reaching default rates of between 25% and 30% in the three different geographical areas. A mean growth velocity of only 1.00 g/kg/day (SD = 1.17) was recorded, with only two (1%) and 20 (15%) children achieving moderate growth velocity and catch-up growth, respectively.
As summarised in Table , the mean WAZ, WHZ, HAZ and MUAC scores improved significantly during the intervention period. However, despite this improvement, 124 (70.5%) children remained within the same nutritional classification, only 46 (26.1%) recovered and six (3.4%) worsened. Deterioration to SAM (WHZ < –3 if before above) occurred in five (2.8%) children in the sample. A paired t-test showed that the final growth velocity ( = 0.79 g/kg/day) differed significantly from the initial growth velocity (
= 1.52 g/kg/day) (t = 4.34, df = 113, p < 0.01) as shown in Figure .
Despite a strong trend that we observed, no significant difference in the mean growth velocity for the groups of children with SAM (1.93 g/kg/day ± 4.07 SD), MAM (0.89 g/kg/day ± 1.30 SD) and underweight but still above –2 WHZ (0.49 ± 0.91 SD) were found (p = 0.058) during the intervention period.
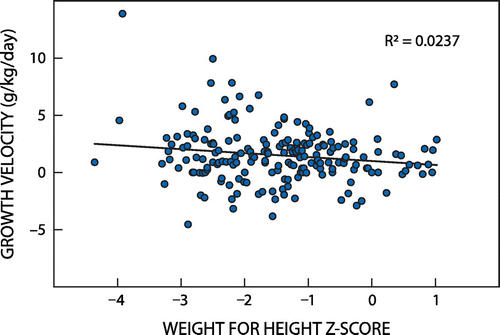
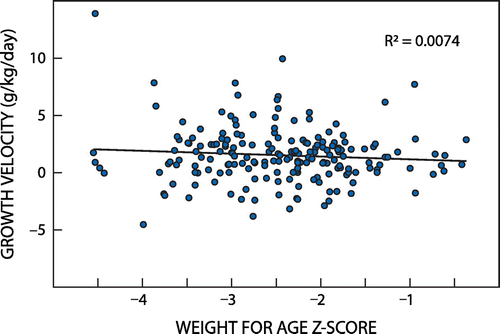
A Pearson correlation, however, showed that the initial WHZ was significantly and negatively correlated with growth velocity (r = –0.15, p < 0.05), indicating that children with lower weight-for-height status at admission tended to have a higher growth velocity during treatment (Figure ). The baseline WAZ, however, was not significantly correlated with growth velocity, as shown in Figure .
Discussion
Intervention with RUSF in this study resulted in improvement of all the anthropometrical indicators. However, recovery rates at 20% to 25% were less than the recovery demonstrated in similar programmes elsewhere in the world.Citation16,17 Growth velocity proved to be higher immediately after admission to the programme and tapered off the longer the children stayed on the intervention programme. Children with a lower initial WHZ with wasting responded better to the aggressive supplementation with RUSF than those who had higher WHZ and were therefore more affected by stunting and underweight. After six weeks of supplementation the mean growth velocity dropped to 1 g/kg/day, which does not even result in normal growth rates in that particular age group. Results indicated that both the growth velocity and recovery decreased with time and that continued supplementation for periods longer than the 14 to 40 days suggested by the WHOCitation10 will benefit only a minority of a sample in a food-insecure environment.
Although only a small number of children were known to be HIV-infected, a number of children failed to respond to aggressive supplementation in the first two weeks. It is therefore recommended that healthcare professionals include routine HIV-testing in treatment protocols to rule out HIV infection as a possible confounding factor complicating catch-up growth.
Only a small percentage of children that were retained in the programme did not benefit from the supplementation and deteriorated. However, it should be acknowledged that success, as defined by a growth velocity of more than 5 g/kg/day,Citation10 could not be demonstrated in this study, which was based in a non-emergency setting. It is imperative to investigate reasons for high default rates, inadequate growth velocity with RUSF supplementation and the slowing of growth velocity among children with moderate malnutrition during nutrition intervention periods in Southern Africa, especially in areas with sufficient nutrition staff. As the majority of these children come from food-insecure households, it is also suggested to investigate the impact of targeted RUSF supplementation in the context of blanket food-distribution programmes to support household food security during the period of intended catch-up growth in these children.
As children in this study did not benefit from targeted supplementation beyond six weeks, the question may well be asked whether it can be justified in providing expensive supplements to children with moderate malnutrition resulting from food insecurity in households. Channelling of those funds to support community-based interventions for the improvement of household food production may be of more value in this particular group, rather than attempting to achieve catch-up growth in children residing in hungry households.
Disclosure
Two of the researchers (LS and RL) do contract work for a manufacturer of RUTFs.
Acknowledgements
This study would not have been possible without a Research Theme Grant from Nelson Mandela Metropolitan University, and Nadia Gous, Louise Cilliers and Craig van Wyk, all registered dietitians, who coordinated the research sites and research activities with staff from the Western Cape, Northern Cape and Free State Departments of Health. The authors also acknowledge Dr Daleen Struwig, medical writer, Faculty of Health Sciences, University of the Free State, for technical and editorial preparation of the manuscript.
Additional information
Funding
References
- Berry L, Dawes A, Biersteker L. Getting the basics right: an essential package of services and support for ECD. S Afr Child Gauge. 2013 [cited 2014 Dec 1];2:26–33. Available from: http://www.ci.org.za/depts/ci/pubs/pdf/general/gauge2013/Gauge2013EssentialPackage.pdf.
- Walker SP, Wachs TD, Grantham-McGregor S, et al. Inequality in early childhood: risk and protective factors for early child development. Lancet. 2011;378(9799):1325–38.10.1016/S0140-6736(11)60555-2
- Grantham-McGregor S, Cheung YB, Cueto S, et al. Developmental potential in the first 5 years for children in developing countries. Lancet. 2007;369(9555):60–70.10.1016/S0140-6736(07)60032-4
- World Health Organization. Nutrition: child malnutrition country estimates (WHO global database). 2013 [cited 2014 July 28]. Available from: http://apps.who.int/gho/data/view.main.1731.
- National Department of Health South Africa. Severe malnutrition under 5 years incidence (per 1 000). District Health Information System Database. 2012 [cited 2014 Jul 28]. Available from: http://indicators.hst.org.za/healthstats/235/data.
- Department of Health South Africa. Roadmap for nutrition in South Africa 2013–2017. 2013 [cited 2014 Dec 1]. Available from: https://extranet.who.int/nutrition/gina/sites/default/files/ZAF%202013%20Roadmap%20for%20Nutrition%20in%20South%20Africa%20.pdf.
- Hendricks M, Goeiman H, Hawkridge A. Promoting healthy growth: strengthening nutritional support for mothers, infants and children. S Afr Child Gauge. 2013 [cited 2014 Dec 1];2:44–49. Available from: http://www.ci.org.za/depts/ci/pubs/pdf/general/gauge2013/Gauge2013Nutrition.pdf.
- Briend A. Highly nutrient-dense spreads: a new approach to delivering multiple micronutrients to high-risk groups. Br J Nutr. 2001;85(Suppl 2):S175–9.10.1079/BJN2000311
- Michaelsen KL, Hoppe C, Roos N, et al. Choice of foods and ingredients for moderately malnourished children 6 months to 5 years of age. Food Nutr Bull. 2009;30(Suppl 3):S343–404.
- Golden MH. Proposed recommended nutrient densities for moderately malnourished children. Food Nutr Bull. 2009;30(Suppl 3):S267–342.
- Bhutta ZA, Das JK, Rizvi A, et al. Evidence-based interventions for improvement of maternal and child nutrition: what can be done and at what cost? Lancet. 2013;382(9890):452–77.10.1016/S0140-6736(13)60996-4
- Sikorski C, Kerac M, Fikremariam M, et al. Preliminary evaluation of the Moyo chart—a novel, low-cost, weight-for-height slide chart for the improved assessment of nutritional status in children. Trans R Soc Trop Med Hyg. 2010;104(11):743–5.10.1016/j.trstmh.2010.07.013
- Kimani F, Sharif SK. National guideline for integrated management of acute malnutrition. Version 1: 2009 June [cited 2014 Dec 1]. Ministry of Medical Services and Ministry of Public Health & Sanitation, Kenya. Available from: http://www.cmamforum.org/Pool/Resources/Kenya-MoH-IMAM-Guideline-June-2009.pdf.
- Gibson RS. Principles of nutrition assessment. 2nd ed. New York, NY: Oxford University Press; 2005.
- Ashworth A, Kanum S, Jackson A, et al. WHO guidelines for the inpatient treatment of severely malnourished children. Geneva: World Health Organization; 2003 [cited 2014 Dec 1]. Available from: http://www.who.int/nutrition/publications/guide_inpatient_text.pdf.
- Lenters LM, Wazny K, Webb P, et al. Treatment of severe and moderate acute malnutrition in low- and middle-income settings: a systematic review, meta-analysis and Delphi process. BMC Pub Health. 2013;13(Suppl 3):1–15.10.1186/1471-2458-13-S3-S23
- Taneja G, Dixit S, Khatri AK, et al. A study to evaluate the effect of nutritional intervention measures on admitted children in selected nutrition rehabilitation centers of Indore and Ujjain divisions of the state of Madhya Pradesh (India). Indian J Community Med. 2010;37(2):107–15.