ABSTRACT
We conducted a 2.5-year field experiment to test the effects of straw incorporated evenly into the soil (EIS) on soil fungal community, SOC chemical composition, and particulate organic matter fractions via comparing with no straw returning (CK), straw mulching (SM), straw plowed into the soil (SP), and identified the linkages between soil fungal community as well as organic C accumulation and POM formation. Our results showed that EIS treatment significantly increased the concentrations of SOC and the proportion of carbohydrate C, di-O-alkyl C, and O-alkyl C in SOC structure, increased the mass proportion and OC contents of MA(c)POM and mM-POM in the upper 40 cm of soil. Meanwhile, EIS treatment increased the relative abundance of Ascomycota, Zygomycota, Chytridiomycota, and Dothideomycetes in 0–20 cm depths, and also had the highest relative abundance of Glomeromycetes and Dothideomycetes in the 20–40 cm soil. Also, our study suggests that straw return enhanced the relative abundances of fungi involved in the carbon cycle and sequestration, including Zygomycota, Chytridiomycota, and Glomeromycota, and Ascomycota. The shifts in fungal community structure can accelerate organic C accumulation and the formation of soil particulate organic matter, especially in EIS treatment.
Introduction
Northeast China is the major maize-growing and grain-producing area of China, and the grain-producing this area accounts for 25% of China’s total annual grain-producing (Zhao et al. Citation2016). As one of the main cultivated soils in northeast China, chernozem accounts for 27% of the total cultivated area in this region. Chernozems are characterized by a surface layer that is rich in humus (with up to 16% humus by mass) and in available calcium ions bound to soil particles, resulting in a well-aggregated structure with abundant natural grass vegetation (Demyan et al. Citation2012). However, over the years, the high-intensity utilization of chernozem resources has led to serious degradation of the quality of chernozem cultivated land. Nevertheless, the huge demand for grain output in China also makes it urgent to increase the grain output and soil fertility of chernozem. Extensive studies have shown that increasing the C seques btration in soil plays a key role in increasing crop yields and improving the function of chernozem, and the turnover of soil organic carbon (SOC) is mainly affected by the physicochemical protection provided by aggregates and the structure of soil microbial community (Plaza et al. Citation2013; Garcia-Franco et al. Citation2015; Han et al. Citation2017). Here, we intend to increase our knowledge of this response, by using physical fraction methods and high sequencing techniques to provide a detailed analysis of soil aggregates structure and fungal communities diversity in chernozem under dryland farming.
Soil aggregates play a coating role on organic C and make the organic C inside them get physical protection, thus helping SOC resist decomposition by soil microorganisms, which is an important mechanism of soil C fixation (Chivenge et al. Citation2011). Also, soil aggregates have a hierarchical organization on each scale. At the micro-scale, individual soil primary particles (sand, silt, clay) bound together in packets to create microaggregates (53–250 μm) (Guan et al. Citation2018). Microaggregates combine to form macroaggregates (>250 μm) by root exudation and mycorrhizal products (O’Brien and Jastrow Citation2013). Meanwhile, soil primary particles also will be combined with SOC to form particulate organic matter (POM) in various ways, while the newly formed POM will be stable in microaggregates and macroaggregates (Sarker et al. Citation2018). Six et al. (Citation2000) and Garcia-Franco et al. (Citation2015) reported that O-alkyl C groups (such as carbohydrates) in SOC play a critical role in the formation of POM. As an effective indicator of soil quality, POM is closely linked to aggregation and its decomposition can enhance soil microbial activity and consequently the production of transient-binding agents (Chen et al. Citation2018).
Soil microorganisms also play an important role in the formation of soil aggregates and the C sequestration, and their effects are mainly in the following two aspects. On the one hand, fungal hyphae are beneficial to the mechanical bonding of soil particles; on the other hand, the exudation of microbial by-products can promote the aggregation of primary particles (Vrdoljak and Sposito Citation2002). In general, fungi are more important than bacteria in the formation of soil aggregates (Yang, Liu, and Zhang Citation2019). And compared with bacteria, fungi can release more extracellular enzymes than bacteria, so they have a greater ability to degrade recalcitrant organic C such as lignin and cellulose than bacteria (Zhao et al. Citation2016). This also implies that enhance C sequestration capacity should include increasing the fungal component of the soil microbial community (Yang et al. Citation2020). However, at this stage, because of the existing cropping system has a series of shortcomings in C sequestration (e.g., intensive tillage, residue removal, soil erosion, and high soil moisture loss), it is difficult to directly change the composition and structure of soil microbial communities through existing technologies. Numerous studies have shown that the application of soil organic amendments (e.g., crop straw, manure, and compost) has a significant effect on the soil microbial community structure, function, and stability (Garcia-Franco et al. Citation2015; Liang et al. Citation2018; Bu et al. Citation2020). Therefore, it is possible to choose the indirect method of adding soil organic amendments to promote fungal reproduction, thereby improving soil structure by stabilizing soil aggregates.
As one of the major organic wastes, approximately 8.75 Tg of crop straws are produced each year in China but only 20% are returned to the field (Jiang et al. Citation2012). Crop straw consists mainly of cellulose (30–40%), hemicellulose (25–35%), and lignin (4–8%) (Zhang et al., Citation2015). Straw return to the field can increase the inputs of nutrients and organic C and has great potential in improving soil fertility and microbial community (Chen et al., Citation2017; Yan et al. Citation2020; Qiu et al. Citation2020). In recent years, with the increasing promotion of straw returning by the Chinese government, different straw returning methods (e.g., straw mulching and straw plowed down into the soil) is being widely promoted and applied in NE China. However, different straw returning methods also have various effects on soil organic carbon and soil structure. Previous studies have only mainly focused on the effects of SOC, soil aggregation, soil microbial community, and other single indicators by different straw returning methods, but few studies explored the linking of particulate organic matter and fungal community on organic C accumulation (Larkin et al. Citation2011; Zhao et al. Citation2016; Guan et al. Citation2018). Therefore, to evaluate the changes in soil particulate organic matter and fungal community on soil C accumulation under different straw returning methods, we experimented with four different straw returning methods on chernozem in northeast China. Consequently, we examined the effects of different straw returning methods and soil depths on 1) changes of soil particulate organic matter in hierarchical aggregates and 2) changes in the soil fungal community structure. We hypothesise that the different straw returning methods could have different effects on soil organic carbon accumulation and soil fungal communities’ function and diversity, and which could lead to the difference in the formation of particulate organic matter in hierarchical aggregates structure.
Methods and materials
Site description
The experiments were conducted in Nong’ an County (44°26ʹN 125°21ʹE), which is located at Jilin Province in Northeast China. The annual average temperature is 5°C, and the average annual precipitation is 332 mm. The natural vegetation cover types are Aneurolepidium Chinensis and Stipa baicalensis. The soil is classified as Calciboroll (Soil Survey Staff Citation1999) or Gleyic Chernozem (IUSS Working Group WRB Citation2015)), which developed in loess-derived sediments. The basic properties of the soil are shown in .
Table 1. Main properties of the bulk soil at 0–20 and 20–40 cm soil depths
Experimental design and soil sampling
The field experiment was conducted in May 2016 and followed a randomized design with four treatments and three replications. The area of each plot was 5 m × 10 m. The four treatments were as follows: no straw return (CK), straw that was chopped and plowed into the approximately 0–20 cm soil depth range (SP), return of straw as mulch after plowing (SM) and straw incorporated evenly into the soil (EIS). EIS consists of the following steps: 1) straw is crushed into pieces, 1–1.5 cm in length and spread over the field by the combine harvesting the crop and 2) the straw is plowed evenly into the 0–20 cm soil depth, using the crashing–ridging technology (Fan et al. Citation2018). The maize variety was XianYu 335. Each straw return plot had a similar maize straw application rate (9500 kg ha−1). Each plot was fertilized with inorganic fertilizers (N, P, and K) at doses of 165 kg N ha−1, 82.5 kg P2O ha−1 and 82.5 kg K2O ha−1 respectively. Soil samples were collected from two soil layers (0–20 cm and 20–40 cm) in October 2018. Each plot was randomly selected five points following the core method. Then, the samples were removed stones and plant residues and subsequently divided into two parts: one sub-sample was stored at −20°C for DNA extraction, while the other was air-dried and passed through an 8-mm sieve for the analyses as required.
Analytical methods
Soil organic matter fractionation
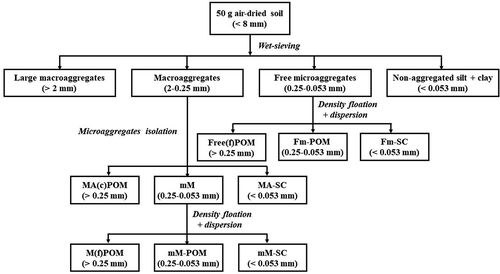
According to the research of O’Brien and Jastrow (Citation2013) and Plaza et al. (Citation2013), the method used for organic matter physical fractionation was isolated by three steps (). In the first step, air-dried soil sample (< 8 mm) was wet-sieved into four aggregate-size classes (i.e., >2000, 250–2000, 53–250, and <53 µm). In the second step, the macroaggregates (250–2000 µm) were placed on the 0.25- and 0.053-mm sieves and shaken with 50 stainless steel beads (4-mm diameter). After a continuous and steady water flow through the sieve, the coarse POM inside macroaggregates (MA(c)POM) and microaggregates-within-macroaggregates (mM) were retained on the 0.25- and 0.053-mm sieves, respectively. In the third step, free microaggregates (53–250 µm) and mM were separated by NaI solution (1.85 g cm−3), the floating material was obtained by centrifugation, the heavy fraction was dispersed by shaking with 0.5% sodium hexametaphosphate for 18 h, and then the dispersed heavy fraction was passed through a 53-µm sieve. Thus, free microaggregates from wet sieving were fractionated into non-occluded fine POM (Free(f)POM) and POM inside free microaggregates (Fm-POM), mM was fractionated into non-occluded fine POM (M(f)POM) and POM inside mM (mM-POM). The organic carbon content of each POM fraction was determined by H2SO4-K2Cr2O7 oxidation followed by titration with FeSO4.
Characterization of the organic C fraction
The solid-state 13C cross-polarization magic-angle-spinning (CPMAS) NMR spectra of soil samples were obtained on an AVANCE III 400 WB spectrometer (Bruker BioSpin AG, Fällanden, Switzerland) at 100.6 MHz with a spinning rate of 8 kHz, an acquisition time of 20 ms, a recycle time of 3 s, and a contact time of 2 ms. Chemical shift values were externally referenced to the methylene resonance of the adamantane standard at 38.4 ppm. Semi-quantification was performed by integration using MestReNova 5.3.1 software (Mestrelab Research S.L., Santiago de Compostela, Spain).
Solid DNA extraction and illumina HiSeq sequencing
Soil DNA was extracted from 0.5 g of the soil samples, using a Fast DNA SPIN Kit for soil, according to the manufacturer’s instructions. The DNA was quantified on a Nanodrop spectrometer and was stored at −80°C before use. The fungal ITS1 region was amplified using the primers ITS1-F (5ʹ-CTTGGTCATTTAGAGGAAGTAA-3ʹ) and ITS2-R (5ʹ-GCTGCGTTCTTCATCGATGC-3ʹ). The ITS gene fragments were sequenced using the Miseq platform. Amplicons were extracted from 2% agarose gels and purified using the AxyPrep DNA Gel Extraction Kit (Axygen Biosciences) according to the manufacturer instructions and were quantified with Quanti FluorTM-ST (Promega). Purified amplicons were pooled in an equimolar manner, and were then paired end-sequenced (2*300) on a MiSeq platform. MiSeq sequencing was performed at Genewiz Genomic Institute (Soochow, China).
Sequence analysis was conducted with the software QIIME. The primers were removed, and any sequences of a length < 220 or > 500 bp, mean quality score < 20, and the number of nitrogenous bases > 3 were removed from further analysis. Operational taxonomic units (OTUs) with a 97% similarity cutoff were clustered using UPARSE (version 7.1); USEARCH was used to filter chimeras, and the remaining sequences were clustered to generate OTUs at the 97% similarity level (Edgar Citation2010). The Mothur software was used for the calculation of species diversity (Chao and Shannon indices) using 35,000 sequence reads randomly selected from each sample. A representative sequence of each OTU was assigned to a taxonomic level in the Ribosomal Database Project (RDP) database using the RDP classifier (Maidak et al. Citation1997).
Statistical analysis
The SPSS 22.0 analytical software package and Excel 2016 were used for the statistical analyses. One-way analysis of variance (ANOVA) with a least significant difference (LSD) test was used to evaluate the differences of dependent variables. The P < 0.05 level was considered to be significant. In addition, multivariate analyses of the correlations between POM fractions and soil fungal communities composition were conducted using RDA with CANOCO 5.0 software. A manual forward-selection procedure was implemented in the RDA to determine the significance of the influence of environmental variables on the microbial community structure (P < 0.05) by a Monte Carlo test with 999 permutations.
Results
Changes in soil organic carbon and its chemical composition
As shown in , straw return significantly increased the SOC content. Relative to control, EIS treatment had the highest SOC content (17.31 and 14.55 g kg−1) in 0–20 cm and 20–40 cm depths, followed by SM (16.15 and 13.89 g kg−1) and SP (16.11 and 13.94 g kg−1). In addition, some differences in the chemical composition of SOC were also found between different treatments in 0–20 cm and 20–40 cm soil. In 0–20 cm soil, straw return treatments increased the percentages of O-alkyl C, Carbohydrate C, Di-O-alkyl C, whereas decreased Aryl C compared to control. For each straw return treatment, EIS had the highest percentages of Methoxyl C, Carbohydrate C, Di-O-alkyl C, and O-alkyl C, while SM and SP had similar percentages of each composition. In 20–40 cm, only EIS had higher relative contents of carbohydrate C, di-O-alkyl C, and O-alkyl C and lower percentages of aryl C than control. Moreover, the Aryl C in each straw returning treatments were more increased in 20–40 cm than 0–20 cm soil depth, while the opposite results were obtained in alkoxy C.
Table 2. Soil organic carbon contents and relative C distributions of Alkyl-C, O-alkyl-C, Aryl-C, and Carbonyl-C under different straw returning methods at 0–20 and 20–40 cm depth
Changes in particulate organic matter
In each particulate organic matter (POM) fractions, the fine POM in free microaggregates and microaggregates-within-macroaggregates had the lowest proportion of total soil in 0–20 and 20–40 cm depth, but the proportion of M(f)POM (0.53%-1.61%) was higher than Free(f)POM (0.16%-0.37%) in 0–20 cm soil depth ()). Compared with CK, straw return significantly increased the proportions of MA(c)POM, Fm-POM, M(f)POM, and mM-POM in 0–20 and 20–40 cm soil depth. The proportions of the mM-POM showed the highest increment in the SP, EIS, and SM treatments, which was 3.2, 5.2, and 3.1 times greater than those in the control in 0–20 cm and 2.1, 2.4, and 2.85 times in 20–40 cm.
Figure 2. Mass proportion (a) and organic carbon contents (b) of different particulate organic matter (POM) fractions under different straw returning methods at 0–20 and 20–40 cm depths
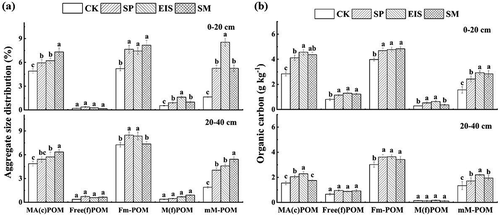
Across all experimental treatments, the C content of each POM fraction was much lower in 20–40 cm than 0–20 cm soil depth ()). For each POM fractions, Fm-POM had the highest C content (3.02–5.57 g kg−1) in 0–40 cm soil depth, followed by MA(c)POM (1.61–4.57 g kg−1), mM-POM (1.34–2.94 g kg−1), Free(f)POM (0.66–1.30 g kg−1) and M(f)POM (0.25–0.64 g kg−1). Relative to the control, all straw return treatments increased C concentrations in all POM fractions except M(f)POM in 20–40 cm soil depth. Interestingly, EIS was observed to have the highest C concentration in MA(c)POM and Mm-POM in 0–20 and 20–40 cm soil depth, which 38.7% and 86.6% higher than CK in 0–20 cm depth, respectively; in the 20–40 cm depth, the corresponding values were 19.5% and 23.1%.
Soil fungal community diversity
The relative abundance (RAs) of the dominant fungal phyla were Ascomycota (47.59–67.71%), Basidiomycota (5.57–10.89%), and Zygomycota (3.2–6.38%) in all soil samples ()). Compared with CK, Ascomycota was more abundant in EIS and scantier in SP in 0–20 cm, but there were lower in the straw return treatments in the 20–40 cm depth. The Basidiomycota and Zygomycota were more abundant in EIS and SM than in CK and SP in 0–20 cm, while in 20–40 cm, their abundance in EIS did not differ from that in CK or SP, only SM had a higher abundance of Basidiomycetes and Zygomycota. For other fungal taxa, the phyla of Chytridiomycota and Glomeromycota have a lower abundance in CK. However, EIS and SM increased by 67.5% and 34.6% abundance of Chytridiomycota in 0–20 cm,and increased 7.3 times and 17.5 times the abundance of Glomeromycota in 20–40 cm.
Figure 3. Relative abundance of soil fungal (a) phyla and (B) class with different straw returning methods at 0–20 and 20–40 cm depths
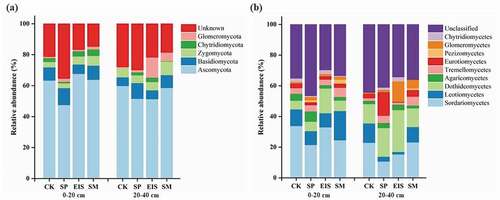
At the class level, Sordariomycetes (21.52–33.88%) and Leotiomycetes (9.04–19.1%) were the dominant fungal class in 0–20 cm soil depth, whereas Sordariomycetes (10.69%-23.17%) and Dothideomycetes (12.2%-27.32%) had high levels of fungi in 20–40 cm ()). Compared with CK, SM and SP have lower Sordariomycetes abundance in 0–20 cm, while SP and EIS also have a lower abundance of Sordariomycetes and Leotiomycetes in 20–40 cm. In addition, Dothideomycetes were more frequently observed in EIS in 0–20 and 20–40 cm soil depths (16.16% and 26.2%, respectively), and Glomeromycetes were also dominant in EIS in 20–40 cm (12.96%). Moreover, SP has the highest abundance of Eurotiomycetes in 20–40 cm relative to all treatments (15.31%).
Relationships between the soil fungal community and soil organic carbon structure
The redundancy analysis (RDA) was conducted to further investigate the influences of different soil fungal communities on soil organic carbon structure. In 0–20 cm soil depth, the abundance of fungal phylum-Zygomycota was positively correlated with O-alkyl C and Carbohydrate C, and negatively correlated with aryl C; Ascomycota was positively correlated with Alkyl C and negatively correlated with Carbonyl C (). In contrast, Basidiomycota was positively correlated with Carbonyl C. Meanwhile, the abundance of Glomeromycota was positively correlated with Alkyl C accumulation, whereas negatively correlated with aryl C in 20–40 cm soil; and Chytridiomycota was positively correlated with Di-O-alkyl C and O-alkyl C.
Figure 4. Redundancy analysis (RDA) of soil fungal communities and SOC fractions under different straw returning methods at 0–20 and 20–40 cm depths
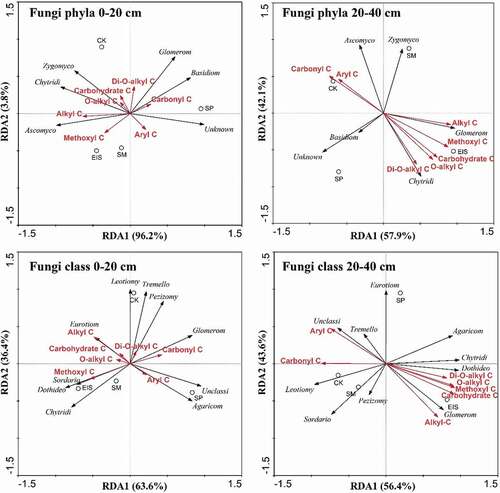
With the fungal class level, the abundance of Sordariomycetes and Dothideomycetes were positively correlated with Methoxyl C and negatively correlated with Carbonyl C in 0–20 cm depth, but Glomeromycetes was positively correlated with Carbonyl C in 0–20 cm depth. Meanwhile, Eurotiomycetes significantly influenced Alkyl C, O-alkyl C, and Carbohydrate C, and Agaricomycetes significantly affected Aryl C in 0–20 cm depth. In 20–40 cm depth, Dothideomycetes and Glomeromycetes were significantly influenced Alkyl C, O-alkyl C, Di-O-alkyl C, Methoxyl C, and Carbohydrate C, and no affected-on Aryl C.
Relationships between the soil fungal community and soil particulate organic matter
RDA showed that the influences of different soil fungal communities on soil particulate organic matter. In 0–20 cm soil depth, the abundance of fungal phylum-Zygomycota was positively correlated with mM-POM, Free(f)POM, and MA(c)POM, and negatively correlated with M(f)POM; Ascomycota and Chytridiomycota were positively correlated with M(f)POM (). In 20–40 cm soil depth, Glomeromycota positively correlated with mM-POM, and Chytridiomycota was positively correlated with MA(c)POM, Fm-POM, Free(f)-POM, M(f)POM. In contrast, Ascomycota almost negatively correlated with all soil POM fractions. Moreover, almost each POM fraction was positively correlated with the Chytridiomycota and Glomeromycota in all depths under EIS treatment but negatively correlated under CK treatment.
Figure 5. Redundancy analysis (RDA) of soil fungal communities and POM fractions under different straw returning methods at 0–20 and 20–40 cm depths
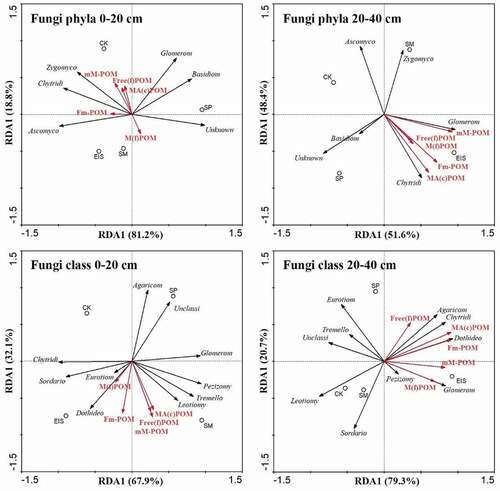
With the fungal class level, Dothideomycetes and Eurotiomycetes were positively correlated with M(f)POM and Fm-POM, Leotiomycetes was positively correlated with MA(c)POM, mM-POM, Free(f)-POM, but Agaricomycetes almost negatively correlated with all soil POM fractions. In 20–40 cm soil depth, Glomeromycetes was positively correlated with M(f)POM and mM-POM, Dothideomycetes, Agaricomycetes, and Chytridiomycetes were positively correlated with Fm-POM, MA(c)POM and Free(f)-POM.
Discussion
Effects of straw returning methods on SOC accumulation
Soil organic carbon (SOC) is an important index to evaluate soil fertility and soil quality, numerous studies have shown that returning crop straw to the field is beneficial to the storage and accumulation of SOC in soil (Han et al. Citation2016). In this study, each straw returning treatment significantly increased the SOC content, especially in the 0–20 cm soil layer. This result is similar to the finding of Kubar et al. (Citation2019), they found that the surface soil had more air and water circulation than deeper soil. Jin et al. (Citation2020) also found that the higher air and water content can help in the growth of the aerobic organisms present in the soil and accelerate the straw decomposition and SOC content increased. Meanwhile, EIS significantly increased SOC content than other straw returning treatments in all soil depths, which may be related to the higher crushing degree of straw by crashing-ridging technology. Cui, Li, and Wang (Citation2017) reported that the original organizational structure of the straw is broken to a certain extent by increasing the crushing degree of the straw, which destroys the protective effect of the cuticle on the outer surface of the straw, accelerates the decomposition, and nutrient release of the straw, and finally shortens the duration of straw decomposition. Moreover, the lower SOC contents in SP and SM in 20–40 cm soil layer also suggested that the traditional straw returning method has a weak influence on deep soil.
Besides quantitative changes in SOC, the quality of SOC fractions also plays an important role in soil quality and C sequestration. Hu et al. (Citation2018) and Kubar et al. (Citation2019) found the application of maize straw decreased the relative contents of alkyl C and aromatic C, whereas increased O-alkyl C of SOC fractions in the long-term fertilization. In our results, straw returning increased the percentages of O-alkyl C, Carbohydrate C, and Di-O-alkyl C in soil, whereas decreased aryl C compared to control in 0–20 cm soil depth. This result is mainly related to straw decomposition progressed. When straw is decomposed, the relative decreases were found in O-alkyl C and di-O-alkyl C contents, and relative increases in phenolic C and aromatic C contents (Zhang et al. Citation2011). Xu et al. (Citation2017) demonstrated that straw decomposition was mainly due to losses of O-alkyl C, di-O-alkyl C, and carbonyl C, and this process also effectively promoted the increase of O-alkyl C, di-O-alkyl C in soil. The aryl C was found significantly increased in 20–40 cm in straw returning treatments. This is similar to the finding of Rumpel, Eusterhues, and Kogel-Knabner (Citation2010) who observed that the soil SOC fractions contain a high proportion of aromatic carbon with strong inertness in deep soil, and the content of aryl C will increase with the increase of soil depth, which also shows that the influence of straw returning on SOC fractions will gradually weaken with the increase of soil depth. For each straw return treatment, EIS had the highest percentages of Methoxyl C, Carbohydrate C, Di-O-alkyl C, and O-alkyl C in both soil depths. This result suggested that the SOC fractions in EIS contain more structures with low oxidation degree and easily decomposable (O-alkyl C). O-alkyl C is considered to be a relatively easily decomposable fraction, which is mainly due to a large amount of fresh organic matter entering into the surface soil (Zhang et al., Citation2015). The carbohydrates produced by the decomposition of cellulose and hemicellulose in crop straws are beneficial to the production of O-alkyl C in SOC fractions (Plaza et al. Citation2012). EIS through crashing-ridging technology effectively promotes the close contact between crushed straw and soil, promotes the growth of crop roots, and stimulates the activity of soil microorganisms. Therefore, compared with other methods of straw returning to the field, EIS is more conducive to the accumulation of soil organic carbon.
Effects of straw returning methods on particulate organic matter
Particulate organic matter (POM) is the product of organic matter decomposed in the soil, which is stabilized on the mineral surface through various organo-mineral binding reactions (such as ligand exchange, cation bridging, hydrogen bonding, and Van der Waals force) (Xie et al. Citation2014). POM occupies most of the total soil carbon and has the characteristics of long turnover time in the soil (Feng et al. Citation2014). Therefore, it is considered as a passive or stable carbon pool with chemically protected (Benbi, Boparai, and Brar Citation2014). In our study, straw return significantly increased the proportions of MA(c)POM, Fm-POM, M(f)POM, and mM-POM in 0–20 and 20–40 cm soil depth in free microaggregates and macroaggregates, and it also significantly increases the C content of these POM fractions compared to CK. These findings demonstrated that interactions of maize straw and soil minerals indeed occurred, and the input of exogenous organic carbon plays a very important role in the formation of POM in macroaggregates and microaggregates (Guan et al. Citation2019). Meanwhile, our results also showed that the C content and proportions of light POM fraction (M(f)POM and Free(f)POM) was relatively lower in soil macroaggregates and microaggregates, which was consistent with earlier findings by Leifeld and Kogel-Knabner (Citation2005). Light fractions within the occluded and free microaggregates consist of less decomposed organic residues and are possessed in higher POC concentrations (Zhang et al. Citation2018). However, they usually showed a higher degree of degradation and faster C turnover, and therefore had lower proportions inside the occluded and free microaggregates (Zaccone et al. Citation2018). In this study, SP, EIS, and SM had a similar increment of the Fm-POM proportions and C contents. Fm-POM is considered as an inert organic carbon pool with relatively strong stability, which could act as a cementing agent for the formation of stable microaggregates (Watteau et al. Citation2012). Previous studies have demonstrated that the physical blockage of Fm-POM in the microaggregate enhances its protective effect on microbial decomposition, and ultimately promotes long-term C sequestration (Guan et al. Citation2018). This suggested that Fm-POM is less affected by different cultivation measures and straw returning methods. In addition, the present study showed that EIS had the highest C contents of MA(c)POM and mM-POM, SM had the highest proportions of MA(c)POM than other treatments in 0–20 cm and 20–40 cm soil depth. MA(c)POM and mM-POM are mainly comprised of newly plant residue input, which is relatively stable, has a faster turnover rate and strong physical protection effects on POC, and important for C sequestration (O’Brien and Jastrow Citation2013). Therefore, these results indicate that EIS crushed the straw and mixed it fully with the soil to better promote the combination of straw and soil primary particles, and effectively accelerate the enrichment of organic carbon to coarse POM and microaggregates within macroaggregates. SM increased the soil water contents and reduced the soil disturbance, helping the formation and stability of coarse POM in macroaggregates.
Effects of straw returning methods on soil fungal community
Fungi are very important in the soil ecosystem and perform a series of important ecological functions, such as pathogenesis, symbiotic, and decomposition, and also affect plant growth, nutrient transfer, and the recycling of soil organic matter and mineral nutrients (Yang et al. Citation2020). In our study, different straw returning methods changed the soil fungal community at different taxonomic levels. At the phylum level, the greater abundance of Ascomycota was recorded in EIS treatment and the lowest was recorded in CK and SP treatments, and the higher abundance of Basidiomycota and Zygomycota were found in EIS and SM treatment in 0–20 cm. Ascomycota and Basidiomycota are considered to be the key decomposer in agricultural soils (Sun et al. Citation2016). Lentendu et al. (Citation2014) found that the abundance of Ascomycota and Basidiomycota can be flourished by the input of organic matter into the soil. At our sampling soil depth, EIS has accelerated the maize straw thoroughly mixed with soils under the crashing-ridging technology, thus possibly stimulating the growth and proliferation of Ascomycota and Basidiomycota. The change in soil water storage and soil temperature also important for the abundance of the fungal community (Qiu et al. Citation2020). Dong et al. (Citation2017) recorded that the greater relative abundance of Zygomycota was found in the straw mulching treatment due to increased soil temperature. The application of straw mulch has the advantage of storing more soil water and increased soil temperature on the surface soil effectively increased the abundance of Basidiomycota and Zygomycota in 0–20 cm soil depth. In addition, EIS also increased the abundance of Glomeromycota in 20–40 cm soil depth. Some recent studies reported that both Glomeromycota are negatively correlated with soil bulk density, and they can survive anaerobic conditions but need to grow under high oxygen concentrations (Qiu et al. Citation2020). EIS through the crashing-ridging technology decreased the soil bulk density and increased the soil porosity, accelerate the increase of oxygen concentrations at deep soil, thus promoting the increased Glomeromycota in 0–20 cm.
At the class level, Sordariomycetes, Leotiomycetes, and Dothideomycetes were the three dominant fungal classes in 0–20 cm and 20–40 cm soil depth. Similar results were found in the saline-sodic soils and black soils by Wang et al. Citation2019) and Zhou et al. (Citation2016). Previous studies reported that Sordariomycetes and Dothideomycetes have a significant positive correlation with soil organic carbon structure and are important to global carbon cycling as a saprophyte and degrader of plant biomass (Ren et al. Citation2018). We found that EIS had the highest abundance of Dothideomycetes in 0–20 and 20–40 cm soil depths, this indicated that EIS effectively promotes the accumulation and increase of soil organic matter and improves the structure of soil organic matter. Meanwhile, EIS also had the highest abundance of Glomeromycetes in 20–40 cm soil depth, this result is similar to the increase of Glomeromycota, they are all affected by the soil oxygen concentrations. In this study, SM treatment contained more abundance of Leotiomycetes in 0–20 cm soil depth. Leotiomycetes contain common root fungi and have been shown to harvest and provide their host plants with nutrients immobilized in organic matter (Zheng et al. Citation2018). Zheng et al. (Citation2018) also found that Leotiomycetes can secrete β-xylosidase to degrade cover crop residues. This result indicated that straw mulching promoted the growth of Leotiomycetes, and the growth of Leotiomycetes also significantly promoted the decomposition of straw. Also, SP has the highest abundance of Eurotiomycetes in 20–40 cm relative to all treatments, which is probably related to the change of soil pH after straw plowing (Mothapo et al. Citation2015; Wang et al. Citation2019).
Relating fungal community structure to organic C accumulation and particulate organic matter in the soil
In this study, we investigated the effects of different straw returning methods on soil fungal communities and their relationship to SOC fractions and structure. The RDA showed that the fungal phyla of Zygomycota and Chytridiomycota and the fungal class of Eurotiomycetes, Dothideomycetes and Leotiomycetes (Ascomycota) were positively correlated with O-alkyl C and Carbohydrate C, and positively correlated with mM-POM, Free(f)POM, and MA(c)POM in 0–20 cm soil depth. These results suggested that Ascomycota, Zygomycota, and Chytridiomycota have a close relationship with the formation of soil macroaggregates and the increase of aliphatic structure in soil organic carbon. This finding is somewhat different from previous studies, some studies have believed that only Ascomycota is an important factor affecting the formation of soil aggregates and carbon sequestration. However, Xiao et al. (Citation2018) found that Zygomycota also had a key role affect O-alkyl C and Carbohydrate C in the soil organic carbon structure, which indirectly affects the combination of soil organic carbon and soil primary particles, thereby affecting the distribution of particulate organic carbon in aggregates of different sizes. In contrast to the above results, Agaricomycetes significantly affected Aryl C, and almost negatively correlated with all soil POM fractions. Agaricomycetes were the most conspicuous group among Basidiomycota, which have the broadest impact in the forest ecosystem (Ren et al. Citation2018). The previous study has shown that Agaricomycetes were negatively correlated with SOC fractions and respiration, which was consistent with our results, indicated that Agaricomycetes were not conducive to the SOC accumulation in surface soil (Wang et al. Citation2019).
Different from the results shown in the surface soil, in the 20–40 cm soil depth, the RDA results showed that the Chytridiomycota and Glomeromycota have a positive correlation with almost all the soil particle organic matter components, and at the same time, they also have a significant positive correlation with all soil organic carbon functional groups other than the Aryl C and Carbonyl C. Although Chytridiomycota and Glomeromycota account for a relatively less proportion of the fungal community, the study of Garcia-Franco et al. (Citation2015) found that Chytridiomycota and Glomeromycota have a strong enrichment effect on soil organic carbon content and positive correlation with SOC contents. Further research has shown that Chytridiomycota and Glomeromycota can accelerate the decompose of cellulose and lignin and the associated production of binding agents, such as polysaccharides, and they themselves have enhanced the formation of macroaggregates from microaggregates (Cui et al. Citation2016; Qiu et al. Citation2020). Our results also indicate that O-alkyl C and carbohydrate C and soil particulate organic matter were most favorable under EIS treatments below 20 cm of soil respectively. This may be the same as our previous inferences. EIS increases soil porosity, reduces soil bulk density, and promotes air circulation in the deep soil. These changes promote the reproduction of these two fungi, which significantly promotes the alkylation of organic matter structure and the formation of soil particulate organic matter compared with other treatments.
Conclusion
Our study demonstrated that straw return significantly increased bulk soil C content, affected the chemical composition of SOC, accelerated the mass proportion and OC contents of each POM fractions, and changed the soil fungal community composition in the different soil depths. Comparing the straw returning treatments, EIS treatment significantly increased the concentrations of SOC and the proportion of Carbohydrate C, Di-O-alkyl C, and O-alkyl C in SOC structure, increased the mass proportion and OC contents of MA(c)-POM and mM-POM in the upper 40 cm of soil. Meanwhile, EIS treatment increased the relative abundance of Ascomycota, Zygomycota, Chytridiomycota, and Dothideomycetes in 0–20 cm depths, and also had the highest relative abundance of Glomeromycetes and Dothideomycetes in the 20–40 cm soil compared with the other treatments. SP treatment did not have any significant impact on SOC and POM fractions, only has the highest abundance of Eurotiomycetes in 20–40 cm relative to all treatments. SM treatment presented similar results to EIS treatment in terms of the C concentrations of SOC, MA(c)-POM, mM-POM, and Fm-POM, and had the higher fungal diversity in the topsoil and subsoil. Our study also provides evidence that straw return enhanced the relative abundances of fungi involved in the carbon cycle and sequestration, including Zygomycota, Chytridiomycota, and Glomeromycota, and Ascomycota. The shifts in fungal community structure can accelerate organic C accumulation and the formation of soil particulate organic matter, especially in EIS treatment.
Additional information
Funding
References
- Benbi, D. K., A. K. Boparai, and K. Brar. 2014. “Decomposition of Particulate Organic Matter Is More Sensitive to Temperature than the Mineral Associated Organic Matter.” Soil Biology & Biochemistry 70: 183–13. doi:https://doi.org/10.1016/j.soilbio.2013.12.032.
- Bu, R., T. Ren, M. Lei, B. Liu, X. Li, R. Cong, and J. Lu. 2020. “Tillage and Straw-returning Practices Effect on Soil Dissolved Organic Matter, Aggregate Fraction and Bacteria Community under Rice-rice-rapeseed Rotation System.” Agriculture, Ecosystems & Environment 287: 106681.
- Chen, X., Y. Xu, H. J. Gao, J. Mao, W. Chu, and M. L. Thompson. 2018. “Biochemical Stabilization of Soil Organic Matter in Straw-amended, Anaerobic and Aerobic Soils.” Science of the Total Environment 625: 1065–1073. doi:https://doi.org/10.1016/j.scitotenv.2017.12.293.
- Chen, Z., H. Wang, X. Liu, X. Zhao, D. Lu, J. Zhou, and C. Li. 2017. “Changes in Soil Microbial Community and Organic Carbon Fractions under Short-term Straw Return in a Rice–wheat Cropping System.” Soil and Tillage Research 165: 121–127. doi:https://doi.org/10.1016/j.still.2016.07.018.
- Chivenge, P., B. Vanlauwe, R. Gentile, and J. Six. 2011. “Organic Resource Quality Influences Short-term Aggregate Dynamics and Soil Organic Carbon and Nitrogen Accumulation.” Soil Biology & Biochemistry 43 (3): 657–666. doi:https://doi.org/10.1016/j.soilbio.2010.12.002.
- Cui, T., Z. Li, and S. Wang. 2017. “Effects of In-situ Straw Decomposition on Composition of Humus and Structure of Humic Acid at Different Soil Depths.” Journal of Soils and Sediments 17 (10): 2391–2399. doi:https://doi.org/10.1007/s11368-017-1704-6.
- Cui, X., J. Hu, J. Wang, J. Yang, and X. Lin. 2016. “Reclamation Negatively Influences Arbuscular Mycorrhizal Fungal Community Structure and Diversity in Coastal Saline-alkaline Land in Eastern China as Revealed by Illumina Sequencing.” Applied Soil Ecology 98: 140–149. doi:https://doi.org/10.1016/j.apsoil.2015.10.008.
- Demyan, M. S., F. Rasche, E. Schulz, M. Breulmann, T. Müller, and G. Cadisch. 2012. “Use of Specific Peaks Obtained by Diffuse Reflectance Fourier Transform Mid‐infrared Spectroscopy to Study the Composition of Organic Matter in a Haplic Chernozem.” European Journal of Soil Science 63 (2): 189–199. doi:https://doi.org/10.1111/j.1365-2389.2011.01420.x.
- Dong, W. Y., E. K. Liu, C. R. Yan, J. Tian, H. H. Zhang, and Y. Q. Zhang. 2017. “Impact of No Tillage Vs Conventional Tillage on the Soil Bacterial Community Structure in a Winter Wheat Cropping Succession in Northern China.” European Journal of Soil Biology 80: 35–42.
- Edgar, R. C. 2010. “Search and Clustering Orders of Magnitude Faster than BLAST.” Bioinformatics 26: 2460–2461. doi:https://doi.org/10.1093/bioinformatics/btq461.
- Fan, W., J. Wu, J. Li, and J. Hu. 2018. “Comparative Effects of Different Maize Straw Returning Modes on Soil Humus Composition and Humic Acid Structural Characteristics in Northeast China.” Chemistry and Ecology 34 (4): 355–370. doi:https://doi.org/10.1080/02757540.2018.1437147.
- Feng, W., A. F. Plante, A. K. Aufdenkampe, and J. Six. 2014. “Soil Organic Matter Stability in Organo-mineral Complexes as a Function of Increasing C Loading.” Soil Biology & Biochemistry 69: 398–405. doi:https://doi.org/10.1016/j.soilbio.2013.11.024.
- Garcia-Franco, N., M. Martínez-Mena, M. Goberna, and J. Albaladejo. 2015. “Changes in Soil Aggregation and Microbial Community Structure Control Carbon Sequestration after Afforestation of Semiarid Shrublands.” Soil Biology & Biochemistry 87: 110–121. doi:https://doi.org/10.1016/j.soilbio.2015.04.012.
- Guan, S., N. An, N. Zong, Y. He, P. Shi, J. Zhang, and N. He. 2018. “Climate Warming Impacts on Soil Organic Carbon Fractions and Aggregate Stability in a Tibetan Alpine Meadow.” Soil Biology & Biochemistry 116: 224–236. doi:https://doi.org/10.1016/j.soilbio.2017.10.011.
- Guan, S., S. J. Liu, R. Y. Liu, J. J. Zhang, J. Ren, H. G. Cai, and X. X. Lin. 2019. “Soil Organic Carbon Associated with Aggregate-size and Density Fractions in a Mollisol Amended with Charred and Uncharred Maize Straw.” Journal of Integrative Agriculture 18: 1496–1507. doi:https://doi.org/10.1016/S2095-3119(19)62643-2.
- Han, L., K. Sun, J. Jin, and B. Xing. 2016. “Some Concepts of Soil Organic Carbon Characteristics and Mineral Interaction from a Review of Literature.” Soil Biology & Biochemistry 94: 107–121. doi:https://doi.org/10.1016/j.soilbio.2015.11.023.
- Han, X., F. Zhao, X. Tong, J. Deng, G. Yang, L. Chen, and D. Kang. 2017. “Understanding Soil Carbon Sequestration following the Afforestation of Former Arable Land by Physical Fractionation.” Catena 150: 317–327. doi:https://doi.org/10.1016/j.catena.2016.11.027.
- Hu, J., J. Wu, X. Qu, and J. Li. 2018. “Effects of Organic Wastes on Structural Characterizations of Humic Acid in Semiarid Soil under Plastic Mulched Drip Irrigation.” Chemosphere 200: 313–321. doi:https://doi.org/10.1016/j.chemosphere.2018.02.128.
- IUSS Working Group WRB. 2015. World Reference Base for Soil Resources 2014. Rome, Italy: update 2015 International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. Food and Agriculture Organization of the United Nations.
- Jiang, D., D. Zhuang, J. Fu, Y. Huang, and K. Wen. 2012. “Bioenergy Potential from Crop Residues in China: Availability and Distribution.” Renewable and Sustainable Energy Reviews 16 (3): 1377–1382. doi:https://doi.org/10.1016/j.rser.2011.12.012.
- Jin, Z., T. Shah, L. Zhang, H. Liu, S. Peng, and L. Nie. 2020. “Effect of Straw Returning on Soil Organic Carbon in Rice-wheat Rotation System: A Review.” Food and Energy Security 9: 200. doi:https://doi.org/10.1002/fes3.200.
- Kubar, K. A., L. Huang, J. Lu, X. Li, B. Xue, and Z. Yin. 2019. “Long-term Tillage and Straw Returning Effects on Organic C Fractions and Chemical Composition of SOC in Rice-rape Cropping System.” Archives of Agronomy and Soil Science 65 (1): 125–137. doi:https://doi.org/10.1080/03650340.2018.1490726.
- Larkin, R. P., C. W. Honeycutt, T. S. Griffin, O. M. Olanya, J. M. Halloran, and Z. He. 2011. “Effects of Different Potato Cropping System Approaches and Water Management on Soilborne Diseases and Soil Microbial Communities.” Phytopathology 101: 58–67. doi:https://doi.org/10.1094/PHYTO-04-10-0100.
- Leifeld, J., and I. Kogel-Knabner. 2005. “Soil Organic Matter Fractions as Early Indicators for Carbon Stock Changes under Different Land-use?” Geoderma. 124: 143–155.
- Lentendu, G., T. Wubet, A. Chatzinotas, C. Wilhelm, F. Buscot, and M. Schlegel. 2014. “Effects of Long-term Differential Fertilization on Eukaryotic Microbial Communities in an Arable Soil: A Multiple Barcoding Approach.” Molecular Ecology 23: 3341–3355. doi:https://doi.org/10.1111/mec.12819.
- Liang, B., C. Ma, L. Fan, Y. Wang, and Y. Yuan. 2018. “Soil Amendment Alters Soil Physicochemical Properties and Bacterial Community Structure of a Replanted Apple Orchard.” Microbiological Research 216: 1–11. doi:https://doi.org/10.1016/j.micres.2018.07.010.
- Maidak, B. L., G. J. Olsen, N. Larsen, R. Overbeek, M. J., . McCaughey, and C. R. Woese. 1997. “The RDP (Ribosomal Database Project).” Nucleic Acids Research 25: 109–111. doi:https://doi.org/10.1093/nar/25.1.109.
- Mothapo, N., H. Chen, M. A. Cubeta, J. M. Grossman, F. Fuller, and W. Shi. 2015. “Phylogenetic, Taxonomic and Functional Diversity of Fungal Denitrifiers and Associated N2O Production Efficacy.” Soil Biology & Biochemistry 83: 160–175. doi:https://doi.org/10.1016/j.soilbio.2015.02.001.
- O’Brien, S. L., and J. D. Jastrow. 2013. “Physical and Chemical Protection in Hierarchical Soil Aggregates Regulates Soil Carbon and Nitrogen Recovery in Restored Perennial Grasslands.” Soil Biology & Biochemistry 61: 1–13. doi:https://doi.org/10.1016/j.soilbio.2013.01.031.
- Plaza, C., D. Courtier-Murias, J. M. Fernández, A. Polo, and A. J. Simpson. 2013. “Physical, Chemical, and Biochemical Mechanisms of Soil Organic Matter Stabilization under Conservation Tillage Systems: A Central Role for Microbes and Microbial By-products in C Sequestration.” Soil Biology & Biochemistry 57: 124–134. doi:https://doi.org/10.1016/j.soilbio.2012.07.026.
- Plaza, C., J. M. Fernández, E. I. Pereira, and A. Polo. 2012. “A Comprehensive Method for Fractionating Soil Organic Matter Not Protected and Protected from Decomposition by Physical and Chemical Mechanisms. CLEAN–Soil.” Air, Water 40 (2): 134–139. doi:https://doi.org/10.1002/clen.201100338.
- Qiu, Y., W. Lv, X. Wang, Z. Xie, and Y. Wang. 2020. “Long-term Effects of Gravel Mulching and Straw Mulching on Soil Physicochemical Properties and Bacterial and Fungal Community Composition in the Loess Plateau of China.” European Journal of Soil Biology 98: 103188. doi:https://doi.org/10.1016/j.ejsobi.2020.103188.
- Ren, C., T. Wang, Y. Xu, J. Deng, F. Zhao, G. Yang, G. Ren, Y. Feng, and G. Ren. 2018. “Differential Soil Microbial Community Responses to the Linkage of Soil Organic Carbon Fractions with Respiration across Land-use Changes.” Forest Ecology and Management 409: 170–178. doi:https://doi.org/10.1016/j.foreco.2017.11.011.
- Rumpel, C., K. Eusterhues, and I. Kogel-Knabner. 2010. “Non-cellulosic Neutral Sugar Contribution to Mineral Associated Organic Matter in Top- and Subsoil Horizons of Two Acid Forest Soils.” Soil Biology & Biochemistry 42: 379–382. doi:https://doi.org/10.1016/j.soilbio.2009.11.004.
- Sarker, J. R., B. P. Singh, A. L. Cowie, Y. Fang, D. Collins, W. J. Dougherty, and B. K. Singh. 2018. “Carbon and Nutrient Mineralisation Dynamics in Aggregate-size Classes from Different Tillage Systems after Input of Canola and Wheat Residues.” Soil Biology & Biochemistry 116: 22–38. doi:https://doi.org/10.1016/j.soilbio.2017.09.030.
- Six, J. Α. Ε. Τ., Elliott, E. T., & Paustian, K. 2000. “Soil Macroaggregate Turnover and Microaggregate Formation: A Mechanism for C Sequestration under No-tillage Agriculture.” Soil Biology & Biochemistry 32 (14): 2099–2103.
- Soil Survey Staff. 1999. Soil Taxonomy: A Basic System of Soil Classification for Making and Interpreting Soil Surveys. 2nd ed. Washington D.C., USA: Natural Resources Conservation Service. U.S. Department of Agriculture Handbook 436.
- Sun, R., M. Dsouza, J. A. Gilbert, X. Guo, D. Wang, Z. Guo, H. Chu, and H. Chu. 2016. “Fungal Community Composition in Soils Subjected to Long‐term Chemical Fertilization Is Most Influenced by the Type of Organic Matter.” Environmental Microbiology 18 (12): 5137–5150. doi:https://doi.org/10.1111/1462-2920.13512.
- Vrdoljak, G., and G. Sposito. 2002. “Soil Aggregate Hierarchy in a Brazilian Oxisol.” Developments in Soil Science. Vol. 28, 197–217.
- Wang, S., S. Luo, X. Zhou, C. Chang, L. Tian, X. Li, C. Tian, Q. Gao, and C. Tian. 2019. “Soil Ameliorants Alter Physicochemical Properties and Fungal Communities in Saline-sodic Soils of Northeast China.” Archives of Agronomy and Soil Science 65 (8): 1147–1159. doi:https://doi.org/10.1080/03650340.2018.1555707.
- Watteau, F., G. Villemin, F. Bartoli, C. Schwartz, and J. L. Morel. 2012. “0-20 μm Aggregate Typology Based on the Nature of Aggregative Organic Materials in a Cultivated Silty Topsoil.” Soil Biology & Biochemistry 46: 103–114. doi:https://doi.org/10.1016/j.soilbio.2011.11.021.
- Xiao, X., Y. Liang, S. Zhou, S. Zhuang, and B. Sun. 2018. “Fungal Community Reveals Less Dispersal Limitation and Potentially More Connected Network than that of Bacteria in Bamboo Forest Soils.” Molecular Ecology 27 (2): 550–563. doi:https://doi.org/10.1111/mec.14428.
- Xie, H., J. Li, P. Zhu, C. Peng, J. Wang, H. He, and X. Zhang. 2014. “Long-term Manure Amendments Enhance Neutral Sugar Accumulation in Bulk Soil and Particulate Organic Matter in a Mollisol.” Soil Biology & Biochemistry 78: 45–53. doi:https://doi.org/10.1016/j.soilbio.2014.07.009.
- Xu, J., B. Zhao, W. Chu, J. Mao, and J. Zhang. 2017. “Chemical Nature of Humic Substances in Two Typical Chinese Soils (Upland Vs Paddy Soil): A Comparative Advanced Solid State NMR Study.” The Science of the Total Environment 576: 444. doi:https://doi.org/10.1016/j.scitotenv.2016.10.118.
- Yan, S., J. Song, J. Fan, C. Yan, S. Dong, C. Ma, and Z. Gong. 2020. “Changes in Soil Organic Carbon Fractions and Microbial Community under Rice Straw Return in Northeast China.” Global Ecology and Conservation 22: 00962. doi:https://doi.org/10.1016/j.gecco.2020.e00962.
- Yang, C., N. Liu, and Y. Zhang. 2019. “Soil Aggregates Regulate the Impact of Soil Bacterial and Fungal Communities on Soil Respiration.” Geoderma 337: 444–452. doi:https://doi.org/10.1016/j.geoderma.2018.10.002.
- Yang, H., Y. Li, S. Zhai, C. Fang, J. Liu, and Q. Zhang. 2020. “Long Term Ditch-buried Straw Return Affects Soil Fungal Community Structure and Carbon-degrading Enzymatic Activities in a Rice-wheat Rotation System.” Applied Soil Ecology 155: 103660. doi:https://doi.org/10.1016/j.apsoil.2020.103660.
- Zaccone, C., L. Beneduce, C. Lotti, G. Martino, and C. Plaza. 2018. “DNA Occurrence in Organic Matter Fractions Isolated from Amended, Agricultural Soils.” Applied Soil Ecology 130: 134–142. doi:https://doi.org/10.1016/j.apsoil.2018.06.011.
- Zhang, J., F. Hu, H. Li, Q. Gao, X. Song, X. Ke, and L. Wang. 2011. “Effects of Earthworm Activity on Humus Composition and Humic Acid Characteristics of Soil in a Maize Residue Amended Rice–wheat Rotation Agroecosystem.” Applied Soil Ecology 51: 1–8. doi:https://doi.org/10.1016/j.apsoil.2011.08.004.
- Zhang, L., H. Ma, H. Zhang, L. Xun, G. Chen, and L. Wang. 2015. “Thermomyces Lanuginosus Is the Dominant Fungus in Maize Straw Composts.”Bioresource Technology 197: 266–275. doi:https://doi.org/10.1016/j.biortech.2015.08.089
- Zhang, X., X. Xin, A. Zhu, W. Yang, J. Zhang, S. Ding, and L. Shao. 2018. “Linking Macroaggregation to Soil Microbial Community and Organic Carbon Accumulation under Different Tillage and Residue Managements.” Soil and Tillage Research 178: 99–107. doi:https://doi.org/10.1016/j.still.2017.12.020.
- Zhang, Y., S. Yao, J. Mao, D. C. Olk, X. Cao, and B. Zhang. 2015. “Chemical Composition of Organic Matter in a Deep Soil Changed with a Positive Priming Effect Due to Glucose Addition as Investigated by 13C NMR Spectroscopy.” Soil Biology & Biochemistry 85: 137–144. doi:https://doi.org/10.1016/j.soilbio.2015.03.013
- Zhao, S., K. Li, W. Zhou, S. Qiu, S. Huang, and P. He. 2016. “Changes in Soil Microbial Community, Enzyme Activities and Organic Matter Fractions under Long-term Straw Return in North-central China.” Agriculture, Ecosystems & Environment 216: 82–88. doi:https://doi.org/10.1016/j.agee.2015.09.028.
- Zheng, W., Z. Zhao, Q. Gong, B. Zhai, and Z. Li. 2018. “Effects of Cover Crop in an Apple Orchard on Microbial Community Composition, Networks, and Potential Genes Involved with Degradation of Crop Residues in Soil.” Biology and Fertility of Soils 54 (6): 743–759. doi:https://doi.org/10.1007/s00374-018-1298-1.
- Zhou, J., X. Jiang, B. Zhou, B. Zhao, M. Ma, D. Guan, J. Li, et al.. 2016. “Thirty-four Years of Nitrogen Fertilization Decreases Fungal Diversity and Alters Fungal Community Composition in Black Soil in Northeast China”. Soil Biology & Biochemistry 95: 135–143. doi:https://doi.org/10.1016/j.soilbio.2015.12.012.