Adipose lipolysis not regulated by estradiol
pp. 76–86
The steroid hormone estradiol's (E2) role in adipose lipolysis modulation is still unclear, however it is well known that E2 can influence physical activity, food intake and energy expenditure. Additionally, there is evidence of estradiol loss leading to shifts in subcutaneous to visceral fat storage. This research paper by Macdonald et al. explored the role of E2 with regards to adipose tissue lipolysis regulation through testing in both subcutaneous and visceral adipose depots in vivo through ovariectomized rats and ex vivo via direct acute E2 exposure. Estradiol did not seem to directly regulate adipose tissue lipolysis based on these tests.
Adipose morphology during collagen-induced arthritis
pp. 87–101
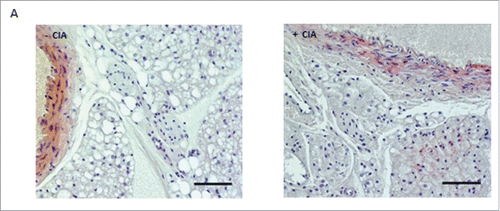
Patients with rheumatoid arthritis are often afflicted with a change in fat/lean mass ratio, which can reduce the effectiveness of anti-rheumatic drugs as well as anti-TNF therapy. This research paper by Sime, Choy and Williams presents a new model using collagen-induced arthritis (CIA) in DBA/1 mice in order to investigate the altered adipose function in hosts with inflammatory associated disease. Their novel approach describes morphological and molecular changes in perivascular adipose tissue (PVAT), white adipose tissue and brown adipose tissue in DBA/1 arthritis mice and identifies early stage biomarkers which may help detect or treat vascular dysfunction during arthritis ().
Adipocyte flow cytometry
pp. 112–123
Often used to identify pre-adipocytes, flow cytometry is able to analyze cellular heterogeneity in mixed populations. Authors Boumelhem et al. present a new flow cytometric protocol which improves live adipocyte analysis at the single cell level. This protocol requires no fixation or permeabilization, can quantify live adipocyte phenotype and frequency, and can characterize live adipocytes according to numerous factors such as lipid content, nuclear content, mitochondrial membrane depolarization and adipocyte surface protein phenotype ()
Obesity-dependent DNA methylation in adipocytes
pp. 124–133
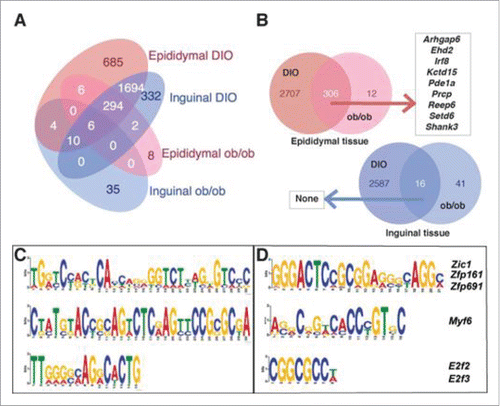
DNA methylation can be effected by obesity as well as insulin resistance, and an individual may be predisposed to obesity and diabetes due to excess of nutrients during fetal development. This study by Sonne et al. sought to identify genes which had changes associated with obesity in DNA methylation as well as gene expression in adipose tissue. Changes were identified using a genetic and diet-induced model, enabling the authors to differentiate between alterations due to high fat feeding from those associated with genetic obesity. Overall, nine genes were identified, including Irf8 which may be related to immune type 1/type 2 balance ()
Predictors to bariatric surgery success
pp. 134–140
Although bariatric surgery has shown to be effective, final weight loss results can still be unpredictable with many patients achieving suboptimal weight loss. This brief report by Muir et al. examines whether pre-bariatric surgery identified adipose tissue phenotypic features that were related to remodeling showed any correlation to optimal post-surgical weight loss. Several factors seemed to be associated with less weight loss such as type 2 diabetes, age, and male sex. Reduced adipose tissue preadipocytes was related with less weight loss in women but not men. These data support the hypothesis that certain phenotypic features of adipose tissue remodeling can serve as predictors and may help select optimal patients.
BMP4 may impair BAT differentiation
pp. 141–146
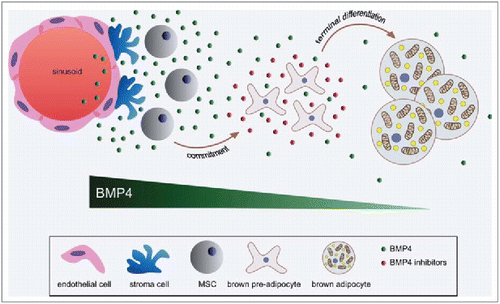
In this commentary by Modica and Wolfrum, the authors expand on their recent findings of additional bone morphogenic protein 4 (BMP4) function during adipogenesis. While its role in guiding mesenchymal stem cells into adipogenic and osteogenic linages is well known, it was found that BMP4 promotes white fat cell terminal differentiation during adipogenesis, instead of brown fat cells. Additionally, BMP4 can either promote or repress oxidative metabolism depending on cell context. Research such as this exploring factors that can impair brown adipose tissue (BAT) formation may lead to further breakthroughs in BAT regulation ()