ABSTRACT
Stimulator of interferon response cGAMP interactor 1 (STING1, best known as STING) is an endoplasmic reticulum-sessile protein that serves as a signaling hub, receiving input from several pattern recognition receptors, most of which sense ectopic DNA species in the cytosol. In particular, STING ensures the production of type I interferon (IFN) in response to invading DNA viruses, bacterial pathogens, as well as DNA leaking from mitochondria or the nucleus (e.g., in cells exposed to chemotherapy or radiotherapy). As a type I IFN is critical for the initiation of anticancer immune responses, the pharmaceutical industry has generated molecules that directly activate STING for use in oncological indications. Such STING agonists are being tested in clinical trials with the rationale of activating STING in tumor cells or tumor-infiltrating immune cells (including dendritic cells) to elicit immunostimulatory effects, alone or in combination with a range of established chemotherapeutic and immunotherapeutic regimens. In this Trial Watch, we discuss preclinical evidence and accumulating clinical experience shaping the design of Phase I and Phase II trials that evaluate the safety and preliminary efficacy of STING agonists in cancer patients.
Introduction
Stimulator of interferon response cGAMP interactor 1 (STING1, best known as STING) was first described in 2008 as a transmembrane component of the endoplasmic reticulum (ER) that senses cytosolic double-stranded DNA (dsDNA).Citation1–Citation4 This key adaptor protein in innate immune signalingCitation5–Citation8 can be activated by several cytoplasmic DNA sensorsCitation9–Citation11 including cyclic GMP-AMP synthase (CGAS),Citation12–Citation15 Z-DNA binding protein 1 (ZBP1, best known as DAI), DEAD-box helicase 41 (DDX41), interferon-gamma inducible protein 16 (IFI16),Citation16–Citation18 LRR binding FLII interacting protein 1 (LRRFIP1),Citation19,Citation20 MRE11 homolog, double-strand break repair nuclease (MRE11),Citation21 and perhaps protein kinase, DNA-activated, catalytic subunit (PRKDC, best known as DNA-PK),Citation22,Citation23 Among these sensors, CGAS has been studied in an extensive fashion. Mechanistically, the accumulation of ectopic dsDNA in the cytosol activates the enzymatic function of CGAS to generate cyclic GMP-AMP (cGAMP),Citation15,Citation24 as well as other cyclic dinucleotides (CDNs)Citation25,Citation26 bind to and activate STING, triggering a signal transduction pathway that culminates in the initiation of interferon regulatory factor 3 (IRF3)- or NF-κB-dependent transcriptional programs.Citation27–Citation30 Notably, CDN-bound STING also stimulates autophagy, an evolutionarily conserved mechanism for the preservation of cellular and organismal homeostasis,Citation31–Citation33 and such a function appears to be more ancient than the initiation of IRF3 and NF-κB transcriptional activity.Citation34
Figure 1. Overview of STING signaling in cancer cells. Accumulation of double-stranded DNA (dsDNA) in the cytosol of cancer cells responding to some chemotherapeutics or radiation therapy boosts the enzymatic functions of cyclic GMP-AMP synthase (CGAS), resulting in the cyclic GMP-AMP (cGAMP)-dependent oligomerization and activation of stimulator of interferon response cGAMP interactor 1 (STING1, best known as STING) at the endoplasmic reticulum (ER). Activated STING promotes a TANK binding kinase 1 (TAK1)-dependent signal transduction cascade that initiates interferon regulatory factor 3 (IRF3)- and NF-κB-dependent transcription, potentially culminating with the secretion of numerous cytokines including type I interferon (IFN), interleukin 6 (IL-6) and tumor necrosis factor (TNF). CDN, cyclic dinucleotide; IκBα (official name: NFKBIA), NFKB inhibitor alpha; IKKα (official name: CHUK), component of inhibitor of nuclear factor-kappa B kinase complex; IKKβ (official name: IKBKB), inhibitor of nuclear factor-kappa B kinase subunit beta; PARP, poly(ADP-ribose) polymerase.
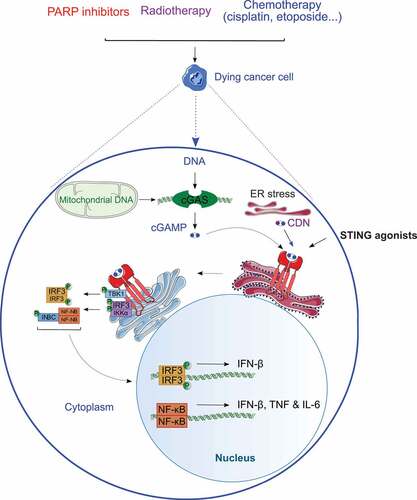
STING is particularly prone to activation in the context of viral or bacterial infection, at least in part reflecting (1) the elevated sensitivity of CGAS for histone-free DNA,Citation35,Citation36 and (2) the direct contribution of bacterial CDNs to STING.Citation25,Citation26 However, STING can also be triggered by the cytosolic accumulation of endogenous DNA of both nuclearCitation37–Citation39 and more so mitochondrialCitation40–Citation44 origin. Thus, cancer cells undergoing DNA damage and mitochondrial outer membrane permeabilization (MOMP)Citation45 in response to chemotherapy or radiation therapy are likely to secrete type I interferon (IFN) and other STING-dependent cytokines,Citation46–Citation48 although the MOMP-driven activation of apoptotic caspases considerably inhibits the process.Citation49–Citation54 Moreover, dying cancer cells appear to deliver STING-activatory DNA species to dendritic cells (DCs),Citation55–Citation57 which are the key initiators of anticancer immune responses,Citation58–Citation62 at least in some cases via exosome releaseCitation63 or GAP junctions.Citation64,Citation65
In summary, STING occupies a central role in the activation of tumor-targeting immune responses,Citation5,Citation66,Citation67 which generated considerable attention around the possibility to develop chemical STING agonists for use in oncological indications.Citation68 In this Trial Watch, we discuss recent preclinical and clinical advances in the development of STING agonists for cancer immunotherapy.
STING signaling in preclinical tumor models
Accumulating preclinical evidence documents the relevance of STING signaling in malignant cells and innate immune effectors of the tumor microenvironment for the initiation of anticancer immunity.Citation69–Citation71 Initially, it has been proposed that the engulfment of dying cancer cells by tumor-infiltrating CD8α+ DCs would trigger STING signaling in the latter, culminating in the abundant secretion of type I IFN and consequent activation of autocrine and paracrine pathways supporting the cross-priming of tumor-specific CD8+ cytotoxic T lymphocytes (CTLs).Citation72–Citation79 Consistent with this model, Sting1−/- mice are unable to mount efficient T cell immunity against syngeneic melanomasCitation57 and gliomas,Citation80 correlating with deficient type I IFN production. Similarly, Goldenticket mice – which harbor a single nucleotide polymorphism in Sting1 (T596A) that mimics the effects of a loss-of-function mutation – cannot establish efficient IFN-dependent immune responses against Listeria monocytogenes.Citation81,Citation82 Subsequent works, however, suggested that STING signaling in neoplastic cells, responding to some chemotherapeutic agents and radiotherapy, when delivered according to optimal doses and fractionation schedules,Citation83–Citation86 also contributes to anticancer immunity, at least in some settings.Citation46 In line with this model, the short-hairpin RNA-mediated depletion of CGAS or STING in mouse mammary carcinoma cells exposed to hypofractionated radiationCitation87 abolishes their ability to establish a tumor-specific immune response with systemic outreach (so-called ‘abscopal response’) in the presence of an immunostimulatory agent.Citation46 Moreover, recent data suggest that STING signaling in DCs may also be initiated by CGAS-activatory or STING-activatory molecules that accumulate in cancer cells responding to treatment and are transferred to DCs via exosomes or GAP junctions.Citation64 Thus, STING activation in both malignant and immune components of the tumor microenvironmentCitation88–Citation90 has been linked to superior anticancer immunity in a variety of preclinical tumor models.
Over the past few years, these observations prompted an intense wave of investigation aimed at the identification and development of pharmacological STING agonists for use in cancer patients.Citation9,Citation68,Citation91,Citation92 Historically, flavone acetic acid (FAA) has been the first of such molecules to be investigated for its anticancer properties, although FAA was not known to trigger STING activation at that time.Citation93 Indeed, FAA was originally characterized as a vascular-disrupting agent that showed some antitumoral activity against murine colon tumors.Citation93 Although these results encouraged further testing, FAA failed to display robust anticancer activity in murine tumor models and in Phase I clinical trials.Citation94–Citation96 In an attempt to improve efficacy, various modifications were introduced into the molecular structure of FAA, resulting in a battery of derivatives including 5,6-dimethylxanthenone-4-acetic acid (DMXAA, also known as ASA404 or vadimezan).Citation97,Citation98 Well before the identification of STING, intratumoral or systemic DMXAA administration was shown to exhibit IFN- and tumor necrosis factor (TNF)-dependent anticancer activity against multiple mouseCitation99–Citation102 and rat carcinomas,Citation103,Citation104 especially (but not exclusively) when tumors were grown in immunocompetent, syngeneic hosts (de facto suggesting to a mode of action not limited to vascular disruption).Citation101 Moreover, DMXAA turned out to efficiently synergize with various other anticancer regimens in vivo, including (but not limited to): radiotherapy,Citation105 thermoradiotherapy,Citation106 radioimmunotherapy,Citation107 chemotherapy (with a particular emphasis on taxanes),Citation108,Citation109 immunomodulatory drugs such as thalidomideCitation110,Citation111 and immunotherapy.Citation112
Corroborating initial findings, the intratumoral or systemic administration of DMXAA or other STING agonists, alone or combined with other therapeutic agents, have ultimately been attributed pronounced therapeutic effects in numerous murine models of fibrosarcoma, glioma,Citation80 melanoma,Citation66,Citation113 as well as breast,Citation114–Citation117 colorectalCitation118 and prostate carcinoma.Citation119 Moreover, various CDNs have been shown to boost the therapeutic activity of anticancer vaccines in a variety of tumor models, including (1) mouse 4T1 triple-negative mammary carcinomas treated with a Listeria monocytogenes-based vaccine;Citation120–Citation122 (2) mouse B16 melanomas treated with the TRIVAX vaccine, which consists of synthetic peptides, the Toll-like receptor 3 (TLR3) agonist polyinosinic:polycytidylic acid (polyI:C)Citation123 and co-stimulatory antibodiesCitation124,Citation125 targeting CD40,Citation126 or the STINGVAX vaccine, a cellular vaccine engineered to secrete colony-stimulating factor 2 (CSF2, best known as GM-CSF),Citation127 plus an immune checkpoint blocker targeting programmed cell death 1 (PDCD1, best known as PD-1),Citation128–Citation130 and (3) mouse CT26 colorectal carcinoma treated with the STINGVAX vaccine plus a PD-1 blocker.Citation128,Citation130
However, natural CDNs are rapidly degraded by circulating and cell-bound enzymes, including ectonucleotide pyrophosphatase/phosphodiesterase 1 (ENPP1),Citation131 calling for the development of molecules with improved stability for clinical applications. Novel STING agonists developed to circumvent the limited half-life of natural CDNs include both CDN derivatives (e.g., GSK 532)Citation132 and CDN-unrelated agents (e.g., TTI-10001, CRD5500 and amidobenzimidazole derivatives)Citation133–Citation135 In vitro, GSK 532 can induce cytokine responses in human peripheral blood mononuclear cells (PBMCs) bearing various STING haplotypes,Citation132 which is not the case of DXMAA and all natural CDNs.Citation66 Intratumoral administration of GSK 532 mediated robust anticancer effects against CT26 colorectal carcinomas growing in immunocompetent syngeneic mice, culminating with the eradication of existing lesions and the establishment of protective immunity against rechallenge with the same cancer cell line.Citation132 Such an antitumor effect was largely compromised after CD8+ T cell depletion,Citation132,Citation136 further corroborating the ability of GSK 532 to initiate adaptive anticancer immunity. Intratumoral TTI-10001 administration induced the expression of various pro-inflammatory cytokines along with T cell activation in mice bearing MC38 colorectal carcinomas or A20 B-cell lymphoma, culminating with disease eradication in a significant fraction of animals.Citation135 CRD5500 (also called LB-061) administered intratumorally or systemically induced tumor regression in BALB/c mice bearing syngeneic CT26 colorectal tumors engineered to express human STING, a therapeutic effect that was further increased when mice concomitantly received an immune checkpoint blocker. Finally, intravenous administration of an amidobenzimidazole (ABZI)-based compound to immunocompetent mice bearing syngeneic colorectal cancers resulted in complete and long-lasting disease control.Citation133
Additional lines of evidence supported the potential benefit of (re)activating STING for cancer therapy. For instance, the tumor suppressor MUS81 structure-specific endonuclease subunit (MUS81) has been shown to promote cytosolic DNA accumulation in prostate cancer cells, ultimately activating a STING-dependent program for cancer immunosurveillance.Citation137,Citation138 Along similar lines, the oncogene E7 from human papillomavirus (HPV) and E1A from adenovirus inhibit CGAS-STING signalingCitation139 to subvert antiviral immunity in the host, which eventually facilitate malignant transformation. Finally, STING and/or CGAS expression is silenced by epigenetic mechanisms in various cancer types including colorectal tumors,Citation140 further supporting the advantage obtained by developing neoplasms as a consequence of STING inactivation. Taken together, these preclinical findings provide robust grounds for testing STING agonists in cancer patients.
STING agonists as cancer therapeutics
DMXAA
Arguably, DMXAA is the best characterized of all STING agonists, largely reflecting its capacity to induce apoptosis in endothelial cells,Citation141 and hence the initial development of DMXAA as a STING-unrelated vascular-disrupting agent.Citation142,Citation143 When this Trial Watch was being redacted (April 2020), official sources listed only 18 clinical trials that investigate the safety and therapeutic profile of DMXAA in cancer patients (source www.clinicaltrials.gov) ()
Table 1. Clinical trials testing STING agonists in oncological indications.
DMXAA has preferentially been tested in patients with solid tumors, mainly in cohorts of individuals with advanced or chemorefractory neoplasms. The Phase I trial NCT00863733, aiming to evaluate pharmacodynamics, pharmacokinetics, toxicity and preliminary antitumor efficacy of DMXAA, demonstrated a good safety profile and tolerability over a large range of doses.Citation144 Notably, neither this study nor another Phase I clinical trial conducted in the United Kingdom documented significant degrees of myelosuppression in patients receiving DMXAA intravenously,Citation145 pointing to DMXAA as to a good combinatorial partner for conventional chemotherapy. Nevertheless, additional investigations were required to elucidate the therapeutic potential of the compound.
Two other Phase I clinical trials (NCT00856336 and NCT00003697) tested intravenous DMXAA as a single agent in patients with various solid tumors, demonstrating some benefits only at doses higher than the ones generally tested in the clinic.Citation146 Moreover, these studies confirmed that DMXAA perturbed retinal function, consistent with the inhibition of phosphodiesterases.Citation146,Citation147 Alongside, the tolerability, safety, efficacy and pharmacokinetics of DMXAA, combined with the microtubular poison docetaxelCitation148,Citation149 have been tested in Japanese patients affected by advanced or recurrent solid tumors.Citation150,Citation151 This Phase I clinical study (NCT01285453) proved that DMXAA plus docetaxel are generally well tolerated, documenting only Grade 1–2 adverse events including constipation, decreased appetite, alopecia, fatigue and neutropenia.Citation150 These results led to the design of a Phase III clinical study (ATTRACT-2)Citation143 testing DMXAA plus docetaxel as a second-line therapy for advanced non-small cell lung carcinoma (NSCLC).Citation143 Unfortunately, the ATTRACT-2 trial has been prematurely terminated for undisclosed reasons probably linked to preliminary efficacy.Citation152,Citation153 A similar Phase II study was conducted in 70 patients affected by hormone-refractory metastatic prostate cancer (NCT00111618), demonstrating good safety and some degree of clinical activity.Citation154 In the context of NSCLC, an additional Phase I trial (NCT00674102) aimed at investigating the safety and efficacy of DMXAA combined with carboplatin and paclitaxel in patients affected by squamous or non-squamous NSCLC.Citation155 Despite some limitations, such as the restricted number of patients, DMXAA was associated with comparable safety and efficacy independent from the tumor histology, thus allowing for the inclusion of all patients in a Phase II expansion assay (NCT00832494). Additionally, this combinatorial chemotherapeutic regimen has been tested as first-line chemotherapy in a Phase II trial (NCT01057342) enrolling patients affected by advanced squamous small cell lung carcinoma (SCLC).Citation156 This study was prematurely stopped due to lack of efficacy based on progression-free survival.
Thus, the addition of DMXAA to conventional chemotherapeutic agents with immunostimulatory effectsCitation151 has been consistently associated with poor preliminary efficacy or an increased rate of adverse reactions, as demonstrated by the consistent fraction of prematurely terminated clinical trials (8 out of 18). In most such cases, DMXAA was combined with taxanes (docetaxel or paclitaxel) alone (NCT01290380 and NCT00738387) or in the context of carboplatin-based chemotherapyCitation157 (NCT01240642, NCT00674102, NCT00662597 and NCT01299415), in patients with non-selected, advanced or chemorefractory solid tumors (NCT01285453, NCT01299701, NCT01278849, NCT01240642, NCT01290380, NCT01278758, NCT00856336, NCT01031212), or in a restricted oncological indication such as NSCLC (NCT00738387 and NCT00662597) or SCLC (NCT01057342). Two additional clinical assays testing DMXAA in cancer patients are currently listed as “withdrawn” (source www.clinicaltrials.gov). The first of such trials intended to test intravenous DMXAA as second-line chemotherapy in combination with docetaxel in patients affected by advanced urothelial carcinoma (NCT01071928). The second of such studies aimed to test DMXAA plus carboplatin, paclitaxel and the endothelial growth factor receptor (EGFR)-targeting agent cetuximabCitation158–Citation160 in patients with chemorefractory solid tumors (NCT01031212). Importantly, recent structure-function studies of mouse and human STING demonstrated that DMXAA does not bind to human STING, but only to its mouse counterparts,Citation161,Citation162 likely explaining the limited activity documented in clinical testing. That said, DMXAA appears to potently inhibit phosphodiesterases, which has been invoked to explain its vascular-disrupting properties.Citation146,Citation147 As phosphodiesterase inhibition is expected to favor the accumulation of endogenous CDNs, at least to some extent, whether the (limited) therapeutic effects of DMXAA are entirely STING-independent remains to be elucidated.
Other STING agonists in clinical evaluation
At least 15 different STING agonists other than DMXAA have been developed to circumvent the limited efficacy of the latter. E7766, belonging to a novel class of macrocycle-bridged STING agonists (MBSAs)Citation163 is the only STING agonist currently being tested in cancer patients as a standalone intravenous intervention. In particular, the tolerability, safety and preliminary activity of this molecule have been investigated in patients with advanced solid tumors or lymphomas (NCT04144140) as well as in individuals affected by non-muscle invasive bladder cancer (NCT04109092). Both these Phase I clinical studies forecast one dose-escalation part and a second dose-expansion phase. GSK3745417Citation164 is currently being tested as monotherapy or combined with the PD-1 blocking antibody pembrolizumab,Citation165 in patients with advanced, refractory/relapsed solid tumors (NCT03843359, NCT03010176) or lymphomas (NCT03010176). Preliminary results suggest that this combinatorial regimen is safe and mediates some clinical activity.Citation166 MK-1454 is being tested in patients with metastatic or unresectable, recurrent head and neck squamous cell carcinoma (HNSCC) (NCT04220866), while MK-2118 administered i.t. or s.c. (together with pembrolizumab) is being investigated in individuals affected by advanced/metastatic solid tumor or lymphomas (NCT03249792). ADU-S100 (also known as MIW815) is currently being tested in combination with pembrolizumab for CD274 (PD-L1)-positive recurrent or metastatic HNSCC patients (NCT03937141), as well in individuals with advanced solid tumors concomitantly receiving a PD-1 blocker other than pembrolizumab (NCT03172936) or the cytotoxic T-lymphocyte associated protein 4 (CTLA4) blocker ipilimumab (NCT02675439). Preliminary results from NCT03172936 suggest that the combination of ADU-S100 plus the PD-1 blocker spartalizumab is well tolerated and mediates some clinical activity in patients with solid tumors,Citation167 notably PD-1-naïve triple-negative breast cancerCitation168 and PD-1-relapsed/refractory melanoma.Citation167,Citation169,Citation170 The success of this regimen might have a particular positive impact on melanoma patients refractory to PD-1 blockers.Citation171–Citation173 The PD-1 blocker nivolumabCitation174 is also being tested in combination with the STING agonist SB11285 in subjects with advanced solid tumors (NCT04096638). Only one clinical trial, enrolling patients with advanced solid tumors (NCT03956680), evaluated the safety and preliminary efficacy of ipilimumab and nivolumab together,Citation175–Citation178 co-administered with the STING agonist BMS-986301.
Indirect STING activation by cancer therapies
Accumulating preclinical and clinical evidence demonstrates that numerous anticancer agents currently employed in the clinical practice or under clinical development can activate CGAS-STING signaling downstream of the accumulation of endogenous DNA in the cytosol, which may potentially provide an underestimated contribution to therapeutic efficacy. Among others, these agents include (1) radiation therapy, at least when administered according to optimal dose and fractionation schedules that do not favor the upregulation of the dsDNA-degrading enzyme three prime repair exonuclease 1 (TREX1),Citation46,Citation55,Citation63,Citation179-Citation181 (2) molecules that cause DNA damage, such as cisplatinCitation182,Citation183 and topoisomerase inhibitors like etoposide,Citation183–Citation186 or compromise the DNA damage response, including the clinically employed poly(ADP)-ribose polymerase 1 (PARP) inhibitor olaparib,Citation187–Citation190 as well as experimental inhibitors of ATR serine/threonine kinase (ATR),Citation191 or cause mitotic disturbances, such as paclitaxel and other taxanes.Citation35,Citation192
Taken together, these observations suggest that several commonly used anticancer agents trigger STING-dependent cytokine responses. Besides contributing to therapeutic efficacy (at least to some degree), such responses may be actionable therapeutically, and hence need to be taken under attention consideration when combinatorial regimens are conceived.
Concluding remarks
Mounting preclinical and clinical evidence suggests that carefully designed agonists of human STING (as well as molecules that trigger STING signaling in an indirect fashion) activate therapeutically relevant type I IFN-dependent responses in cancer or immune cells, in particular DCs. Although to the best of our knowledge no Phase III registration studies have yet been launched, several completed and ongoing Phase II studies have detected signs of clinical activity for STING agonists, though at the cost of non-negligible side effects. It will be interesting to learn whether mechanism-linked undesired effects can be minimized by favoring intratumoral over systemic delivery routes and whether this procedure would induce clinically exploitable systemic responses. If so, STING agonists may have a bright future.
Acknowledgments
LG is supported by a Breakthrough Level 2 grant from the US Department of Defense (DoD), Breast Cancer Research Program (BRCP) (#BC180476P1), by the 2019 Laura Ziskin Prize in Translational Research (#ZP-6177, PI: Formenti) from the Stand Up to Cancer (SU2C), by a Mantle Cell Lymphoma Research Initiative (MCL-RI, PI: Chen-Kiang) grant from the Leukemia and Lymphoma Society (LLS), by a startup grant from the Dept. of Radiation Oncology at Weill Cornell Medicine (New York, US), by a Rapid Response Grant from the Functional Genomics Initiative (New York, US), by industrial collaborations with Lytix (Oslo, Norway) and Phosplatin (New York, US), and by donations from Phosplatin (New York, US), the Luke Heller TECPR2 Foundation (Boston, US) and Sotio a.s. (Prague, Czech Republic). GK is supported by the Ligue contre le Cancer (équipe labellisée); Agence National de la Recherche (ANR) – Projets blancs; ANR under the frame of E-Rare-2, the ERA-Net for Research on Rare Diseases; AMMICa US23/CNRS UMS3655; Association pour la recherche sur le cancer (ARC); Association “Le Cancer du Sein, Parlons-en!”; Cancéropôle Ile-de-France; Chancelerie des universités de Paris (Legs Poix), Fondation pour la Recherche Médicale (FRM); a donation by Elior; European Research Area Network on Cardiovascular Diseases (ERA-CVD, MINOTAUR); Gustave Roussy Odyssea, the European Union Horizon 2020 Project Oncobiome; Fondation Carrefour; High-end Foreign Expert Program in China (GDW20171100085), Institut National du Cancer (INCa); Inserm (HTE); Institut Universitaire de France; LeDucq Foundation; the LabEx Immuno-Oncology (ANR-18-IDEX-0001); the RHU Torino Lumière; the Seerave Foundation; the SIRIC Stratified Oncology Cell DNA Repair and Tumor Immune Elimination (SOCRATE); and the SIRIC Cancer Research and Personalized Medicine (CARPEM).
Supplementary material
Supplemental data for this article can be accessed on the publisher’s website.
References
- Ishikawa H, Barber GN. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature. 2008;455:674–12. doi:10.1038/nature07317.
- Vanpouille-Box C, Hoffmann JA, Galluzzi L. Pharmacological modulation of nucleic acid sensors - therapeutic potential and persisting obstacles. Nat Rev Drug Discov. 2019;18:845–867. doi:10.1038/s41573-019-0043-2.
- Paludan SR, Bowie AG. Immune sensing of DNA. Immunity. 2013;38:870–880. doi:10.1016/j.immuni.2013.05.004.
- Abe T, Harashima A, Xia T, Konno H, Konno K, Morales A, Ahn J, Gutman D, Barber GN. STING recognition of cytoplasmic DNA instigates cellular defense. Mol Cell. 2013;50:5–15. doi:10.1016/j.molcel.2013.01.039.
- Barber GN. STING: infection, inflammation and cancer. Nat Rev Immunol. 2015;15:760–770. doi:10.1038/nri3921.
- Chen Q, Sun L, Chen ZJ. Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat Immunol. 2016;17:1142–1149. doi:10.1038/ni.3558.
- Galluzzi L, Vanpouille-Box C, Bakhoum SF, Demaria S. SnapShot:CGAS-STING signaling. Cell. 2018;173:276–e1. doi:10.1016/j.cell.2018.03.015.
- Vanpouille-Box C, Galluzzi L. Nucleic acid sensing at the interface between innate and adaptive immunity. Int Rev Cell Mol Biol. 2019;345:ix–xiii.
- Motwani M, Pesiridis S, Fitzgerald KA. DNA sensing by the cGAS-STING pathway in health and disease. Nat Rev Genet. 2019;20:657–674. doi:10.1038/s41576-019-0151-1.
- Vanpouille-Box C, Demaria S, Formenti SC, Galluzzi L. Cytosolic DNA sensing in organismal tumor control. Cancer Cell. 2018;34:361–378. doi:10.1016/j.ccell.2018.05.013.
- Unterholzner L. The interferon response to intracellular DNA: why so many receptors? Immunobiology. 2013;218:1312–1321. doi:10.1016/j.imbio.2013.07.007.
- Ablasser A, Goldeck M, Cavlar T, Deimling T, Witte G, Rohl I, Hopfner K-P, Ludwig J, Hornung V. cGAS produces a 2ʹ-5ʹ-linked cyclic dinucleotide second messenger that activates STING. Nature. 2013;498:380–384. doi:10.1038/nature12306.
- Civril F, Deimling T, de Oliveira Mann CC, Ablasser A, Moldt M, Witte G, Hornung V, Hopfner K-P. Structural mechanism of cytosolic DNA sensing by cGAS. Nature. 2013;498:332–337. doi:10.1038/nature12305.
- Diner EJ, Burdette DL, Wilson SC, Monroe KM, Kellenberger CA, Hyodo M, Hayakawa Y, Hammond M, Vance R. The innate immune DNA sensor cGAS produces a noncanonical cyclic dinucleotide that activates human STING. Cell Rep. 2013;3(5):1355–1361. doi:10.1016/j.celrep.2013.05.009.
- Sun L, Wu J, Du F, Chen X, Chen ZJ. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science. 2013;339:786–791. doi:10.1126/science.1232458.
- Almine JF, O’Hare CA, Dunphy G, Haga IR, Naik RJ, Atrih A, Connolly DJ, Taylor J, Kelsall IR, Bowie AG, et al. IFI16 and cGAS cooperate in the activation of STING during DNA sensing in human keratinocytes. Nat Commun. 2017;8(1):14392. doi:10.1038/ncomms14392.
- Dunphy G, Flannery SM, Almine JF, Connolly DJ, Paulus C, Jonsson KL, Jakobsen MR, Nevels MM, Bowie AG, Unterholzner L, et al. Non-canonical activation of the DNA sensing adaptor STING by ATM and IFI16 mediates NF-kappaB signaling after nuclear DNA damage. Mol Cell. 2018;71:745–60 e5. doi:10.1016/j.molcel.2018.07.034.
- Jonsson KL, Laustsen A, Krapp C, Skipper KA, Thavachelvam K, Hotter D, Egedal JH, Kjolby M, Mohammadi P, Prabakaran T, et al. IFI16 is required for DNA sensing in human macrophages by promoting production and function of cGAMP. Nat Commun. 2017;8(1):14391. doi:10.1038/ncomms14391.
- Nguyen JB, Modis Y. Crystal structure of the dimeric coiled-coil domain of the cytosolic nucleic acid sensor LRRFIP1. J Struct Biol. 2013;181:82–88. doi:10.1016/j.jsb.2012.10.006.
- Yang P, An H, Liu X, Wen M, Zheng Y, Rui Y, Cao X. The cytosolic nucleic acid sensor LRRFIP1 mediates the production of type I interferon via a beta-catenin-dependent pathway. Nat Immunol. 2010;11:487–494. doi:10.1038/ni.1876.
- Kondo T, Kobayashi J, Saitoh T, Maruyama K, Ishii KJ, Barber GN, Komatsu K, Akira S, Kawai T. DNA damage sensor MRE11 recognizes cytosolic double-stranded DNA and induces type I interferon by regulating STING trafficking. Proc Natl Acad Sci U S A. 2013;110:2969–2974. doi:10.1073/pnas.1222694110.
- Ferguson BJ, Mansur DS, Peters NE, Ren H, Smith GL. DNA-PK is a DNA sensor for IRF-3-dependent innate immunity. Elife. 2012;1:e00047. doi:10.7554/eLife.00047.
- Burleigh K, Maltbaek JH, Cambier S, Green R, Gale M Jr., James RC, Stetson DB. Human DNA-PK activates a STING-independent DNA sensing pathway. Sci Immunol. 2020;5(43):eaba4219. doi:10.1126/sciimmunol.aba4219.
- Gao P, Ascano M, Wu Y, Barchet W, Gaffney BL, Zillinger T, Serganov A, Liu Y, Jones R, Hartmann G, et al. Cyclic [G(2ʹ,5ʹ)pA(3ʹ,5ʹ)p] is the metazoan second messenger produced by DNA-activated cyclic GMP-AMP synthase. Cell. 2013;153:1094–1107. doi:10.1016/j.cell.2013.04.046.
- Burdette DL, Monroe KM, Sotelo-Troha K, Iwig JS, Eckert B, Hyodo M, Hayakawa Y, Vance RE. STING is a direct innate immune sensor of cyclic di-GMP. Nature. 2011;478(7370):515–518. doi:10.1038/nature10429.
- Dey B, Dey RJ, Cheung LS, Pokkali S, Guo H, Lee JH, Bishai WR. A bacterial cyclic dinucleotide activates the cytosolic surveillance pathway and mediates innate resistance to tuberculosis. Nat Med. 2015;21:401–406. doi:10.1038/nm.3813.
- Abe T, Barber GN. Cytosolic-DNA-mediated, STING-dependent proinflammatory gene induction necessitates canonical NF-kappaB activation through TBK1. J Virol. 2014;88:5328–5341. doi:10.1128/JVI.00037-14.
- Ishikawa H, Ma Z, Barber GN. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature. 2009;461:788–792. doi:10.1038/nature08476.
- Bakhoum SF, Ngo B, Laughney AM, Cavallo JA, Murphy CJ, Ly P, Shah P, Sriram RK, Watkins TBK, Taunk NK, et al. Chromosomal instability drives metastasis through a cytosolic DNA response. Nature. 2018;553:467–472. doi:10.1038/nature25432.
- Zhong Z, Sanchez-Lopez E, Karin M. Autophagy, inflammation, and immunity: A troika governing cancer and its treatment. Cell. 2016;166:288–298. doi:10.1016/j.cell.2016.05.051.
- Galluzzi L, Green DR. Autophagy-independent functions of the autophagy machinery. Cell. 2019;177:1682–1699. doi:10.1016/j.cell.2019.05.026.
- Anderson CM, Macleod KF. Autophagy and cancer cell metabolism. Int Rev Cell Mol Biol. 2019;347:145–190.
- Rybstein MD, Bravo-San Pedro JM, Kroemer G, Galluzzi L. The autophagic network and cancer. Nat Cell Biol. 2018;20:243–251. doi:10.1038/s41556-018-0042-2.
- Moretti J, Roy S, Bozec D, Martinez J, Chapman JR, Ueberheide B, Lamming DW, Chen ZJ, Horng T, Yeretssian G, et al. STING senses microbial viability to orchestrate stress-mediated autophagy of the endoplasmic reticulum. Cell. 2017;171:809–23 e13. doi:10.1016/j.cell.2017.09.034.
- Zierhut C, Yamaguchi N, Paredes M, Luo JD, Carroll T, Funabiki H. The cytoplasmic DNA sensor cGAS promotes mitotic cell death. Cell. 2019;178:302–15 e23. doi:10.1016/j.cell.2019.05.035.
- Abe T, Shapira SD. Negative regulation of cytosolic sensing of DNA. Int Rev Cell Mol Biol. 2019;344:91–115.
- Harding SM, Benci JL, Irianto J, Discher DE, Minn AJ, Greenberg RA. Mitotic progression following DNA damage enables pattern recognition within micronuclei. Nature. 2017;548:466–470. doi:10.1038/nature23470.
- Mackenzie KJ, Carroll P, Martin CA, Murina O, Fluteau A, Simpson DJ, Olova N, Sutcliffe H, Rainger JK, Leitch A, et al. cGAS surveillance of micronuclei links genome instability to innate immunity. Nature. 2017;548:461–465. doi:10.1038/nature23449.
- Vitale I, Manic G, De Maria R, Kroemer G, Galluzzi L. DNA damage in stem cells. Mol Cell. 2017;66:306–319. doi:10.1016/j.molcel.2017.04.006.
- Riley JS, Tait SW. Mitochondria and pathogen immunity: from killer to firestarter. Embo J. 2019;38. doi:10.15252/embj.2019102325.
- Aarreberg LD, Esser-Nobis K, Driscoll C, Shuvarikov A, Roby JA, Gale M Jr. Interleukin-1beta induces mtDNA release to activate innate immune signaling via cGAS-STING. Mol Cell. 2019;74:801–15 e6. doi:10.1016/j.molcel.2019.02.038.
- Sliter DA, Martinez J, Hao L, Chen X, Sun N, Fischer TD, Burman JL, Li Y, Zhang Z, Narendra DP, et al. Parkin and PINK1 mitigate STING-induced inflammation. Nature. 2018;561:258–262. doi:10.1038/s41586-018-0448-9.
- McArthur K, Whitehead LW, Heddleston JM, Li L, Padman BS, Oorschot V, Geoghegan ND, Chappaz S, Davidson S, San Chin H, et al. BAK/BAX macropores facilitate mitochondrial herniation and mtDNA efflux during apoptosis. Science. 2018;359. doi:10.1126/science.aao6047.
- Porporato PE, Filigheddu N, Pedro JMB, Kroemer G, Galluzzi L. Mitochondrial metabolism and cancer. Cell Res. 2018;28:265–280. doi:10.1038/cr.2017.155.
- Galluzzi L, Vitale I, Warren S, Adjemian S, Agostinis P, Martinez AB, Chan TA, Coukos G, Demaria S, Deutsch E, et al. Consensus guidelines for the definition, detection and interpretation of immunogenic cell death. J Immunother Cancer. 2020;8. doi:10.1136/jitc-2019-000337.
- Vanpouille-Box C, Alard A, Aryankalayil MJ, Sarfraz Y, Diamond JM, Schneider RJ, Inghirami G, Coleman CN, Formenti SC, Demaria S, et al. DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Nat Commun. 2017;8:15618. doi:10.1038/ncomms15618.
- West AP, Khoury-Hanold W, Staron M, Tal MC, Pineda CM, Lang SM, Bestwick M, Duguay BA, Raimundo N, MacDuff DA, et al. Mitochondrial DNA stress primes the antiviral innate immune response. Nature. 2015;520(7548):553–557. doi:10.1038/nature14156.
- Corrales L, Gajewski TF. Molecular pathways: targeting the stimulator of interferon genes (STING) in the immunotherapy of cancer. Clin Cancer Res. 2015;21:4774–4779. doi:10.1158/1078-0432.CCR-15-1362.
- Rongvaux A, Jackson R, Harman CC, Li T, West AP, de Zoete MR, Wu Y, Yordy B, Lakhani S, Kuan C-Y, et al. Apoptotic caspases prevent the induction of type I interferons by mitochondrial DNA. Cell. 2014;159:1563–1577. doi:10.1016/j.cell.2014.11.037.
- White MJ, McArthur K, Metcalf D, Lane RM, Cambier JC, Herold MJ, van Delft M, Bedoui S, Lessene G, Ritchie M, et al. Apoptotic caspases suppress mtDNA-induced STING-mediated type I IFN production. Cell. 2014;159:1549–1562. doi:10.1016/j.cell.2014.11.036.
- Rodriguez-Ruiz ME, Buque A, Hensler M, Chen J, Bloy N, Petroni G, Sato A, Yamazaki T, Fucikova J, Galluzzi L, et al. Apoptotic caspases inhibit abscopal responses to radiation and identify a new prognostic biomarker for breast cancer patients. Oncoimmunology. 2019;8(11):e1655964. doi:10.1080/2162402X.2019.1655964.
- Buque A, Rodriguez-Ruiz ME, Fucikova J, Galluzzi L. Apoptotic caspases cut down the immunogenicity of radiation. Oncoimmunology. 2019;8:e1655364. doi:10.1080/2162402X.2019.1655364.
- Ning X, Wang Y, Jing M, Sha M, Lv M, Gao P, Zhang R, Huang X, Feng J-M, Jiang Z, et al. Apoptotic caspases suppress type I interferon production via the cleavage of cGAS, MAVS, and IRF3. Mol Cell. 2019;74:19–31 e7. doi:10.1016/j.molcel.2019.02.013.
- Han C, Liu Z, Zhang Y, Shen A, Dong C, Zhang A, Moore C, Ren Z, Lu C, Cao X, et al. Tumor cells suppress radiation-induced immunity by hijacking caspase 9 signaling. Nat Immunol. 2020;21(5):546–554. doi:10.1038/s41590-020-0641-5.
- Deng L, Liang H, Xu M, Yang X, Burnette B, Arina A, Li X-D, Mauceri H, Beckett M, Darga T, et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity. 2014;41(5):843–852. doi:10.1016/j.immuni.2014.10.019.
- Lee YS, Radford KJ. The role of dendritic cells in cancer. Int Rev Cell Mol Biol. 2019;348:123–178.
- Woo SR, Fuertes MB, Corrales L, Spranger S, Furdyna MJ, Leung MY, Duggan R, Wang Y, Barber G, Fitzgerald K, et al. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity. 2014;41:830–842. doi:10.1016/j.immuni.2014.10.017.
- Sprooten J, Agostinis P, Garg AD. Type I interferons and dendritic cells in cancer immunotherapy. Int Rev Cell Mol Biol. 2019;348:217–262.
- Wculek SK, Cueto FJ, Mujal AM, Melero I, Krummel MF, Sancho D. Dendritic cells in cancer immunology and immunotherapy. Nat Rev Immunol. 2020;20:7–24. doi:10.1038/s41577-019-0210-z.
- Martinek J, Wu TC, Cadena D, Banchereau J, Palucka K. Interplay between dendritic cells and cancer cells. Int Rev Cell Mol Biol. 2019;348:179–215.
- Woo SR, Corrales L, Gajewski TF. Innate immune recognition of cancer. Annu Rev Immunol. 2015;33:445–474. doi:10.1146/annurev-immunol-032414-112043.
- Gajewski TF, Corrales L, Williams J, Horton B, Sivan A, Spranger S. Cancer immunotherapy targets based on understanding the T cell-inflamed versus non-T cell-inflamed tumor microenvironment. Adv Exp Med Biol. 2017;1036:19–31.
- Diamond JM, Vanpouille-Box C, Spada S, Rudqvist NP, Chapman JR, Ueberheide BM, Pilones KA, Sarfraz Y, Formenti SC, Demaria S, et al. Exosomes shuttle TREX1-sensitive IFN-stimulatory dsDNA from irradiated cancer cells to DCs. Cancer Immunol Res. 2018;6:910–920. doi:10.1158/2326-6066.CIR-17-0581.
- Schadt L, Sparano C, Schweiger NA, Silina K, Cecconi V, Lucchiari G, Yagita H, Guggisberg E, Saba S, Nascakova Z, et al. Cancer-cell-intrinsic cGAS expression mediates tumor immunogenicity. Cell Rep. 2019;29(5):1236–48 e7. doi:10.1016/j.celrep.2019.09.065.
- Torralba D, Baixauli F, Villarroya-Beltri C, Fernandez-Delgado I, Latorre-Pellicer A, Acin-Perez R, Martín-Cófreces NB, Jaso-Tamame ÁL, Iborra S, Jorge I, et al. Priming of dendritic cells by DNA-containing extracellular vesicles from activated T cells through antigen-driven contacts. Nat Commun. 2018;9(1):2658. doi:10.1038/s41467-018-05077-9.
- Corrales L, Glickman LH, McWhirter SM, Kanne DB, Sivick KE, Katibah GE, Woo S-R, Lemmens E, Banda T, Leong J, et al. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 2015;11(7):1018–1030. doi:10.1016/j.celrep.2015.04.031.
- Zhu Y, An X, Zhang X, Qiao Y, Zheng T, Li X. STING: a master regulator in the cancer-immunity cycle. Mol Cancer. 2019;18:152. doi:10.1186/s12943-019-1087-y.
- Medler T, Patel JM, Alice A, Baird JR, Hu HM, Gough MJ. Activating the nucleic acid-sensing machinery for anticancer immunity. Int Rev Cell Mol Biol. 2019;344:173–214.
- Humbert M, Hugues S. Warming up the tumor microenvironment in order to enhance immunogenicity. Oncoimmunology. 2019;8:e1510710. doi:10.1080/2162402X.2018.1510710.
- Xiao Z, Locasale JW, Dai Z. Metabolism in the tumor microenvironment: insights from single-cell analysis. Oncoimmunology. 2020;9:1726556. doi:10.1080/2162402X.2020.1726556.
- Yang M, Du W, Yi L, Wu S, He C, Zhai W, Yue C, Sun R, Menk AV, Delgoffe GM, et al. Checkpoint molecules coordinately restrain hyperactivated effector T cells in the tumor microenvironment. Oncoimmunology. 2020;9:1708064. doi:10.1080/2162402X.2019.1708064.
- Diamond MS, Kinder M, Matsushita H, Mashayekhi M, Dunn GP, Archambault JM, Lee H, Arthur CD, White JM, Kalinke U, et al. Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med. 2011;208:1989–2003. doi:10.1084/jem.20101158.
- Balan S, Saxena M, Bhardwaj N. Dendritic cell subsets and locations. Int Rev Cell Mol Biol. 2019;348:1–68.
- Fuertes MB, Kacha AK, Kline J, Woo SR, Kranz DM, Murphy KM, Gajewski TF. Host type I IFN signals are required for antitumor CD8+ T cell responses through CD8{alpha}+ dendritic cells. J Exp Med. 2011;208:2005–2016. doi:10.1084/jem.20101159.
- Klarquist J, Hennies CM, Lehn MA, Reboulet RA, Feau S, Janssen EM. STING-mediated DNA sensing promotes antitumor and autoimmune responses to dying cells. J Immunol. 2014;193:6124–6134. doi:10.4049/jimmunol.1401869.
- Zitvogel L, Galluzzi L, Kepp O, Smyth MJ, Kroemer G. Type I interferons in anticancer immunity. Nat Rev Immunol. 2015;15:405–414. doi:10.1038/nri3845.
- Dunn GP, Bruce AT, Sheehan KC, Shankaran V, Uppaluri R, Bui JD, Diamond MS, Koebel CM, Arthur C, White JM, Schreiber RD. A critical function for type I interferons in cancer immunoediting. Nat Immunol. 2005;6:722–729. doi:10.1038/ni1213.
- Leylek R, Idoyaga J. The versatile plasmacytoid dendritic cell: function, heterogeneity, and plasticity. Int Rev Cell Mol Biol. 2019;349:177–211.
- Liu J, Rozeman EA, O’Donnell JS, Allen S, Fanchi L, Smyth MJ, Blank CU, Teng MWL. Batf3(+) DCs and type I IFN are critical for the efficacy of neoadjuvant cancer immunotherapy. Oncoimmunology. 2019;8:e1546068. doi:10.1080/2162402X.2018.1546068.
- Ohkuri T, Ghosh A, Kosaka A, Zhu J, Ikeura M, David M, Watkins SC, Sarkar SN, Okada H. STING contributes to antiglioma immunity via triggering type I IFN signals in the tumor microenvironment. Cancer Immunol Res. 2014;2(12):1199–1208. doi:10.1158/2326-6066.CIR-14-0099.
- Sauer JD, Sotelo-Troha K, von Moltke J, Monroe KM, Rae CS, Brubaker SW, Hyodo M, Hayakawa Y, Woodward JJ, Portnoy DA, et al. The N-ethyl-N-nitrosourea-induced goldenticket mouse mutant reveals an essential function of sting in the in vivo interferon response to listeria monocytogenes and cyclic dinucleotides. Infect Immun. 2011;79:688–694. doi:10.1128/IAI.00999-10.
- Hansen K, Prabakaran T, Laustsen A, Jorgensen SE, Rahbaek SH, Jensen SB, Nielsen R, Leber JH, Decker T, Horan KA, et al. Listeria monocytogenes induces IFNβ expression through an IFI16-, cGAS- and STING-dependent pathway. Embo J. 2014;33:1654–1666. doi:10.15252/embj.201488029.
- Deutsch E, Chargari C, Galluzzi L, Kroemer G. Optimising efficacy and reducing toxicity of anticancer radioimmunotherapy. Lancet Oncol. 2019;20:e452–e63. doi:10.1016/S1470-2045(19)30171-8.
- Ko EC, Benjamin KT, Formenti SC. Generating antitumor immunity by targeted radiation therapy: role of dose and fractionation. Adv Radiat Oncol. 2018;3:486–493. doi:10.1016/j.adro.2018.08.021.
- Krombach J, Hennel R, Brix N, Orth M, Schoetz U, Ernst A, Schuster J, Zuchtriegel G, Reichel CA, Bierschenk S, et al. Priming anti-tumor immunity by radiotherapy: dying tumor cell-derived DAMPs trigger endothelial cell activation and recruitment of myeloid cells. Oncoimmunology. 2019;8:e1523097. doi:10.1080/2162402X.2018.1523097.
- Ngwa W, Irabor OC, Schoenfeld JD, Hesser J, Demaria S, Formenti SC. Using immunotherapy to boost the abscopal effect. Nat Rev Cancer. 2018;18:313–322. doi:10.1038/nrc.2018.6.
- Mohamad O. Diaz de Leon A, Schroeder S, Leiker A, Christie A, Zhang-Velten E, et al. Safety and efficacy of concurrent immune checkpoint inhibitors and hypofractionated body radiotherapy. Oncoimmunology. 2018;7:e1440168.
- Fridman WH, Zitvogel L, Sautes-Fridman C, Kroemer G. The immune contexture in cancer prognosis and treatment. Nat Rev Clin Oncol. 2017;14:717–734. doi:10.1038/nrclinonc.2017.101.
- Galluzzi L, Chan TA, Kroemer G, Wolchok JD, Lopez-Soto A. The hallmarks of successful anticancer immunotherapy. Sci Transl Med. 2018;10. doi:10.1126/scitranslmed.aat7807.
- Cauwels A, Van Lint S, Garcin G, Bultinck J, Paul F, Gerlo S, Van der Heyden J, Bordat Y, Catteeuw D, De Cauwer L, et al. A safe and highly efficient tumor-targeted type I interferon immunotherapy depends on the tumor microenvironment. Oncoimmunology. 2018;7:e1398876. doi:10.1080/2162402X.2017.1398876.
- Su T, Zhang Y, Valerie K, Wang XY, Lin S, Zhu G. STING activation in cancer immunotherapy. Theranostics. 2019;9:7759–7771. doi:10.7150/thno.37574.
- Wu JJ, Zhao L, Hu HG, Li WH, Li YM. Agonists and inhibitors of the STING pathway: potential agents for immunotherapy. Med Res Rev. 2020;40:1117–1141. doi:10.1002/med.21649.
- Plowman J, Narayanan VL, Dykes D, Szarvasi E, Briet P, Yoder OC, Paull KD. Flavone acetic acid: a novel agent with preclinical antitumor activity against colon adenocarcinoma 38 in mice. Cancer Treat Rep. 1986;70:631–635.
- Cummings J, Smyth JF. Flavone 8-acetic acid: our current understanding of its mechanism of action in solid tumours. Cancer Chemother Pharmacol. 1989;24:269–272. doi:10.1007/BF00304756.
- Dodion PF, Abrams J, Gerard B, Crespeigne N, Peeters B, Van Berchem C, Kenis Y. Clinical and pharmacokinetic phase I trial with the diethylaminoester of flavone acetic acid (LM985, NSC 293015). Eur J Cancer Clin Oncol. 1987;23:837–842. doi:10.1016/0277-5379(87)90288-4.
- Kerr DJ, Kaye SB, Graham J, Cassidy J, Harding M, Setanoians A, McGrath JC, Vezin WR, Cunningham D, Forrest G, et al. Phase I and pharmacokinetic study of LM985 (flavone acetic acid ester).. Cancer Res. 1986;46(6):3142–3146.
- Daei Farshchi Adli A, Jahanban-Esfahlan R, Seidi K, Samandari-Rad S, Zarghami N. An overview on vadimezan (DMXAA): the vascular disrupting agent. Chem Biol Drug Des. 2018;91:996–1006. doi:10.1111/cbdd.13166.
- Zwi LJ, Baguley BC, Gavin JB, Wilson WR. Necrosis in non-tumour tissues caused by flavone acetic acid and 5,6-dimethyl xanthenone acetic acid. Br J Cancer. 1990;62:932–934. doi:10.1038/bjc.1990.412.
- Baguley BC, Ching LM. Immunomodulatory actions of xanthenone anticancer agents. BioDrugs. 1997;8:119–127. doi:10.2165/00063030-199708020-00005.
- Joseph WR, Cao Z, Mountjoy KG, Marshall ES, Baguley BC, Ching LM. Stimulation of tumors to synthesize tumor necrosis factor-alpha in situ using 5,6-dimethylxanthenone-4-acetic acid: a novel approach to cancer therapy. Cancer Res. 1999;59:633–638.
- Laws AL, Matthew AM, Double JA, Bibby MC. Preclinical in vitro and in vivo activity of 5,6-dimethylxanthenone-4-acetic acid. Br J Cancer. 1995;71:1204–1209. doi:10.1038/bjc.1995.234.
- Philpott M, Baguley BC, Ching LM. Induction of tumour necrosis factor-alpha by single and repeated doses of the antitumour agent 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemother Pharmacol. 1995;36:143–148. doi:10.1007/BF00689199.
- McPhail LD, McIntyre DJ, Ludwig C, Kestell P, Griffiths JR, Kelland LR, Robinson SP. Rat tumor response to the vascular-disrupting agent 5,6-dimethylxanthenone-4-acetic acid as measured by dynamic contrast-enhanced magnetic resonance imaging, plasma 5-hydroxyindoleacetic acid levels, and tumor necrosis. Neoplasia. 2006;8:199–206. doi:10.1593/neo.05739.
- Liu JJ, Ching LM, Goldthorpe M, Sutherland R, Baguley BC, Kirker JA, McKeage MJ. Antitumour action of 5,6-dimethylxanthenone-4-acetic acid in rats bearing chemically induced primary mammary tumours. Cancer Chemother Pharmacol. 2007;59:661–669. doi:10.1007/s00280-006-0321-7.
- Wilson WR, Li AE, Cowan DS, Siim BG. Enhancement of tumor radiation response by the antivascular agent 5,6-dimethylxanthenone-4-acetic acid. Int J Radiat Oncol Biol Phys. 1998;42:905–908. doi:10.1016/S0360-3016(98)00358-7.
- Murata R, Horsman MR. Tumour-specific enhancement of thermoradiotherapy at mild temperatures by the vascular targeting agent 5,6-dimethylxanthenone-4-acetic acid. Int J Hyperthermia. 2004;20:393–404. doi:10.1080/02656730310001619370.
- Pedley RB, Boden JA, Boden R, Boxer GM, Flynn AA, Keep PA, Begent RH. Ablation of colorectal xenografts with combined radioimmunotherapy and tumor blood flow-modifying agents. Cancer Res. 1996;56:3293–3300.
- Pruijn FB, van Daalen M, Holford NH, Wilson WR. Mechanisms of enhancement of the antitumour activity of melphalan by the tumour-blood-flow inhibitor 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemother Pharmacol. 1997;39:541–546. doi:10.1007/s002800050611.
- Siim BG, Lee AE, Shalal-Zwain S, Pruijn FB, McKeage MJ, Wilson WR. Marked potentiation of the antitumour activity of chemotherapeutic drugs by the antivascular agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA). Cancer Chemother Pharmacol. 2003;51:43–52. doi:10.1007/s00280-002-0529-0.
- Ching LM, Browne WL, Tchernegovski R, Gregory T, Baguley BC, Palmer BD. Interaction of thalidomide, phthalimide analogues of thalidomide and pentoxifylline with the anti-tumour agent 5,6-dimethylxanthenone-4-acetic acid: concomitant reduction of serum tumour necrosis factor-alpha and enhancement of anti-tumour activity. Br J Cancer. 1998;78:336–343. doi:10.1038/bjc.1998.495.
- Cao Z, Joseph WR, Browne WL, Mountjoy KG, Palmer BD, Baguley BC, Ching L-M. Thalidomide increases both intra-tumoural tumour necrosis factor-alpha production and anti-tumour activity in response to 5,6-dimethylxanthenone-4-acetic acid. Br J Cancer. 1999;80:716–723. doi:10.1038/sj.bjc.6690415.
- Kanwar JR, Kanwar RK, Pandey S, Ching LM, Krissansen GW. Vascular attack by 5,6-dimethylxanthenone-4-acetic acid combined with B7.1 (CD80)-mediated immunotherapy overcomes immune resistance and leads to the eradication of large tumors and multiple tumor foci. Cancer Res. 2001;61:1948–1956.
- Demaria O, De Gassart A, Coso S, Gestermann N, Di Domizio J, Flatz L, Gaide O, Michielin O, Hwu P, Petrova TV, et al. STING activation of tumor endothelial cells initiates spontaneous and therapeutic antitumor immunity. Proc Natl Acad Sci U S A. 2015;112:15408–15413. doi:10.1073/pnas.1512832112.
- Chandra D, Quispe-Tintaya W, Jahangir A, Asafu-Adjei D, Ramos I, Sintim HO, Zhou J, Hayakawa Y, Karaolis DKR, Gravekamp C, et al. STING ligand c-di-GMP improves cancer vaccination against metastatic breast cancer. Cancer Immunol Res. 2014;2(9):901–910. doi:10.1158/2326-6066.CIR-13-0123.
- Cheng N, Watkins-Schulz R, Junkins RD, David CN, Johnson BM, Montgomery SA, Peine KJ, Darr DB, Yuan H, McKinnon KP, et al. A nanoparticle-incorporated STING activator enhances antitumor immunity in PD-L1-insensitive models of triple-negative breast cancer. JCI Insight. 2018;3. doi:10.1172/jci.insight.120638.
- Yang H, Lee WS, Kong SJ, Kim CG, Kim JH, Chang SK, Kim S, Kim G, Chon HJ, Kim C, et al. STING activation reprograms tumor vasculatures and synergizes with VEGFR2 blockade. J Clin Invest. 2019;130:4350–4364. doi:10.1172/JCI125413.
- Weiss JM, Guerin MV, Regnier F, Renault G, Galy-Fauroux I, Vimeux L, Feuillet V, Peranzoni E, Thoreau M, Trautmann A, et al. The STING agonist DMXAA triggers a cooperation between T lymphocytes and myeloid cells that leads to tumor regression. Oncoimmunology. 2017;6(10):e1346765. doi:10.1080/2162402X.2017.1346765.
- Zhu Q, Man SM, Gurung P, Liu Z, Vogel P, Lamkanfi M, Kanneganti T-D. Cutting edge: STING mediates protection against colorectal tumorigenesis by governing the magnitude of intestinal inflammation. J Immunol. 2014;193(10):4779–4782. doi:10.4049/jimmunol.1402051.
- Ager CR, Reilley MJ, Nicholas C, Bartkowiak T, Jaiswal AR, Curran MA. Intratumoral STING activation with T-cell checkpoint modulation generates systemic antitumor immunity. Cancer Immunol Res. 2017;5:676–684. doi:10.1158/2326-6066.CIR-17-0049.
- Gravekamp C, Chandra D. Targeting STING pathways for the treatment of cancer. Oncoimmunology. 2015;4(12):e988463. doi:10.4161/2162402X.2014.988463.
- Kim SH, Castro F, Gonzalez D, Maciag PC, Paterson Y, Gravekamp C. Mage-b vaccine delivered by recombinant Listeria monocytogenes is highly effective against breast cancer metastases. Br J Cancer. 2008;99(5):741–749. doi:10.1038/sj.bjc.6604526.
- Sauer JD, Pereyre S, Archer KA, Burke TP, Hanson B, Lauer P, Portnoy DA. Listeria monocytogenes engineered to activate the Nlrc4 inflammasome are severely attenuated and are poor inducers of protective immunity. Proc Natl Acad Sci U S A. 2011;108:12419–12424. doi:10.1073/pnas.1019041108.
- Smith M, Garcia-Martinez E, Pitter MR, Fucikova J, Spisek R, Zitvogel L, Kroemer G, Galluzzi L. Trial watch: toll-like receptor agonists in cancer immunotherapy. Oncoimmunology. 2018;7(12):e1526250. doi:10.1080/2162402X.2018.1526250.
- Aranda F, Vacchelli E, Eggermont A, Galon J, Fridman WH, Zitvogel L, Kroemer G, Galluzzi L. Trial watch: immunostimulatory monoclonal antibodies in cancer therapy. Oncoimmunology. 2014;3(2):e27297. doi:10.4161/onci.27297.
- Han X, Vesely MD. Stimulating T cells against cancer with agonist immunostimulatory monoclonal antibodies. Int Rev Cell Mol Biol. 2019;342:1–25.
- Wang Z, Celis E. STING activator c-di-GMP enhances the anti-tumor effects of peptide vaccines in melanoma-bearing mice. Cancer Immunol Immunother. 2015;64(8):1057–1066. doi:10.1007/s00262-015-1713-5.
- Koster BD, de Jong TD, van den Hout M, Sluijter BJR, Vuylsteke R, Molenkamp BG, Vosslamber S, van den Tol MP, van den Eertwegh AJM, de Gruijl TD, et al. In the mix: the potential benefits of adding GM-CSF to CpG-B in the local treatment of patients with early-stage melanoma. Oncoimmunology. 2020;9(1):1708066. doi:10.1080/2162402X.2019.1708066.
- Fu J, Kanne DB, Leong M, Glickman LH, McWhirter SM, Lemmens E, Mechette K, Leong JJ, Lauer P, Liu W, et al. STING agonist formulated cancer vaccines can cure established tumors resistant to PD-1 blockade. Sci Transl Med. 2015;7:283ra52.
- Vitale I, Sistigu A, Manic G, Rudqvist NP, Trajanoski Z, Galluzzi L. Mutational and antigenic landscape in tumor progression and cancer immunotherapy. Trends Cell Biol. 2019;29:396–416. doi:10.1016/j.tcb.2019.01.003.
- Kim YJ. STINGing the tumor’s immune evasion mechanism. Oncoimmunology. 2018;7:e1083673. doi:10.1080/2162402X.2015.1083673.
- Li L, Yin Q, Kuss P, Maliga Z, Millan JL, Wu H, Mitchison TJ. Hydrolysis of 2ʹ3’-cGAMP by ENPP1 and design of nonhydrolyzable analogs. Nat Chem Biol. 2014;10:1043–1048. doi:10.1038/nchembio.1661.
- Yang J, Adam M, Clemens J, Creech K, Schneck J, Pasikanti K, Tran JL, Joglekar D, Hopson C, Pesiridis, S, et al. Preclinical characterization of GSK532, a novel STING agonist with potent anti-tumor activity. American Association for Cancer Research Annual Meeting 2018.
- Banerjee M, Basu S, Middya S, Shrivastava R, Ghosh R, Pryde DC, Yadav D, Bhattacharya G, Soram T, Puniya K, et al. CRD5500: A versatile small molecule STING agonist amenable to bioconjugation as an ADC. American Association for Cancer Research Annual Meeting 2019.
- Ramanjulu JM, Pesiridis GS, Yang J, Concha N, Singhaus R, Zhang SY, Tran J-L, Moore P, Lehmann S, Eberl HC, et al. Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature. 2018;564:439–443. doi:10.1038/s41586-018-0705-y.
- Wang Z, Dove P, Rosa D, Bossen B, Helke S, Charbonneau M, Brinen L, Dodge K, Lin GH, Galligan C, et al. Preclinical characterization of a novel non-cyclic dinucleotide small molecule STING agonist with potent antitumor activity in mice. American Association of Cancer Research Annual Meeting 2019.
- Borrie AE, Maleki Vareki S. T lymphocyte-based cancer immunotherapeutics. Int Rev Cell Mol Biol. 2018;341:201–276.
- Ho SW, Zhang WL, Tan NY, Khatoo M, Suter MA, Tripathi S, Cheung FG, Lim W, Tan P, Ngeow J, et al. The DNA structure-specific endonuclease MUS81 mediates DNA sensor STING-dependent host rejection of prostate cancer cells. Immunity. 2016;44(5):1177–1189. doi:10.1016/j.immuni.2016.04.010.
- Rao S, Gharib K, Han A. Cancer immunosurveillance by T cells. Int Rev Cell Mol Biol. 2019;342:149–173.
- Lau L, Gray EE, Brunette RL, Stetson DB. DNA tumor virus oncogenes antagonize the cGAS-STING DNA-sensing pathway. Science. 2015;350:568–571. doi:10.1126/science.aab3291.
- Xia T, Konno H, Ahn J, Barber GN. Deregulation of STING signaling in colorectal carcinoma constrains DNA damage responses and correlates with tumorigenesis. Cell Rep. 2016;14:282–297. doi:10.1016/j.celrep.2015.12.029.
- Ching L-M, Cao Z, Kieda C, Zwain S, Jameson MB, Baguley BC. Induction of endothelial cell apoptosis by the antivascular agent 5,6-dimethylxanthenone-4-acetic acid. Br J Cancer. 2002;86(12):1937–1942. doi:10.1038/sj.bjc.6600368.
- Baguley BC. Antivascular therapy of cancer: DMXAA. Lancet Oncol. 2003;4:141–148. doi:10.1016/S1470-2045(03)01018-0.
- Baguley BC, Siemann DW. Temporal aspects of the action of ASA404 (vadimezan; DMXAA). Expert Opin Investig Drugs. 2010;19:1413–1425. doi:10.1517/13543784.2010.529128.
- Jameson MB, Thompson PI, Baguley BC, Evans BD, Harvey VJ, Porter DJ, McCrystal MR, Small M, Bellenger K, Gumbrell L, et al. Clinical aspects of a phase I trial of 5,6-dimethylxanthenone-4-acetic acid (DMXAA), a novel antivascular agent. Br J Cancer. 2003;88(12):1844–1850. doi:10.1038/sj.bjc.6600992.
- Rustin GJ, Bradley C, Galbraith S, Stratford M, Loadman P, Waller S, Bellenger K, Gumbrell L, Folkes L, Halbert G, et al. 5,6-dimethylxanthenone-4-acetic acid (DMXAA), a novel antivascular agent: phase I clinical and pharmacokinetic study. Br J Cancer. 2003;88(8):1160–1167. doi:10.1038/sj.bjc.6600885.
- Jameson MB, Sharp DM, Sissingh JI, Hogg CR, Thompson PI, McKeage MJ, Jeffery M, Waller S, Acton G, Green C, et al. Transient retinal effects of 5,6-dimethylxanthenone-4-acetic acid (DMXAA, ASA404), an antitumor vascular-disrupting agent in phase I clinical trials. Invest Ophthalmol Vis Sci. 2009;50(6):2553–2559. doi:10.1167/iovs.08-2068.
- Zhang SH, Zhang Y, Shen J, Zhang S, Chen L, Gu J, Mruk JS, Cheng G, Zhu L, Kunapuli SP, et al. Tumor vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid inhibits platelet activation and thrombosis via inhibition of thromboxane A2 signaling and phosphodiesterase. J Thromb Haemost. 2013;11:1855–1866. doi:10.1111/jth.12362.
- Vitale I, Galluzzi L, Senovilla L, Criollo A, Jemaa M, Castedo M,Kroemer G. Illicit survival of cancer cells during polyploidization and depolyploidization. Cell Death Differ. 2011;18:1403–1413. doi:10.1038/cdd.2010.145.
- Perez EA. Microtubule inhibitors: differentiating tubulin-inhibiting agents based on mechanisms of action, clinical activity, and resistance. Mol Cancer Ther. 2009;8(8):2086–2095. doi:10.1158/1535-7163.MCT-09-0366.
- Daga H, Hida T, Ishikawa S, Shimizu J, Tokunaga S, Horio Y, Kobayashi K, Takeda K. The safety and tolerability of intravenous ASA404 when administered in combination with docetaxel (60 or 75 mg/m2) in Japanese patients with advanced or recurrent solid tumors. Jpn J Clin Oncol. 2011;41(9):1067–1073. doi:10.1093/jjco/hyr110.
- Galluzzi L, Buque A, Kepp O, Zitvogel L, Kroemer G. Immunological effects of conventional chemotherapy and targeted anticancer agents. Cancer Cell. 2015;28:690–714. doi:10.1016/j.ccell.2015.10.012.
- Aggarwal C, Somaiah N, Simon G. Antiangiogenic agents in the management of non-small cell lung cancer: where do we stand now and where are we headed? Cancer Biol Ther. 2012;13:247–263. doi:10.4161/cbt.19594.
- Lara PN Jr., Douillard JY, Nakagawa K, von Pawel J, McKeage MJ, Albert I, Losonczy G, Reck M, Heo D-S, Fan X, et al. Randomized phase III placebo-controlled trial of carboplatin and paclitaxel with or without the vascular disrupting agent vadimezan (ASA404) in advanced non-small-cell lung cancer. J Clin Oncol. 2011;29:2965–2971. doi:10.1200/JCO.2011.35.0660.
- McKeage MJ. The potential of DMXAA (ASA404) in combination with docetaxel in advanced prostate cancer. Expert Opin Investig Drugs. 2008;17:23–29. doi:10.1517/13543784.17.1.23.
- McKeage MJ, Jameson MB, Investigators ASSG. Comparative outcomes of squamous and non-squamous non-small cell lung cancer (NSCLC) patients in phase II studies of ASA404 (DMXAA) - retrospective analysis of pooled data. J Thorac Dis. 2010;2:199–204. doi:10.3978/j..2072-1439.2010.02.04.1.
- Qiu Z, Lin A, Li K, Lin W, Wang Q, Wei T, Zhu W, Luo P, Zhang J. A novel mutation panel for predicting etoposide resistance in small-cell lung cancer. Drug Des Devel Ther. 2019;13:2021–2041. doi:10.2147/DDDT.S205633.
- Michels J, Vitale I, Galluzzi L, Adam J, Olaussen KA, Kepp O, Senovilla L, Talhaoui I, Guegan J, Enot DP, et al. Cisplatin resistance associated with PARP hyperactivation. Cancer Res. 2013;73(7):2271–2280. doi:10.1158/0008-5472.CAN-12-3000.
- Psyrri A, Burtness B. Targeted therapies: molecular selection for ‘smart’ study design in lung cancer. Nat Rev Clin Oncol. 2010;7:621–622. doi:10.1038/nrclinonc.2010.156.
- de La Motte Rouge T, Galluzzi L, Olaussen KA, Zermati Y, Tasdemir E, Robert T, Ripoche H, Lazar V, Dessen P, Harper F, et al. A novel epidermal growth factor receptor inhibitor promotes apoptosis in non-small cell lung cancer cells resistant to erlotinib. Cancer Res. 2007;67(13):6253–6262. doi:10.1158/0008-5472.CAN-07-0538.
- Kansy BA, Shayan G, Jie H-B, Gibson SP, Lei YL, Brandau S, Lang S, Schmitt NC, Ding F, Lin Y, et al. T cell receptor richness in peripheral blood increases after cetuximab therapy and correlates with therapeutic response. Oncoimmunology. 2018;7(11):e1494112. doi:10.1080/2162402X.2018.1494112.
- Conlon J, Burdette DL, Sharma S, Bhat N, Thompson M, Jiang Z, Rathinam VAK, Monks B, Jin T, Xiao TS, et al. Mouse, but not human STING, binds and signals in response to the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid. J Immunol. 2013;190(10):5216–5225. doi:10.4049/jimmunol.1300097.
- Prantner D, Perkins DJ, Lai W, Williams MS, Sharma S, Fitzgerald KA, Vogel SN. 5,6-dimethylxanthenone-4-acetic acid (DMXAA) activates stimulator of interferon gene (STING)-dependent innate immune pathways and is regulated by mitochondrial membrane potential. J Biol Chem. 2012;287(47):39776–39788. doi:10.1074/jbc.M112.382986.
- Endo A, Kim D, Huang K, Hao M, Mathieu S, Choi H, Majumder U, Zhu X, Shen Y, Sanders K, et al. Discovery of E7766: A representative of a novel class of macrocycle-bridged STING agonists (MBSAs) with superior potency and pan-genotypic activity. American Association for Cancer Research Annual Meeting 2019.
- Flood BA, Higgs EF, Li S, Luke JJ, Gajewski TF. STING pathway agonism as a cancer therapeutic. Immunol Rev. 2019;290(1):24–38. doi:10.1111/imr.12765.
- Kuryk L, Moller AW, Jaderberg M. Combination of immunogenic oncolytic adenovirus ONCOS-102 with anti-PD-1 pembrolizumab exhibits synergistic antitumor effect in humanized A2058 melanoma huNOG mouse model. Oncoimmunology. 2019;8:e1532763. doi:10.1080/2162402X.2018.1532763.
- Harrington KJ, Brody J, Ingham M, Strauss J, Cemerski S, Wang M, Tse A, Khilnani A, Marabelle A, Golan T. Preliminary results of the first-in-human (FIH) study of MK-1454, an agonist of stimulator of interferon genes (STING), as monotherapy or in combination with pembrolizumab (pembro) in patients with advanced solid tumors or lymphomas. ESMO. 2018.
- Pelster MS, Amaria RN. Combined targeted therapy and immunotherapy in melanoma: a review of the impact on the tumor microenvironment and outcomes of early clinical trials. Ther Adv Med Oncol. 2019;11:1758835919830826. doi:10.1177/1758835919830826.
- Crosby EJ, Wei J, Yang XY, Lei G, Wang T, Liu C-X, Agarwal P, Korman AJ, Morse MA, Gouin K, et al. Complimentary mechanisms of dual checkpoint blockade expand unique T-cell repertoires and activate adaptive anti-tumor immunity in triple-negative breast tumors. Oncoimmunology. 2018;7(5):e1421891. doi:10.1080/2162402X.2017.1421891.
- Meric-Bernstam F, Kaur Sandhu S, Hamid O, Spreafico A, Kasper S, Dummer R, Shimizu T, Steeghs N, Lewis N, Talluto CC, et al. Phase Ib study of MIW815 (ADU-S100) in combination with spartalizumab (PDR001) in patients (pts) with advanced/metastatic solid tumors or lymphomas. Journal of Clinical Oncology. 2020;37:2507.
- Bommareddy PK, Zloza A, Rabkin SD, Kaufman HL. Oncolytic virus immunotherapy induces immunogenic cell death and overcomes STING deficiency in melanoma. Oncoimmunology. 2019;8:1591875. doi:10.1080/2162402X.2019.1591875.
- Khunger A, Rytlewski JA, Fields P, Yusko EC, Tarhini AA. The impact of CTLA-4 blockade and interferon-alpha on clonality of T-cell repertoire in the tumor microenvironment and peripheral blood of metastatic melanoma patients. Oncoimmunology. 2019;8:e1652538. doi:10.1080/2162402X.2019.1652538.
- Kroemer G, Galluzzi L. Combinatorial immunotherapy with checkpoint blockers solves the problem of metastatic melanoma-an exclamation sign with a question mark. Oncoimmunology. 2015;4:e1058037. doi:10.1080/2162402X.2015.1058037.
- Dupont R, Berard E, Puisset F, Comont T, Delord JP, Guimbaud R, Meyer N, Mazieres J, Alric L. The prognostic impact of immune-related adverse events during anti-PD1 treatment in melanoma and non-small-cell lung cancer: a real-life retrospective study. Oncoimmunology. 2020;9:1682383. doi:10.1080/2162402X.2019.1682383.
- Costantini A, Julie C, Dumenil C, Helias-Rodzewicz Z, Tisserand J, Dumoulin J, Giraud V, Labrune S, Chinet T, Emile J-F, et al. Predictive role of plasmatic biomarkers in advanced non-small cell lung cancer treated by nivolumab. Oncoimmunology. 2018;7:e1452581. doi:10.1080/2162402X.2018.1452581.
- Motzer RJ, Tannir NM, McDermott DF, Aren Frontera O, Melichar B, Choueiri TK, Plimack ER, Barthélémy P, Porta C, George S, et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378(14):1277–1290. doi:10.1056/NEJMoa1712126.
- Hellmann MD, Ciuleanu T-E, Pluzanski A, Lee JS, Otterson GA, Audigier-Valette C, Minenza E, Linardou H, Burgers S, Salman P, et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med. 2018;378(22):2093–2104. doi:10.1056/NEJMoa1801946.
- Wolchok JD, Chiarion-Sileni V, Gonzalez R, Rutkowski P, Grob -J-J, Cowey CL, Lao CD, Wagstaff J, Schadendorf D, Ferrucci PF, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377(14):1345–1356. doi:10.1056/NEJMoa1709684.
- Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, Schadendorf D, Dummer R, Smylie M, Rutkowski P, Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373(1):23–34. doi:10.1056/NEJMoa1504030.
- Formenti SC, Rudqvist N-P, Golden E, Cooper B, Wennerberg E, Lhuillier C, Vanpouille-Box C, Friedman K, Ferrari de Andrade L, Wucherpfennig KW, et al. Radiotherapy induces responses of lung cancer to CTLA-4 blockade. Nat Med. 2018;24(12):1845–1851. doi:10.1038/s41591-018-0232-2.
- Rodriguez-Ruiz ME, Vitale I, Harrington KJ, Melero I, Galluzzi L. Immunological impact of cell death signaling driven by radiation on the tumor microenvironment. Nat Immunol. 2020;21(2):120–134. doi:10.1038/s41590-019-0561-4.
- Burnette BC, Liang H, Lee Y, Chlewicki L, Khodarev NN, Weichselbaum RR, Fu Y-X, Auh SL. The efficacy of radiotherapy relies upon induction of type i interferon-dependent innate and adaptive immunity. Cancer Res. 2011;71(7):2488–2496. doi:10.1158/0008-5472.CAN-10-2820.
- Grabosch S, Bulatovic M, Zeng F, Ma T, Zhang L, Ross M, Brozick J, Fang Y, Tseng G, Kim E, et al. Cisplatin-induced immune modulation in ovarian cancer mouse models with distinct inflammation profiles. Oncogene. 2019;38(13):2380–2393. doi:10.1038/s41388-018-0581-9.
- Maekawa H, Inoue T, Ouchi H, Jao T-M, Inoue R, Nishi H, Fujii R, Ishidate F, Tanaka T, Tanaka Y, et al. Mitochondrial damage causes inflammation via cGAS-STING signaling in acute kidney injury. Cell Rep. 2019;29(5):1261–73 e6. doi:10.1016/j.celrep.2019.09.050.
- Unterholzner L, Dunphy G. cGAS-independent STING activation in response to DNA damage. Mol Cell Oncol. 2019;6:1558682. doi:10.1080/23723556.2018.1558682.
- Wilkinson RD, Johnston DI, Parkes EE, McCabe N, Kennedy RD Exploring the effect of chemotherapies on STING-dependent cytokine release. American Association for Cancer Research Annual Meeting 2018.
- Wang Z, Chen J, Hu J, Zhang H, Xu F, He W, Wang X, Li M, Lu W, Zeng G, et al. cGAS/STING axis mediates a topoisomerase II inhibitor-induced tumor immunogenicity. J Clin Invest. 2019;130:4850–4862. doi:10.1172/JCI127471.
- Chabanon RM, Muirhead G, Krastev DB, Adam J, Morel D, Garrido M, Lamb A, Hénon C, Dorvault N, Rouanne M, et al. PARP inhibition enhances tumor cell-intrinsic immunity in ERCC1-deficient non-small cell lung cancer. J Clin Invest. 2019;129:1211–1228. doi:10.1172/JCI123319.
- Ding L, Kim H-J, Wang Q, Kearns M, Jiang T, Ohlson CE, Li BB, Xie S, Liu JF, Stover EH, Ding L, Kim HJ, Wang Q, Kearns M, Jiang T, Ohlson CE, et al. PARP inhibition elicits STING-dependent antitumor immunity in brca1-deficient ovarian cancer. Cell Rep. 2018;25(11):2972–80 e5. doi:10.1016/j.celrep.2018.11.054.
- Reislander T, Lombardi EP, Groelly FJ, Miar A, Porru M, Di Vito S, Wright B, Lockstone H, Biroccio A, Harris A, et al. BRCA2 abrogation triggers innate immune responses potentiated by treatment with PARP inhibitors. Nat Commun. 2019;10(1):3143. doi:10.1038/s41467-019-11048-5.
- Shen J, Zhao W, Ju Z, Wang L, Peng Y, Labrie M, Yap TA, Mills GB, Peng G. PARPi triggers the STING-dependent immune response and enhances the therapeutic efficacy of immune checkpoint blockade independent of BRCAness. Cancer Res. 2019;79(2):311–319. doi:10.1158/0008-5472.CAN-18-1003.
- Dillon MT, Bergerhoff KF, Pedersen M, Whittock H, Crespo-Rodriguez E, Patin EC, Pearson A, Smith HG, Paget JTE, Patel RR, et al. ATR inhibition potentiates the radiation-induced inflammatory tumor microenvironment. Clin Cancer Res. 2019;25(11):3392–3403. doi:10.1158/1078-0432.CCR-18-1821.
- Lohard S, Bourgeois N, Maillet L, Gautier F, Fetiveau A, Lasla H, Nguyen F, Vuillier C, Dumont A, Moreau-Aubry A, et al. STING-dependent paracriny shapes apoptotic priming of breast tumors in response to anti-mitotic treatment. Nat Commun. 2020;11(1):259. doi:10.1038/s41467-019-13689-y.
