ABSTRACT
In September 2020, the Japanese government approved cetuximab saratolacan (previously known as RM-1929, commercial name: Akalux) for the treatment of unresectable locally advanced or recurrent head and neck cancer. Cetuximab saratolacan is a chemical conjugate of the photosensitizer IR700 with cetuximab, which targets EGFR. The treatment consists in the intravenous injection of cetuximab saratolacan, which binds to head and neck cancer cells expressing high levels of EGFR, followed by illumination of the tumor with red light (690 nm) for photodynamic therapy. This approach causes immunogenic cell death in malignant tissues, thus triggering a potent anticancer immune response.
Photodynamic therapy (PDT) is a promising and noninvasive clinical option for the treatment of solid tumors. It requires the administration of a pro-drug (known as photosensitizer) that after a defined time interval, is activated by illumination with light of a specific wavelength causing the production of reactive oxygen species (ROS) in target cells. The clinical use of PDT started 25 years ago with the FDA approval of porfimer sodium (Photofrin) for the palliative treatment of obstructing esophageal cancer. Photofrin is now also used for other types of cancer namely, non-small cell lung cancer, bladder cancer and Barrett’s esophagus. Additional photosensitizers have received clinical approval for cancer treatment including temoporfin (Foscan), talaporfin (Laserphyrin), 5-aminolevulinic acid (Levulan) and more recently, padeliporfin (TOOKAD).Citation1
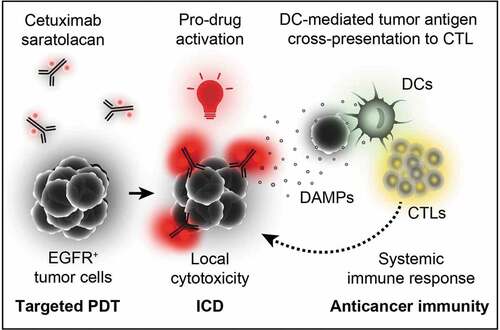
Figure 1. Mechanism of cetuximab saratolacan. Cetuximab-bound IR700 selectively binds to the surface of EGFR+ tumor cells. Its photo-activation at 690 nm selectively kills EGFR-expressing cells, thus allowing for targeted photodynamic therapy (PDT) and the induction of immunogenic cell death (ICD). This elicits a systemic immune response that contributes to the eradication of malignant cells. CTLs, cytotoxic T lymphocytes; DCs, dendritic cells
PDT has proven therapeutic efficacy based on direct cytotoxic effects on tumor cells and the damage of tumor blood vessels. In addition, PDT with photosensitizers such as porfimer sodium, temoporfin, hypericin and redaporfin kill tumor cells while stimulating an anticancer immune response robust enough to destroy metastasis outside of the field of illumination, hence causing an abscopal effect.Citation2–4 In addition, extracorporeal photochemotherapy (ECP) with the photosensitizer 8-methoxypsoralen that is employed for the treatment of cutaneous T cell lymphoma has recently been shown to confer immunogenic effects.Citation5
This type of tumor cell killing, known as immunogenic cell death (ICD), is characterized by the spatially and temporally controlled emission of specific danger associated molecular patterns (DAMPs), such as the exposure of calreticulin on the cellular surface, the secretion of adenosine triphosphate (ATP), the release of annexin A1 (ANXA1), the exodus of high-mobility group box 1 (HMGB1) and the production of type I interferon (IFN), altogether facilitating the presentation of tumor associated antigens by dendritic cells.Citation6
Although the light activation of the photosensitizer prodrug enables a certain degree of local selectivity, limited tumor specificity and poor drug accumulation in target cells are still major limitations of cancer therapies including PDT. In order to overcome these issues, photosensitizers can be conjugated to targeting moieties that specifically bind to receptors overexpressed on tumor cells (active targeting), leading to improved internalization. Several clinically approved monoclonal antibodies (mAbs) have become an appealing option for targeted delivery of anti-cancer drugs owing to their high target-specificity and affinity.Citation7 For instance, the human epidermal growth factor receptor 2 (HER2) targeting antibody trastuzumab conjugated to the topoisomerase I inhibitor deruxtecan or to the microtubule inhibitor emtansine/T-DM1 have received FDA approval in December 2019 for the treatment of patients with HER2-positive breast cancer who have progressed on available therapies.
The first antibody-photosensitizer conjugate (designated cetuximab saratolacan) has now received approval by the Japanese government for the treatment of advanced and recurrent head and neck cancer. It involves the water-soluble silicon-phthalocyanine derivative IRDye700DX (best known as IR700) conjugated to cetuximab. The latter is an FDA approved mAb targeting the epidermal growth factor receptor (EGFR), which is overexpressed in many types of cancer. The therapeutic strategy relies on the systemic administration of the targeting conjugate followed by a 24 h interval that enables tumor-specific accumulation of the targeting conjugate on the surface of EGFR+ cells. Tumor illumination is then performed at 690 nm by using an external laser. The absorption peak of IR700 at the near infra-red constitutes an important advantage of this system as it permits increased light penetration across tissues. Photoactivation of IR700 specifically kills EGFR-expressing tumor cells while sparing the tumor microenvironment as well as the surrounding healthy tissues.Citation8 Cytotoxicity is mediated by light-induced ROS generated by the cetuximab-IR700 attached at the cell surface that culminate in the necrotic disruption of the plasma membrane followed by the release of intracellular content, including tumor antigens and DAMPs,Citation9 which altogether trigger immune responses that largely contribute to long-term disease control. Curiously, necrosis is often observed upon PDT regardless of the photosensitizer. However, anti-tumor immunity triggered by PDT is not expected to be exclusively mediated by the release of cellular content as other modalities inducing accidental necrosis (such as freeze-thaw) are regarded as immunologically silent. Thus, it is reasonable to think that PDT has the capacity to generate novel antigens and/or DAMPs (e.g., oxidation-associate molecular patterns, OAMPs) that enhance the antigenicity and adjuvanticity of PDT-killed cancer cells. Nevertheless, the mechanisms underlying the immunostimulatory properties of PDT are poorly understood and deserve further investigation. As a possibility, apoptotic cell death occurring in the penumbra, at the edge of illuminated regions, may contribute to the immunogenicity of PDT ().Citation6
The aforementioned targeted delivery strategy is highly versatile as alternative antibodies can be used to target IR700 to other tumor antigens or other types of tumors. This has been successfully demonstrated in preclinical models of a range of malignancies including breast cancer (targeting HER2), prostate cancer (targeting PSMA), oral cancer (targeting CD44), glioblastoma (targeting CD133) and melanoma (targeting CD146).Citation8 Furthermore, the combination of cetuximab-IR700 with other immunotherapies, such as immune checkpoint inhibitors, might be advantageous for oncological patients. In fact, preclinical evaluation of cetuximab-targeted IR700 combined with blockade of the PD-1/PD-L1 axis has allowed for the complete eradication of MC38 colon tumors with signs of abscopal effects.Citation10 Currently, phase 1/2 clinical trials (NCT04305795) of cetuximab-targeted IR700 combined with the anti-PD1 antibody pembrolizumab are being conducted in patients with recurrent or metastatic head and neck squamous cell cancer or advanced cutaneous squamous cell carcinoma.
Disclosure of potential conflicts of interest
G.K. is a cofounder of everImmune and Therafast Bio. G.K. and O.K. are cofounders of Samsara Therapeutics.
Additional information
Funding
References
- Dabrowski JM, Arnaut LG. Photodynamic therapy (PDT) of cancer: from local to systemic treatment. Photochem Photobiol Sci. 2015;14(10):1765–3. doi:10.1039/C5PP00132C.
- Garg AD, Krysko DV, Verfaillie T, Kaczmarek A, Ferreira GB, Marysael T, Rubio N, Firczuk M, Mathieu C, Roebroek AJM, et al. A novel pathway combining calreticulin exposure and ATP secretion in immunogenic cancer cell death. Embo J. 2012;31(5):1062–1079. doi:10.1038/emboj.2011.497.
- Gomes-da-Silva LC, Zhao L, Bezu L, Zhou H, Sauvat A, Liu P, Durand S, Leduc M, Souquere S, Loos F, et al. Photodynamic therapy with redaporfin targets the endoplasmic reticulum and Golgi apparatus. Embo J. 2018;37:13. doi:10.15252/embj.201798354.
- Donohoe C, Senge MO, Arnaut LG, Gomes-da-Silva LC. Cell death in photodynamic therapy: from oxidative stress to anti-tumor immunity. Biochim Et Biophys Acta Rev Cancer. 2019;1872(2):188308. doi:10.1016/j.bbcan.2019.07.003.
- Tatsuno K, Yamazaki T, Hanlon D, Han P, Robinson E, Sobolev O, Yurter A, Rivera-Molina F, Arshad N, Edelson RL, et al. Extracorporeal photochemotherapy induces bona fide immunogenic cell death. Cell Death Dis. 2019;10(8):578. doi:10.1038/s41419-019-1819-3.
- Kepp O, Marabelle A, Zitvogel L, Kroemer G. Oncolysis without viruses - inducing systemic anticancer immune responses with local therapies. Nat Rev Clin Oncol. 2020;17(1):49–64. doi:10.1038/s41571-019-0272-7.
- Rosenblum D, Joshi N, Tao W, Karp JM, Peer D. Progress and challenges towards targeted delivery of cancer therapeutics. Nat Commun. 2018;9(1):1410. doi:10.1038/s41467-018-03705-y.
- Kobayashi H, Furusawa A, Rosenberg A, Choyke PL. Near-infrared photoimmunotherapy of cancer: a new approach that kills cancer cells and enhances anti-cancer host immunity. Int Immunol. 2020. doi:10.1093/intimm/dxaa037.
- Sato K, Ando K, Okuyama S, Moriguchi S, Ogura T, Totoki S, Hanaoka H, Nagaya T, Kokawa R, Takakura H, et al. Photoinduced ligand release from a silicon phthalocyanine dye conjugated with monoclonal antibodies: a mechanism of cancer cell cytotoxicity after near-infrared photoimmunotherapy. ACS Cent Sci. 2018;4(11):1559–1569. doi:10.1021/acscentsci.8b00565.
- Nagaya T, Friedman J, Maruoka Y, Ogata F, Okuyama S, Clavijo PE, Choyke PL, Allen C, Kobayashi H. Host immunity following near-infrared photoimmunotherapy is enhanced with PD-1 checkpoint blockade to eradicate established antigenic tumors. Cancer Immunol Res. 2019;7(3):401–413. doi:10.1158/2326-6066.CIR-18-0546.
