ABSTRACT
We report a new class of ruthenium (Ru)-based photosensitizers that induce potent cytotoxicity in melanoma cells following activation with NIR light. In addition to the direct cytotoxic effect, this Ru-based photodynamic therapy induces immunogenic cell death in melanoma cells that can be therapeutically exploited to establish protective antitumor immunity.
We recently reported ruthenium (Ru) coordination complexes derived from three different ligands arranged in a pseudo-octahedral geometry around the metal center.Citation1 Here, each ligand combined with Ru, plays a unique role in achieving the desired chemical and photophysical properties of the overall three-dimensional structure. In the [Ru(NNN)(NN)(L)]Cl2 construct, NNN is a tridentate polypyridyl ligand that is designed to shift the light absorption window for the photosensitizer into the near-infrared (NIR) region []. The bidentate, π-expanded NN ligand provides access to certain ligand-localized triplet states that are extremely good at sensitizing singlet oxygen, which is thought to be the most important mediator of the PDT effect.Citation2 The monodentate L ligand provides a handle for fine-tuning the absorption window and the efficiency of singlet oxygen production but also tunes the overall chemical and photochemical stabilities of the complexes as well as biological toxicity profiles. The modular architecture of the multiligand construct provides a robust platform for exploration of a vast molecular landscape.
Figure 1. Discovery of immunogenic cell death (ICD)-promoting ruthenium (Ru)-based photosensitizers (PS)
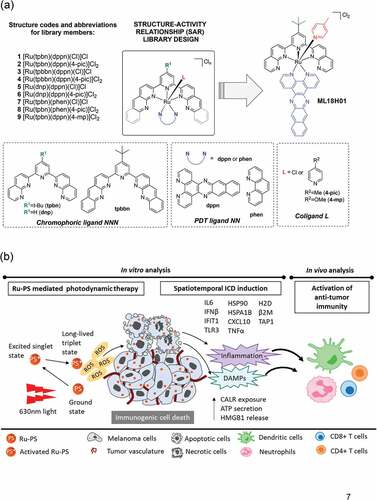
These Ru photosensitizers (PS) represent a marked departure from the traditional tetrapyrrole macrocycles that have been approved and/or clinically investigated for use as PDT agents and have salient advantages over the organic macrocycles. For example, Ru PSs are extremely modular and thus highly tunable for a broad range of applications. Further, the incorporation of the heavy Ru atom leads to near-unity efficiency for the formation of triplet states, which are the states responsible for sensitizing singlet oxygen, from their initially excited singlet states. Specifically, in this study, we have (i) identified the optimal combination of ligands on Ru to achieve NIR-absorbing singlet states and lowest energy triplet states for singlet oxygen production, and (ii) demonstrated a potent cytotoxic effect on melanoma cells with NIR PDT.Citation2
Next, we evaluated the immunological repercussions for cell death within melanoma cells following Ru PDT.Citation2 Considering the positive correlation between antitumor immunity and better melanoma outcomes,Citation3,Citation4 we specifically investigated a phenomenon of immunogenic cell death (ICD) within Ru PDT-treated melanoma cells. In the context of cancers, ICD is a regulated form of cell death that encompasses a cascade of immune responsesCitation5,Citation6 promoting adjuvanticity and antigenicity – both essential for the development of antitumor immunity. For this purpose, we assessed a series of ICD ‘hallmarks’ within melanoma cells following Ru PDT in vitro. First, we investigated reactive oxygen species (ROS), an important mediator of PDT-induced cytotoxicity and known mediator of hypericin PDT-induced ICD.Citation6 We found induction of both mitochondrial and cellular ROS in Ru PDT-treated B16F10 melanoma cells. Additionally, we captured significantly upregulated expression of genes encoding the constituents of type 1 interferon pathway, proinflammatory cytokines, and antigen-presentation machinery. Some of the key markers analyzed here included IL6 (a proinflammatory cytokine important for dendritic cell differentiation and PDT-mediated antitumor immunityCitation7), CXCL10 (involved in T cell chemotaxis), and TLR3Citation6 (involved in amplifying proinflammatory signals such as type I IFNsCitation6). Additionally, significantly enhanced HSP90 and HSPA1B gene expression and calreticulin translocation to plasma membrane, ‘eat-me’ signals enabling the uptake of dead and dying cancer cells by antigen-presenting cells, was observed within Ru PDT-treated melanoma cells.Citation5,Citation6 Finally, increased levels of ICD hallmarks ATP and HMGB1 were also detected in the extracellular media of Ru PDT-treated melanoma cells. Together, these data showed that cytotoxicity within melanoma cells following Ru PDT is accompanied by bona fide ICD markers in vitro.
To demonstrate the clinical utility, we then investigated the capacity of Ru PDT-induced ICD to activate antitumor immunity in vivo. For this purpose, Ru PDT-treated, ICD-undergoing B16F10 melanoma cells were used to vaccinate syngeneic C57BL/6NCrl mice on their left flanks. After 7 days, these mice were challenged with non-treated B16F10 cells on their right flanks. We found that vaccination with Ru-PDT-treated B16F10 cells conferred protection by causing either reduced or delayed tumor growth upon challenge with non-treated B16F10 cells, and lead to an improved tumor-free survival in vaccinated mice as compared to the unvaccinated group. As the vaccination and challenge were performed on the opposite flanks of the mice, the tumor-inhibiting effects of Ru PDT-based vaccinations also indicated the generation of systemic antitumor immunity causing the abscopal effect.
Of note, in light of growing appreciation of the influence of sex, as a biological variable, on cancer progression and therapeutic outcomes,Citation8 our studies included both male and female mice. Interestingly, we found that while tumor growth dynamics were comparable in male and female mice, protection from tumor development upon vaccination and tumor-free survival was higher in female mice as compared to males. These data are in congruence with the observations that male and female melanoma patients display different susceptibilities to therapeutic interventions resulting in sex-biased clinical outcomes.Citation8 Further, these data also agree with the observations reporting superior tumor-specific CD8 + T cell responses against B16F10 melanoma in female mice in response to checkpoint therapy.Citation9 Since CD8 + T cell responses are the ultimate immune effectors within ICD-induced antitumor immune response,Citation6 we believe that similar variations within anti-melanoma CD8 T cell responses occur following Ru PDT.
In summary, our results show that Ru PDT (with clinically approved red light) produces clinically desired anticancer attack on melanoma via two-prongs: 1) by causing direct cytotoxicity in melanoma cells, and 2) via ICD-mediated generation of protective antitumor immunity []. Such a Ru PDT-induced and non-exclusive two-pronged attack on cancers can be harnessed not only to eradicate existing cancer cells but also to establish protection against possible cancer relapse.
Acknowledgments
This work was supported via the National Cancer Institute (NCI) of the National Institutes of Health (NIH)’s Award R01CA222227 to S.A.M. and S.G. The content in this article is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Disclosure statement
S.A.M. has a potential research conflict of interest due to a financial interest with Theralase Technologies, Inc. and PhotoDynamic, Inc. A management plan has been created to preserve objectivity in research in accordance with UTA policy.
Additional information
Funding
- Lifshits LM, Roque III JA, Konda P, Monro S, Cole HD, von Dohlen D, Kim S, Deep G, Thummel RP, Cameron CG. Near-infrared absorbing Ru(ii) complexes act as immunoprotective photodynamic therapy (PDT) agents against aggressive melanoma. Chem Sci. 2020;11(43):11740–3. doi:10.1039/D0SC03875J.
- Monro S, Colón KL, Yin H, Roque J, Konda P, Gujar S, Thummel RP, Lilge L, Cameron CG, McFarland SA. Transition metal complexes and photodynamic therapy from a tumor-centered approach: challenges, opportunities, and highlights from the development of TLD1433. Chem Rev. 2019;119(2):797–828. doi:10.1021/acs.chemrev.8b00211.
- Ralli M, Botticelli A, Visconti IC, Angeletti D, Fiore M, Marchetti P, Lambiase A, de Vincentiis M, Greco A. Immunotherapy in the treatment of metastatic melanoma: current knowledge and future directions. J Immunol Res. 2020;2020:9235638. doi:10.1155/2020/9235638.
- Weiss SA, Wolchok JD, Sznol M. Immunotherapy of melanoma: facts and hopes. Clin Cancer Res. 2019;25(17):5191–5201. doi:10.1158/1078-0432.CCR-18-1550.
- Galluzzi L, Vitale I, Warren S, Adjemian S, Agostinis P, Martinez AB, Chan TA, Coukos G, Demaria S, Deutsch E. Consensus guidelines for the definition, detection and interpretation of immunogenic cell death. J Immunother Cancer. 2020;8(1):e000337. doi:10.1136/jitc-2019-000337.
- Galluzzi L, Buqué A, Kepp O, Zitvogel L, Kroemer G. Immunogenic cell death in cancer and infectious disease. Nat Rev Immunol. 2017;17(2):97–111. doi:10.1038/nri.2016.107.
- Gollnick SO, Evans SS, Baumann H, Owczarczak B, Maier P, Vaughan L, Wang WC, Unger E, Henderson BW. Role of cytokines in photodynamic therapy-induced local and systemic inflammation. Br J Cancer. 2003;88(11):1772–1779. doi:10.1038/sj.bjc.6600864.
- Conforti F, Pala L, Bagnardi V, De Pas T, Martinetti M, Viale G, Gelber RD, Goldhirsch A. Cancer immunotherapy efficacy and patients’ sex: a systematic review and meta-analysis. Lancet Oncol. 2018;19(6):737–746. doi:10.1016/S1470-2045(18)30261-4.
- Lin P-Y, Sun L, Thibodeaux SR, Ludwig SM, Vadlamudi RK, Hurez VJ, Bahar R, Kious MJ, Livi CB, Wall SR. B7-H1-dependent sex-related differences in tumor immunity and immunotherapy responses. J Immunol. 2010;185(5):2747–2753. doi:10.4049/jimmunol.1000496.
