ABSTRACT
There has been some concern among parents and in the media that vaccinating children against human papillomavirus could be seen as giving children permission to engage in risky sexual behaviors (also known as sexual disinhibition). Several studies have found this concern to be unfounded but there have been no attempts to synthesize the relevant studies in order to assess if there is evidence of sexual disinhibition. The aim of this study was to synthesize recent literature examining sexual behaviors and biological outcomes (e.g., sexually transmitted infections) post-HPV vaccination. We reviewed literature from January 1, 2008-June 30, 2015 using PubMed, CINAHL, and Embase with the following search terms: [(sex behavior OR sex behavior OR sexual) AND (human papillomavirus OR HPV) AND (vaccines OR vaccine OR vaccination)] followed by a cited reference search. We included studies that examined biological outcomes and reported behaviors post-vaccination in both males and females. Studies were reviewed by title and abstract and relevant studies were examined as full-text articles. We identified 2,503 articles and 20 were eventually included in the review. None of the studies of sexual behaviors and/or biological outcomes found evidence of riskier behaviors or higher rates of STIs after HPV vaccination. Instead, the studies found that vaccinated compared to unvaccinated individuals were less likely to report vaginal intercourse without a condom (OR = 0.5; 95%CI = 0.4–0.6) and non-use of contraception (OR = 0.27; 95%CI = 0.15–0.48) and unvaccinated participants had higher rates of Chlamydia (OR = 2.3; 95%CI = 1.06–5.00). These results should be reassuring to parents and health care providers.
Introduction
In 2015, the Centers for Disease Control and Prevention (CDC) reported that there are approximately 79 million Americans who are currently infected with Human Papillomavirus (HPV) and 14 million new infections occur every year, making it the most common sexually transmitted infection (STI).Citation1 It is estimated that the worldwide prevalence of HPV infection in women without cervical abnormalities is 11–12%. This varies by region with higher rates in sub-Saharan Africa (24%), Eastern Europe (21%) and Latin America (16%).Citation2 Infection with HPV is a risk factor for serious health issues including genital warts, cervical cancer, anal cancer, penile cancer, oropharyngeal cancers, and recurrent respiratory papillomatosis.Citation1 Currently, there are 3 different HPV vaccines: 1) A bivalent vaccine (2vHPV) that protects against HPV types 16 and 18, 2 types that are responsible for about 70% of cervical cancer diagnoses;Citation3 2) A quadrivalent vaccine (4vHPV) that protects against HPV-16 and 18 as well as HPV-6 and 11, 2 types that cause about 90% of the cases of genital warts;Citation4 and 3) A nine-valent HPV vaccine (9vHPV) was recently licensed by the US. Food and Drug Administration, and protects against the 4 HPV types in 4vHPV as well as 5 additional oncogenic types (HPV types 31, 33, 45, 52, and 58). 9vHPV has the potential to prevent 80–90% of cervical cancers, and many vulvar, vaginal, and anal cancers in addition to 90% of genital warts.Citation5
Despite the recommendations made by national immunization advisory committees around the world, HPV vaccination rates, especially in the United States, remain suboptimal.Citation6,7 For example, only 60.0% of adolescent girls and 41.7% of adolescent boys between the ages of 13 and 17 received one or more doses in the HPV vaccine series in the United States in 2014.Citation8 These rates are even lower when examining the percentages of adolescents who have completed the series, which is necessary in order to receive the maximum protection from the vaccine.Citation9 Several barriers to HPV vaccination exist including cost of the vaccination, lack of knowledge about HPV transmission, and parental concerns about vaccinating their children against an STI.Citation10
One concern among parents, clinicians, and public health officials, which has received particular attention in the media, is that the introduction of the HPV vaccine will lead to risk compensation, a concept introduced by Wilde in the 1980s that he called Risk Homeostasis Theory (RHT).Citation11 RHT suggests that individuals have innate set points for risk tolerance and that they change their behaviors based on perceived risk in order to maintain their set point. According to RHT, then, if an individual believes that a measure protects him/her from a certain risk, then he/she would be more likely to engage in risky behaviors.Citation11 Critics of the HPV vaccine have expressed concern that the receipt of the vaccine could cause adolescents to engage in riskier sexual behavior due to perceived protection from sexually transmitted infections.Citation12 This is a concern that has arisen throughout the world. For example, several Roman Catholic bishops in Canada discouraged vaccination, stating that school-based vaccination “sends a message that early sexual intercourse is allowed”Citation13 and that abstinence is the “only truly healthy choice.”Citation14,15 One study using qualitative interviews of mothers in the United Kingdom found it described as giving girls a “carte blanche” to engage in behaviors that put them at risk for pregnancy, HIV, and STIs.Citation16
These parental concerns might have been influenced by stories they encountered in the media.Citation17-19 A US study found a majority of news articles about HPV vaccination were neutrally toned but the results also indicated that there were several important informational facts that were omitted in the articles, namely information about side effects, duration of protection, and information about catch-up vaccination.Citation20 Internet use and accuracy of internet information regarding the HPV vaccine are important considerations because some studies have found that vaccine concerns can be influenced by negative (and often inaccurate) media reports.Citation21 Media reports and inaccurate information could cause parents concern prior to getting their children vaccinated, specifically in regard to risk compensation.
Across several studies between 16% and 26% of parents indicate that they were concerned that HPV vaccination would increase the likelihood that their child would engage in riskier sexual behavior.Citation22-25 However, this worry about sexual disinhibition was not usually listed as a reason for non-vaccination, and only 3–6% of parents cited this concern as a reason for refusing vaccination.Citation26,27
The objective of the present study was to conduct a systematic review of research literature to evaluate whether there is evidence (via either self-report of sexual behaviors or biological outcomes such as sexually transmitted infections) of sexual disinhibition following HPV vaccination. Our aims were to answer the following research questions: 1) Is there consistent, replicated evidence of increased self-reported risky sexual behaviors after HPV vaccination? and 2) Is there consistent, replicated evidence of increased incidence of sexually transmitted infections or pregnancies after HPV vaccination?
Results
Study characteristics
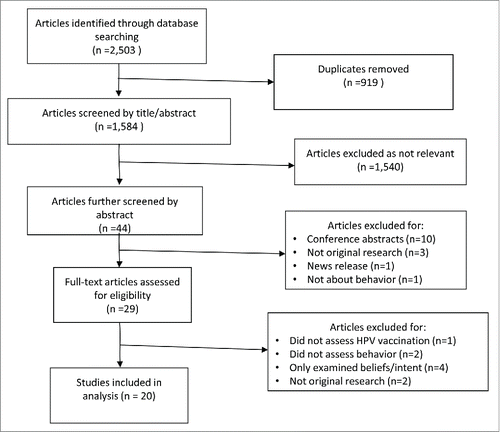
The initial search of the 3 databases resulted in 2,503 returns. Once duplicates were deleted, 1,584 articles remained. After screening by title and abstract, there were 29 full-text articles to be reviewed by all of the authors. The final selection resulted in 20 articles being included in the study. For the full PRISMA flow diagram, see .
Of the 20 studies included in the systematic review, 2 were qualitative, 12 were cross-sectional, and 8 were longitudinal studies. It is important to note that these categories are not mutually exclusive. If a study used more than one study design and the results were presented separately, it was counted in both relevant categories. Although we searched for studies between 2008 and 2015, the relevant studies for this review were published between 2011 and 2015. Furthermore, although we did not restrict by age or gender, all of the studies examined exclusively female populations and all of them studied populations within the 10 to 46 year-old age range. The descriptive statistics of the included studies are reported in . The full list of qualitative, cross-sectional, and longitudinal study results from the review are included as , respectively.
Table 1. Descriptive statistics and quality assessment.
Table 2. Qualitative studies.
Table 3. Cross-sectional studies.
Table 4. Longitudinal studies.
Critical appraisal of the studies
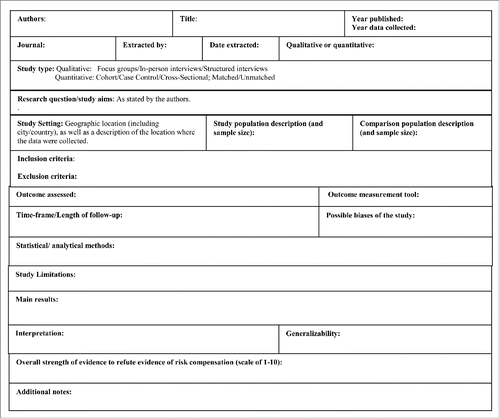
The 20 studies were divided among all of the co-authors, such that each study was reviewed by 2 co-authors, who then filled out corresponding data extraction sheets (the data extraction sheet used for this study is provided as Appendix A). The data extraction sheet contained items from previously validated data quality assessment tools including the Agency for Healthcare Research and Quality's (AHRQ), Cochrane, PRISMA, and the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies. Items on the data extraction sheet included assessment of study design, inclusion/exclusion criteria, study population and comparison group, length of follow-up, outcomes, limitations, generalizability, and an assessment of overall study quality. Overall study quality was assessed by each coauthor on a scale of 1–10 with higher numbers indicating greater study quality. The authors were given criteria with which to judge the studies and if the quality assessment from each author varied by more than 2 points, disagreements were resolved through discussion. The study's quality score was then averaged between the 2 reviewers' scores. Average study quality score ranged from 1.8 to 9.0 (mean = 6.2; standard deviation = 2.0).
Behavioral outcomes
All but 3 studies examined self-reported behavioral outcomes (n = 17, 85%). A widely studied behavior was sexual activity, which was assessed in many of the studies included in the review (n = 12) but was only statistically evaluated in 10 of the studies. Sexual activity was defined differently across studies, and included reported sexual intercourse any time after receipt of HPV vaccine (or any time after 10 y of age for the unvaccinated controls),Citation28 ever having sex with someone of the opposite sex,Citation29 and any experience of intercourse.Citation30,31 Other behaviors examined in the studies were: age at sexual debut (n = 7), number of sexual partners (n = 13), use of contraception (n = 16), and other sexual risk behaviors (n = 5).
Self-reported sexual activity
One study by Aujo et al. (2014) used both qualitative and quantitative methods to examine self-reported sexual activity in 2 different communities (one vaccinated and one unvaccinated community) in Uganda.Citation28 The qualitative component of the study found that most girls indicated they were not engaged in sexual activity themselves but they knew of others who were. The cross-sectional survey portion of the study found that young girls engaged in sexual activity in both the vaccinated and unvaccinated districts, but vaccinated girls had a lower reported rate of sexual intercourse than unvaccinated girls (1.5% vs. 2.5%).Citation28
The majority of the studies examining self-reported sexual activity were cross-sectional in nature and some of these cross-sectional studies did find that vaccinated participants were more likely to have engaged in sexual intercourse than unvaccinated participants.Citation30,32,33 However, these studies also noted that many of their participants (between 45% and 62%) were sexually active prior to HPV vaccination.Citation29,30 Due to the cross-sectional nature of these studies, these results may be more indicative of a woman engaging in sexual behavior and then seeking out protective measures as opposed to the vaccination causing the increased sexual behavior. Furthermore, previous studies have shown that providers are more likely to offer or strongly recommend the vaccine to patients they believe are sexually active or are not in a monogamous relationship.Citation34-36 Other cross-sectional studies found no statistical difference in sexual activity between vaccinated and unvaccinated participants (odds ratios between 0.88 and 1.07).Citation37-40
The longitudinal studies did not demonstrate that vaccinated women were more sexually active than unvaccinated women when post-vaccination behavior change was assessed. Some of these studies found that there was no statistically significant difference in baseline to follow-up sexual activity between vaccinated and unvaccinated women,Citation37,41,42 while others found decreases in sexual activity after vaccination. One study of female sex workers conducted by Brown et al. (2013) in Peru found female sex workers decreased sexual activity with all clients after vaccination (p < 0.001).Citation43 And another study (Mayhew et al., 2014) of a fully vaccinated sample found that participants between 16 and 21 y of age who inappropriately perceived lower risk for non-HPV STIs after vaccination were actually less likely to initiate sex over the following 6 month period (OR = 0.13; 95% CI = 0.03–0.69).Citation31
Self-reported age at sexual debut
Seven of the studies examined age at first intercourse as a risky behavior post-vaccination. The studies defined the variable differently with some looking at age as a continuous variable and some categorizing the variable as sexual debut before or after a certain age (usually 15 or 16). Regardless of how the variable was defined, none of the studies that examined age at sexual debut found a statistically significant difference between vaccinated and unvaccinated groups.Citation29,33,38,44-46 Only one study (Liddon et al., 2012) found an association between HPV vaccine and age at first intercourse. But this was only significant in the bivariate model of a subset of 20–24 y olds and was not significant in the multivariable model.Citation39
Self-reported number of sexual partners
Most of the studies examining the number of sexual partners between groups found no association between vaccination status and number of partners. This result held true in cross-sectional studies when the researchers examined number of lifetime partners,Citation29,33,37-40,44,45 number of partners in the last 12 months,Citation44 and number of partners before 18 y of age.Citation38 It also held true in studies that followed cohorts longitudinally, which found vaccinated women did not have more sexual partners at follow-up.Citation37,41,42 A Mayhew et al. (2014) study of a fully vaccinated sample did not see a significant increase in the number of sexual partners after the sample was vaccinated.Citation31
In fact, some of the studies found that vaccinated women had fewer sexual partners than unvaccinated women. One population-based study of Nordic women (Hansen et al., 2014) found that women who were vaccinated opportunistically (i.e., those that were vaccinated but not during an organized vaccination program) were significantly less likely than unvaccinated participants to have had 4 or more partners before reaching age 18 (AOR = 0.56; 95% CI = 0.40-0.78).Citation38 A similar finding was reported in a study conducted by Sadler et al. (2015) in the United Kingdom that found that non-vaccination was positively associated with having 3 or more partners in the last 6 months (OR = 2.12; 95% CI = 1.08-4.17).Citation46 A study conducted by Lutringer-Magnin et al. (2013) in France, found that vaccinated girls between the ages of 17 and 20 had had fewer partners than unvaccinated girls (p = 0.01).Citation40 Furthermore, a study of Peruvian female sex workers by Brown et al. (2013) found that participants had a significantly lower frequency of intercourse with new clients after vaccination (p < 0.001).Citation43
Use of contraception
Several of the studies examined use of contraception. Some assessed condom use while others examined use of hormonal contraception or counseling on contraception. While there has been some concern that an erroneous belief that the HPV vaccine protects against all STIs would result in a decrease in condom use, a study examining risk perceptions found that this erroneous belief did not result in decreased condom use.Citation31 A qualitative study by Ports et al. (2014) found that none of the women they interviewed reported that the HPV vaccine had an influence on their use of condoms during sexual activity.Citation47 Cross-sectional studies assessing condom use utilized different definitions for their studies including an assessment of general condom use,Citation30,44,45 consistent condom use in the last month,Citation39 condom use during most recent intercourse,Citation40 condom use during first intercourse,Citation40,48 and consistent condom use during all sexual encounters.Citation29,32,33,44 Regardless of how condom use was defined, none of the aforementioned studies found any statistically significant relationship between HPV vaccination and condom use. However, some studies did note that vaccinated women were more likely to take protective measures and found a positive association between condom use and vaccination. A study by Hansen et al. (2014) found that non-use of contraception during first intercourse was significantly less frequent among women who were vaccinated before sexual debut as opposed to their matched unvaccinated counterparts (OR=0.27; 95%CI=0.15-0.48 for those vaccinated during an organized vaccination program; OR=0.69; 95%CI=0.52-0.93 for those vaccinated opportunistically).Citation38 Liddon et al. (2012) found that a higher percentage of those who reported always or at least inconsistently using condoms reported being vaccinated as opposed to those who never used condoms.Citation39 Ruiz-Sternberg et al. (2014) reported a similar finding, that vaccinated women were more likely to report consistent condom use than unvaccinated women.Citation33 Additionally, Sadler et al. (2015) reported condom use at first intercourse was positively associated with HPV vaccination (OR = 0.55; 95%CI = 0.32-0.96).Citation48 There were 2 studies that examined use of emergency hormonal contraception (Lutringer-Magnin et al., 2013 and Sadler et al., 2015). Neither study found any differences in the use of emergency contraception between those who had been vaccinated and those who had not.Citation40,48
Contraceptive behavior was also examined by several studies longitudinally. Bednarczyk et al. (2012) used data from a managed care organization to assess the relationship between a patient seeking counseling on contraceptives and HPV vaccine. This study found that vaccinated participants were more likely to seek counseling on contraceptive use as opposed to unvaccinated participants, although the adjusted incident rate ratio was not significant (OR=2.31; 95%CI = 0.99-5.38).Citation49 This result is consistent with a study by Forster et al. (2012) that found no change in condom use from baseline to follow up by vaccination group (OR=0.88; 95%CI = 0.58-1.33).Citation37 A study of female sex workers found no change in condom use with all clients after vaccination but they did note that condom use with non-paying partners increased, although not significantly (p = 0.38).Citation43 Another longitudinal study (Cummings et al., 2012) found instances of vaginal intercourse without a condom over the last 2 months was significantly lower in the vaccinated group (p < 0 .001).Citation41 The findings from all of the studies combined demonstrated that there was either no association between HPV vaccination and condom use/contraception counseling or they demonstrated that vaccinated participants engaged in safer behaviors than unvaccinated participants.
Other risk behaviors
Many of the included studies also examined risky behaviors that did not fit in the above categories. Several of the studies examined relationship status. Most of them defined being in a monogamous relationship as a “safe” behavior and being in non-exclusive relationships or having one-night stands as constituting “unsafe” behavior. One study conducted in Uganda that used dating and sexual activity as measures of risky behavior found that unvaccinated girls were dating at higher rates than vaccinated girls (5% vs. 2.5%) but the authors did not assess if those specific dating relationships involved risky sexual behaviors.Citation28 Another study (Mather et al., 2012) compared “safe sexual behavior” between vaccinated and unvaccinated individuals by creating a composite measure that included condom use, use of other contraception, and having been in a monogamous relationship for at least 3 months. Using this measure, authors found no relationship between HPV vaccination and engaging in safe sexual behavior.Citation32 Rysavy et al. (2014) used a similar technique to examine “high risk sexual behaviors” such as frequency of condom use, number of partners, as well as experience of anal and oral intercourse and age at first anal and oral intercourse to create a risk behavior score. This study found that there were no differences between the vaccinated and unvaccinated groups on the composite risk behavior score.Citation45 This relationship was still not significant when the high risk behaviors were examined individually. Another study (Mattebo et al., 2014) examined high risk behaviors individually and assessed the effect of experiencing one-night stands, group sex, “friends with benefits” relationships, oral sex, and anal sex. In this study the authors did find that vaccinated women were more likely to have experienced a one-night stand (p = 0.046) but this cross-sectional study noted that 62% of their sample had reached sexual debut before vaccination so the temporal relationship between sexual experience and vaccination cannot be assessed and it is possible a person engaging in high-risk sexual behaviors was more likely to seek vaccination.Citation30 Furthermore, this study found no differences when comparing vaccinated and unvaccinated individuals with respect to experiencing a “friends with benefits” relationship, group sex, giving oral sex, receiving oral sex, or anal sex.Citation30 Lastly, a cross-sectional study examined several risk behaviors including anal intercourse, sexual experiences abroad, use of drugs or alcohol, and being a current smoker. Among other risk behaviors previously discussed (i.e., contraception use, number of partners, etc.) this study found that the unvaccinated group was more likely to have experienced anal intercourse as their last sexual contact (OR = 4.43; 95%CI = 1.23-14.29) and to be a current smoker.Citation48 Smoking status is an important factor to examine because smoking is a risk factor for cervical cancer, even when controlling for the effects of HPV infection and other potential cofactors.Citation50,51
Biological outcomes
Along with reporting behavioral outcomes, several studies also examined biological outcomes (n = 9, 45%). For the purposes of this review, they have been divided into two general categories: STI testing/diagnosis and pregnancy (including a composite measure of STI and pregnancy)/abortions.
STI/HIV testing or diagnosis
All of the studies in this subset had some measure of STI testing or diagnosis. Of the studies that examined STI or HIV testing or diagnosis, none of them found HPV vaccination to increase STI rates. Of the cross-sectional studies, one (Liddon et al., 2012) found no association between HPV vaccination and receiving STI services in the past year in both the 15-19 and the 20-24 y old age groups.Citation39 Two cross-sectional studies examining reported STI diagnosesCitation30,45 and one examining HIV serologyCitation48 all found no significant differences between the vaccinated and unvaccinated groups. Additionally, one study (Sadler et al., 2015) found that unvaccinated women were more likely to have received a positive Chlamydia trachomatis diagnosis (OR = 2.3; 95% CI = 1.06-5.00).Citation48
Two longitudinal studies (Cummings et al., 2012 and Jena et al., 2015) examined the association between STIs and HPV vaccination.Citation41,52 One longitudinal study by Jena et al. (2015) found that the difference-in-difference odds ratios in the year after vaccination was similar between the vaccinated and unvaccinated groups.Citation52 This held true for both the 12 to 14 and the 15 to 18 year-old groups. Another cohort study found no differences in Chlamydia or Trichomonas infection rates between vaccinated and matched unvaccinated groups.Citation41
Abortion/Pregnancy or a composite measure of STI and pregnancy
Several studies used composite measures of STI diagnosis and pregnancies. One cohort study (Bednarczyk et al., 2012) looked at a composite measure of testing or diagnosis of Chlamydia trachomatis infection, pregnancy, or venereal disease not otherwise specified (referred to as testing/diagnosis/counseling) found that girls receiving HPV vaccine did not have significantly higher rates of testing/diagnosis/counseling. They found the same results when they examined diagnosis-only rates between vaccinated and unvaccinated groups.Citation49 Another cohort study (Smith et al., 2015) found no statistically significant increase in a composite measure of STIs and pregnancy in relation to HPV vaccination. This result held true when STIs and pregnancy were examined separately as well.Citation53
Studies also examined pregnancy and abortions individually. Sadler et al. (2015) found no association between vaccination status and abortion (p = 0.85).Citation48 Another (Lutringer-Magnin et al., 2013) assessed abortion rates between vaccinated and unvaccinated groups found that 7.1% of their population had had an abortion and all of these participants were unvaccinated.Citation40 Additionally, a study with a population between 13 and 23 y of age found that unvaccinated women were more likely to have been pregnant (20% vs. 8.6%, p = 0.016), a result that runs contrary to the notion of risk compensation.Citation45
Discussion
There have been several studies examining the association between HPV vaccination using self-report or biological markers of increased risky sexual behaviors. Twenty studies were identified for this systematic review. The included studies were all published between 2011 and 2015 and varied in study design and sample size. Each of the studies had limitations and the results of each individual research effort should be interpreted with caution. However, when this body of literature is examined as a whole, with similar findings reported across studies, the consistent and replicated evidence indicates that HPV vaccination does not lead to risk compensation/sexual disinhibition. The finding that adolescents do not appear to respond to vaccination with increased risky sexual behavior suggests no support for RHT as applied to HPV vaccination. This conclusion is supported both by studies that focused on self-reported sexual behavior as well as studies that examined biological markers of risk (e.g., STI diagnosis).
Furthermore, there appeared to be more support for the fact that vaccinated women actually showed less involvement in risky behaviors than unvaccinated women, which was evidenced by lower numbers of sexual partners and increased use of contraception. This finding is not entirely surprising in light of previous research reporting that pro-health behaviors tend to cluster together, such that a person who engages in one protective health behavior (e.g., getting vaccinated) is more likely to engage in another (e.g., using condoms).Citation54 Additionally, an HPV vaccination visit to a healthcare provider may present families and providers with opportunities to discuss and promote health and disease prevention behaviors.
Several studies that examined risk perception merit discussion, even though they did not meet the inclusion criteria for the systematic review, and therefore were not presented in the Results section. Some of these studies examined the perception of risk in a completely vaccinated sample and compared a decreased risk perception post-vaccination to subsequent sexual behaviors. These studies found that vaccinated individuals perceived a need for safer sexual behavior after vaccination.Citation55-57 It is notable that these studies' findings were consistent with the results of studies included in this review. Furthermore, authors of a recent review of the literature on HPV vaccine attitudes and uptake found that the concern about risk compensation following vaccination was a “myth” rather than a valid concern.Citation58
This systematic review does have limitations so conclusions must be drawn with caution. First, the studies included are heterogeneous in both population and outcome definitions, which could reduce the specificity and precision of the findings. This issue is common in systematic reviews of this nature. For instance, one recent systematic review examining HPV vaccination or vaccine intent found varied evidence between HPV vaccination or vaccine intent and sexual behavior primarily due to the heterogeneous nature of the included studies.Citation59 At the same time, the fact that similar results are reported across studies that varied so widely in methodology and population, suggests that the findings of no association between vaccination and sexual risk compensation are robust. Secondly, the desire to engage in risky sexual behavior might cause a person to seek out vaccination which would make it appear that the vaccinated group practices more risky sexual behavior. This can be difficult to assess, particularly in cross-sectional studies because, as some research has shown, there are times when physicians have vaccinated their patients based on perceived risk status.Citation34-36 This should not be confused with an implication that the vaccination is the cause of the risky behavior. Finally, this review of the literature is reliant on the findings of the studies included in the review. Each of these individual studies had limitations of their own including: a lack of a comparison group, recall bias, and social desirability of responses among others. Nevertheless, this study thoroughly examined 3 different databases with relevant search terms in order to capture the appropriate studies. To our knowledge, this is the first systematic review examining the association (or lack thereof) between HPV vaccination and subsequent sexual behaviors and adds credibility to the literature by combining the results and showing there is no consistent, replicated evidence of sexual disinhibition after HPV vaccination.
Materials and methods
A systematic review of the literature was performed using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) protocol and searching the PubMed, CINAHL, and Embase databases. The following search terms were used: [(sex behavior OR sex behavior OR sexual) AND (human papillomavirus OR HPV) AND (vaccines OR vaccine OR vaccination)] and the results were limited to studies from January 1, 2008 to June 30, 2015. The January 1, 2008 start date was chosen in order to capture articles examining behaviors post-vaccination. 4vHPV was first licensed in the US in mid-2006 and the Advisory Committee on Immunization Practices (ACIP) voted to routinely recommend it for girls in June 2006, at which point it was covered by the majority of insurers.Citation60 These recommendations were published in the Morbidity and Mortality Weekly Report in March 2007.Citation61 Therefore, relevant research examining post-vaccination behaviors was not published until 2010. Nevertheless, the 2008 start date for the search was chosen as a conservative estimate in order to ensure that we captured all relevant research.
To answer the outlined research questions, study outcomes were divided into 2 groups: self-reported behaviors (age at sexual initiation, reported number of sexual partners, and reported condom usage), and biological outcomes (pregnancy and STI diagnoses). Articles were included if they examined reported behaviors and/or biological outcomes post-vaccination. Studies were not restricted by age, gender, or geographic location. Articles were excluded if they examined worry or behavioral intent but not actual behaviors. Studies were also excluded if they were not original, peer-reviewed research articles (e.g. published abstracts for conferences, press releases, or commentaries on other articles).
First, studies were screened for inclusion by reviewing the titles and abstracts (MLK & GKS). Additional studies were excluded by a closer examination of the remaining abstracts (MLK & GDZ). The remaining studies were examined as full-text articles. Two authors independently reviewed each study to determine the relevance for inclusion. Each reviewer completed a data extraction and quality assessment sheet for each article. Most of the quality assessment tools for systematic reviews were developed and validated for intervention research.Citation62-65 The data extraction sheet used in this systematic review was developed by combining relevant portions of previously validated data quality assessment tools from the Agency for Healthcare Research and Quality's (AHRQ), Cochrane, PRISMA, and the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies.
Conclusion
The consistent, replicated evidence found across the 20 studies examined in this systematic review provides a strong body of evidence refuting that there is an association between HPV vaccination and risky sexual behavior. The 20 different studies, utilizing at least 4 distinct study designs, and including a total of 521,879 participants, found no evidence of increased numbers of sexual partners, younger age of sexual initiation, decreased use of contraception (including both condoms and hormonal contraceptives), increased STI diagnoses, increased pregnancy rates, or increased history of abortion among those vaccinated against HPV. In fact, some studies found vaccinated women showed lower risky behaviors than unvaccinated women, indicating a tendency toward less risky health behaviors. These findings should alleviate parental and provider concerns that HPV vaccination will lead to risky sexual behaviors. Furthermore, as others have noted, even if risk compensation was identified as an issue related to HPV vaccination, this would not be justification for withholding vaccination, but would argue for effective pre- and post-vaccination counseling.Citation58
Disclosure of potential conflicts of interest
Dr. Kahn chaired the review committee for a grant to the Society for Adolescent Health and Medicine evaluating public health demonstration project proposals to improve adolescent vaccination; grant funding for this program was from Merck, Inc. Dr. Kahn co-chaired two HPV vaccine clinical trials in HIV-positive individuals, for which Merck & Co., Inc., provided vaccine and immunogenicity titers. Dr. Rosberger has served as a consultant for Merck at a meeting in October 2015 and as a lecturer to family physicians in April 2015. Dr. Zimet is an investigator on investigator-initiated research funded by Merck and by Roche and he served as a consultant to Merck in 2014.
Acknowledgments
GS is a Vanier Canada Graduate Scholar and a Canadian Queen Elizabeth II Diamond Jubilee Scholar.
Funding
MK is supported by the National Cancer Institute of the National Institutes of Health under Award Number R25CA117865. Its content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, including the National Cancer Institute or the National Institute of Nursing Research. Effort on this manuscript was supported, in part, by the Center for HPV Research, which is funded by the Indiana University–Purdue University Indianapolis Signature Centers Initiative in conjunction with the Indiana University School of Medicine Department of Pediatrics and the Indiana University Melvin and Bren Simon Cancer Center.
References
- Centers for Disease Control and Prevention. Genital HPV infection—CDC Fact Sheet. Atlanta, GA, 2014. Retrieved from: http://www.cdc.gov/std/hpv/hpv-factsheet-march-2014.pdf
- Forman D, de Martel C, Lacey CJ, Soerjomataram I, Lortet-Tieulent J, Bruni L, Vignat J, Ferlay J, Bray F, Plummer M, et al. Global burden of human papillomavirus and related diseases. Vaccine 2012; 30 Suppl 5:F12–23; PMID:23199955; http://dx.doi.org/10.1016/j.vaccine.2012.07.055
- Centers for Disease Control Prevention. FDA licensure of bivalent human papillomavirus vaccine (HPV2, Cervarix) for use in females and updated HPV vaccination recommendations from the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2010; 59:626–9; PMID:20508593.
- Centers for Disease Control and Prevention. HPV vaccine information for clinicians—Fact sheet. Atlanta, GA 2012. Retrieved from: http://www.cdc.gov/std/hpv/stdfact-hpv-vaccine-hcp.htm
- United States Food and Drug Administration. FDA approves Gardasil 9 for prevention of certain cancers caused by five additional types of HPV. 2014; Retrieved from: http://www.fda.gov/NewsEvents/ Newsroom/PressAnnouncements/ucm426485.htm
- Stokley S, Jeyarajah J, Yankey D, Cano M, Gee J, Roark J, Curtis RD, Markowitx L. Human papillomavirus vaccination coverage among adolescents, 2007-2013, and post-licensure vaccine safety monitoring, 2006-2014 - United States. MMWR Morb Mortal Wkly Rep. 2014; 63: 620–624; PMID:25055185.
- Wigle J, Fontenot H, Zimet G. Global delivery of human papillomavirus vaccines. Pediatr Clin North Am 2016; 63:81–95; PMID:26613690; http://dx.doi.org/10.1016/j.pcl.2015.08.004
- Reagan-Steiner S, Yankey D, Jeyarajah J, Elam-Evans LD, Singleton JA, Curtis CR, MacNeil J, Markowitz LE, Stokley S. National, regional, state, and selected local area vaccination coverage among adolescents aged 13–17 years—United States, 2014. MMWR Morb Mortal Wkly Rep; 64:784–92; http://dx.doi.org/10.15585/mmwr.mm6429a3
- United States Department of Health and Human Services. HPV (Human Papillomavirus) vaccine. 2015; Retrieved from: http://www.vaccines.gov/diseases/hpv/
- Dempsey AF, Davis MM. Overcoming barriers to adherence to HPV vaccination recommendations. Am J Manag Care 2006; 12:S484–91; PMID:17203992.
- Wilde GJ. Risk homeostasis theory: an overview. Inj Prev 1998; 4:89–91; PMID:9666358; http://dx.doi.org/10.1136/ip.4.2.89
- Pollitt K. Virginity or death! The Nation. New York, NY: Nation Co Inc 33 Irving Place, 8th Floor, New York, NY 10003 USA, 2005.
- Guichon JR, Mitchell I, Buffler P, Caplan A. Citizen intervention in a religious ban on in-school HPV vaccine administration in Calgary, Canada. Prev Med 2013; 57:409–13; PMID:23769899; http://dx.doi.org/10.1016/j.ypmed.2013.06.005
- HPV vaccine not as effective as abstinence, B.C. bishop tells parents. CBC News. Sep 24, 2015. Retrieved from: http://www.cbc.ca/news/canada/british-columbia/hpv-vaccine-abstinence-bishop-bc-1.3242076
- Smith R, Motiuk D, Henry F, Pettipas G, Bouchard L, Chatlain M. A message to Catholic parents/ guardians and the Catholic educational community 2008. Available:http://ccsdmedia.s3.amazonaws.com/wp-content/uploads/2014/08/Alberta_Bishops_letter.pdf
- Waller J, Marlow LA, Wardle J. Mothers’ attitudes towards preventing cervical cancer through human papillomavirus vaccination: a qualitative study. Cancer Epidemiol Biomarkers Prev 2006; 15:1257–61; PMID:16835320; http://dx.doi.org/10.1158/1055-9965.EPI-06-0041
- Brewer NT, Fazekas KI. Predictors of HPV vaccine acceptability: a theory-informed, systematic review. Prev Med 2007; 45:107–14; PMID:17628649; http://dx.doi.org/10.1016/j.ypmed.2007.05.013
- Friedman AL, Shepeard H. Exploring the knowledge, attitudes, beliefs, and communication preferences of the general public regarding HPV findings from CDC focus group research and implications for practice. Health Educ Behav 2007; 34:471–85; PMID:17000622; http://dx.doi.org/10.1177/1090198106292022
- Olshen E, Woods ER, Austin SB, Luskin M, Bauchner H. Parental acceptance of the human papillomavirus vaccine. J Adolesc Health 2005; 37:248–51; PMID:16109349; http://dx.doi.org/10.1016/j. jadohealth.2005.05.016
- Habel MA, Liddon N, Stryker JE. The HPV vaccine: a content analysis of online news stories. J Womens Health 2009; 18:401–7; http://dx.doi.org/10.1089/jwh.2008.0920
- Rosenthal D, Dyson S, Pitts M, Garland S. Challenges to accepting a human papilloma virus (HPV) vaccine: a qualitative study of Australian women. Women & Health 2007; 45:59–73; PMID:18019286; http://dx.doi.org/10.1300/J013v45n02_04
- Mayer MK, Reiter PL, Zucker RA, Brewer NT. Parents’ and sons’ beliefs in sexual disinhibition after human papillomavirus vaccination. Sex Transm Dis 2013; 40:822–8; PMID:24275737; http://dx.doi. org/10.1097/OLQ.0000000000000021
- Ogilvie GS, Remple VP, Marra F, McNeil SA, Naus M, Pielak KL, Ehlen TG, Dobson SR, Money DM, Patrick DM. Parental intention to have daughters receive the human papillomavirus vaccine. Can Med Assoc J 2007; 177:1506–12; http://dx.doi.org/10.1503/cmaj.071022
- Schuler CL, Reiter PL, Smith JS, Brewer NT. Human papillomavirus vaccine and behavioural disinhibition. Sex Transm Infect 2011; 87:349–353; PMID:21357601; http://dx.doi.org/10.1136/sti.2010.048017
- Marlow LAV, Forster AS, Wardle J, Waller J. Mothers’ and adolescents’ beliefs about risk compensation following HPV vaccination. J Adolesc Health 2009; 44:446–51; PMID:19380091; http://dx.doi.org/10.1016/j.jadohealth.2008.09.011
- Brabin L, Roberts SA, Stretch R, Baxter D, Chambers G, Kitchener H, McCann R. Uptake of first two doses of human papillomavirus vaccine by adolescent schoolgirls in Manchester: prospective cohort study. BMJ 2008; 336:1056–8; PMID:18436917; http://dx.doi.org/10.1136/bmj.39541.534109.BE
- Etter DJ, Zimet GD, Rickert VI. Human papillomavirus vaccine in adolescent women: a 2012 update. Curr Opin Obstet Gynecol 2012; 24:305–10; PMID:22781077; http://dx.doi.org/10.1097/ GCO.0b013e3283567005
- Aujo JC, Bakeera-Kitaka S, Kiguli S, Mirembe F. No difference in sexual behavior of adolescent girls following Human Papilloma Virus vaccination: a case study two districts in Uganda; Nakasongola and Luwero. BMC Public Health 2014; 14:155; PMID:24520841; http://dx.doi.org/10.1186/1471-2458-14-155
- Marchand E, Glenn B, Bastani R. HPV vaccination and sexual behavior in a community college sample. J Commun Health 2013; 38:1010–4; http://dx.doi.org/10.1007/s10900-013-9710-0
- Mattebo M, Grun N, Rosenblad A, Larsson M, Haggstrom-Nordin E, Dalianis T, Tyden T. Sexual experiences in relation to HPV vaccination status in female high school students in Sweden Eur J Contracept Reprod Health Care 2014; 19:86–92; PMID:24555926; http://dx.doi.org/10.3109/13625187.2013.878021
- Mayhew A, Mullins TL, Ding L, Rosenthal SL, Zimet GD, Morrow C, Kahn JA. Risk perceptions and subsequent sexual behaviors after HPV vaccination in adolescents. Pediatrics 2014; 133:404–11; PMID:24488747; http://dx.doi.org/10.1542/peds.2013-2822
- Mather T, McCaffery K, Juraskova I. Does HPV vaccination affect women's attitudes to cervical cancer screening and safe sexual behaviour? Vaccine 2012; 30:3196–201; PMID:22425789; http://dx.doi.org/10.1016/j.vaccine.2012.02.081
- Ruiz-Sternberg AM, Pinzon-Rondon AM. Risk perception and sexual behavior in HPV-vaccinated and unvaccinated young Colombian women. Int J Gynaecol Obstet 2014; 126:205–8; PMID:24996685; http://dx.doi.org/10.1016/j.ijgo.2014.03.033
- Gilkey MB, Malo TL, Shah PD, Hall ME, Brewer NT. Quality of physician communication about Human Papillomavirus vaccine: Findings from a national survey. Cancer Epidemiol Biomarkers Prev 2015; 24:1673–9; PMID:26494764; http://dx.doi.org/10.1158/1055-9965.EPI-15-0326
- Zimet GD, Stupiansky NW, Weiss TW, Rosenthal SL, Good MB, Vichnin MD. Influence of patient's relationship status and HPV history on physicians’ decisions to recommend HPV vaccination. Vaccine 2011; 29:378–81; PMID:21111781; http://dx.doi.org/10.1016/j.vaccine.2010.11.027
- Kepka D, Berkowitz Z, Yabroff KR, Roland K, Saraiya M. Human papillomavirus vaccine practices in the USA: do primary care providers use sexual history and cervical cancer screening results to make HPV vaccine recommendations? Sex Transm Infect 2012; 88:433–5; PMID:22522751; http://dx.doi.org/10.1136/sextrans-2011-050437
- Forster AS, Marlow LA, Stephenson J, Wardle J, Waller J. Human papillomavirus vaccination and sexual behaviour: cross-sectional and longitudinal surveys conducted in England. Vaccine 2012; 30:4939–44; PMID:22664223; http://dx.doi.org/10.1016/j.vaccine.2012.05.053
- Hansen BT, Kjaer SK, Arnheim-Dahlstrom L, Liaw KL, Jensen KE, Thomsen LT, Munk C, Nygard M. Human papillomavirus (HPV) vaccination and subsequent sexual behaviour: evidence from a large survey of Nordic women. Vaccine 2014; 32:4945–53; PMID:25045810; http://dx.doi.org/10.1016/j.vaccine.2014.07.025
- Liddon NC, Leichliter JS, Markowitz LE. Human Papillomavirus vaccine and sexual behavior among adolescent and young women. Am J Prev Med 2012; 42:44–52; PMID:22176845; http://dx.doi.org/ 10.1016/j.amepre.2011.09.024
- Lutringer-Magnin D, Kalecinski J, Cropet C, Barone G, Ronin V, Regnier V, Leocmach Y, Jacquard AC, Vanhems P, Chauvin F, et al. Prevention of sexually transmitted infections among girls and young women in relation to their HPV vaccination status. Eur J Public Health 2013; 23:1046–53; PMID:23430762; http://dx.doi.org/10.1093/eurpub/ckt018
- Cummings T, Zimet GD, Brown D, Tu W, Yang Z, Fortenberry JD, Shew ML. Reduction of HPV infections through vaccination among at-risk urban adolescents. Vaccine 2012; 30:5496–9; PMID:22750043; http://dx.doi.org/10.1016/j.vaccine.2012.06.057
- Al Romaih WRR, Srinivas A, Shahtahmasebi S, Omar HA. No significant change in sexual behavior in association with human papillomavirus vaccination in young girls. Int J Child Adolesc Health 2011; 4:351–5.
- Brown B, Blas MM, Heidari O, Carcamo C, Halsey NA. Reported changes in sexual behaviour and human papillomavirus knowledge in Peruvian female sex workers following participation in a human papillomavirus vaccine trial. Int J STD AIDS 2013; 24:531–5; PMID:23970767; http://dx.doi.org/10.1177/0956462412473891
- Ratanasiripong NT. Human papillomavirus vaccine increases high-risk sexual behaviors: a myth or valid concern. J Sch Nurs 2014; 30:456–63; PMID:24414172; http://dx.doi.org/10.1177/ 1059840513520042
- Rysavy MB, Kresowik JD, Liu D, Mains L, Lessard M, Ryan GL. Human papillomavirus vaccination and sexual behavior in young women. J Pediatr Adolesc Gynecol 2014; 27:67–71; PMID:24405635; http://dx.doi.org/10.1016/j.jpag.2013.08.009
- Sadler L, Roberts SA, Hampal G, McManus D, Mandal D, Brabin L. Comparing risk behaviours of human papillomavirus-vaccinated and non-vaccinated women. J Fam Plann Reprod Health Care 2015; PMID:25605479.
- Ports KA, Barnack-Tavlaris JL, Mosavel M, Murithi LK. Young women's sexual and reproductive health post HPV vaccination. Womens Reprod Health 2014; 1:43–55; http://dx.doi.org/10.1080/ 23293691.2014.901804
- Sadler L, Roberts SA, Hampal G, McManus D, Mandal D, Brabin L. Comparing risk behaviours of human papillomavirus-vaccinated and non-vaccinated women. J Fam Plann Reprod Health Care 2015; 41:255–8; PMID:25605479; http://dx.doi.org/10.1136/jfprhc-2014-100896
- Bednarczyk RA, Davis R, Ault K, Orenstein W, Omer SB. Sexual activity–related outcomes after human papillomavirus vaccination of 11-to 12-year-olds. Pediatrics 2012; 130:798–805; PMID:23071201; http://dx.doi.org/10.1542/peds.2012-1516
- Fonseca-Moutinho JA. Smoking and cervical cancer. ISRN obstetrics and gynecology 2011; 2011:847684; PMID:21785734; http://dx.doi.org/10.5402/2011/847684
- International Agency for Research on Cancer. Tobacco smoke and involuntary smoking. IARC monographs on the evaluation of carcinogenic risks to humans / World Health Organization, International Agency for Research on Cancer 2004; 83:1–1438.
- Jena AB, Goldman DP, Seabury SA. Incidence of sexually transmitted infections after human papillomavirus vaccination among adolescent females. JAMA Int Med 2015; 175:617–23; http://dx.doi.org/10.1001/jamainternmed.2014.7886
- Smith LM, Kaufman JS, Strumpf EC, Levesque LE. Effect of human papillomavirus (HPV) vaccination on clinical indicators of sexual behaviour among adolescent girls: the Ontario Grade 8 HPV Vaccine Cohort Study. Can Med Assoc J 2015; 187:E74–81; http://dx.doi.org/10.1503/cmaj.140900
- Fortenberry JD, Costa FM, Jessor R, Donovan JE. Contraceptive behavior and adolescent lifestyles: A structural modeling approach. J Res Adolesc 1997; 7:307–29; PMID:12292602; http://dx.doi.org/10.1207/s15327795jra0703_4
- Kahn JA, Xu J, Zimet GD, Liu N, Gonin R, Dillard ME, Squires K. Risk perceptions after Human Papillomavirus Vaccination in HIV-infected adolescents and young adult women. J Adolesc Health 2012; 50:464–70; PMID:22525109; http://dx.doi.org/10.1016/j.jadohealth.2011.09.005
- Mullins TL, Zimet GD, Rosenthal SL, Morrow C, Ding L, Shew M, Fortenberry JD, Bernstein DI, Kahn JA. Adolescent perceptions of risk and need for safer sexual behaviors after first human papillomavirus vaccination. Arch Pediatr Adolesc Med 2012; 166:82–8; PMID:22213755; http://dx.doi.org/10.1001/archpediatrics.2011.186
- Mullins TLK, Widdice LE, Rosenthal SL, Zimet GD, Kahn JA. Risk perceptions, sexual attitudes, and sexual behavior after HPV vaccination in 11–12 year-old girls. Vaccine 2015; 33:3907–12; PMID:26116249; http://dx.doi.org/10.1016/j.vaccine.2015.06.060
- Zimet GD, Rosberger Z, Fisher WA, Perez S, Stupiansky NW. Beliefs, behaviors and HPV vaccine: Correcting the myths and the misinformation. Prev Med 2013; 57:414–8; PMID:23732252; http://dx.doi.org/10.1016/j.ypmed.2013.05.013
- Coles VA, Patel AS, Allen FL, Keeping ST, Carroll SM. The association of human papillomavirus vaccination with sexual behaviours and human papillomavirus knowledge: a systematic review. Int J STD AIDS 2014:777–88; PMID:25300588.
- Hunsaker J, Veselovskiy G, Gazmararian JA. Health insurance plans and immunization: assessment of practices and policies, 2005-2008. Pediatrics 2009; 124 Suppl 5:S532–9; PMID:19948585; http://dx.doi.org/10.1542/peds.2009-1542M
- Markowitz LE, Dunne E, Saraiya M, Lawson H, Chesson H, Unger E. Quadrivalent human papillomavirus vaccine. MMWR Morb Mortal Wkly Rep 2007; 56:1–24; PMID:17218934.
- Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. J Clin Epidemiol 2009; 62:1006–12; PMID:19631508; http://dx.doi.org/10.1016/j.jclinepi.2009.06.005
- Higgins JP, Green S. Cochrane handbook for systematic reviews of interventions. Wiley Online Library, 2008.
- West S, King V, Carey TS, Lohr KN, McKoy N, Sutton SF, Lux L. Systems to rate the strength of scientific evidence. Evid Rep Technol Assess (Summ) 2002:1–11; PMID:11979732.
- National Institutes of Health. Quality assessment tool for observational cohort and cross-sectional studies. 2014. Available: https://www.nhlbi.nih.gov/health-pro/guidelines/in-develop/cardiovascularrisk-reduction/tools/cohort