ABSTRACT
Immune checkpoint inhibitors have shown efficacy in the treatment of different cancers by stimulating the antitumoral activity of the patient’s immune system, representing a major breakthrough in the field of cancer therapy. Monoclonal antibodies including anti-cytotoxic T-lymphocyte–associated protein 4, anti–programmed cell death protein 1 and its ligand inhibitors have been approved for advanced melanoma among other solid cancers. Although immunotherapy demonstrated a good safety profile, a new spectrum of multisystemic immune-related adverse events has been recently reported due to their use. Cutaneous reactions represent one of the leading adverse events, often reported in literature as “skin rash”, and rarely further characterized in distinct dermatologic entities. Herein we describe the distinctive cutaneous rashes occurring during immunotherapies for advanced melanoma, discussing implications in the treatment management.
Introduction
The introduction of immunotherapy has established a milestone in the treatment of several cancers, and novel anticancer therapies targeting immune checkpoints (ICs) pathways have shown remarkable clinical activity.Citation1 The upregulation of ICs and their ligands in several cancers, associated to the ICs role in tumor immune evasion has led to the development of new agents blocking the interaction between ICs and their ligands. Both cytotoxic T-lymphocyte–associated protein-4 (CTLA-4) and programmed cell death protein-1 (PD-1) belong to the class of negative key regulators of T cell-activation, modulating cellular mechanism of priming, activation and proliferation.Citation1 The main biological activity of immune-checkpoint inhibitors (ICIs) is to revert the immune-inhibitory tumor microenvironment balance toward a pro-inflammatory state mediating the activation of a cellular immune response addressed to specific tumor antigens.Citation1,Citation2 The use of ICIs is increasing among different cancer typesCitation3–5 including advanced melanoma;Citation6 however, their use is associated to nonspecific immune activation leading to possible different immune-related adverse events (irAEs).Citation7 Cutaneous irAEs (irCAEs) are one of the most commonly reported immune side effects in patients treated with ICIs, either anti-CTLA-4 (ipilimumab, 43%–45% of the patients) or anti-PD-1 (nivolumab and pembrolizumab in 34%) usually develop early in the course of treatment, within the first 2–8 weeks after initiation.Citation8,Citation9 Overall irCAEs represent more than 30% of all AEs, especially skin rash, pruritus, and vitiligo,Citation9 more likely occuring during combined therapy (anti-CTLA-4 and anti-PD-1) rather than monotherapy.Citation10 The Common Terminology Criteria for Adverse Events (CTCAE) classification is commonly used to evaluate the severity of the irCAE. This scale considers physical manifestations and their psychosocial impact for the patient, ranging from 1 to 5, where grade-1 indicate a mild reaction and grade-5 the patient’s death due to irCAE. The onset of irAEs may strongly impair patient’s quality of life, worsening functional scores and increasing symptoms burden, like fatigue, aching joints and aching muscles.Citation11,Citation12 The generic term “skin rash” has been widely used so far to define very different skin reactions without further distinctions of single dermatologic entities; clinical and histopathological findings were partly reported in different case reports or expert opinions.Citation8,Citation13 Chronic lichenoid interface dermatitis and spongiotic dermatitis represent the most common histological reported diagnoses in case of irCAEs in melanoma patients according to the literature, reflecting acute or chronic inflammation associated with variable epidermal changes.Citation14,Citation15 We report the main distinct dermatologic entities indicated so far as “skin rash”, describing clinical and histologic features, with a proposal therapeutic management.
Type of skin manifestations
Maculopupular rash (MPR)
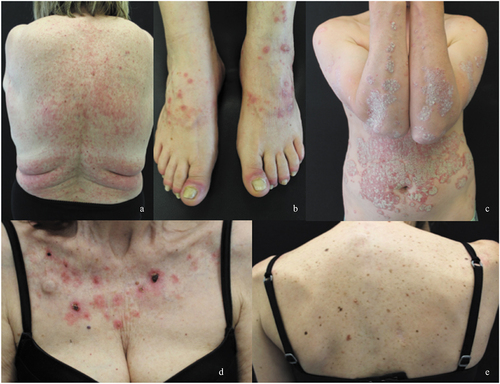
Represents the most prevalent irCAE observed during ICIs therapy, with a higher incidence during anti-CTLA-4 therapy (49–68%) compared to anti-PD-1 monotherapy (20%).Citation8,Citation16,Citation17 MPR is clinically characterized by multiple pruritic erythematous macules and papules () coalescing in plaques, slightly palpable, which typically develop on trunk and extensor surfaces of the extremities, sparing the face.Citation8,Citation18,Citation19 Cutaneous lesions are usually associated with pruritus but can be asymptomatic and sometimes show an exacerbation after each cycle of treatment.Citation14 The onset of MPR usually occurs early in the course of treatment, within 3–6 weeks after the initial dose, often appearing as the first cutaneous adverse reaction;Citation8 however, the timing of this skin eruption can vary between 3 weeks and 2 years after ICI starting date.Citation8 They are often self-limited and easily managed with topical corticosteroid.Citation18 The clinical presentation of MPR may resemble exanthema-like drug eruptions due to antibiotics, therefore an accurate differential diagnosis is fundamental.Citation14 Moreover, it is important to consider that pruritic maculo-papular eruption can be rarely the initial manifestation of blistering disease, including bullous pemphigoid (BP), DRESS syndrome or the more severe skin reactions Steven-Johnson syndrome and toxic epidermal necrolysis. Patients developing erythroderma, mucositis, blisters, localized tenderness or swelling and skin pain in a clinical scenario of fever, lymphadenopathy and eosinophilia, should be carefully assessed, monitored and promptly treated.Citation20 Reports on the development of BP during ICI have been rapidly increasing, leading to a better recognition and management of this entity.Citation21,Citation22
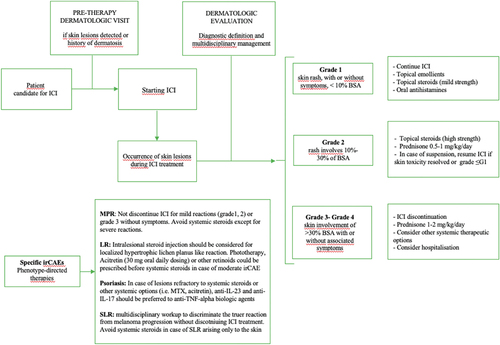
The therapeutic strategies of MPR () are based on the severity of the ICIs-related: grade 1–2 MPR are managed with mid- or high-potency topical corticosteroids without interrupting or changing immunotherapy dose. Severe skin manifestations (grade 3–4) generally require systemic corticosteroids (prednisone 0.5–1 mg/kg/day) and ICIs should be withheld until rash is grade 1 or less.Citation7,Citation23 Recurrences of MPR with high-grade toxicity require ICI treatment discontinuation.Citation24
Lichenoid eruption (LE)
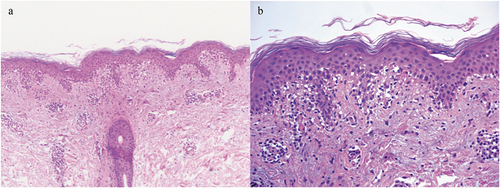
Is a common irCAE which develops in 24% to 30% of cases developing during ICIs treatment, particularly PD‐1 inhibitors,Citation25,Citation26 with a mean latency from the initial dose of 4–18 months.Citation27,Citation28 Lichenoid lesions exhibit clinical aspects of violaceous scaly papules, pruritic hypertrophic plaques or patches, with a focal or generalized distribution. Less frequently, skin lesions are distributed exclusively to palmoplantar areas or mucosal foldsCitation8,Citation14,Citation18 and therefore physical examination of oral and genital mucosae is recommended. Histologic findings of skin biopsies demonstrate a typical band-like lymphocytic infiltrate at the papillary dermis with large number of eosinophils and epidermal orthokeratosisCitation29 (). In lichen planus-like reaction due to immunotherapies, the histologic detection of parakeratosis, spongiosis, and eosinophils could facilitate the distinction of this lichenoid drug reaction from a typical lichen planus, but the disclosure of these features is not mandatory for a definite diagnosis.Citation15 LE is usually treated with topical or systemic corticosteroids, with an initial dose varying depending on the grade and clinical severity and is tapered slowly over 4–6 weeks if clinical improvement is shown.Citation8,Citation23 Palmoplantar manifestations can be treated with phototherapy, either UVA or narrow-band UVB.Citation15,Citation18 It is well-known that high doses of systemic steroids, as well as other immune-modulating agents should not be concomitantly used with immunotherapy, because of the risk of downgrading the therapeutic potency of ICIs. Given that LE is not life-threatening even when >30% BSA, preferably oral retinoids (adopted from classic lichen planus treatment recommendations) ± phototherapy could be prescribed before high doses of systemic steroids trying to avoid the delaying or discontinuation of ICIs (). In cases of severe corticosteroid‐refractory cutaneous lesions, other systemic options such as cyclosporine (2–3 mg/kg/die), methotrexate (single 15 mg dose) and anti-TNF alpha should be considered as third-line therapies withholding or discontinuing immunotherapy until toxicity grade ≤1 or ≤2 without symptomsCitation30,Citation31 ().
Psoriasiform rash
Has been frequently reported as a cutaneous adverse event during ICIs, appearing 5 to 10 weeks after the first dose of the drug. Patients with a previous history of psoriasis may show an even earlier onset.Citation28,Citation32 Although psoriasiform rash may clinically appear in different clinical subtypes, plaque psoriasis with well-defined erythematous and scaly papules/plaques over extensor extremities is the most frequent; plaques located on the mammary folds and inguinal regions (so called inverse psoriasis), seborrheic plaques on the face and scalp, palmoplantar keratotic plaques, pustular psoriasis and generalized guttate papules have been also observed during ICIs treatment. Nails can be involved with clinically typical psoriatic signs as thickening, crumbling, pitting.Citation32 Psoriatic arthritis can also occur, even in setting of a negative personal or family history of psoriasis.Citation33,Citation34 In case of unsatisfactory response to topical corticosteroids, traditional systemic therapies such as acitretin, apremilast and methotrexate are recommended prior to systemic corticosteroids, in order not to impair the efficacy of ICIs drugs (), as well as flares of psoriasis upon steroids discontinuation. In addition, biological drugs (anti-TNF alpha, anti-IL-17 and anti-IL-23) can be effective in case of psoriatic lesions refractory to traditional systemic treatments.Citation32,Citation34
IL-17 inhibitors may act as an additional immunotherapy risk factor for the development of colitis as severe irAE, thus IL-17 inhibitors tend not to be recommended as first-line biologic treatment in case of severe psoriatic reaction. Opposed to that, anti-TNF alpha agents are prescribed in combination with ICI in patients at risk for colitis.Citation35 In a more holistic management approach anti-IL-17 drugs should be at least recommended after failure or IL-23. Indeed, based on the literature and our experience, the use of drugs targeting IL-23 and IL-17 in refractory psoriatic patients should be preferred to anti-TNF-alpha agents regarding efficacy.Citation34
Xerotic/eczematous rash
Is a frequent cutaneous adverse event with both anti-CTLA-4 and anti-PD-1 agents, generally exhibiting a widespread distribution on the trunk and extremities.Citation28 Eczematous reactions consist of multiple erythematous scaly papules, sometimes arising in the form of localized patches/plaques, nummular plaques or dyshidrotic vesicles. This type of skin reaction is often misdiagnosed and clinically included in maculopapular or lichenoid rashes, thus, a diagnostic biopsy is needed in any case of clinical doubt. In a cohort of patients affected by different advanced malignancies, namely lung cancer, renal cell carcinoma, urothelial carcinoma and melanoma, the onset of rash/eczema was observed in 60.7% cases of advanced melanoma, with anti-CTLA-4 (ipilimumab) administration being associated to a higher incidence cutaneous toxicity, while anti-PD-1 (nivolumab) blocker was mainly involved in the development of more severe cases.Citation36 Hwang et al. evaluated 82 patients treated with anti-PD-1 therapy for metastatic melanoma proving a significant tendency of eczema to appear in combination with vitiligo.Citation37 Histologic examination of skin affected areas revealed epidermal spongiosis, and perivascular CD4 + T-cell infiltrates with eosinophils. Treatment of eczematous rash includes the use of topical corticosteroids or a combination of antihistamines and topical steroid.Citation36
Neutrophilic dermatoses
Encompass a heterogeneous group of diseases unified by the predominance of neutrophils as assessed by histologic examination, and a close association to both hematological and solid malignancies, as well as infections, autoimmune disorders, or as adverse event of drugs or idiopathic entity.Citation38 To the best of our knowledge, the onset of a neutrophilic dermatosis as a cutaneous adverse event of ICIs in the treatment of advanced melanoma is relatively rare and based upon single case-reports.Citation39 Sweet’s syndrome is an irCAE belonging to this group. Five patients presenting this cutaneous condition after treatment with ipilimumab for metastatic melanoma have been described,Citation40–44 estimating a median and average interval to clinical onset of 8.9 weeks (SD = 3.38 within a range of 6–12 weeks), after the first administration. Complete regression was achieved with oral/intravenous administration of corticosteroids in all cases (one patient received dapsone simultaneously), without recurrence of symptoms. Three of 5 cases reported a permanent discontinuation of the involved checkpoint inhibitor, while one case described a class switch to anti-PD-1 (nivolumab) following complete resolution of Sweet’s syndrome.
A single case of pyoderma gangrenosum (PG) was seen in one patient with metastatic melanoma treated with ipilimumab 16 weeks after the first dose of the drug.Citation45 PG had a peristomal localization well treated with a simultaneous administration of steroids and immunosuppressant (infliximab), after relapsing with only high-dose systemic steroids (200 mg twice daily).Citation45 The definitive interruption rather than a resumption of the causative drug was unreported.
The development of pustular eruptions during ICIs treatment in melanoma patients, was rarely reported including two cases of acute generalized exanthematous pustulosis (AGEP), one case of acute localized exanthematous pustulosis (ALEP) and one cases of intracorneal pustular drug eruption.Citation46–49 Median interval time of development of cutaneous lesions was 9 weeks (SD = 3.4 with a range of 3.3–12 weeks), that is later than other more frequent irCAEs. Both cases of AGEP were treated with either topical or systemic corticosteroids, choosing a loading dose of 50 mg/day oral prednisone or 120 mg/day oral prednisolone followed by a slowly tapering down, thus achieving a complete clinical resolution. In the case of ALEP, an improvement was observed using intermittent topical steroid (betamethasone dipropionate) after 4 months follow-up evaluation, instead of a more rapid resolution reported in the management of intracorneal pustular drug eruption, through a 4 week course of topical therapy (betamethasone valerate). Of note, cases of ALEP and intracorneal pustular drug eruption were treated without discontinuing checkpoint inhibitors and in one of two mentioned reports of AGEP a pharmaceutical switch from anti-CTLA-4 to anti-PD-1 (pembrolizumab) drug, after completing steroid tapering, was chosen.
Other less common reported immune-related skin rash
- Urticarial lesions have been mainly observed as prodromic signs of bullous eruptions.Citation13,Citation18 Moreover, two reports described the development of a localized angioedema secondary to ICI. In details, one case was related to nivolumab and occurred after 20 weeks of therapy,Citation50 while the other case was associated with ipilimumab occurring relatively early during treatment, i.e. after the third infusion of the drug (9 weeks). A short course of systemic corticosteroids and the discontinuation of the involved drug allowed a gradual and complete resolution of the clinical signs.Citation50,Citation51
- Grover’s disease has occasionally been reported during ICIs treatment.Citation52–54 Skin lesions consist of small pruritic, polymorphic, papulovesicular lesions distributed on the trunk, back, and extremities. Histologic examinations show acantholysis with or without dyskeratosis. Cutaneous rash is usually self-limited and should be managed symptomatically. The treatment is based on topical corticosteroids, phototherapy and topical or oral retinoids for more severe or refractory cases.Citation55
- Dermatomyositis is an autoimmune disease characterized by distinctive skin eruption and symmetrical proximal muscle weakness. Immunotherapy-induced dermatomyositis is a rarely reported adverse event.Citation56,Citation57 Given its frequent paraneoplastic nature, an accurate differential diagnosis including medical history of the patient, history of the disease, multisystem involvement, detection of specific antibodies, suggestive imaging, muscle/skin biopsy, neoplastic cells activity, is fundamental in terms of prognosis and choice of therapeutic approaches. Dermatomyositis eruption is typically characterized by reddish-purple skin rash on or around the eyelids (heliotrope rash), painful erythematous papules on bilateral dorsal hands and plaques symmetrically distributed on sun-exposed areas, associated to muscle weakness in the proximal upper and/or lower extremities.Citation57 Treatment with intravenous immunoglobulins was preferred to systemic corticosteroids in most reported cases, also representing a steroid-sparing therapy option. Notably, patients who have been restarted on ICI therapy after developing dermatomyositis experienced a recurrence of the skin lesions, however this kind of irCAE has been correlated to a better oncologic treatment outcome.Citation58
- Sarcoidosis like reaction (SLR) has been described during treatment with ICIs in melanoma patients.Citation59,Citation60 SLR usually affects the lungs, lymph nodes and skin, and typically improves after discontinuation of ICI.Citation61 Cutaneous lesions are characterized by erythematous/violaceus nodules and plaques, asymptomatic or sometimes painful, usually located on the extremities. SLR may also arise as erythema nodosum-like or lupus-like lesions. Histology typically shows ill-defined non-caseating granulomas involving the entire dermis, nevertheless lesional skin can show nonspecific histologic signs associated with systemic sarcoidosis. The cutaneous lesions as well as lymph nodes can mimic cancer metastasis at the functional nuclear medicine imaging (PET scan), thus a correct diagnosis is required.Citation60 The majority of SLR cases was successfully treated with topical high-potent corticosteroids, but a significant proportion of patients discontinued ICIs because of the lungs or lymph nodes involvement.Citation62
Conclusion
A variety of irCAE may occur during immunotherapy treatment. Cutaneous lesions greatly differ in terms of clinical aspects and degree of severity. A dermatologic evaluation would be recommended before the first dose of immunotherapy, due to the possibility of drug making a preexisting dermatosis worse (). The most frequent skin manifestations during immunotherapy include maculopapular rash, psoriasiform and lichenoid eruptions; they are usually mild and can be successfully treated with topical corticosteroids, but more diffuse and/or exfoliative patterns may need cancer treatment interruption. Indeed, the pruritic maculopapular and xerotic skin rashes can also represent the initial manifestation of a more characteristic skin disorder induced by ICIs, including psoriasis, bullous pemphigoid or neutrophilic dermatoses. In very few cases, initial skin rash can quickly worsen into a severe cutaneous drug reaction (Steven Johnson Syndrome, Toxic Epidermal Necrolysis).Citation23 As opposed to other organs’ irAEs (e.g. hypophysitis, pneumonia) and with the exception of Toxic Epidermal Necrolysis and bullous disorders for the skin, life-threatening cutaneous toxicity is extremely unlikely even in the scenario of skin rashes >30% BSA; thus, depending on the patient’s profile (symptomatic or not) and the skin lesions phenotype, maintenance of immunotherapy is potentially feasible.Citation63 The term “skin rash” frequently used in clinical trials is too vague and unprecise and should be avoided; it is probably due to the fact that patients are not routinely seen and systematically subjected to a full-body skin examination by a dermatologist. Given the frequency of the irCAEs and their impact on treatment course, dermatologists have an important role in evaluating and managing skin toxicities, including skin biopsies for any atypical, severe, persistent, recurrent or poorly tolerated rash. In addition, each skin disease may require specific treatment recommendations. In literature there are multiple guidelines based on the severity of the skin rash,Citation24 but not so many based on different types of reactions. Few authors highlighted the possible different management of the cutaneous skin rashes occurring during ICI treatment.Citation8,Citation13,Citation18 Hereby we reviewed the different irCAEs which have been indicated as “skin rash” and emphasized the importance of a complete and accurate dermatological diagnosis of each irCAE particularly for treatment decision, including the possibility to not discontinue the ICI in case of maculopapular mild or moderate reaction, the use of local field directed therapies for most types of skin reactions, the predilection of specific systemic agents instead of systemic corticosteroids. Thus, increased awareness and early recognition of the specific skin reaction could lead to a prompt and effective management reducing the need for unnecessary immunotherapy interruption improving oncological outcome.
Declaration of Interest
The authors declare that the scientific paper was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest
Acknowledgments
We sincerely thank the patients and their families for granting permission to publish this information. Alessandro Di Stefani, MD (Istituto di Dermatologia, Università Cattolica del Sacro Cuore, Rome, Italy), for his contribution to examine histopathology. These contributors were not financially supported.
References
- Hegde PS, Chen DS. Top 10 challenges in cancer immunotherapy. Immunity. 2020 Jan;14;52(1):17–8. doi:https://doi.org/10.1016/j.immuni.2019.12.011.
- Toor SM, Sasidharan Nair V, Decock J, Elkord E. Immune checkpoints in the tumor microenvironment. Semin Cancer Biol. 2020 Oct;65:1–12. doi:https://doi.org/10.1016/j.semcancer.2019.06.021.
- Yokota T, Homma A, Kiyota N, Tahara M, Hanai N, Asakage T, Matsuura K, Ogawa T, Saito Y, Sano D, et al. Immunotherapy for squamous cell carcinoma of the head and neck. Jpn J Clin Oncol. 2020 Sep 28;50(10):1089–96. doi:https://doi.org/10.1093/jjco/hyaa139.
- Ruiz-Cordero R, Devine WP. Targeted Therapy and Checkpoint immunotherapy in lung cancer. Surg Pathol Clin. 2020;13(1):17–33. doi:https://doi.org/10.1016/j.path.2019.11.002.
- Zayac A, Almhanna K. Esophageal, gastric cancer and immunotherapy: small steps in the right direction? Transl Gastroenterol Hepatol. 2020;5:9. doi:https://doi.org/10.21037/tgh.2019.09.05.
- Livingstone A, Agarwal A, Stockler MR, Menzies AM, Howard K, Morton RL. Preferences for immunotherapy in melanoma: a systematic review. Ann Surg Oncol. 2020 Feb;27(2):571–84. doi:https://doi.org/10.1245/s10434-019-07963-y.
- Magee DE, Hird AE, Klaassen Z, Sridhar SS, Nam RK, Wallis CJD, Kulkarni GS. Adverse event profile for immunotherapy agents compared with chemotherapy in solid organ tumors: a systematic review and meta-analysis of randomized clinical trials. Ann Oncol. 2020 Jan;31(1):50–60. doi:https://doi.org/10.1016/j.annonc.2019.10.008 .
- Geisler AN, Phillips GS, Barrios DM, Wu J, Leung DYM, Moy AP, Kern JA, Lacouture ME. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020 Nov;83(5):1255–1268. doi:https://doi.org/10.1016/j.jaad.2020.03.132 .
- Almutairi AR, McBride A, Slack M, Erstad BL, Abraham I. Potential immune-related adverse events associated with monotherapy and combination therapy of ipilimumab, nivolumab, and pembrolizumab for advanced melanoma: a systematic review and meta-analysis. Front Oncol. 2020 Feb 11;10:91. doi:https://doi.org/10.3389/fonc.2020.00091.
- Hodi FS, Chiarion-Sileni V, Gonzalez R, Grob -J-J, Rutkowski P, Cowey CL, Lao CD, Schadendorf D, Wagstaff J, Dummer R, et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018;19(11):1480–92. doi:https://doi.org/10.1016/S1470-2045(18)30700-9.
- Boekhout AH, Rogiers A, Jozwiak K, Boers-Sonderen MJ, van den Eertwegh AJ, Hospers GA, de Groot JWB, Aarts MJB, Kapiteijn E, Ten Tije AJ et al. Health-related quality of life of long-term advanced melanoma survivors treated with anti-CTLA-4 immune checkpoint inhibition compared to matched controls. Acta Oncol. 2021 Jan;60(1):69–77. doi:https://doi.org/10.1080/0284186X.2020.1818823.s Epub 2020 Sep 14
- Mamoor M, Postow MA, Lavery AJ, Baxi SS, Khan N, Mao JJ, Rogak LJ, Sidlow R, Thom B, Wolchok JA, et al. Quality of life in long-term survivors of advanced melanoma treated with checkpoint inhibitors. J Immunother Cancer. 2020;8(1):e000260. doi:https://doi.org/10.1136/jitc-2019-000260.
- Belum VR, Benhuri B, Postow MA, Hellmann MD, Lesokhin AM, Segal NH, Motzer RJ, Wu S, Busam KJ, Wolchok JD, et al. Characterisation and management of dermatologic adverse events to agents targeting the PD-1 receptor. Eur J Cancer. 2016;60:12–25. doi:https://doi.org/10.1016/j.ejca.2016.02.010.
- Shi VJ, Rodic N, Gettinger S, Leventhal JS, Neckman JP, Girardi M, Bosenberg M, Choi JN. Clinical and histologic features of lichenoid mucocutaneous eruptions due to anti-programmed cell death 1 and anti-programmed cell death ligand 1 immunotherapy. JAMA Dermatol. 2016;152(10):1128–36. doi:https://doi.org/10.1001/jamadermatol.2016.2226.
- Tetzlaff MT, Nagarajan P, Chon S, Huen A, Diab A, Omar P, Aung PP, Torres-Cabala CA, Mays SR, Prieto VG, et al. Lichenoid dermatologic toxicity from immune checkpoint blockade therapy. A detailed examination of the clinicopathologic features. Am J Dermatopathol. 2017;39(2):121–29. doi:https://doi.org/10.1097/DAD.0000000000000688.
- Curry JL, Tetzlaff MT, Nagarajan P, Drucker C, Diab A, Hymes SR, Duvic M, Hwu W-J, Wargo JA, Torres-Cabala CA, et al. Diverse types of dermatologic toxicities from immune checkpoint blockade therapy. J Cutan Pathol. 2017;44(2):158–76. doi:https://doi.org/10.1111/cup.12858.
- Postow MA. Managing immune checkpoint-blocking anti- body side effects. Am Soc Clin Oncol Educ Book. 2015;35:76–83. doi:https://doi.org/10.14694/EdBook_AM.2015.35.76.
- Sibaud V. Dermatologic reactions to immune checkpoint inhibitors: skin toxicities and immunotherapy. Am J Clin Dermatol. 2018;19(3):345–61. doi:https://doi.org/10.1007/s40257-017-0336-3.
- Shoushtari AN, Friedman CF, Navid-Azarbaijani P, Postow MA, Callahan MK, Momtaz P, Panageas KS, Wolchok JD, Chapman PB. Measuring toxic effects and time to treatment failure for nivolumab plus ipilimumab in melanoma. JAMA Oncol. 2018;4(1):98–101. doi:https://doi.org/10.1001/jamaoncol.2017.2391.
- Apalla Z, Lallas A, Delli F, Lazaridou E, Papalampou S, Apostolidou S, Gerochristou M, Rigopoulos D, Stratigos A, Nikolaou V, et al. Management of immune checkpoint inhibitor-induced bullous pemphigoid. J Am Acad Dermatol. 2021 Feb;84(2):540–43. doi:https://doi.org/10.1016/j.jaad.2020.05.045.
- Lopez AT, Geskin LA. Case of nivolumab-induced bullous pemphigoid: review of dermatologic toxicity associated with programmed cell death protein-1/programmed death ligand-1 inhibitors and recommendations for diagnosis and management. Oncologist. 2018 Oct;23(10):1119–26. doi:https://doi.org/10.1634/theoncologist.2018-0128.
- Nelson CA, Singer S, Chen T, Puleo AE, Lian CG, Wei EX, Giobbie-Hurder A, Mostaghimi A, LeBoeuf NR. Bullous pemphigoid after anti-PD-1 therapy: a retrospective case-control study evaluating impact on tumor response and survival outcomes. J Am Acad Dermatol. 2020 Jan;10:S0190-9622(20)30048–7. doi:https://doi.org/10.1016/j.jaad.2019.12.068 .
- Sibaud V, Meyer N, Lamant L, Vigarios E, Mazieres J, Delord JP. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28(4):254–63. doi:https://doi.org/10.1097/CCO.0000000000000290.
- Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, Jordan K. ESMO guidelines committee. Management of toxicities from immunotherapy: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv119–iv142. doi:https://doi.org/10.1093/annonc/mdx225.
- Robert C, Ribas A, Schachter J, Arance A, Grob -J-J, Mortier L, Daud A, Carlino MS, McNeil CM, Lotem M, et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 2019;20(9):1239–51. doi:https://doi.org/10.1016/S1470-2045(19)30388-2.
- Ascierto PA, Del Vecchio M, Mandalà M, Gogas H, Arance AM, Dalle S, Cowey CL, Schenker M, Grob -J-J, Chiarion-Sileni V, et al. Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. 2020 Nov;21(11):1465–77. doi:https://doi.org/10.1016/S1470-2045(20)30494-0.
- Wang LL, Patel G, Chiesa-Fuxench ZC, McGettigan S, Schuchter L, Mitchell TC, Ming ME, Chu EY. Timing of onset of adverse cutaneous reactions associated with programmed cell death protein 1 inhibitor therapy. JAMA Dermatol. 2018;154(9):1057–61. doi:https://doi.org/10.1001/jamadermatol.2018.1912.
- Coleman E, Ko C, Dai F, Tomayko MM, Kluger H, Leventhal JS. Inflammatory eruptions associated with immune checkpoint inhibitor therapy: a single-institution retrospective analysis with stratification of reactions by toxicity and implications for management. J Am Acad Dermatol. 2019;80(4):990–97. doi:https://doi.org/10.1016/j.jaad.2018.10.062.
- Min Lee CK, Li S, Tran DC, Zhu GA, Kim J, Kwong BY, Chang ALS. Characterization of dermatitis after PD-1/PD-L1 inhibitor therapy and association with multiple oncologic outcomes: a retrospective case-control study. J Am Acad Dermatol. 2018;79(6):1047–52. doi:https://doi.org/10.1016/j.jaad.2018.05.035.
- Fixsen E, Patel J, Selim MA, Kheterpal M. Resolution of pembrolizumab-associated steroid-refractory lichenoid dermatitis with cyclosporine. Oncologist. 2019;24(3):e103–e105. doi:https://doi.org/10.1634/theoncologist.2018-0531.
- Varma A, Friedlander P, De Moll EH, Desman G, Levitt J. Resolution of pembrolizumab-associated lichenoid dermatitis with a single dose of methotrexate. Dermatol Online J. 2020;26:13030/qt6348767t
- Nikolaou V, Sibaud V, Fattore D, Sollena P, Ortiz-Brugués A, Giacchero D, Romano MC, Riganti J, Lallas K, Peris K et al. Immune checkpoint-mediated psoriasis: a multicentric European study of 115 patients from European network for cutaneous ADverse event to oncologic drugs (ENCADO) group. J Am Acad Dermatol. 2020 Dec;3:S0190-9622(20)33115–7. Epub ahead of print. PMID: 33279646. doi:https://doi.org/10.1016/j.jaad.2020.08.137 .
- Menzies AM, Johnson DB, Ramanujam S, Atkinson VG, Wong ANM, Park JJ, McQuade JL, Shoushtari AN, Tsai KK, Eroglu Z, et al. Anti-PD-1 therapy in patients with advanced melanoma and preexisting autoimmune disorders or major toxicity with ipilimumab. Ann Oncol. 2017;28(2):368–76. doi:https://doi.org/10.1093/annonc/mdw443.
- Tanaka R, Okiyama N, Okune M, Ishitsuka Y, Watanabe R, Furuta J, Ohtsuka M, Otsuka A, Maruyama H, Fujisawa Y, et al. Serum level of interleukin-6 is increased in nivolumab-associated psoriasiform dermatitis and tumor necrosis factor-a is a biomarker of nivolumab recativity. J Dermatol Sci. 2017;86(1):71–73. doi:https://doi.org/10.1016/j.jdermsci.2016.12.019.
- Esfahani K, Elkrief A, Calabrese C, Lapointe R, Hudson M, Routy B, Miller Jr WH, Calabrese L. Moving towards personalized treatments of immune-related adverse events. Nat Rev Clin Oncol. 2020 Aug;17(8):504–15. doi:https://doi.org/10.1038/s41571-020-0352-8
- Rovers JFJ, Bovenschen HJ. Dermatological side effects rarely interfere with the continuation of checkpoint inhibitor immunotherapy for cancer. Int J Dermatol. 2020;59(12):1485–90. doi:https://doi.org/10.1111/ijd.15163.
- Hwang S, Carlos G, Wakade D, Byth K, Kong BY, Chou S, Carlino MS, Kefford R, Fernandez-Penas P. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single institution cohort. J Am Acad Dermatol. 2016;74(3):455–61. doi:https://doi.org/10.1016/j.jaad.2015.10.029.
- Alavi A, Sajic D, Cerci FB, Ghazarian D, Rosenbach M, Jorizzo J. Neutrophilic dermatoses: an update. Am J Clin Dermatol. 2014 Oct;15(5):413–23. doi:https://doi.org/10.1007/s40257-014-0092-6.
- Ravi V, Maloney NJ, Worswick S. Neutrophilic dermatoses as adverse effects of checkpoint inhibitors: a review. Dermatol Ther. 2019 Sep;32(5):e13074. doi:https://doi.org/10.1111/dth.13074.
- Pintova S, Sidhu H, Friedlander PA, Holcombe RF. Sweet’s syndrome in a patient with metastatic melanoma after ipilimumab therapy. Melanoma Res. 2013;23(6):498–501. doi:https://doi.org/10.1097/CMR.0000000000000017.
- Adler NR, Murray WK, Brady B, McCormack C, Pan Y. Sweet syndrome associated with ipilimumab in a patient with metastatic melanoma. Clin Exp Dermatol. 2018;43(4):497–99. doi:https://doi.org/10.1111/ced.13399.
- Kyllo RL, Parker MK, Rosman I, Musiek AC. Ipilimumab-associated Sweet syndrome in a patient with high-risk melanoma. J Am Acad Dermatol. 2014;70(4):e85–e86. doi:https://doi.org/10.1016/j.jaad.2013.11.022.
- Gormley R, Wanat K, Elenitsas R, Giles J, McGettigan S, Schuchter L, Takeshita J. Ipilimumab-associated Sweet syndrome in a melanoma patient. J Am Acad Dermatol. 2014;71(5):e211–3. doi:https://doi.org/10.1016/j.jaad.2014.06.042.
- Yaşar HA, Akkus E, Heper A, Akay BN, Urun Y, Utkan G. Sweet’s syndrome under ipilimumab therapy and a brief comparison of the cases in literature. J Oncol Pharm Pract. 2020;26(7):1762–64. doi:https://doi.org/10.1177/1078155220906885.
- Rudolph BM, Staib F, Von Stebut E, Hainz M, Grabbe S, Loquai C. Neutrophilic disease of the skin and intestines after ipilimumab treatment for malignant melanoma - simultaneous occurrence of pyoderma gangrenosum and colitis. Eur J Dermatol. 2014;24(2):268–69. doi:https://doi.org/10.1684/ejd.2014.2297.
- Hwang SJE, Carlos G, Wakade D, Sharma R, Fernandez-Penas P. Ipilimumab-induced acute generalized exanthematous pustulosis in a patient with metastatic melanoma. Melanoma Res. 2016;26(4):417–20. doi:https://doi.org/10.1097/CMR.0000000000000261.
- Page B, Borradori L, Beltraminelli H, Yawalkar N, Hunger RE. Acute generalized exanthematous pustulosis associated with ipilimumab and nivolumab. J Eur Acad Dermatol Venereol. 2018;32(7):e256–e257. doi:https://doi.org/10.1111/jdv.14282.
- Zhao CY, Consuegra G, Chou S, Fernández-Peñas P. Intracorneal pustular drug eruption, a novel cutaneous adverse event in anti-programmed cell death-1 patients that highlights the effect of anti-programmed cell death-1 in neutrophils. Melanoma Res. 2017;27(6):641–44. doi:https://doi.org/10.1097/CMR.0000000000000397.
- Wang CY, Khoo C, McCormack CJ, Xu W, Pan Y. Acute localized exanthematous pustulosis secondary to pembrolizumab. Australas J Dermatol. 2017;58(4):322–23. doi:https://doi.org/10.1111/ajd.12594.
- Ratra A, Dasanu CA. Angioedema late in the course of adjuvant nivolumab therapy for melanoma. J Oncol Pharm Pract. 2020;26(4):1019–21. doi:https://doi.org/10.1177/1078155219881181.
- Kreckè N, Zimmer A, Friesenhahn-Ochs B, Müller CSL, Vogt T, Pföhler C. Sneaky side effects and ineffectiveness of an immunotherapy with ipilimumab in a case of metastatic melanoma. Dermatoendocrinol. 2016;8(1):e1199307. doi:https://doi.org/10.1080/19381980.2016.1199307.
- Munoz J, Guillot B, Girard C, Dereure O, Du-Thanh A. First report of ipilimumab-induced Grover disease. Br J Dermatol. 2014;171:1236–37
- Koelzer VH, Buser T, Willi N, Rothschild SI, Wicki A, Schiller P, Cathomas G, Zippelius A, Mertz KD. Grover’s-like drug eruption in a patient with metastatic melanoma under ipilimumab therapy. J Immunother Cancer. 2016;4(1):47. doi:https://doi.org/10.1186/s40425-016-0151-z.
- Uemura M, Faisal F, Haymaker C, McQuail N, Sirmans E, Hudgens CW, Barbara L, Bernatchez C, Curry JL, Hwu P, et al. A case report of Grover’s disease from immunotherapy-a skin toxicity induced by inhibition of CTLA-4 but not PD-1. J Immunother Cancer. 2016;4(1):55. doi:https://doi.org/10.1186/s40425-016-0157-6.
- Muntyanu A, Netchiporouk E, Gerstein W, Gniadecki R, Litvinov IV. Cutaneous immune-related adverse events (irAEs) to immune checkpoint inhibitors: a dermatology perspective on management. J Cutan Med Surg. 2020 Aug;3:1203475420943260. Epub ahead of print. PMID: 32746624. doi:https://doi.org/10.1177/1203475420943260.
- Kosche C, Stout M, Sosman J, Lukas RV, Choi JN. Dermatomyositis in a patient undergoing nivolumab therapy for metastatic melanoma: a case report and review of the literature. Melanoma Res. 2020 Jun;30(3):313–16. PMID: 31567590. doi:https://doi.org/10.1097/CMR.0000000000000642.
- Messer A, Drozd B, Glitza IC, Lu H, Patel AB. Dermatomyositis associated with nivolumab therapy for melanoma: a case report and review of the literature. Dermatol Online J. 2020 Aug;15;26(8):13030/qt4c21b068. PMID: 32941716.
- Martini DJ, Hamieh L, McKay RR, Harshman LC, Brandao R, Norton CK, Steinharter JA, Krajewski KM, Gao X, Schutz FA, et al. Durable clinical benefit in metastatic renal cell carcinoma patients who discontinue PD-1/PD-L1 therapy for immune-related adverse events (irAEs). Cancer Immunol Res. 2018;6(4):402–08. doi:https://doi.org/10.1158/2326-6066.CIR-17-0220.
- Apalla Z, Kemanetzi C, Papageorgiou C, Bobos M, Manoli M, Fotiadou C, Hatzibougias D, Boukovinas I, Stergiou E, Levva S et al. Challenges in sarcoidosis and sarcoid-like reactions associated to immune checkpoint inhibitors: a narrative review apropos of a case. Dermatol Ther. 2020 Dec 2:e14618. Epub ahead of print. PMID: 33263945. doi:https://doi.org/10.1111/dth.14618 .
- Chorti E, Kanaki T, Zimmer L, Hadaschik E, Ugurel S, Gratsias E, Roesch A, Bonella F, Wessendorf TE, Wälscher J et al. Drug-induced sarcoidosis-like reaction in adjuvant immunotherapy: increased rate and mimicker of metastasis. Eur J Cancer. 2020 May;131:18–26. Epub 2020 Apr;2. PMID: 32248071. doi:https://doi.org/10.1016/j.ejca.2020.02.024 .
- Chopra A, Nautiyal A, Kalkanis A, Judson MA. Drug-induced sarcoidosis-like reactions. Chest. 2018;154:664e77.
- Tetzlaff MT, Nelson KC, Diab A, Staerkel GA, Nagarajan P, Torres-Cabala CA, Chasen BA, Wargo JA, Prieto VG, Amaria RN et al. Granulomatous/sarcoid-like lesions associated with checkpoint inhibitors: a marker of therapy response in a subset of melanoma patients. J Immunother Cancer. 2018 Feb;12;6(1):14. PMID: 29433571; PMCID: PMC5810034. doi:https://doi.org/10.1186/s40425-018-0323-0
- Apalla Z, Sibaud V. Immunotherapy-mediated dermatological adverse events: the urgent need for a common, clinically meaningful, management strategy. Support Care Cancer. 2020 Dec;28(12):5597–99. doi:https://doi.org/10.1007/s00520-020-05701-9.
Appendix
Figure A1. Cutaneous immune-related adverse events usually reported as “skin rash”: A maculopapular rash; B lichenoid reaction; C psoriatic rash; D neutrophilic rash; E Grover’s disease.