ABSTRACT
There is a paucity of evidence linking pneumococcal infection and influenza with SARS-CoV-2 and COVID-19. There is circumstantial evidence of the possibility of an association between S. pneumoniae and SARS-CoV-2 such as the increased binding of S. pneumoniae to coronavirus-infected human airway epithelium, the frequent use of broad-spectrum antibiotics in the management of COVID-19 which could mask secondary bacterial infection, and the observation that pneumococcal vaccination is associated with decreased SARS-CoV-2 nasopharyngeal swab positivity. We performed a targeted literature review for the year 2020, using search terms S. pneumoniae, influenza, SARS-CoV-2, and found 25 relevant articles of a total of 291. Pneumococcal and influenza vaccinations have the potential to contribute toward efforts aimed at reducing the health burden of SARS-CoV-2, especially by reducing preventable admissions to hospital for pneumonia and the consequent risk of nosocomial SARS-CoV-2 transmission.
Background and rationale
Pneumococcal infections historically have played a role in pandemics, namely influenza pandemics.Citation1 However, the evidence for the role of pneumococcal infections in SARS/MERS is equivocal.Citation2 There is laboratory experimental evidence that non-SARS coronaviruses can facilitate the binding of S. pneumoniae onto respiratory epithelium ().Citation3 However, associations between pneumococcal infection and SARS-CoV-2 infections are thus far not clear. The use of antimicrobials may be “masking” underlying bacterial infections in SARS-CoV-2 and possibly the very population that is at risk of SARS-CoV-2/COVID-19 has a level of pneumococcal and influenza vaccination that may be contributing toward lowering the morbidity in SARS-CoV-2/COVID-19. Pneumococcal and influenza vaccination have the potential to contribute toward efforts aimed at reducing the health burden of SARS-CoV-2, especially by reducing preventable admissions to hospital for pneumonia and the consequent risk of nosocomial SARS-CoV-2 transmission. Performing a clinical trial to explore co-infections and the role of pneumococcal and influenza vaccination in SARS-CoV-2 morbidity would be impractical. We therefore sought to perform a targeted literature review with the following aims:
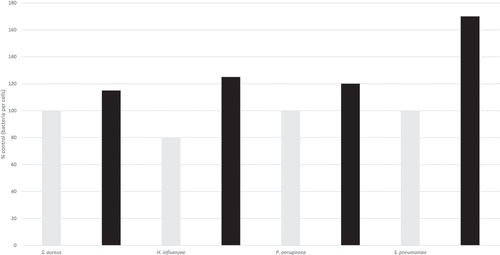
Figure 1. Number of bacteria that attached to mock (gray) or HCoV-NL63 infected LLC-MK2 (black) cells as a percentage of the control.Citation3 The differences for S. aureus, H. influenzae and P. aeruginosa are not significant; the difference for S. pneumoniae is significant.
Aims
Explore the role of S. pneumoniae and influenza in the current SARS-CoV-2 pandemic
Explore the role of pneumococcal and influenza vaccination in preventing hospital admissions for pneumonia in the current SARS-CoV-2 pandemic, and thereby the possibility of preventing subsequent morbidity and mortality
Inform future and further research
Objectives
Perform a targeted literature review (Medline/OVID and PubMed) using the search terms: Streptococcus pneumoniae, influenza, SARS-CoV-2
Include all study designs peer-reviewed or in the process of being peer-reviewed, and performed from 1 January to 31 December 2020, published in English
Inform future and further research in S. pneumoniae, influenza and SARS-CoV-2
Methodology
We performed a targeted literature review using (Medline/OVID and PubMed) and using the following search terms for the period 1 January to 31 December 2020: Streptococcus pneumoniae, influenza, SARS-CoV-2. We included all study designs which were peer-reviewed or in the process of being peer-reviewed, performed in the year 2020. Our search yielded 291 articles of which 25 were relevant.
The review is structured as follows:
Documentation of the extent to which nosocomial COVID-19 is a problem
The extent to which pneumococcal infections and influenza are problems in hospitalized COVID-19 patients, both those primarily admitted with COVID-19 and those with nosocomial COVID-19
The extent to which pneumococcal and influenza vaccination might diminish these problems
The extent to which nosocomial COVID-19 is a problem
Risk of nosocomial SARS-CoV-2 infection
Even with a safe and effective vaccine against SARS-CoV-2, it may be some time until there is sufficient community-wide protection for those at highest risk of contracting COVID-19 to be protected. In the meantime, avoiding unnecessary contact with the healthcare setting constitutes one way of decreasing exposure risk and preventing nosocomial infection.Citation4–6 In the 2020 UK study by Rickman et al. of 435 cases of PCR-positive COVID-19 inpatients over a six-week period, 47 (11%) had met the definition for definite hospital acquisition whilst a further 19 (4%) probably acquired the infection in hospital.Citation6 The median time from admission to symptom onset in these 66 hospital-acquired cases was 26 days (IQR 13 to 55 days) indicating that the patients were in hospital for a substantial amount of time before contracting COVID-19.
In China, the proportion of nosocomial COVID-19 infection has been reported to be 44%.Citation7 The authors concluded: “A large proportion of confirmed cases of COVID-19 were infected within healthcare facilities.” However, another study which was designed to determine the frequency of nosocomially-acquired COVID-19 infection found that, over a 12-week period and considering 9149 US patients suspected of having COVID-19 of which 697 were confirmed, only two cases were thought to be acquired nosocomially.Citation8
All SARS-CoV-2 positive patients at a south-east London tertiary care facility, from 1 March to 18 April 2020, who developed symptoms >14 days after admission were classified as having hospital-associated COVID-19.Citation9 Approximately 5000 patients were tested for SARS-CoV-2 and of those 1729 tested positive. Of these, 865 (50%) were admitted to hospital within 14 days of testing. A total of 58 (7.1%) of COVID-19 cases in hospital were classified as having been acquired in hospital and a potential source patient was identified in 44 instances. Underlying risk factors for hospital-acquired infection included neurological, musculoskeletal, respiratory, renal, gastrointestinal, liver, cardiac, hematological and endocrine conditions, as well as dementia and HIV. Study results demonstrated that hospital transmission of SARS-CoV-2 could be initiated by “carriers” who may not have been showing symptoms and thus would not have been placed under respiratory precautions. In their conclusions, the authors recommended admission screening for all patients.
The risk of healthcare-associated acquisition of SARS-CoV-2 by healthcare workers has also been studied.Citation10 At the University Hospital Münster in Germany, from February to July 2020, 7760 healthcare workers, inpatients and outpatients were tested for SARS-CoV-2, of whom 357 were positive. Twenty-seven index cases of hospital-associated SARS-CoV-2 infection could be identified and were followed up to investigate any further transmission the hospital. Of these cases, 23 were healthcare workers and 4 were incidentally detected in patients who were asymptomatic on admission. In other words, healthcare workers played an important role as a link in the introduction of SARS-CoV-2 from the community to the hospital.
Another study of the potential for nosocomial transmission of SARS-CoV-2, performed at three hospitals in the south of The Netherlands in March 2020, involved the screening of 1,796 healthcare workers.Citation11 Of the 1,796 healthcare workers screened (15% of a population of 12,022 healthcare workers), 96 (5%) tested positive for SARS-CoV-2. Further, it was concluded that widespread nosocomial transmission as the source of infection in patients and healthcare workers was not occurring. Although nosocomial COVID-19 is definable, identifiable and measurable, the extent of the problem may not be extensive.
The extent to which pneumococcal infections and influenza are problems in hospitalized COVID-19 patients
Co-infections and SARS-CoV-2
In Wuhan, China, co-infections by other respiratory pathogens in patients with COVID-19 were found to be relatively common.Citation12 Of 250 patients hospitalized with COVID-19, 39 (15%) tested positive for at least one respiratory pathogen in addition to SARS-CoV-2. However, Streptococcus pneumoniae did not feature amongst these pathogens. Similarly, in 116 SARS-CoV-2 positive respiratory samples from California, 24 (20.7%) were positive for one or more additional pathogens with Streptococcus pneumoniae again not featured amongst them.Citation13
In a retrospective, multicentre cohort study including all adult inpatients (≥18 years old) with laboratory-confirmed COVID-19 from Jinyintan Hospital and Wuhan Pulmonary Hospital (Wuhan, China) who had been discharged or had died by Jan 31, 2020, demographic, clinical, treatment, and laboratory data, including serial samples for viral RNA detection, were extracted from electronic medical records and compared between survivors and non-survivors.Citation14 The investigators used univariate and multivariate logistic regression methods to explore the risk factors for death and reported that of 191 COVID-19 patients, 27 of 54 (50%) of patients that died had evidence of secondary bacterial infections (although the organisms were not documented).
Over a slightly longer time-course, 1 January to 17 April 2020, a systematic review and meta-analysis was designed to document the proportion of COVID-19 patients with bacterial, fungal or viral co-infections.Citation15 Patients of all ages in all settings were included. The majority of the 3834 patients included in the 30 studies were adults (n = 3748) with a range of median ages from 42 to 63 years, but only three small studies reported data exclusively for children (n = 86). Antibiotic use was reported in 17 of the 30 studies, with >90% of patients in 10 studies receiving empirical antibiotics. For hospitalized patients with COVID-19, the overall pooled proportion of patients who had laboratory-confirmed bacterial co-infections was 7% (95% confidence interval 3% to 12%). Specific co-infecting pathogens were identified in 17 studies but S. pneumoniae did not feature amongst these.
A living rapid review and meta-analysis, which included 24 studies (of 1308 publications reviewed from December 2019 to March 2020) and 3338 patients (predominantly adult), to explore data on bacterial co-infection and secondary infection in patients with COVID-19 found that bacterial co-infection was evident in 3.5% of patients on presentation and that secondary bacterial infection occurred in 14.3% of the patients (although S. pneumoniae was not represented)Citation16
In addition to the above there are case studies. For example, a female COVID-19 patient in her 80s with several underlying conditions had a positive urinary antigen test for S. pneumoniae and succumbed to respiratory infection despite antimicrobial treatment.Citation17
In a review of laboratory results from inpatient and outpatient facilities in New York City, USA, 8990 (54.8%) of 16,408 patients tested positive for SARS-CoV-2.Citation18 Among a subset of 1204 who also tested positive by multiplex PCR, only 36 (3%) had concurrent influenza or RSV infection. The investigators did not determine whether those with co-infection had more severe illness. This question was addressed by UK investigators who found that patients infected with both influenza virus and SARS-CoV-2 were at greater risk of more severe illness.Citation19 Among 19,256 COVID-19 patients, the likelihood of those with COVID-19 was 58% lower among those with influenza compared with those without influenza. However, the 58 patients with co-infection had an increased risk of mortality compared to those with COVID-19 alone (OR = 5.92; 95% CI 3.32% to 10.91%). Among those with co-infection, 43.1% died compared with 26.9% of those with only SARS-CoV-2 infection. Mortality rates in co-infected patients rose in those 70–79 years of age and 80 years of age and older (57.1% and 66.7% respectively). In Turkey, the rate of influenza co-infection with COVID-19 in adult patients was reported to be 0.54%.Citation20
Although S. pneumoniae is an infrequent cause of secondary or concomitant bacterial infection, cases of invasive pneumococcal disease (IPD) in England were 30% lower in 2019–202 compared with a year earlier (7.7 compared with 10.9 per 100,000; incidence ratio 0.7; 95% confidence interval 0.18 to 2.67 ().Citation21 Although IPD and COVID-19 co-infections were rare, they were associated with higher case fatality rates (CFRs) <28 days (10/21, 47.6%) compared with ≥28 days after (9/27, 33.3%) the diagnosis of IPD (p < .001). Causative serotypes were not documented. CFRs in co-infected patients were higher than average yearly CFRs in the UK (19%) during the period 2006–2016.Citation22
Table 1. Number of invasive pneumococcal disease (IPD) and COVID-19 cases in England (February to June 2020) along with the case fatality rates.Citation21.
From 12 March to 16 April 2020, 26 of 92%28%) adult ICU patients with severe COVID-19 in Argenteuil, France, who were cultured within 48 hours of admission were considered to have a co-infection with a pathogenic bacterium.Citation23 Among all 32 isolates, S. pneumoniae (6/32, 31%) ranked with MRSA (n = 10/32, 31%), H. influenzae (n = 7/32; 22%) and Enterobacteriaceae (n = 5/32; 16%) as the most common bacterial co-infections.
The extent to which pneumococcal and influenza vaccination might diminish these problems
Role of pneumococcal vaccine in preventing hospitalization and pneumonia
In a Japanese study of hospitalized patients ≥65 years of age during 2014–2018, 1355 patients were retrospectively enrolled including 1045 who had not been vaccinated against S. pneumoniae and 315 who had.Citation24 Prior pneumococcal vaccination was associated with shorter all-cause hospital stays (adjusted RR = 0.66, 95% CI 0.57 to 0.76) and with reduced all-cause in-hospital mortality (adjusted OR = 0.42, 95% CI 0.22 to 0.83).
In a US study of 62,918 consecutive adults hospitalized with community-acquired pneumonia from 1999 to 2003 at 109 community and teaching hospitals, 7390 (12%) had a record of prior pneumococcal vaccination.Citation25 Pneumococcal vaccinees were less likely to die of any cause during hospitalization when compared with those with no vaccination (adjusted OR 0.50, 95% CI 0.43 to 0.59). Another important finding was that pneumococcal vaccination reduced the median length of hospital stay by two days compared with no vaccination (p < .001).
A Spanish study evaluated the effectiveness of 23-valent pneumococcal polysaccharide vaccine (PPSV23) in preventing hospital admission for community-acquired pneumonia (CAP) in adults ≥65 years of age over the period 1 May 2005 to 31 January 2007.Citation26 Cases with CAP were matched with three age- and sex-matched controls. Among 489 cases and 1,467 controls, the overall adjusted effectiveness of PPSV23 was 23.6% (95% CI 0.9% to 41.0%).
Potential role of pneumococcal vaccination in preventing SARS-CoV-2
A national Italian web-based survey was established in April 2020 using the European Commission’s open-source official EUSurvey management tool to collect data about behaviors and vaccination history.Citation27 The inclusion criteria were:
18 years of age or older
Having access to a mobile phone, computer or tablet with internet connectivity
Giving informed consent on-line to participate in the study
The self-administered questionnaire covered socio-demographic variables, clinical factors including chronic conditions and medications, vaccinations (including unspecified pneumococcal vaccines and unspecified influenza vaccines in the preceding 12 months), lifestyle and behaviors, including behaviors during lockdowns. From April to June 2020 there were 198,828 respondents with a mean age of 48 years, a standard deviation of 14.7 years and of whom 59.7% were female; 170,731 were aged between 18 and 64 years whilst 28,097 were aged from 65 to 104 years. Of those aged 18 to 64 years of age, 16% had received influenza vaccination and 2% pneumococcal vaccination, whilst 53% and 13% respectively of those aged 65 to 104 years had received influenza vaccination and pneumococcal vaccination. shows the characteristics of study participants associated with positive versus negative SARS-CoV-2 nasopharyngeal swab results (disease not specified).Citation27
Table 2. Probability of a SARS-CoV-2 positive nasopharyngeal swab according to influenza and pneumococcal vaccination status.Citation27.
The study reported that the EPICOVID19 respondents who had received a pneumococcal vaccination in the previous year had a lower probability of having a positive SARS-CoV-2 test in comparison with their non-vaccinated counterparts. History of pneumococcal vaccination in the preceding 5 years was not included in the questionnaire. Such a question may have revealed higher rates of pneumococcal vaccination. What cannot be excluded, however, is the possibility that there had been an increasing vaccination coverage with pneumococcal conjugate vaccine in children resulting in a decline in adult pneumococcal infection through indirect protection. The “healthy user” effect may also have been a potential confounder (vide infra). Whilst the findings need to be confirmed in further studies such as a propensity score matched case-control study, pneumococcal vaccination was associated with a 39% and 44% lower probability of a positive SARS-CoV-2 nasopharyngeal swab in younger and older participants respectively.
Potential role of influenza vaccination in preventing SARS-CoV-2
The EPICOVID19 study showed that individuals 65 years of age and older appeared not to have been protected against SARS-CoV-2 infection through influenza vaccination although there was a marginal protection in those less than 65 years of age ().Citation27 However, it is not clear from the study whether or not the influenza vaccine was well-matched with the circulating influenza virus strains.
By contrast, a US study of 27,201 patients tested for SARS-CoV-2 found that the odds of testing positive for SARS-CoV-2 were reduced by 24% in patients who had received influenza vaccine compared with those who had not (Odds Ratio of 0.76 with a 95% confidence interval 0.68 to 0.86, p < .001).Citation28 Those receiving influenza vaccination had higher rates of comorbidities, such as chronic pulmonary disease, than those who were unvaccinated. However, the authors note that the observed protective association may have been confounded by differences in behavior, response to social distancing and mask orders, and by socioeconomic factors. There are subtle ways, such as dysregulation of the innate immune response, that the influenza virus may increase the susceptibility to SARS-CoV-2 virus infection at the level of the receptors in the lower respiratory tract.Citation29 There are also possible immunological mechanisms, such as the “bystander” effect, by which influenza vaccination could at least decrease the severity of COVID-19.Citation30
Discussion
Pneumococcal infection has historically contributed to morbidity and mortality associated with pandemic respiratory viruses. However, its contribution to COVID-19 morbidity and mortality through co-infection has thus far not been clearly elucidated, based upon the available evidence. Although it has been previously concluded that the majority of deaths associated with the 1918–19 influenza pandemic were the direct result of secondary bacterial pneumonia, it has not yet been explained why children who were infected with the same influenza virus and colonized with the same bacteria as adults had relatively few deaths.Citation1 Then and now with COVID-19, it could well be the host response and not the bacterial infection or the virus itself that is more directly responsible for mortality. There needs to improved data confirming or not confirming the roles of pneumococcal infection and influenza as risk factors for COVID-19 hospitalization and nosocomial COVID-19. This is especially important for influenza because the COVID-19 pandemic has been associated with a low occurrence of influenza in both the Southern and Northern hemispheres.Citation31 Whether influenza virus infection returns to its usual seasonal pattern in countries with high SARS-CoV-2 vaccination rates remains to be seen.
It should be noted that definitive pathogen identification was not available in several cited studies that reported suspected bacterial co-infection. Whilst co-infection results are equivocal, our review suggests that preventing pneumococcal disease and influenza may reduce morbidity and perhaps mortality arising from COVID-19 by reducing the risk for hospital-acquired infection and the severity of COVID-19.
Whilst the Rickman et al. 2020 study suggests that a substantial period of time in hospital may need to elapse before nosocomial acquisition of SARS-CoV-2 occurs, nevertheless decreasing contact with the healthcare system in general and hospitalizations in particular are documented benefits of pneumococcal vaccination.Citation6 There is thus the potential for pneumococcal vaccination to decrease the likelihood of the nosocomial acquisition of SARS-CoV-2, especially those at highest risk with underlying conditions and/or immunosuppression. However, whilst pneumococcal vaccination may be associated with fewer cases of fatal COVID-19 in hospitalized patients, it is unclear whether potential confounding variables such as the “healthy user” effect have been ruled out.
Secondly, pneumococcal vaccination by potentially reducing the circulation of resistant S. pneumoniae could assist in the rationalization of antimicrobial chemotherapy in COVID-19 patients and in efforts to institute antimicrobial stewardship.
Thirdly, there are both theoretical immunological explanations and circumstantial evidence which point to the potential protective effect of influenza vaccination against SARS-CoV-2.
Our approach does have limitations and may not exhaustively represent all available evidence that is continuing rapidly to evolve over the course of the SARS-CoV-2 pandemic, for example a report from Spain that pneumococcal pneumonia had occurred in 3% of COVID-19 patients.Citation32 There may also be studies in languages other than English which address these questions but which we have not included. We were unable to find any randomized controlled trials which address the inter-relationships between S. pneumoniae, influenza and SARS-CoV-2 nor such trials comparing vaccination versus non-vaccination and outcomes such as co-infection or nosocomial SARS-CoV-2 infection. Lastly, our approach may have missed salient search terms which could have inserted bias into our search results.
Targeted interventions other than population-based pneumococcal and influenza vaccination are likely to have a greater impact on COVID-19, for example, active treatment of PCR-positive individuals that reduce symptomatic and severe COVID-19, and of course SARS-CoV-2 vaccination itself.
Further research is required, including the use of indirect and molecular methods for diagnosis, to document the likely burden of pneumococcal infection in those exposed to SARS-CoV-2 and, in turn, those with COVID-19. Comparative studies, comparing jurisdictions with relatively high adult pneumococcal and influenza vaccination coverage with those with relatively low coverage, and yet similar overall exposure to SARS-CoV-2, could elucidate any links as could case-control studies.
More basic research at the microbiome level could confirm whether or not S. pneumoniae is more adherent to the human respiratory epithelium infected with SARS-CoV-2 and what are the interactions of influenza and SARS-CoV-2 at the receptor level. More widespread use of the EUSurvey management tool could confirm the finding from Italy that pneumococcal vaccination is associated with a lower probability of a positive SARS-CoV-2 nasopharyngeal swab.Citation27
Another interesting avenue of research is to explore the possibility that cross-reactivity between SARS-CoV-2 proteins, the carrier protein CRM197 and proteins in pneumococcal vaccines could deliver some degree of protection against SARS-CoV-2.Citation33
Finally, the need for more widespread use of pneumococcal and influenza vaccination does not need to be justified by the presence of a SARS-CoV-2 pandemic. By a systematic review of coverage and factors associated with uptake of these vaccines, as was undertaken in Australia for example, improved programs can be designed which include official national vaccination coverage targets, incentives, key performance indicators and better coverage in highly at-risk and vulnerable populations.Citation34
Conclusions
National and international guidelines on adult pneumococcal and influenza vaccination are already in place, noting at the same time that pediatric pneumococcal and influenza vaccination can impact rates of adult disease by means of reducing transmission. Pneumococcal and influenza vaccination also have the potential to decrease the nosocomial transmission of SARS-CoV-2 by decreasing hospitalizations for pneumonia and decreasing the severity of COVID-19. With a safe and effective SARS-CoV-2 vaccine, the co-administration of such a vaccine with pneumococcal and influenza vaccines needs to be explored.
Acknowledgments
We would like to thank Karyn Davis of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA for editorial assistance.
Disclosure of potential conflicts of interest
EDGM and KF are employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA and may own stock or stock options in Merck & Co., Inc., Kenilworth, NJ, USA, a company that develops and manufactures pneumococcal vaccines, and which is developing vaccines against SARS-CoV-2
Additional information
Funding
References
- Morens DM, Taubenberger JK, Fauci AS. Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness. J Infect Dis. 2008;198(7):962–70. Predominant Role of Bacterial Pneumonia as a Cause of Death in Pandemic Influenza: Implications for Pandemic Influenza Preparedness (nih.gov). doi:10.1086/591708.
- Al-Osail AM, Al-Wazzah MJ. The history and epidemiology of Middle East Respiratory Syndrome corona virus. Multidiscip Respir Med. 2017;12:20. https://mrmjournal.biomedcentral.com/articles/10.1186/s40248-017-0101-8 .
- Golda A, Malek N, Dudek B, Zeglen S, Wojarski J, Ochman M, Kucewicz ZM, Potempa J, Pyrc K. Infection with human coronavirus NL63 enhances streptococcal adherence to epithelial cells. J Gen Virol. 2011;92(Pt6):1358–68. doi:10.1099/vir.0.028381-0.
- Wong SCY, Kwong RT-S, Wu TC, Chan JWM, Chu MY, Lee SY, Wong HY, Lung DC. Risk of nosocomial transmission of coronavirus disease 2019: an experience in a general ward setting in Hong Kong. J Hosp Infect. 2020;105(2):119–27. doi:10.1016/j.jhin.2020.03.036.
- Thindwa D, Quesada MG, Liu Y, Bennett J, Cohen C, Knoll MD, von Gottberg A, Hayford K, Flasche S Use of seasonal influenza and pneumococcal polysaccharide vaccines in older adults to reduce COVID-19 mortality (non-peer-reviewed). Centre for Mathematical Modelling of Infectious Diseases published on-line 25 April 2020 [accessed 2020 Sep 3]. https://cmmid.github.io/topics/covid19/covid19_siv_ppv23.html
- Rickman HM, Rampling T, Shaw K, Martinez-Garcia G, Hail L, Coen P, Shahmanesh M, Shin GY, Nastouli E, Houlihan CF. Nosocomial transmission of COVID-19: a retrospective study of 66 hospital-acquired cases in a London teaching hospital. Clin Infect Dis published on-line. 2020 Jun 20. https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciaa816/5860253
- Zhou Q, Gao Y, Wang X, Liu R, Du P, Wang X, Zhang X, Lu S, Wang Z, Shi Q, et al. Nosocomial infections among patients with COVID-19, SARS and MERS: a rapid review and meta-analysis. Ann Trans Med. 8(10):2020. http://atm.amegroups.com/article/view/42877/html
- Rhee C, Baker M, Vaidya V, Tucker R, Resnick A, Ca M, Klompas M. for the CDC prevention epicenters program. Incidence of nosocomial COVID-19 in patients hospitalized at a large US academic medical center. JAMA Network Open. 2020;3(9):e2020498. doi:10.1001/jamanetworkopen.2020.20498.
- Khonyongwa K, Taori SK, Soares A, Desai N, Sudhanva M, Bernal W, Schelenz S, Curran LA. Incidence and outcomes of healthcare-associated COVID-19 infections: significance of delayed diagnosis and correlation with staff absence. J Hosp Infect. 2020;106:663–72. doi:10.1016/j.jhin.2020.10.006.
- Correa-Martínez CL, Schwierzeck V, Mellmann A, Hennies M, Kampmeier S. Healthcare-associated SARS-CoV-2 transmission – experiences from a German University Hospital. Microorganisms. 2020;8(9):1378. doi:10.3390/microorganisms8091378.
- Sikkema RS, Pas SD, Nieuwenhuijse DF, Á O, Verweij J, Van Der Linden A. COVID-19 in health-care workers in three hospitals in the south of The Netherlands: a cross-sectional study. Lancet Infect Dis. 2020;20(11):1273–80. COVID-19 in health-care workers in three hospitals in the south of the Netherlands: a cross-sectional study (nih.gov). doi:10.1016/S1473-3099(20)30527-2.
- Ma L, Wang W, Le Grange JM, Wang X, Du S, Li C, Wei J, Zhang J-N. Coinfection of SARS-CoV-2 and other respiratory pathogens. Infect Drug Resist. 2020;2020:3045–53. doi:10.2147/IDR.S267238.
- Kim D, Quinn J, Pinsky B, Shah NH, Brown I. Rates of co-infection between SARS-CoV-2 and other respiratory pathogens (letter). JAMA. published on-line. 2020 Apr 15;323(20):2085. doi: 10.1001/jama.2020.6266
- Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020 March 11;395(10229):1054–62. published on-line. doi: 10.1016/S0140-6736(20)30566-3
- Lansbury L, Lim B, Baskaran V, Lim WS. Co-infections in people with COVID-19: a systematic review and meta-analysis. J Infect. 2020;81(2):266–75. doi:10.1016/j.jinf.2020.05.046.
- Langford BJ, So M, Raybardhan S, Leung V, Westwood D, MacFadden DR, Soucy J-P, Daneman N. Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis. Clin Microbiol Infect. published online. 2020 July 22;26(12):1622–29. doi: 10.1016/j.cmi.2020.07.016
- Sohal S, Rodriguez-Nava G, Khabbaz R, Chaudry S, Musurakis C, Chitraka S, Chundi VV, Friedman HJ. SARS-CoV-2 and co-infections: a review of two cases. Case Rep Infect Dis. 2020:article ID 8882348. https://www.hindawi.com/journals/criid/2020/8882348/ .
- Nowak MD, Sordillo EM, Gitman MR, Paniz Mondolfi AE. Co-infection in SARS-CoV-2 infected patients: where are influenza virus and rhinovirus/enterovirus? J Med Virol. 2020 April 30;92(10):1699–700. doi:10.1002/jmv.25953.
- Stowe J, Tessier E, Zhao H, Guy R, Muller-Pebody B, Zambon M, Andrews N, Ramsay M, Lopez Bernal J. Interactions between SARS-CoV-2 and influenza, and the impact of coinfection on disease severity: a test-negative design. Int J Epidemiol published on-line. 22 Sept 2020. Interactions between SARS-CoV-2 and influenza, and the impact of coinfection on disease severity: a test-negative design | International Journal of Epidemiology | Oxford Academic (oup.com). doi:10.1093/ije/dyab081
- Ozaras R, Cirpin R, Duran A, Duman H, Arslan O, Bakcan Y, Kaya M, Mutlu H, Isayeva L, Kebanlı F, et al. Influenza and COVID-19 coinfection: report of six cases and review of the literature. J Med Virol 2020 Jun 4;92(11):2657–65. published online. Influenza and COVID‐19 coinfection: Report of six cases and review of the literature - Ozaras - 2020 - Journal of Medical Virology - Wiley Online Library. doi: 10.1002/jmv.26125
- Amin-Chowdhury Z, Aiano F, Mensah A, Sheppard C, Litt D, Fry NK, Andrews N, Ramsay ME, Ladhani SN. Impact of the COVID-19 pandemic on invasive pneumococcal disease and risk of pneumococcal coinfection with SARS-CoV-2: prospective national cohort study, England. Clin Infect Dis ciaa1728 published on-line. 16 Nov 2020. Impact of the COVID-19 Pandemic on Invasive Pneumococcal Disease and Risk of Pneumococcal Coinfection with SARS-CoV-2: prospective national cohort study, England | Clinical Infectious Diseases | Oxford Academic (oup.com). March 2021;72(5):e65–e75.
- Houseman C, Chapman KE, Manley P, Gorton R, Wilson D, Hughes GJ. Decreasing case fatality rate following invasive pneumococcal disease, North East England, 2006-2016. Epidemiol Infect. 2019;147:e175. Decreasing case fatality rate following invasive pneumococcal disease, North East England, 2006–2016 (nih.gov). doi:10.1017/S0950268819000657.
- Contou D, Claudinon A, Pajot O, Micaëlo M, Longuet Flandre P, Dubert M, Cally R, Logre E, Fraissé M, Mentec H, et al. Bacterial and viral co-infections in patients with severe SARS-CoV-2 pneumonia admitted to a French ICU. Ann Intensive Care published on-line. 7 September 2020;10. Bacterial and viral co-infections in patients with severe SARS-CoV-2 pneumonia admitted to a French ICU | Annals of Intensive Care | Full Text (springeropen.com). doi: 10.1186/s13613-020-00736-x
- Naito T, Suzuki M, Kanazawa A, Takahashi H, Fujibayashi K, Yokokawa H, Kuwatsuru R, Watanabe A. Pneumococcal vaccination reduces in-hospital mortality, length of stay and medical expenditure in hospitalized elderly patients. J Infect Chemother. 2020;26(7):715–21. doi:10.1016/j.jiac.2020.03.016.
- Fisman DN, Abrutyn E, Spaude KA, Kim A, Kirchner C, Daley J. Reduced death, complications, and length of stay among hospitalized adults with community-acquired pneumonia. Clin Infect Dis. 2006;42(8):1093–101. doi:10.1086/501354.
- Domínguez A, Izquierdo C, Salleras L, Ruiz L, Sousa D, Bayas J-M, Benot M, Varona W, Celorrio J-M, Carratalà J. For the working group for the study of prevention of CAP in the elderly. Eur Res J. 2010;36:608–14. https://erj.ersjournals.com/content/36/3/608 .
- Noale M, Trevisan C, Maggi S, Incalzi RA, Pedone C, Di Bari M, Adorni F, Jesuthasan N, Sojic A, Galli M, et al., on behalf of the EPICOVID19 Working Group. The association between influenza and pneumococcal vaccinations and SARS-CoV-2 infection: data from the EPICOVID19 web-based survey. Vacc MDPI. 2020;8(3):471. doi: 10.3390/vaccines8030471
- Conlon A, Ashur C, Washer L, Eagle KA, Hofmann Bowman MA. Impact of the influenza vaccine on COVID-19 infection rates and severity. Am J Infect Control published on-line. 22 Feb 2021;49(6):694–700. Impact of the influenza vaccine on COVID-19 infection rates and severity - American Journal of Infection Control (ajicjournal.org). doi: 10.1016/j.ajic.2021.02.012
- Subbarao K, Mahanty S. Respiratory virus infections: understanding COVID-19. Immunity. 2020;52(6):905–09. Respiratory Virus Infections: Understanding COVID-19 (nih.gov). doi:10.1016/j.immuni.2020.05.004.
- Salem ML, El-Hennawy D. The possible beneficial adjuvant effect of influenza vaccine to minimize the severity of COVID-19. Med Hypotheses. 2020;140:109752. The possible beneficial adjuvant effect of influenza vaccine to minimize the severity of COVID-19 (nih.gov). doi:10.1016/j.mehy.2020.109752.
- Olsen SJ, Azziz-Baumgartner E, Budd AP, Brammer L, Sullivan S, Fasce Pineda R, Cohen C, Fry AM. Decreased influenza activity during the COVID-19 pandemic – United States, Australia, Chile, and South Africa, 2020. Morbidity Mort Weekly Rep. 2020;69(37):1305–09. Decreased Influenza Activity During the COVID-19 Pandemic — United States, Australia, Chile, and South Africa, 2020 | MMWR (cdc.gov). doi:10.15585/mmwr.mm6937a6.
- Anton-Vazques V, Clivillé R. Streptococcus pneumoniae coinfection in hospitalised patients with COVID-19. Eur J Clin Microbiol Infect Dis. 2021;40:1353–55. Streptococcus pneumoniae coinfection in hospitalised patients with COVID-19 (springer.com). doi:10.1007/s10096-021-04166-w.
- Root-Bernstein R. Possible cross-reactivity between SARS-CoV-2 proteins, CRM197 and proteins in pneumococcal vaccines may protect against symptomatic SARS-CoV-2 disease and death. Vacc MDPI. 2020;8(4):559. doi:10.3390/vaccines8040559.
- Dyda A, Karki S, Hayen A, MacIntyre CR, Menzies R, Banks E, Kaldor JM, Liu B. Influenza and pneumococcal vaccination in Australian adults: a systematic review of coverage and factors associated with uptake. BMC Infect Dis. 2016;16. article number 515. Influenza and pneumococcal vaccination in Australian adults: a systematic review of coverage and factors associated with uptake | BMC Infectious Diseases | Full Text (biomedcentral.com). doi: 10.1186/s12879-016-1820-8
