ABSTRACT
Trained immunity is a renewed concept of innate immune memory that facilitates the innate immune system to have the capacity to remember and train cells via metabolic and transcriptional events to enable them to provide nonspecific defense against the subsequent encounters with a range of pathogens and acquire a quicker and more robust immune response, but different from the adaptive immune memory. Reversing the epigenetic changes or targeting the immunological pathways may be considered potential therapeutic approaches to counteract the hyper-responsive or hypo-responsive state of trained immunity. The efficient regulation of immune homeostasis and promotion or inhibition of immune responses is required for a balanced response. Trained immunity-based vaccines can serve as potent immune stimuli and help in the clearance of pathogens in the body through multiple or heterologous effects and confer protection against nonspecific and specific pathogens. This review highlights various features of trained immunity and its applications in developing novel therapeutics and vaccines, along with certain detrimental effects, challenges as well as future perspectives.
Introduction
Host immune responses are classified broadly into innate immune and adaptive immune responses. Both are involved in disease prevention and maintain the homeostasis in the immune response. However, there are significant knowledge gaps to be filled regarding the underlying mechanisms associated with these responses. The innate arm is the first line of host defense that provides immediate nonspecific response against threats, such as foreign pathogens. Meanwhile, the adaptive arm takes time to respond to such threats, but it is specific, provides long-term protection, and helps in developing immunological memory. Innate immunity is regulated by physical barriers, chemical barriers, blood proteins, dendritic cells, natural killer (NK) cells, phagocytic cells (neutrophils and macrophages), and innate lymphoid cells, while the adaptive immunity is mediated by T and B lymphocytes.Citation1–3 NK, γδT, and natural killer T (NKT) cells bridge the gap between the innate and adaptive immune responses despite being from the lineage of αβT cells, and they identify antigens less specifically than T cells, exhibit rapid action against pathogens, and secrete cytokines that regulate the B and T cells.Citation4 Immunological memory is a crucial evolutionary adaptation of the immune system to recognize and remember pathogens that the body has previously encountered and aids in commencing a quicker immune response in a more robust manner. Ever since the discovery of vaccination against smallpox by Edward Jenner (1796), rabies vaccine by Louis Pasteur (1885), and passive immunization by Emil von Behring and Shibasaburo Kitasato (1890), the capability to induce immune memory to confer protection against pathogens was exclusively attributed to antibodies and immune cells (B and T cells). The cells of our innate immune system possess the unique capacity to “remember” things. Therefore, the previous dogma, “Only adaptive immunity can produce immunological memory,” has now been challenged by many recent reports demonstrating that innate immunity in plants and invertebrate organisms (those without adaptive immune responses) can confer resistance to re-infections. Moreover, some vaccine trials in mammalian models have also shown that the mechanism of protection from re-infection is independent of the functions of B and T lymphocytes.Citation5,Citation6
Recently, the classical dogma of the adaptive and innate immune system was challenged by Netea and van der Meer,Citation7 who reported that the innate immune system could train and “remember” earlier encounters with pathogens, as demonstrated by the rapid and more efficient immunological response observed after secondary exposure to the antigens. Many innate immune cells, including monocytes, neutrophils, and dendritic cells, may be affected by inflammatory factors and undergo adaptive reprogramming via genetic changes, leading to a renewed response against potential threats.Citation8 However, the myeloid progenitor and NK cells, which are essential innate immune cells, can also respond to previous antigen encounters via metabolic and epigenetic reprogramming. Moreover, certain infectious agents and vaccines can trigger a wide array of immune responses against a range of pathogens via the innate immune system.Citation7,Citation9–12 This emerging concept of a immune memory is referred to as “trained immunity” or “innate immune memory”.Citation12–14 Immunological memory is not limited to innate immune cells, but can also be exhibited by nonimmune cells.Citation15 As both innate immune and nonimmune cells can induce immunological memory, the term “trained immunity” can be used instead of “innate immune memory” to avoid confusion and harmonize the concept of immunological memory.
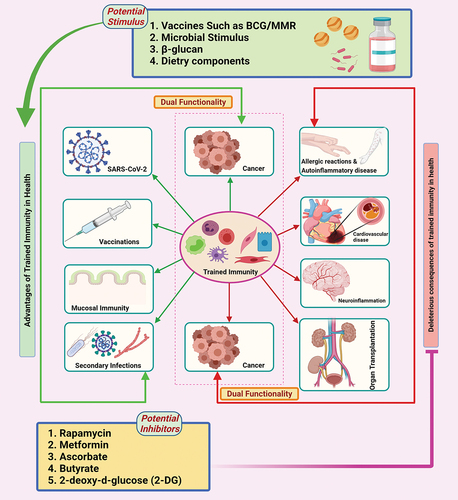
The trained immunity plays significant roles in preventing infections, activating various nonspecific protective responses of vaccination, and tackling many inflammatory diseases or hyperinflammatory reactionsCitation12,Citation16,Citation17 (). Trained immunity is also reported to play significant roles in a variety of pathological processes.Citation12 Moreover, the unique mechanisms that drive trained immunity are considered to be promising novel therapeutic targets for modulating the functions of immune cells, specifically the cells of the innate immune system.Citation12,Citation18 However, the stimulation and inhibition of trained immune responses need to be studied further. Several potential stimulants and inhibitors have been exploited to develop novel therapeutic drugs against various ailments or diseases such as autoinflammatory diseases/allergic reactions, cardiovascular disease, neuroinflammatory disorders and cancers (). In this review, we discuss the basics and applied aspects of trained immunity as a renewed concept of innate immune memory and provide therapeutic insights into the development of novel vaccines, while also outlining the limitations and challenges in its application as well as the future perspectives. We have specifically discussed the metabolic and epigenetic reprogramming pathways and the potential mechanisms by which they interact with each other. In addition, we have also highlighted the therapeutic potential of trained immunity, especially in relation to the emerging coronavirus disease 2019 (COVID-19).
Figure 1. The link between trained immunity with a variety of health and disease conditions. Stimulating or inhibiting the trained immunity with potential stimulators or inhibitors, respectively, has therapeutic potential. The increased heterogeneous immune response can be elicited via exploiting the stimulation of trained immunity. This heterogeneous response against various infections can be used to improve vaccines in the future. Some chronic autoimmune disorders, cardiovascular disease, food allergies, transplantation rejection are characterized via an inappropriately trained immune phenotype. Inhibiting trained immunity via potential inhibitors like rapamycin, metformin, ascorbate, butyrate, and 2-deoxy-d-glucose will be more useful in such cases. Figure was designed by Biorender.Com program (https://biorender.Com/) Accessed on 13 May 2021.
Trained immunity
The concept of trained immunity was established in 2011 by Netea and coworkers, which states that the immune memory is formed by the long-lasting functional reprogramming of innate immune cells. This is induced by the primary exposure to the exogenous or endogenous antigens. Innate immune cells are trained to remember the threat and respond quickly to the secondary nonspecific stimulus. The main difference between innate immunological memory and classical adaptive memory is the involvement of the cells and the specificity of the response against the antigen. Trained immunity involves innate immune cells, such as the NK, myeloid, and innate lymphoid cells.Citation7,Citation10,Citation11,Citation13–15,Citation19 Trained memory lasts from a week to several months/years, which may not be possible only with innate immune cells as they exhibit a short life span in blood. A recent study suggests that nonimmune cells have a long life span, such as stem cells (mesenchymal stromal cells, hematopoietic stem cells, epithelial stem cells, and intestinal stromal cells), microglial cells, and fibroblasts, are associated with long-lasting memory. An essential role of nonimmune cells is releasing anti-microbial factors in response to pathogens. In addition, they execute certain putative functions, such as the production and maintenance of tissue homeostasis and long-term inflammation, indicating the programming of nonimmune cells to produce long-lasting immune memory in the body.Citation18 The long-term adaptation of innate immune or nonimmune cells to the stimulus is responsible for conferring protection against a secondary encounter, facilitated by metabolic and epigenetic reprogramming. Furthermore, recent research has also shown that trained immunity is dependent on two central mechanisms: epigenetic and metabolic reprogramming of cells.Citation20,Citation21
It is important to understand the mechanisms that drive, sustain, and modulate innate immune memory as they can be used as therapeutic targets in the future. Epigenetic and metabolic reprogramming are the two main mechanisms that govern immune memory. Even though these mechanisms are mediated by different sets of epigenetic and metabolic enzymes, there are strong bilateral correlations between metabolic alterations and epigenetic changes.Citation21 Therefore, in the following sections, we have discussed the different fundamental metabolic and epigenetic modifications observed in trained cells as well the two mechanisms by which they communicate.
Metabolic reprogramming
Cellular metabolism is a key mediator of epigenetic reprogramming of trained immune cells and their progenitors.Citation8,Citation22 The ability of metabolites to modulate the function of chromatin-modifying enzymes has long been known;Citation23 therefore, the metabolic reconfiguration of innate immune cells or their progenitors can control their plasticity and epigenetic reprogramming in the form of trained immunity.Citation12,Citation22 Moreover, the extensive changes in cellular metabolism may be associated with the activation and differentiation of the innate immune cells. Cellular metabolism consists of intertwined catabolic and anabolic pathways involved in the regulation of energy as well as biosynthetic intermediates, which are mainly found in the mitochondria. Besides, it can also maintain a balance among the production of reactive oxygen species (ROS), immune mediators, and the antioxidant activities that alter the epigenetic framework and signal transduction mechanisms.Citation24,Citation25 ATP molecules provide energy to the cells in both their active and quiescent states to sustain their viability and functions. Glucose is very important for cell metabolism and it is associated with two prominent metabolic pathways, such as glycolysis that occurs in the cytoplasmic matrix and oxidative phosphorylation (OXPHOS) that occurs in the mitochondria. The intermediates generated during these processes are essential for epigenetic reprogramming. Pyruvate is formed by the glycolytic pathway upon glucose consumption by the cells via the glucose transporter, Glut-1, present on the plasma membrane, releasing two ATPs for every glucose molecule. Pyruvate will then either be metabolized to lactate or enter into the tricarboxylic acid (TCA) cycle, leading to the activation of OXPHOS in the mitochondria. This OXPHOS pathway generates 30–36 ATP molecules by consuming one glucose molecule, which is much higher than glycolysis, producing two ATPs. In this way, the OXPHOS pathway is more advantageous for the production of ATPs over glycolysis.Citation25
In contrast to normal differentiated cells, which rely primarily on mitochondrial oxidative phosphorylation to generate the energy needed for cellular processes, most cancer cells instead rely on aerobic glycolysis, a phenomenon termed “the Warburg effect.” The glycolytic pathway produces energy faster and is active during early infection.Citation26 Since neutrophils are the first responders to infection, they utilize the glycolytic pathway both in the quiescent and activated states.Citation26 In addition, the metabolic pathway also plays a role in distinguishing the macrophage phenotypes. The classical activated (M1) macrophages, which are of the pro-inflammatory phenotype, are involved in the activation of the pentose phosphate pathway (PPP) that supports inflammatory responses by providing glucose 6-phosphate for nucleotide biosynthesis along with nicotinamide adenine dinucleotide phosphate (NADPH) to produce ROS and induce glycolysis presenting low mitochondrial density. Both activated macrophages and neutrophils exhibit increased glycolysis with low oxygen consumption.Citation27 The shift in metabolism from OXPHOS to Warburg effect in the highly proliferative or tumor cells increases ATP production and PPP despite the presence of oxygen. In this Warburg effect, pyruvate generated by glycolysis is converted directly into a lactate molecule instead of entering the TCA cycle.Citation26 Glycolytic metabolism plays a vital role in producing trained immunity in response to beta-glucan and BCG stimulation.Citation25,Citation28,Citation29 The gene expression levels of the glycolytic enzymes are upregulated after exposure to a stimulus.Citation29 After 24 hours and 6 days of exposure, trained macrophages developed more lactate concentrations due to increased glucose consumption, as shown by a higher NAD+ to NADH ratio.Citation25 Furthermore, NMR (nuclear magnetic resonance) experiments were used to examine the glucose flux, with radiolabeled 13C-glucose showing an improvement in 13C integration into lactate after 24 h of lipopolysaccharide (LPS) re-stimulation. In NMR experiments, carbon 12 was replaced by carbon 13 in glucose molecule for signal detection. Therefore, detection of 13C in glucose and lactate indicates utilization of these molecules. In addition, integration of the 13C-labels was enhanced in ribosyl-1, suggesting that the pentose phosphate pathway was also triggered.Citation25
It is to be considered that the BCG and beta-glucan stimulation upregulates glycolysis which in turn activates the serine/threonine kinase (Akt)–mammalian target of rapamycin (mTOR)–hypoxia-inducible-factor 1 alpha (HIF1α) pathway for the cytokine production and cytokines producing capability of trained immune cells abolishes when this pathway or glycolysis with 2-deoxyglucose is pharmacologically inhibited.Citation25,Citation29 Eventually, the rationale for the role of glycolysis in trained immunity comes from the findings that single nucleotide polymorphisms (SNPs) in essential glycolytic enzymes are related to the activation of cytokines in ex vivo trained immune cells in healthy subjects. Furthermore, the activation efficacy of trained immune cells with BCG vaccination was associated with SNPs in two regulatory enzymes of glycolysis, such as hexokinase-2 (HK-2) and phosphofructokinase (PFKP) enzymes.Citation25 Furthermore, mononuclear cells isolated from peripheral blood with trained immune response showed higher expression of HK2 and PFKP.Citation25
TCA cycle and OXPHOS are the most critical and extremely effective mechanisms for ATP production in the resting or anti-inflammatory immune cells, including M2-macrophages and Treg cells.Citation29 In addition, alternative (M2) macrophages also rely on the OXPHOS pathway and play pivotal roles in wound healing.Citation30 TCA cycle intermediates play an important role in epigenetic transcription and take part in the long-term reprogramming of innate immune memory-like succinate and fumarate. These intermediates generated from the TCA cycle and glycolysis act as enzyme cofactors involved in epigenetic changes.Citation31 It is found that fumarate and succinate are raised in trained macrophages and beta-glucan in trained monocytes, and this stabilizes the effect of HIF-1α by elevating the glycolysis.Citation32 When stimulated with β-glucan, monocytes shift the metabolism from OXPHOS toward aerobic glycolytic pathway via the activation of the Akt-mTOR-HIF-1α pathway. This metabolic transition accelerates the activation of immune cells, enhancing the abilities of innate immune cells to function as trained cells within the system. Consistently, inhibition of the Akt–mTOR–HIF1α pathway abolishes the induction of such training of the innate immune cells.Citation29 Gene networks in yeast, bacteria, and other microorganisms can have different phenotypes. In many situations, these microbes adopt a certain phenotype in response to an environmental stimulus, and this phenotype can persist even after the stimulus is removed.Citation33 Sporulating cells carry a cellular protein that is required for germination. The transfer of cellular components from progenitor cells to spore is termed as “phenotypic memory”. This phenotypic memory is due to the stable inheritance of Lac proteins from the original cell to progenitor cells in the absence of the inducer molecule. When cells are grown in a continually changing environment, they retain a memory of earlier lactose exposure and do not require additional lag periods for subsequent lactose stimulation.Citation34 Dr. Bischofs demonstrated that the metabolic enzyme alanine dehydrogenase contributes to this effect; bacteria produce this enzyme when L-alanine amino acid is accessible and stop producing it when it is no longer available. This enzyme is stored in spores, which are passed down from generation to generation and remain dormant until nutrients enter the cell. As a result, the spores develop a stable phenotypic memory of their growth and gene expression of the progenitor cells.Citation35
In addition, crosstalk between glycolysis and glutaminolysis in trained immunity was discovered in subsequent studies using combined metabolomic and genomic analysis of beta-glucan-trained innate immune cells. Trained immune cells accumulate fumarate, a TCA cycle intermediate that affects epigenetic reprogramming by inhibiting the demethylases function of the KDM5 (lysine demethylase 5) histone.Citation32 Furthermore, a range of TCA metabolites have various impacts on the functioning of innate immune cells. For example, alpha-ketoglutarate enhanced the epigenetic reprogramming regulated by the H3K27 demethylase, JMJD3 (Jumonji domain-containing protein D3), promoting the anti-inflammatory activation of macrophages. In addition, alpha-ketoglutarate promotes endotoxin tolerance after conventional LPS- or Interferon gamma (INF-γ)-mediated activation of innate immune cells.Citation36
An illustration of the metabolic and reprogramming pathways with regard to trained immunity is presented in .
Figure 2. Overview of a metabolic pathway in trained immunity. Glucose is a key substrate for cell metabolism and is responsible for two distinct metabolic pathways. Training of immune cells by the stimulus induces complex metabolic pathways, and accumulation of TCA cycle intermediates (fumarate, succinate, and α-ketoglutarate) acts as a cofactor in histone modification and induction of trained immunity. Metabolic products in glycolysis or OXPHOS pathway linking to immune metabolic activation and production result into a faster and more robust trained immune response.

Epigenetic reprogramming
Inflammatory stimulus can cause metabolic changes, which are speculated to be involved in epigenetic reprogramming, resulting in trained immunity. Chromatin conformation regulates the expression of genes by increasing or decreasing the DNA accessibility to transcription factors. The chromosome chromatin is compartmentalized into units called nucleosomes, in which DNA is wrapped around histone proteins. In dimers, the histones are octameric structures formed by four core histone proteins, including H3, H4, H2A, and H2B. The post translational modifications like acetylation, methylation or deacetylation, and demethylation are usually observed at C-terminal of the histones. Genes are poorly accessible when nucleosomes are tightly packed, which affects the transcription process. However, genes and other transcriptional machinery can be readily accessed when nucleosomes are unfolded, enabling transcriptional activation and gene expression. The most common histone modifications like acetylation and methylation either promote or prevent the expression of underlying genes by modifying various histone-modifying enzymes, and these modifications are reversible.Citation37 The ability to trigger a more potent transcriptional response to the antigen is a qualitative and quantitative characteristic feature of the trained innate immune cells. But, in the quiescent cell, the pro-inflammatory genes are in repressed status, and the transcriptional machinery cannot be accessible to the regulatory sites, which favors the expression of inflammatory factors.Citation38 The metabolic products derived from the TCA cycle, glycolysis, and OXPHOS, act as enzyme cofactors and are involved in the acetylation and methylation of histones, viz., flavin adenine dinucleotide (FAD) and α-ketoglutarate for histone and DNA demethylases and acetyl-CoA for histone acetyltransferases. Stimulation of immune cells with pathogens leads to the alteration of histones in monocytes and macrophages, which can create an epigenetic scar at activated genes responsible for long-term memory and the immediate response of the cells to the threat (). Overall, this suggests that histone modifications with metabolic changes are the two critical components of trained immunity.Citation39
Figure 3. Trained immunity of immune cells with respect to epigenetic modification. Stimulation of naive immune cells causes metabolic reprogramming and histone modification. It would leave an epigenetic mark on activated genes. These genes are repressed later in the resting period, and the transcriptional machinery is inaccessible. Since the chromatin mark is partially removed following primary stimulation. During re-infection, trained immune cells may undergo rapid gene transcription as well as increased trimethylation and acetylation at promoter sites, establishing a link between immunometabolic activation and long-term epigenetic modification. Figure was designed by Biorender.Com program (https://biorender.Com/) Accessed on 13 May 2021.
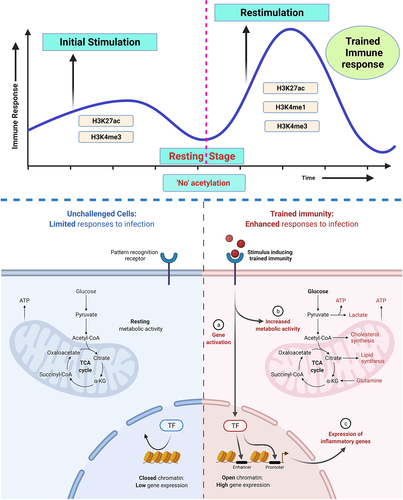
Enrichment of acetylation at lysine 27 (H3K27ac) and methylation at lysine 4 (H3K4me1) position of histone 3 are the epigenetic marks at the distal enhancer region, and, tri-methylation at lysine 4 (H3K4me3) of histone 3 is an epigenetic mark at the promoter region of stimulated genes, and these are considered as very important characteristics of the trained immunity. The chromatin mark (H3K4me3) is partially removed, following the primary stimulation, with the development of a latent (de novo) enhancer, leading to the rapid transcription of genes and producing a more effective and quicker immune response during re-infection. Therefore, opened chromatin marks of mono-methylation (H3K4me1) are easily accessible to transcription factors.Citation31 Epigenetic modifications in different phases/stages of the cell, such as un-stimulated, activated, resting, and restimulated stages, are described in . To improve the relationship between metabolic and epigenetic reprogramming, the inhibition of histone marks, H3K4me3 and also H3K9me3 (tri-methylation at lysine 9 of histone 3), by blocking glycolytic enzymes may be considered.Citation18 Trained immunity features are H3K4me3 enrichment in the promoter regions and enhanced gene transcription coding for pro-inflammatory cytokines. It has also been established as a characteristic feature of the BCG vaccine in providing heterologous immunity to an unrelated pathogen.Citation5,Citation40 BCG vaccination also suggests a clear decrease in the extent of DNA methylation within immune pathways at gene promoters compared to the individuals categorized as non-responders.Citation41
Table 1. Epigenetic changes in trained immunity 12,62
The enzymes play pivotal roles in epigenetic rewiring in cells undergoing training in the presence of a stimulus. In a recent study, two distinct enzymes, including KDM5 and Set7, have been identified, which control the epigenetic remodeling in immune cells. First, KDM5 belongs to the histone demethylases family, is responsible for H3K4 demethylation and plays an essential role in trained immunity. Beta-glucan training of monocytes results in the decreased biological activity of KDM5 demethylases after six days of training, which is controlled by intracellular changes in fumarate.Citation25 Set7 is a lysine methyltransferase that is responsible for changes in H3K4me1. Set7 has been reported as a crucial enzyme required for beta-glucan-induced trained immunity under in vitro as well as in vivo conditions.Citation42 To determine the difference between training and priming, in vitro and in vivo models were used. Human peripheral monocytes were used as in vitro model to show that exposing cells to stimuli (training) for a short period of time (24 hours) induces an immunological response, which then returns to a steady state when there is no stimulus. If epigenetic changes occur during the primary training period, then the response to subsequent stimuli will be enhanced. In contrast to training, priming to a stimulus is not retained for a long time, and does not respond quickly to the secondary stimulus; in fact, cells may not even return to their steady state before the secondary stimulation. Even though the epigenetic events, kinetics and dynamics differ between models, administration of training agents in BM-progenitor cells (in vivo model) causes hematopoietic stem cells (HSCs) proliferation initially and then returns to steady state before secondary stimulation.Citation43,Citation44
Role of nonimmune cells in innate immune memory
Although innate immune memory was first explained in myeloid cells, it may not be possible to transfer memory phenotypes to their descendants and deliver long-term protection due to the limited life span of the cells,Citation12,Citation45 Mature myeloid cells, such as dendritic cells and monocytes, have an average half-life time of 5–7 days.Citation45 This indicates explicitly that long life span cells could be responsible for trained immunity, such as epithelial stem cells, HSCs , intestinal stromal cells (ISCs), mesenchymal stromal cells, microglial cells, and fibroblasts.Citation15 As a result, the output of the immune response in the target tissue depends not only on the immune cells but also on the diverse immune and nonimmune cell networks and signals.Citation46 Nonimmune cells typically have a distinctive function in generating and sustaining tissue homeostasis. These also play a significant role in the defense against pathogens by releasing long-term inflammatory anti-microbial factors, indicating that nonimmune cells may be programmed to have durable immune memory.Citation15 Microglial cells go through training and provide an inflammatory response against bacterial antigens.Citation47–49 It has been postulated that microglial cells develop innate immune memory constituted of various inflammatory pathways orchestrated by epigenetic rewiring.Citation47,Citation50 In a sample of naive mice versus mice primed with attenuated Salmonella typhimurium containing its lipopolysaccharide (LPS), the latter group showed enhanced microglial immunoreactivity in response to a second LPS stimulation four weeks later. There was no elevated immunoreactivity in the microglial cells of naive mice.Citation51 However, the epigenetic pathways that can be related to the long-term activation of microglial cells in response to infection have not yet been elucidated.Citation15
Fibroblasts and epithelial cells establish a functional and physical barrier against foreign particles. In innate immunity, they represent the front line of the defense system.Citation52 In the mice model, a study using imiquimod (IMQ) to induce skin inflammation reported that the skin exposed to inflammatory agents responded quicker to an unknown secondary threat, with quick wound curative ability in primed mice than the naive mice.Citation53 Intestinal stromal cells (ISCs) are nonprofessional immune cells that display immune characteristics and contribute significantly to trained immunity or several other related processes. ISCs such as mesenchymal stem cells (MSCs), fibroblasts, epithelial cells, and endothelial cells express TLRs 1–9 and release certain pro-inflammatory cytokines in exposure to pathogens.Citation54,Citation55 It has been documented that the ISCs also release sustained pro-inflammatory cytokines during the second contact to attract other immune cells at the infection site.Citation56 ISCs exhibit recollection features during human allergic inflammatory disorder, indicating differences in accessibility of chromatin as the stimulus is removed.Citation57 Moreover, MSCs primed with LPS or tumor necrosis factor (TNF) was also reported to develop a robust immune reaction by releasing a range of pro-inflammatory cytokines, including IL-8, MCP-1, and IL-6, at the time of restimulation. Primed MSCs exert a more significant therapeutic effect than unprimed MSCs in a diabetic rat model.Citation19 Trained immunity may occur in bone marrow progenitor cells, tissue macrophages, and blood monocytes (peripheral trained immunity). Hematopoietic stem cells (HSCs) divide asymmetrically, eventually forming the lymphoid and myeloid cells entire repertoire, which are long-lasting cells with self-renewal processes.
Monocytes acquired from trained HSCs move to peripheral organs, leading to macrophages originating from monocytes with improved effector functions against different forms of the pathogen.Citation12 BCG vaccine primed HSCs reach the bone marrow and generate a strong immune memory against the pathogenic bacterial strains.Citation20 Beta-glucan can induce increased glycolysis in trained HSCs.Citation58 Latent gamma herpesvirus and adenovirus infections are given to the lungs to test trained immunity levels in individual tissues. Studies have shown reduced house dust-induced asthma in the lungs of mice previously infected with gamma-herpesvirus that might be ascribed to the development of long-term monocyte-derived regulatory alveolar macrophages, which played a role in protecting the emergence of a lung allergic reaction.Citation59,Citation60 It has been demonstrated a trained phenotype in hematopoietic stem cells of mice, which are indicated by the high production capacity of cytokine and myeloid skewing following the administration of β-glucan.Citation58 BCGʻs occurrence in the bone marrow milieu results in trained phenotype in multipotent progenitors, bone marrow-derived macrophages, and hematopoietic stem cells, thus imparting augmented protection against infection by Mycobacterium tuberculosis.Citation20 This result has recently been corroborated in the BCG vaccinated human volunteers, in whom the trained immunity was detected in the bone marrow after 3 months of BCG immunization, inducing peripheral trained immunity.Citation61
Trained immunity vs tolerance
Innate immunity exhibits two contrasting adaptive effects. Functional reprogramming of innate immune cells caused by their early activation can either result in a more intense (trained immunity) or less potent (trained tolerance) response to the subsequent encounter of the pathogen. Trained immunity requires the metabolic and epigenetic remodeling of immune cells to enable a balanced response against subsequent infections. However, misleading trained immune strategies may trigger the development of diseases, leading to either a chronic hyper-inflammatory disorder or permanent immunological tolerance by suppressing the role of the immune system to sustain homeostasis and avoid tissue/organ injuries.Citation12 Priming with Candida albicans or beta-glucan induces monocytes and increases the production of cytokines upon re-stimulation.Citation62 Monocytes are functionally programmed based on the type and concentration of the ligand. Engagement of NOD-like receptor (NLR) induces trained immunity. Long-term immune function can be achieved with high doses of muramyl dipeptide (MDP) or Tri-DAP ligands, but it may vanish at lower concentrations of the ligand. Tolerance is mainly caused by the presence of high inflammatory levels of pattern recognition receptor (PRR) ligands in toll-like receptors (TLRs), except for CpG, for which every dosage appears to be inherently tolerant.Citation63 Moreover, the pre-stimulation of LPS is considered to induce tolerance.Citation64 Interestingly, this kind of tolerance-induced effect was not only diminished but reversed at low concentrations of TLR ligands, and the monocytes were trained to be maintained in an enhanced pro-inflammatory state. Anti-inflammatory pathways are activated in combination with pro-inflammatory responses to stop excessive inflammation and tissue destruction while also reducing the time of action of such inflammatory responses.Citation63
Detrimental effects of trained immunity
Trained immunity is thought to be an effective immune defense against infections. Furthermore, trained immunity dictates the vaccineʻs heterologous effects, resulting in improved defense against secondary infections, but can play a dysfunctional role in chronic inflammatory diseases due to long-term activation.Citation39,Citation65 Trained immunity has maladaptive consequences in chronic inflammatory conditions, contributing to hyperinflammation and the development of various other complications, such as cardiovascular diseases, autoinflammatory diseases, and neuroinflammation.Citation66
Sterile inflammation forms the base from which systemic inflammatory disorders develop in response to behavioral variations in Western societies. Western lifestyle exerts long-lasting effects on meta-inflammation, chronic metabolic inflammations caused by excessive western diet consumption accompanied by sedentary behavior, and are memorized by innate immune cells via epigenetic reestablishment and remodeling.Citation67 Monocytes and macrophages, which are part of the innate immune system, have crucial roles in the poor prognosis of many diseases, including cardiometabolic illnesses, neuroinflammation, diabetes, and obesity. Numerous findings suggest the deleterious consequences of trained immunity in cardiac disorders.Citation68–70 The epidemiological association and high risks of diseases or increased infection rates can be explained by uncovering the roles of trained immunity in cardiometabolic disorders. In addition, various external and internal stimuli, such as oxidized low-density lipoprotein (oxLDL), catecholamines, and aldosterone, can promote trained innate immunity in monocytes or other cells, including nonimmune cells, which may aggravate the infectious risks of many life-threatening diseases.Citation71–74 Furthermore, atherosclerosis is characterized by endothelial dysfunction and systemic vascular inflammatory responses caused by the exaggerated innate immune system. Long-term activation of the innate immune system in trained immunity may link non-resolving inflammation and atherosclerosis, implying that trained immunity may contribute to atherosclerosis.Citation21,Citation68,Citation70,Citation75–78 Innate immune cells, including macrophages and monocytes, play a pivotal role in immune homeostasis by eradicating pathogens and repairing the tissue damage. Because of innate immune memory these cells are also involved in aggravation of chronic disease conditions.Citation11,Citation13,Citation79 Four weeks of exposure to LDL-containing Western-type diet induced the extreme reprogramming of transcriptional events in the bone marrow myeloid progenitor cells and circulating monocytes in atherosclerosis-prone Ldlr-/-mice.Citation21,Citation67 Pathogen associated molecular patterns (PAMPS) and damage associated molecular patterns (DAMPS) released during tissue injuries and after tissue or organ transplantation allow macrophages to be epigenetically reprogrammed, contributing to the development of trained immunity. The donor allografts upregulate vimentin and high mobility group box 1 (HMGB1) in the mouse heart transplantation model that induces local grafting-infiltration of monocyte-derived cells.Citation80 Both PAMPs and DAMPs are recognized by PRRs expressed by macrophages, which trigger the immune response resulting in the increased production of inflammatory cytokines.Citation81 Murine studies revealed that organ transplantation induces macrophage training, indicating a previously unrecognized mechanism contributing to allograft rejection.Citation82 Sepsis is the uncontrolled production of inflammatory cytokines after infection, which can lead to septic shock. The infection causes a pro-inflammatory response for eliminating the causative agents in physiological conditions, followed by an anti-inflammatory response to circumvent hyper-inflammation and subsequent tissue impairment. Pro-inflammatory and anti-inflammatory responses have largely been compensated. Nevertheless, the homeostasis is disturbed, and the anti-inflammatory response becomes irregular due to the overwhelming pro-inflammatory response. Thus, there is increased stimulation of anti-inflammatory reactions leading to immunoparalysis after 24–48 h of hyper-inflammation.Citation83
A major group of age-associated neurodegenerative disorders is linked with chronic inflammation. Peripheral use of inflammatory stimuli in the mouse model of Alzheimerʻs disease contributes to long-lasting microglia training. These brain-resident macrophages aggravate β-amyloidosis in the brain. Systemic inflammation allows the microglia to be reprogrammed, leading to potentially hyper-responsive ‘trainedʻ brain immune system states.Citation84 Microglia has been recognized as crucial players in developing any disease conditions in the central nervous system. They are resident macrophages that play an essential role in normal and pathological brain activity. Neuroinflammation may be induced by the microglial immune memory.Citation47 Microglia can be programmed to induce biochemical and genomic alterations. Modifications in H3K4me1 and H3K27ac that persist up to 6 months may elicit immune training in neural macrophages.Citation49 In another study, similar observations were found in microglia, demonstrating that histone acetyltransferases HDAC1/2 are required for this process.Citation85 Microglial immunological response may be generated from both microbiological and endogenous triggers, including stress, and is assumed to be transgenerational.Citation51,Citation86–88 In addition, monocytes displayed an inflammatory nature, and patient outcomes were linked to enhanced cytokine release volume in an aged group of old people with the cerebral small-vessel disease.Citation89 Robust and successful immune system activation is required for the elimination of cancer cells from the body. Inflammatory responses that are excessive or protracted can also encourage the progression of tumors as chronic inflammation. Activation of metabolic processes and trained immunity in tumor cells share some typical attributes, such as dependency on glycolysis and upregulation of the signaling of transcription factors, such as HIF1 alpha.Citation12 DAMPS released from dying cells in the donor organ binds to trained immunity associated PRR and induces metabolic changes in innate immune cells during organ transplantation. Trained macrophages produce more proinflammatory cytokines and stimulate the adaptive immune system, which results in allograft rejection.Citation80
Therapeutic targeting of trained immunity
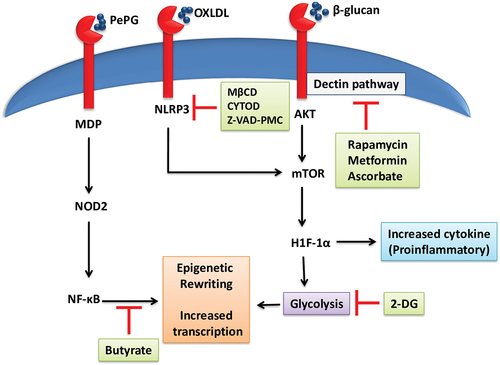
The regulation of trained immunity can be utilized as a novel therapeutic strategy to treat excessive and defective immune activities. Microbial ligands induce trained immunity via various signaling pathways, facilitated by epigenetic, metabolic, and transcriptional events. Βeta glucans are fungal cell wall polysaccharides consisting of β1,3 or β1,3/β1,6 glycosidic bonds. Macrophages recognize these PAMPs via dectin-1 dependent pathway (C-type transmembrane lectin receptor). Relevant epigenetic marks, specifically histone marks, such as H3K27Ac, H3K4me1, and H3K4me3, are activated by the activation of macrophages via dectin 1, resulting in trained immunity that triggers non-targeted immune responses toward exogenous pathogens. Endogenous proteins, vimentin and HMGB1, are evident in sterile inflammation. Dectin-1-expressing macrophages bind to vimentin, which is upregulated in the donor organ following organ transplantation, resulting in macrophage activation and acute rejection. A study to validate the activity of endogenous proteins revealed that dectin-1 or TLR4 deficiency significantly reduced pro-inflammatory cytokines and lactate production by graft infiltrating macrophages.Citation80,Citation82 During chronic rejection, graft infiltrating macrophages express M1 and M2 phenotype markers. Manipulation of M1/M2 polarization can be used as a therapeutic approach to combat allograft rejection.Citation90 Lack of Tumor necrosis factor receptor-associated factor (TRAF6) in macrophages prevents the accumulation of M1 phenotype and deletion of mTOR mitigates the accumulation of M2 that results in long-term allograft survival without chronic rejection.Citation91,Citation92 Dectin pathway is inhibited by rapamycin, metformin, and mTOR, ascorbate targeting AKT/protein kinase B, and HIF1α, respectively.Citation29
Therapeutic approaches against fungal infections such as candidiasis render protection through monocyte-dependent innate immune memory to mice against lethal candidiasis.Citation93 Peptidoglycan (PepG) is an important PAMP that is associated with LPS and triggers inflammatory cytokine production. The smallest PepG derived molecular structure MDP (muramyl dipeptide) engages with NOD2 (Nucleotide oligomerization domain). BCG provides nonspecific defense against secondary infections via Nod-like receptor 2 (NOD2)-dependent epigenetic rewiring of innate immune cells.Citation5 Trained immune cells were observed to increase histone H3 trimethylation at lysine (H3K4me3) at the promoters of crucial inflammatory cytokines such as IL-6 and TNF-alpha, which was correlated with the increased production of these proinflammatory cytokines. NOD2 is an intracellular PRR that acts as a general sensor for the muramyl dipeptide unit of bacteria cell walls and viral RNA.Citation94 NOD2 activation signal passes through the nuclear factor (NF)-κB and further activates epigenetic remodeling, inducing trained immune in macrophages. However, butyrate can be used to block this activation of macrophages and prevent the histone acetylation.Citation29,Citation95
Oxidized LDL is also one of the reasons for trained immunity activation. When immune cells, such as monocytes, are primed with OxLDL, they shift to glycolysis for ATP synthesis and produce higher levels of pro-inflammatory cytokines upon restimulation.Citation65 The OxLDL is a DAMP molecule that binds with CD36 expressed in myeloid cells.Citation71 OxLDL can also help to cholesterol crystals formation and activation of inflammasome (NLRP3) when internalized and released into the cytoplasm, results in the release of IL-1β and other pro-inflammatory cytokines end up with long-lasting inflammatory reaction.Citation96 The stimulation of monocytes by OxLDL has adverse effects in organ transplantation, as it is linked to a higher risk of graft rejection in recipients.Citation97 As a result, trained macrophages may promote chronic rejection by causing atherosclerosis and allograft fibrosis.Citation82 However, methyl-β-cyclodextrin (MβCD), cytochalasin D (CYTOD), and Z-VAD-FMK inhibit the activation of NLRP3 and the formation of cholesterol.Citation98 OxLDL-mediated trained immunity phenotype is eliminated by 5'-deoxy-5'-methylthioadenosine (histone methyl-transferase inhibitor) and reversing histone methylation is a requisite for causing a change in chromatin structure, which indicates increased gene expression. The compound 2-deoxy-d-glucose (2-DG) plays a noteworthy role in blocking glycolysis that induces the trained immunity in monocytes.Citation32 Therapeutic drugs targeting the signaling pathway of trained immunity are presented in .
Figure 4. A schematic representation of trained immunity-signaling pathways and their therapeutic targets. PAMPs targeting a variety of PRRs results in the activation of various signaling pathways that facilitate trained immunity.
As GM-CSF and IL-1β are the main regulators of myeloid-biased progenitor-induced trained immunity, antibodies induced against these molecules are speculated to be beneficial in hindering trained immunity. In a recent CANTOS trial, the adverse impacts of trained immunity were successfully inhibited by a monoclonal antibody targeting IL-1β in cardiovascular disease patients.Citation99 RNA interference (RNAi) is an evolving alternative modality for therapeutic blocking of particular receptors or the expression of trained immunity-related molecules. RNAi has been successfully extended to minimize immune cell mobilization after myocardial infarction.Citation100 Intravenously injecting nanomaterials engaging myeloid cells and their progenitor and stem cells in the bone marrow can achieve long-term therapeutic advantages. By complexing these nanostructured materials with molecular structures that suppress metabolic and epigenetic pathways, the activation of trained immunity can be avoided.Citation101,Citation102 Epigenetic therapy can be used to regulate immune response by modifying gene expression in certain inflammatory and autoimmune disease conditions. Azacytidine and decitabine are DNA methyltransferase inhibitors used in cancer treatment to limit the growth and division of cells.Citation103 Histone deacetylase inhibitors including trichostatin A, givinostat, and valproic acid are used to treat arthritis, sepsis, and diabetes by lowering the levels of proinflammatory cytokines and reducing systemic inflammation.Citation104,Citation105 In one study, individuals were vaccinated at 6 pm and at 8–9 am with the BCG vaccine. Staphylococcus aureus and Mycobacterium tuberculosis were used to activate the peripheral blood mononuclear cells before and two weeks and three months post-immunization. It was found that the individuals vaccinated in the morning exhibited stronger immunity and increased cytokine production in the isolated monocytes than those vaccinated in the evening.Citation106 The innate immune cells have a shorter life span and undergo apoptosis. TNF, phosphatidyl inositol 3-kinase (PI-3K)/Akt, extracellular signal-regulated kinase (ERK), anti-apoptotic molecules and heat shock proteins play essential roles in defining monocyte life span by regulating gene transcription and suppressing the apoptotic pathway.Citation107 Endogenous TNF treatment of monocytes and macrophages induces an immune response and favors prolonged survival of monocytes. LPS stimulation of monocytes induces ERK activation, which then phosphorylates various cytoplasmic and nuclear substrates that regulate cell growth, survival, and death.Citation107 PI-3K/Akt survival signaling modulates Bcl-2 expression via NF-kB and phosphorylating the anti-apoptotic proteins Bad and XIAP. Cytokines such as GM-CSF, Interleukin (IL)-15 and IL-18, can stimulate neutrophils limiting apoptotic activity that further leads to the increased neutrophil effector activities.Citation108 T cell-macrophage interaction is required for heterologous protection of vaccines against infections, and it is also involved in long-term adaptation and antimicrobial activity in innate immune cells.Citation109,Citation110
Trained immunity-based vaccines (TIbVs)
Trained immunity is known to have some advantages in strengthening the host immune responses to infectious pathogens and designing effective vaccines. TIbVs may induce specific and nonspecific immunity by targeting the innate immune cells to stimulate trained immunity. Attenuated or inactivated pathogens can also be an exemplar of TIbVas long as they comprise PAMPs capable of triggering PRRs that can elicit a trained immunity. Different PRR ligands, such as Candida albicans-derived beta-glucan and BCG-derived muramyl di-peptide, are identified as trained immune stimuli.Citation111 It should be noted at this point that it opens up the possibilities of achieving a trained immunity by varying the set of PRR ligands and targeting immune cells. In this context of vaccine designing, trained immune cells are used to activate the adaptive immune response as an emerging therapeutic strategy for the clearance of bystander pathogens. Dendritic cells (DCs) act as links between innate and adaptive systems and enhance the adaptive response via the release of IL-1β.Citation112 Polybacterial sublingual vaccine MV130 designed to prevent chronic infection of the respiratory tract has been reported to increase T cellsʻ in vivo response to unrelated antigens by priming DCs and facilitating the release of IL-1β in vitro.Citation113 BCG vaccine also improves the T cell PRR expression in innate immune cells as well as the release of cytokines, such as IL-1β.Citation114 Further, defense against M. tuberculosis infection by BCG vaccine was correlated with the enhanced production of certain pro-inflammatory cytokines following heterologous stimulation with Streptococcus pneumonia and E. coli,Citation115 an observation reminiscent of trained immunity.Citation116, Citation117 In addition to conferring T cell immunity against bacteria in patients treated with MV130, T cell response to heterologous flu antigen has been reported to be increased via the activation of the NLR and TLR signaling pathways in DCs.Citation112 Likewise, another MV140 sublingual vaccine is intended to circumvent urinary tract infections, inducing TLR and CLR-mediated Th1 and Th17 responses.Citation118 MV140 is effective against urobacteria that are not part of its composition.Citation119
BCG vaccination can also protect against viral diseases. In a placebo-integrated pilot study of BCG vaccination, all volunteers were administered a yellow fever vaccine one month after BCG. BCG-immunized volunteers showed a substantial decrease in viremia comparable to the placebo group, strongly associated with elevated production of IL-1β.Citation32 Two experiments undertaken in Africa showed that the adults vaccinated for smallpox exhibited slightly reduced mortality rates.Citation120 Thy1+ subclass of NK cells displayed certain innate memory features after the Vaccinia viral infection.Citation121 In children, trivalent live attenuated influenza vaccine has provided indirect protection against respiratory diseases.Citation122 In a mice model, nonspecific cross-protection has been achieved against RSV (Respiratory syncytial virus) by cold adapted, live attenuated influenza vaccine (CAIV) as it shares common characteristics, including activation of innate immunity through many PRRs, such as TLR7, TLR3, and retinoic acid-inducible gene I.Citation123 Immunostimulant, OM-85, is an oral bacterial lysate mixture reported to significantly improve resistance to respiratory viral infection in the murine model and decrease viral quantity in the lungs.Citation124 It also reduced the lung epithelial cell infection caused by rhinovirus.Citation125 MTBVAC, the live attenuated strain of M. tuberculosis, can provide trained immunity by inducing glutaminolysis, glycolysis, and accumulating histone methylation marks in pro-inflammatory gene promoters responsible for rendering nonspecific defense against heterologous pathogens. MTBVAC was administered to C57BL/6 mice that were then infected nine weeks later with intranasal streptococcal pneumonia; it was observed that 60% of the vaccinated mice survived, while the unvaccinated group of animals died. Blood bacteria culturing revealed the absence of live bacteria in the surviving animals.Citation126 A considerable reduction in infant mortality was associated with the BCG vaccine. In addition to tuberculosis protection, the inherent adjuvant properties of BCG will enhance the immune-stimulating properties of other neonatal vaccines.Citation127 Oral polio vaccine (OPV) and measles vaccine (MV) have provided strong immunity against heterologous infections and also reduced the fatality rate.Citation128 In addition to the positive heterologous effects of BCG; recombinant MTB-based novel vaccine candidates, like MV, OPV, and VPM1002, also have noteworthy effects. Hence, they are included in the clinical investigations. A sturdy state of innate immune cell activation is caused by enhancing the host defense via the induction of trained immunity. In this case, upon encountering a new pathogen, these cells can exhibit quicker and improved responsiveness to reduce the chances of infection and prevent transmission.Citation12 The BCG, measles, yellow fever and polio vaccines were shown to protect and reduce the other infections. Animals were demonstrated to be protected against Candida albicans, Schistosoma mansoni, and M. tuberculosis after receiving the BCG vaccine.Citation129 BCGʻs ability to induce trained immunity in monocytes and macrophages appears to be critical for its anticancer effects.Citation130 The BPZE1 live attenuated pertussis vaccine has also been shown to protect against various heterologous respiratory infections such as Bordetella, influenza, and respiratory syncytial virus (RSV).Citation131 In mouse models, live-attenuated influenza vaccination provided rapid protection against respiratory syncytial virus.Citation123 Ty21a, a heat-killed salmonella vaccine, provides protection against C. albicans.Citation132 In addition, exposure with the fungal ligand β-glucan protected against infection with Staphylococcus aureus, while muramyl dipeptide, a peptidoglycan component, protected against infections with Streptococcus pneumoniae and Toxoplasma gondii.Citation133,Citation134 Other examples include the use of CpG oligodeoxynucleotide, which protects against sepsis and E. coli meningitis, and flagellin-induced protection against S. pneumoniae and rotavirus.Citation135
Hence, integrating innate immune response in the form of trained immunity into the biological effects of prospective vaccines can enhance the efficacy of the vaccine. Consequently, a vaccine that targets both innate (trained immunity) and adaptive (classical immune memory) immune response will undoubtedly be more successful than the existing vaccines against infections, including the SARS-CoV-2 infection. Further advancements will also increase the vaccine outcomes of groups at a greater risk of illness due to SARS-CoV-2 infection, such as the elderly. Adaptive immune responses deteriorate with age, and incorporating trained immunity into vaccinations for the aged population could help to fix this dilemma.Citation136 Hence, the following section describes the trained immunity in the context of COVID-19.
Trained immunity and COVID-19
The ongoing COVID-19 pandemic is continuously flaring up, with a second wave being observed worldwide.Citation137,Citation138 Recently, few potent vaccines have been developed. A vaccination campaign is in progress at the global level128. Several drugs and therapeutics are undergoing clinical trialsCitation139–141; however, there is limited choice of effective drugs and therapies. Immunomodulatory and immune-enhancing strategies have also been suggested to provide some protection from higher severity of the COVID-19 infection in the affected patients as well as for prophylactic purposes to prevent the spread of the SARS-CoV-2 infection by boosting the immunity of the body.Citation142,Citation143 BCG vaccination at birth may provide defense against COVID-19, though this assertion may not refer to other coronaviruses such as MERS (Middle East respiratory syndrome) or SARS-CoV (severe acute respiratory syndrome coronavirus), because they are closely related.Citation9,Citation144,Citation145 However, trained immunity is not a long-lived immune response and lasts for a short time, implying that trained immunity acquired at birth cannot be sufficient to shield adults from pathogens later in life.Citation146 In addition to BCG, microbial stimulation or other vaccines may help in developing a trained immunity to defend against the SARS-CoV-2 infection. The nonspecific protection of BCG against several infections, especially viral infections, is not mediated by adaptive immune response constituent of lymphocytes, but via the reprogramming of innate immune cells, such as monocytes.Citation9,Citation147–149 BCG vaccinationCitation150,Citation151 and beta-glucan stimulation of monocytesCitation5,Citation152 have been linked with the activation of trained immunity, which can be directly associated with the resistance against infectious diseases, including COVID-19.Citation10,Citation153,Citation154 During the COVID-19 pandemic, BCG vaccination has been linked to a lower rate of COVID-19 illness and severe exhaustion.Citation151 In theory, the activation of BCG or beta-glucan causes innate immune cells and their precursors to reprogram, resulting in heterologous defenses against a range of pathogens, including viruses.Citation32
In viral infections, such as SARS-CoV-2, trained immunity modulates the innate immune response for more beneficial outcomes, especially in all populations at a greater risk of infection.Citation17,Citation155 Early retrospective research on the effects of BCG vaccination in infancy has generated mixed findings in terms of its effectiveness in combating SARS-CoV-2 infection.Citation156–160 Moreover, recent epidemiological findings during initial waves of the COVID-19 pandemic illustrate that the occurrence of persons with positive tuberculin sensitivity tests, as a result of exposure to Mycobacterium spp. or as a result of BCG vaccination, is inversely related to the incidence of SARS-CoV-2 infection.Citation161 While the associations between trained immunity and defense against COVID-19 are still not conclusive, this led to an increase in randomized controlled trials to see if trained immunity has positive consequences on the innate immune response to provide a more favourable response against the SARS-CoV-2 infections.Citation17,Citation155 More than 25 trials have been conducted to assess the protective effects of the BCG vaccination against COVID-19, with many still being developed.Citation162
The trained immunity following BCG vaccination is associated with enhanced production of several pro-inflammatory cytokines, including TNF-α, IL-1, and IL-6, providing significant protection against different viral infections.Citation5,Citation114 However, it is imperative to consider the potential of trained immune cells to produce a range of pro-inflammatory cytokines, such as IL-1, IL-6, and TNF-α.Citation5 These pro-inflammatory cytokines can exaggerate the secretion of inflammatory cytokines in severely affected COVID-19 patients. On the other hand, trained immunity has been postulated with the reprogramming of innate immune cells in such a way that the trained cells of the innate immune system can work more robustly during the early phase of the viral infection, which in turn prevent the poor prognosis of the COVID-19 disease.Citation12,Citation17 The collaboration of the innate immune response with the adaptive immune system is critical in the fight against various viral infections, including SARS-CoV-2. BCG vaccination activates the production of CD4+T cells and IFN-γ. Several recent studies have correlated the trained immunity and the adaptive immune response provided by BCG vaccine against COVID-19. BCG vaccine might generate cross-reactive T cells against SARS-CoV-2 and provide cross-protection against the SARS-CoV-2 infection.Citation163
Previous research has also revealed the link between NK cell activity in the vaccinated population and the specific immunological protection against the influenza virus. NK cell stimulation remains elevated following one month of immunization, which might provide immunity against influenza and other respiratory viral infections.Citation164 Observational epidemiological studies have also revealed that the Measles, Mumps, and Rubella vaccination protects against COVID-19.Citation165 Hanker et al. postulated that age probably declines immunogenicity against measles vaccination which might be a reason for such increasing incidence of COVID-19 in adults because most of the vaccinated persons seemed to have no protective IgG antibody against measles beyond ten years.Citation166 Furthermore, trained immunity may be one of the factors that render youngsters less susceptible to SARS-CoV-2 infection.Citation167
Many adult patients have acquired similar vaccinations, such as BCG in children, during their lives, but there are varying infection rates and fatalities among the different areas and population groups. Therefore, future research should concentrate on the absolute degree of defense provided by trained immunity.Citation168 Large randomized BCG vaccination trials are required to substantiate the protective effects of BCG vaccination against COVID-19.Citation169 It is also necessary to investigate the molecular pathways that inhibit the innate immune cells from reprogramming to a suppressive phenotype in extreme COVID-19 cases. In contrast, investigations on the protective efficacy of the BCG vaccination against COVID-19 have shown contradictory results, with no substantial impact of the BCG vaccine against COVID-19.Citation158,Citation160 In another study, it has been reported that BCG vaccination was more effective against COVID-19 in younger populations than in the elderly.Citation170 From the above contradictory reports on the effects of BCG, further studies may be needed to derive a conclusive opinion on the positive impact of BCG vaccination against COVID-19.
The mRNA and recombinant adenoviral vaccine (AdV) encoding for the SARS-CoV-2 spike (S) protein have been found to induce both innate and adaptive immune response. To ensure maximum protection, a booster dose of vaccine was administered after 3–4 weeks. Mild symptoms such as transient fever, injection site pain etc., associated with vaccination, were aggravated by the second dose. This secondary enhancement of inflammatory response is due to short-term change in macrophages through the concept of trained immunity.Citation60,Citation171
Trained immunity and other diseases
Besides the crucial involvement of trained immunity in cardiovascular and neuroinflammatory illnesses described above, it is also reported to exert adverse consequences in numerous autoinflammatory disorders or allergies.Citation172 Recurring fever episodes and hyper inflammation are reported in patients with hyper-IgD syndrome (HIDS), characterized by Mevalonate Kinase insufficiency.Citation173 The immune cells, such as monocytes, isolated from such individuals have a trained phenotype, as demonstrated by the higher production of several proinflammatory cytokines along with epigenetic and metabolic reprogramming.Citation172 The hyperinflammatory characteristics of other autoinflammatory disorders, including Familial Mediterranean Fever (FMF) and Behçetʻs syndrome, are also probably caused by a similar trained immune phenotype. The participation of trained immune systems in such illnesses must be studied in future investigations.Citation66
BCG immunization protects against TB, provides a short-term immune response against a range of respiratory tract infections, and protects newborns from neonatal sepsis, hence reducing death rates among the young population.Citation12 According to a randomized clinical experiment, BCG vaccination stimulates the genome-wide epigenetic reprogramming of monocytes, leading to protection against yellow fever.
The increase of IL-1β was linked with the generation of trained immunity and reduction of viremia, but not with the specific IFN-γresponse. Genetic, epigenetic, and immunological investigations have all shown the role of IL-1β in the generation of trained immunity. Ultimately, BCG causes epigenetic reprogramming in human monocytes in vivo, which is accompanied by functional programming and resistance toward non-related viral diseases, while IL-1β actively participates as a modulator of trained immune responses.Citation32
According to another phase 1 clinical study, after the challenge of malaria infection, BCG vaccinated volunteers experience more rapid and severe clinical adverse effects and exhibit early expression of the NK cell activation markers. Furthermore, parasitemia is negatively associated with increased phenotypic NK cell and monocyte activation in BCG-vaccinated individuals. According to the pooled results,Citation174 the BCG vaccine changes the symptomatic and immunological response to malaria, which needs to be evaluated in the future with large clinical studies. The anticancer activities of the BCG vaccine, such as those observed in bladder cancer, are associated with training immunity.Citation175 As a result, trained immunity is an adaptive characteristic that improves the fitness of living organisms against harmful microorganisms and several other infections.
The possible significance of trained immunity and epigenetic alterations in food allergy has been identified in recent research. Sensitive children with food allergies exhibited significantly enhanced innate immune responses than healthy children.Citation176–178 Cells of innate immune system, such as monocytes, obtained from children with allergies have shown the changes in the innate immune functioning, exhibiting the trained immune phenotype.Citation179,Citation180 Furthermore, it was proposed that an established balanced condition of trained immunity can be exploited to limit allergic responses.Citation181 An animal model of allergic illness prior to BCG vaccination was found to reduce allergic sensitivity.Citation182 Another research validated this aspect by observing the protection against allergic asthma by herpes viral infection via the innate immune system activation.Citation59
Similarly, the likelihood of IgE-mediated food allergy was reduced with the cellular pertussis vaccine.Citation183 Several clinical trials are being undertaken to test if BCG birth immunization inhibits the emergence of food allergy in countries with less infectious burden.Citation184 Further studies are required to clarify the distinction between the possibility of training immunity to avoid severe allergies as well as the harmful consequences associated with it. Trained immunity is a possible mechanism by which the innate immune cells receive distinct functions from a distinct transcriptional program via long-term epigenetic modifications. In some conditions, few of these alterations might be protective, while others might reflect the poor prognosis of various diseases.Citation66
Future perspectives
In future studies, targeting epigenetic events will be a beneficial strategy to regulate immune response by modifying gene expression in certain autoimmune diseases and immune suppressive conditions. More investigation is needed to understand the interplay between immunological signals and trained immune pathways, as live attenuated vaccines are potent inducers of innate immunity and provide protection during the critical period of unexpected pandemics. Further elucidation of the trained immune pathways will aid in enhancing the strength of trained innate cells and improving the immunization approaches. Moreover, comprehensive studies are essential to understand the specific roles of trained immunity, including strategies to increase the life span of innate immune cells, which may aid in the development of novel diagnostic, therapeutic, and vaccination techniques.
Conclusion
Infection or vaccination initially triggers the innate immune response, which recedes after some time. However, transcriptional events in immune cells induce a stronger activation of the innate host defense against subsequent infections. The evidence from various studies suggests that trained immune memory is an evolving fundamental characteristic of the immune system to defend against foreign agents, including infectious pathogens. Activation of the innate immune cells enhanced the host defense by developing trained immunity. The evolutionary dichotomy of innate and adaptive immunity is the novel concept of trained immunity. The key distinction is that classical immunological memory is antigen-specific and regulated by antibodies and antibody-producing cells and T-cells, while trained immunity is nonspecific and mediated by major shifts in metabolic and transcriptional events in the myeloid and lymphoid cells. Besides that, myeloid (monocytes, macrophages and dendritic cells) and lymphoid (NK cells and innate lymphoid cells) cells have a limited life span. Recent studies suggest that stem cells and other stromal cells may be responsible for long-term immune memory. After primary exposure and subsequent transcriptional events, the typical process of trained immunity leaves certain epigenetic marks on the cells, enabling them to respond more rapidly and robustly to subsequent inflammatory stimuli. Though, some studies have indicated that the adaptive properties of NK cells have long-lasting effects, however trained memory is different from adaptive feature of memory, hence more studies are required to investigate the epigenetic activity of NK cells in relation to trained immunity. Trained immunity is like a double-edged sword. Immunological memory can help to induce a stronger immune response against future threats by upregulating the production of pro-inflammatory cytokines; however, the persistence of these effects can lead to the development of various diseases. In this scenario, immunological tolerance is crucial in preventing such adverse consequences by facilitating a hypoinflammatory state. Furthermore, trained immunity is primarily dependent on the capacity of the cells to undergo certain pathways to produce innate immune memory. Therapeutic approaches can, therefore, target particular sub-populations of cells to either trigger or inhibit such potential responses.
Furthermore, explorative studies could investigate the impact of trained immunity on the modulation of the immune system against various diseases, including cancer, atherosclerosis, cardiovascular events, autoimmune diseases, organ rejection, sepsis, etc., as well as auto-inflammatory diseases using nanobiological antibodies and RNAi technology. TIbVs might be used as immunostimulants against endogenous pathogens as a novel immunotherapeutic approach against noninfectious and infectious immune-related infections. Innate immune cells display PRRs specific to PAMPs and DAMPs that are released by injured cells. The interactions of PRRs with PAMPs or DAMPs stimulate the intracellular signaling pathways. Precise targeting of immune cells or PRR ligands can provide broad protection (specific and nonspecific) against bystander pathogens along with long-term immunity by inducing an adaptive immune response via the release of typical cytokines. Developing promising strategies to target specific cells, receptors, and even pathways mediated by trained immunity may aid in the design of next-generation therapies and vaccines that can induce adaptive and trained immune memory. Moreover, trained innate immune cells may be used as potential candidates to boost various vaccine candidatesʻ efficacy and develop novel therapeutic strategies. Trained immune cells with improved antigen uptake and antigen presentation abilities as well as improved cytokine output, may be used to aid in the re-stimulation of the adaptive immune cells, like B and T cells. Hence, a more robust adaptive immune response can be generated by incorporating the concept of trained immunity in future vaccine candidates.
Author contributions
Conceptualization, S.B., R.M., M.D., K.D., and S.D.; investigation and resources, S.B., R.M., M.D., A.S., P.M., M.D., R.T., M.B., T.B.E., S.M., A.A.R., A.A.M., Z.A.A., S.A., K.D., and S.D.; writing—original draft preparation S.B., R.M., M.D., K.D., and S.D.; writing—review, updation and editing, S.B., A.A.R., S.A., R.M., M.D., M.D., K.D., and S.D.; visualization and supervision, K.D., A.A.R., and S.D.; project administration, K.D., A.A.R., and S.D.; funding acquisition, A.A.R. All authors have read and agreed to the published version of the manuscript.
Acknowledgements
All the authors acknowledge and thank their respective Institutes and Universities.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Bergman P, Raqib R, Rekha RS, Agerberth B, Gudmundsson GH. Host directed therapy against infection by boosting innate immunity. Front Immunol. 2020;11:11. doi:10.3389/fimmu.2020.01209.
- Hewitt RJ, Lloyd CM. Regulation of immune responses by the airway epithelial cell landscape. Nat Rev Immunol. 2021;21(6):347–19. doi:10.1038/s41577-020-00477-9.
- Janeway C, Travers P, Walport M, and Shlomick M. Immunobiology: the immune system in health and disease. 5thed. New York: Garland Science; 2001. p. 10775.
- Mak TW, Saunders ME, and Jett BD. Primer to the immune response. 2nd ed. Burlington (MA): Newnes; 2014. p. 1–674.
- Kleinnijenhuis J, Quintin J, Preijers F, Joosten LA, Ifrim DC, Saeed S, Jacobs C, van Loenhout J, de Jong D, Stunnenberg HG, et al. Bacille Calmette-Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci U S A. 2012;109(43):109. doi:10.1073/pnas.1202870109.
- Bromuro C, Torosantucci A, Chiani P, Conti S, Polonelli L, Cassone A. Interplay between protective and inhibitory antibodies dictates the outcome of experimentally disseminated candidiasis in recipients of a Candida albicans vaccine. Infect Immun. 2002;70(10):5462–70. doi:10.1128/IAI.70.10.5462-5470.2002.
- Netea MG, van der Meer JWM. Trained immunity: an ancient way of remembering. Cell Host Microbe. 2017;21:297–300. doi:10.1016/j.chom.2017.02.003.
- Domínguez-Andrés J, Joosten LA, Netea MG. Induction of innate immune memory: the role of cellular metabolism. Curr Opin Immunol. 2019;56:10–16. doi:10.1016/j.coi.2018.09.001.
- Basak P, Sachdeva N, Dayal D. Can BCG vaccine protect against COVID-19 via trained immunity and tolerogenesis? BioEssays. 2021;43(3):43. doi:10.1002/bies.202000200.
- Mantovani A, Netea MG. Trained innate immunity, epigenetics, and Covid-19. N Engl J Med. 2020;383(11):1078–80. doi:10.1056/NEJMcibr2011679.
- Netea MG, Joosten LAB, Latz E, Mills KHG, Natoli G, Stunnenberg HG, Oʻ-Neill LAJ, Xavier RJ. Trained immunity: a program of innate immune memory in health and disease. Science (80-). 2016;352:427. doi:10.1126/science.aaf1098.
- Netea MG, Domínguez-Andrés J, Barreiro LB, Chavakis T, Divangahi M, Fuchs E, Joosten LAB, van der Meer JWM, Mhlanga MM, Mulder WJM, et al. Defining trained immunity and its role in health and disease. Nat Rev Immunol. 2020;20(6):375–88. doi:10.1038/s41577-020-0285-6.
- Netea MG, Quintin J, Van Der Meer JWM. Trained immunity: a memory for innate host defense. Cell Host Microbe. 2011;9(5):355–61. doi:10.1016/j.chom.2011.04.006.
- Arneth B. Trained innate immunity. Immunol Res. 2021;69(1):69. doi:10.1007/s12026-021-09170-y.
- Hamada A, Torre C, Drancourt M, Ghigo E. Trained immunity carried by non-immune cells. Front Microbiol. 2018;9:10. doi:10.3389/fmicb.2018.03225.
- Dominguez-Andres J, Netea MG. Long-term reprogramming of the innate immune system. J Leukoc Biol. 2019;105(2):329–38. doi:10.1002/JLB.MR0318-104R.
- Oʻ-Neill LAJ, Netea MG. BCG-Induced trained immunity: can it offer protection against COVID-19? Nat Rev Immunol. 2020;20(6):335–37. doi:10.1038/s41577-020-0337-y.
- Mulder WJM, Ochando J, Joosten LAB, Fayad ZA, Netea MG. Therapeutic targeting of trained immunity. Nat Rev Drug Discov. 2019;18(7):553–66. doi:10.1038/s41573-019-0025-4.
- Liu GY, Liu Y, Lu Y, Qin YR, Di GH, Lei YH, Liu HX, Li YQ, Wu C, Hu XW, et al. Short-Term memory of danger signals or environmental stimuli in mesenchymal stem cells: implications for therapeutic potential. Cell Mel Immunol. 2016;13(3):369–78. doi:10.1038/cmi.2015.11.
- Kaufmann E, Sanz J, Dunn JL, Khan N, MendonçMendonçA LE, Pacis A, Tzelepis F, Pernet E, Dumaine A, Grenier JC, et al. BCG educates hematopoietic stem cells to generate protective innate immunity against tuberculosis. Cell. 2018;172(1–2):176–90.e19. doi:10.1016/j.cell.2017.12.031.
- Riksen NP. Trained immunity and atherosclerotic cardiovascular disease. Curr Opin Lipidol. 2019;30(5):395–400. doi:10.1097/MOL.0000000000000628.
- Penkov S, Mitroulis I, Hajishengallis G, Chavakis T. Immunometabolic crosstalk: an ancestral principle of trained immunity? Trends Immunol. 2019;40(1):1–11. doi:10.1016/j.it.2018.11.002.
- Donohoe DR, Bultman SJ. Metaboloepigenetics: interrelationships between energy metabolism and epigenetic control of gene expression. J Cell Physiol. 2012;227(9):3169–77. doi:10.1002/jcp.24054.
- Wilson DF. Regulation of cellular metabolism: programming and maintaining metabolic homeostasis. J pppl Physiol. 2013;115(11):1583–88. doi:10.1152/japplphysiol.00894.2013.
- Arts RJW, Carvalho A, La Rocca C, Palma C, Rodrigues F, Silvestre R, Kleinnijenhuis J, Lachmandas E, Gonçalves LG, Belinha A, et al. Immunometabolic pathways in BCG-induced trained immunity. Cell Rep. 2016;17(10):2562–71. doi:10.1016/j.celrep.2016.11.011.
- Ganeshan K, Chawla A. Metabolic regulation of immune responses. Annu Rev Immunol. 2014;32(1):609–34. doi:10.1146/annurev-immunol-032713-120236.
- Hard GC. Some biochemical aspects of the immune macrophage. Br J Exp Pathol. 1970;51:97–105.
- Riksen NP, Netea MG. Immunometabolic control of trained immunity. Mol Aspects Med. 2021;77:77. doi:10.1016/j.mam.2020.100897.
- Chang PV, Hao L, Offermanns S, Medzhitov R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc Natl Acad Sci U S A. 2014;111(6):2247–52. doi:10.1073/pnas.1322269111.
- Kelly B, Oʻ-Neill LAJ. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res [Internet]. 2015;25(7):771–84. doi:10.1038/cr.2015.68%5Cnpapers3://publication/doi/10.1038/cr.2015.68.
- Incalcaterra S, Dominguez JA. Trained immunity at a glance; A review on the innate immune memory and its potential role in infections, diseases and new therapeutic strategies. Adv J Grad Res. 2020;8(1):68–81. doi:10.21467/ajgr.8.1.68-81.
- Arts RJW, Moorlag SJCFM, Novakovic B, Li Y, Wang SY, Oosting M, Kumar V, Xavier RJ, Wijmenga C, Joosten LAB, et al. BCG vaccination protects against experimental viral infection in humans through the induction of Cytokines associated with trained immunity. Cell Host Microbe. 2018;23(1):89–100.e5. doi:10.1016/j.chom.2017.12.010.
- Acar M, Becskei A, Van Oudenaarden A. Enhancement of cellular memory by reducing stochastic transitions. Nature. 2005;435:228–32. doi:10.1038/nature03524.
- Lambert G, Kussel E. Memory and fitness optimization of bacteria under fluctuating environments. PLoS Genet. 2014;10(9):10. doi:10.1371/journal.pgen.1004556.
- Mutlu A, Trauth S, Ziesack M, Nagler K, Bergeest JP, Rohr K, Becker N, Höfer T, Bischofs IB. Phenotypic memory in Bacillus subtilis links dormancy entry and exit by a spore quantity-quality tradeoff. Nat Commun. 2018;9(1). doi:10.1038/s41467-017-02477-1.
- Liu PS, Wang H, Li X, Chao T, Teav T, Christen S, DI Conza G, Cheng WC, Chou CH, Vavakova M, et al. Α-ketoglutarate orchestrates macrophage activation through metabolic and epigenetic reprogramming. Nat Immunol. 2017;18(9):985–94. doi:10.1038/ni.3796.
- Chen S, Yang J, Wei Y, Wei X. Epigenetic regulation of macrophages: from homeostasis maintenance to host defense. Cell Mol Immunol. 2020;17(1):36–49. doi:10.1038/s41423-019-0315-0.
- Ghisletti S, Barozzi I, Mietton F, Polletti S, De Santa F, Venturini E, Gregory L, Lonie L, Chew A, Wei CL, et al. Identification and characterization of enhancers controlling the inflammatory gene expression program in macrophages. Immunity. 2010;32(3):317–28. doi:10.1016/j.immuni.2010.02.008.
- Keating ST, El-Osta A. Epigenetics and metabolism. Circ Res. 2015;116(4):715–36. doi:10.1161/CIRCRESAHA.116.303936.
- Van Der Heijden CDCC, Noz MP, Joosten LAB, Netea MG, Riksen NP, Keating ST. Epigenetics and trained immunity. Antioxidants Redox Signal. 2018;29(11):1023–40. doi:10.1089/ars.2017.7310.
- Verma D, Parasa VR, Raffetseder J, Martis M, Mehta RB, Netea M, Lerm M. Anti-Mycobacterial activity correlates with altered DNA methylation pattern in immune cells from BCG-vaccinated subjects. Sci Rep. 2017;7(1). doi:10.1038/s41598-017-12110-2.
- Keating ST, Groh L, Thiem K, Bekkering S, Li Y, Matzaraki V, van der Heijden CDCC, van Puffelen JH, Lachmandas E, Jansen T, et al. Rewiring of glucose metabolism defines trained immunity induced by oxidized low-density lipoprotein. J Mol Med (Berl). 2020;98(6):819–31. doi:10.1007/s00109-020-01915-w.
- Divangahi M, Aaby P, Khader SA, Barreiro LB, Bekkering S, Chavakis T, van Crevel R, Curtis N, DiNardo AR, Dominguez-Andres J, et al. Trained immunity, tolerance, priming and differentiation: distinct immunological processes. Nat Immunol. 2021;22(1):2–6. doi:10.1038/s41590-020-00845-6.
- Khan N, Downey J, Sanz J, Kaufmann E, Blankenhaus B, Pacis A, Pernet E, Ahmed E, Cardoso S, Nijnik A, et al. M. tuberculosis reprograms hematopoietic stem cells to limit myelopoiesis and impair trained immunity. Cell. 2020;183(3):752–70.e22. doi:10.1016/j.cell.2020.09.062.
- Patel AA, Zhang Y, Fullerton JN, Boelen L, Rongvaux A, Maini AA, Bigley V, Flavell RA, Gilroy DW, Asquith B, et al. The fate and lifespan of human monocyte subsets in steady state and systemic inflammation. J Exp Med. 2017;214(7):1913–23. doi:10.1084/jem.20170355.
- Takeuchi O, Akira S. Pattern recognition receptors and inflammation. Cell. 2010;140:805–20. doi:10.1016/j.cell.2010.01.022.
- Haley MJ, Brough D, Quintin J, Allan SM. Microglial priming as trained immunity in the brain. Neuroscience. 2019;405:47–54. doi:10.1016/j.neuroscience.2017.12.039.
- Lelios I, Greter M. Trained microglia trigger memory loss. Immunity [Internet]. 2018;48:849–51.
- Wendeln AC, Degenhardt K, Kaurani L, Gertig M, Ulas T, Jain G, Wagner J, Häsler LM, Wild K, Skodras A, et al. Innate immune memory in the brain shapes neurological disease hallmarks. Nature. 2018;556(7701):332–38. doi:10.1038/s41586-018-0023-4.
- Haley SL, Tzvetkov EP, Meuwissen S, Plummer JR, McGettigan JP. Targeting vaccine-induced extrafollicular pathway of B cell differentiation improves rabies postexposure prophylaxis. J Virol. 2017;91(8):91. doi:10.1128/JVI.02435-16.
- Püntener U, Booth SG, Perry VH, Teeling JL. Long-Term impact of systemic bacterial infection on the cerebral vasculature and microglia. J Neuroinflammation. 2012;9(1). doi:10.1186/1742-2094-9-146.
- Ochiel D, Fahey J, Ghosh M, Haddad S, Wira C. Innate immunity in the female reproductive tract: role of sex hormones in regulating uterine epithelial cell protection against pthogens. Curr Womens Health Rev. 2008;4(2):102–17. doi:10.2174/157340408784246395.
- Naik S, Larsen SB, Gomez NC, Alaverdyan K, Sendoel A, Yuan S, Polak L, Kulukian A, Chai S, Fuchs E. Inflammatory memory sensitizes skin epithelial stem cells to tissue damage. Nature. 2017;550:475–80. doi:10.1038/nature24271.
- Owens BMJ, Simmons A. Intestinal stromal cells in mucosal immunity and homeostasis. Mucosal Immunol. 2013;6(2):224–34. doi:10.1038/mi.2012.125.
- Augenlicht LH, Li W, Peregrina K, and Houston M. Abstract 1708: intestinal stem cells are sentinel cells for nutritional exposure. Cancer Res . 2016;76:1708.
- Owens BMJ. Inflammation, innate immunity, and the intestinal stromal cell niche: opportunities and challenges. Front Immunol. 2015;6:319.
- Ordovas-Montanes J, Beyaz S, Rakoff-Nahoum S, Shalek AK. Distribution and storage of inflammatory memory in barrier tissues. Nat Rev Immunol. 2020;20(5):308–20. doi:10.1038/s41577-019-0263-z.
- Mitroulis I, Ruppova K, Wang B, Chen LS, Grzybek M, Grinenko T, Eugster A, Troullinaki M, Palladini A, Kourtzelis I, et al. Modulation of myelopoiesis progenitors is anintegral component of trained immunity. Cell. 2018;172(1–2):147–61.e12. doi:10.1016/j.cell.2017.11.034.
- Machiels B, Dourcy M, Xiao X, Javaux J, Mesnil C, Sabatel C, Desmecht D, Lallemand F, Martinive P, Hammad H, et al. A gammaherpesvirus provides protection against allergic asthma by inducing the replacement of resident alveolar macrophages with regulatory monocytes. Nat Immunol. 2017;18(12):1310–20. doi:10.1038/ni.3857 .
- Yao Y, Jeyanathan M, Haddadi S, Barra NG, Vaseghi-Shanjani M, Damjanovic D, Lai R, Afkhami S, Chen Y, Dvorkin-Gheva A, et al. Induction of autonomous memory alveolar macrophages requires T cell help and is critical to trained immunity. Cell. 2018;175(6):1634–50.e17. doi:10.1016/j.cell.2018.09.042.
- Cirovic B, de Bree LCJ, Groh L, Blok BA, Chan J, van der Velden WJFM, Bremmers MEJ, van Crevel R, Händler K, Picelli S, et al. BCG vaccination in humans elicits trained immunity via the hematopoietic progenitor compartment. Cell Host Microbe. 2020;28(2):322–34.e5. doi:10.1016/j.chom.2020.05.014.
- Quintin J, Saeed S, Martens JHA, Giamarellos-Bourboulis EJ, Ifrim DC, Logie C, Jacobs L, Jansen T, Kullberg BJ, Wijmenga C, et al. Candida albicans infection affords protection against reinfection via functional reprogramming of monocytes. Cell Host Microbe. 2012;12(2):223–32. doi:10.1016/j.chom.2012.06.006.
- Ifrim DC, Quintin J, Joosten LAB, Jacobs C, Jansen T, Jacobs L, Gow NAR, Williams DL, Van Der Meer JWM, Netea MG. Trained immunity or tolerance: opposing functional programs induced in human monocytes after engagement of various pattern recognition receptors. Clin Vaccine Immunol. 2014;21(4):534–45. doi:10.1128/CVI.00688-13.
- Dobrovolskaia MA, Vogel SN. Toll receptors, CD14, and macrophage activation and deactivation by LPS. Microbes Infect. 2002;4(9):903–14. doi:10.1016/S1286-4579(02)01613-1.
- Bekkering S, Blok BA, Joosten LAB, Riksen NP, Van Crevel R, Netea MG. In vitro experimental model of trained innate immunity in human primary monocytes. Clin Vaccine Immunol. 2016;23(12):926–33. doi:10.1128/CVI.00349-16.
- Bekkering S, Domínguez-Andrés J, Joosten LAB, Riksen NP, Netea MG. Trained immunity: reprogramming innate immunity in health and disease. Annu Rev Immunol. 2021;39:39. doi:10.1146/annurev-immunol-102119-073855.
- Christ A, Latz E. The Western lifestyle has lasting effects on metaflammation. Nat Rev Immunol. 2019;19(5):267–68. doi:10.1038/s41577-019-0156-1.
- Bekkering S, Quintin J, Joosten LAB, van der Meer JWM, Netea MG, Riksen NP. Trained innate immunity and atherosclerosis. Clin Ther. 2014;36(8):e3. doi:10.1016/j.clinthera.2014.05.016.
- Bekkering S, Saner C, Riksen NP, Netea MG, Sabin MA, Saffery R, Stienstra R, Burgner DP. Trained immunity: linking obesity and cardiovascular disease across the life-course? Trends Endocrinol Metab. 2020;31(5):378–89. doi:10.1016/j.tem.2020.01.008.
- Thiem K, Stienstra R, Riksen NP, Keating ST. Trained immunity and diabetic vascular disease. Clin Sci (Lond). 2019;133(2):195–203. doi:10.1042/CS20180905.
- Bekkering S, Quintin J, Joosten LAB, Van Der Meer JWM, Netea MG, Riksen NP. Oxidized low-density lipoprotein induces long-term proinflammatory cytokine production and foam cell formation via epigenetic reprogramming of monocytes. Arterioscler Thromb Vasc Biol. 2014;34(8):1731–38. doi:10.1161/ATVBAHA.114.303887.
- Van Der Valk FM, Bekkering S, Kroon J, Yeang C, Van Den Bossche J, Van Buul JD, Ravandi A, Nederveen AJ, Verberne HJ, Scipione C, et al. Oxidized phospholipids on lipoprotein(a) elicit arterial wall inflammation and an inflammatory monocyte response in humans. Circulation. 2016;134:611–24. doi:10.1161/CIRCULATIONAHA.116.020838.
- Van Der Heijden CDCC, Groh L, Keating ST, Kaffa C, Noz MP, Kersten S, Van Herwaarden AE, Hoischen A, Joosten LAB, Timmers HJLM, et al. Catecholamines induce trained immunity in monocytes in vitro and in vivo. Circ Res. 2020;127(2):269–83. doi:10.1161/CIRCRESAHA.119.315800.
- Van Der Heijden CDCC, Keating ST, Groh L, Joosten LAB, Netea MG, Riksen NP. Aldosterone induces trained immunity: the role of fatty acid synthesis. Cardiovasc Res. 2020;116(2):317–28. doi:10.1093/cvr/cvz137.
- Christ A, Bekkering S, Latz E, Riksen NP. Long-Term activation of the innate immune system in atherosclerosis. Semin Immunol. 2016;28(4):384–93. doi:10.1016/j.smim.2016.04.004.
- Leentjens J, Bekkering S, Joosten LAB, Netea MG, Burgner DP, Riksen NP. Trained innate immunity as a novel mechanism linking infection and the development of atherosclerosis. Circ Res. 2018;122(5):664–69. doi:10.1161/CIRCRESAHA.117.312465.
- Van Tuijl J, Joosten LAB, Netea MG, Bekkering S, Riksen NP. Immunometabolism orchestrates training of innate immunity in atherosclerosis. Cardiovasc Res. 2019;115(9):1416–24. doi:10.1093/cvr/cvz107.
- Zhong C, Yang X, Feng Y, and Yu J. Trained immunity: an underlying driver of inflammatory atherosclerosis. Front Immunol. 2020;11:284.
- Netea MG, Latz E, Mills KHG, Oʻ-Neill LAJ. Innate immune memory: A paradigm shift in understanding host defense. Nat Immunol. 2015;16(7):675–79. doi:10.1038/ni.3178.
- Braza MS, van Leent MMT, Lameijer M, Sanchez-Gaytan BL, Arts RJW, Pérez-Medina C, Conde P, Garcia MR, Gonzalez-Perez M, Brahmachary M, et al. Inhibiting inflammation with myeloid cell-specific nanobiologics promotes organ transplant acceptance. Immunity. 2018;49(5):819–28.e6. doi:10.1016/j.immuni.2018.09.008.
- Wang X, Antony V, Wang Y, Wu G, Liang G. Pattern recognition receptor-mediated inflammation in diabetic vascular complications. Med Res Rev. 2020;40(6):2466–84. doi:10.1002/med.21711.
- Ochando J, Fayad ZA, Madsen JC, Netea MG, Mulder WJM. Trained immunity in organ transplantation. Am J Transplant. 2020;20(1):10–18. doi:10.1111/ajt.15620.
- Wentowski C, Mewada N, Nielsen ND. Sepsis in 2018: a review. Anaesth Intensive Care Med. 2019;20(1):6–13. doi:10.1016/j.mpaic.2018.11.009.
- Ransohoff RM. How neuroinflammation contributes to neurodegeneration. Science (80-). 2016;353(6301):777–83. doi:10.1126/science.aag2590.
- Datta M, Staszewski O, Raschi E, Frosch M, Hagemeyer N, Tay TL, Blank T, Kreutzfeldt M, Merkler D, Ziegler-Waldkirch S, et al. Histone deacetylases 1 and 2 regulate microglia function during development, homeostasis, and neurodegeneration in a context-dependent manner. Immunity. 2018;48(3):514–29.e6. doi:10.1016/j.immuni.2018.02.016.
- Williamson LL, Sholar PW, Mistry RS, Smith SH, Bilbo SD. Microglia and memory: modulation by early-life infection. J Neurosci. 2011;31(43):15511–21. doi:10.1523/JNEUROSCI.3688-11.2011.
- Perry VH, Holmes C. Microglial priming in neurodegenerative disease. Nat Rev Neurol. 2014;10(4):217–24. doi:10.1038/nrneurol.2014.38.
- Knuesel I, Chicha L, Britschgi M, Schobel SA, Bodmer M, Hellings JA, Toovey S, Prinssen EP. Maternal immune activation and abnormal brain development across CNS disorders. Nat Rev Neurol. 2014;10(11):643–60. doi:10.1038/nrneurol.2014.187.
- Noz MP, Ter Telgte A, Wiegertjes K, Joosten LAB, Netea MG, De Leeuw FE, Riksen NP. Trained immunity characteristics are associated with progressive cerebral small vessel disease. Stroke. 2018;49(12):2910–17. doi:10.1161/STROKEAHA.118.023192.
- Kim J, Choi SE, Lim BJ, Kim YS, Huh KH, Lee J, Kim SI, Kim MS, Jeong HJ. Clinical significance of macrophage polarization in antibody-mediated rejection of renal allograft. Transplant Proc. 2018;50(4):1005–08. doi:10.1016/j.transproceed.2018.02.037.
- Zhao Y, Chen S, Lan P, Wu C, Dou Y, Xiao X, Zhang Z, Minze L, He X, Chen W, et al. Macrophage subpopulations and their impact on chronic allograft rejection versus graft acceptance in a mouse heart transplant model. Am J Transplant. 2018;18(3):604–16. doi:10.1111/ajt.14543.
- Ordikhani F, Pothula V, Sanchez-Tarjuelo R, Jordan S, Ochando J. Macrophages in organ transplantation. Front Immunol. 2020;11:11. doi:10.3389/fimmu.2020.582939.
- Brown GD, Herre J, Williams DL, Willment JA, Marshall ASJ, Gordon S. Dectin-1 mediates the biological effects of β-glucans. J Exp Med. 2003;197(9):1119–24. doi:10.1084/jem.20021890.
- Girardin SE, Boneca IG, Viala J, Chamaillard M, Labigne A, Thomas G, Philpott DJ, Sansonetti PJ. Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J Biol Chem. 2003;278(11):8869–72. doi:10.1074/jbc.C200651200.
- Lührs H, Gerke T, Müller JG, Melcher R, Schauber J, Boxberger F, Scheppach W, Menzel T. Butyrate inhibits NF-κB activation in lamina propria macrophages of patients with ulcerative colitis. Scand J Gastroenterol. 2002;37(4):458–66. doi:10.1080/003655202317316105.
- Duewell P, Kono H, Rayner KJ, Sirois CM, Vladimer G, Bauernfeind FG, Abela GS, Franchi L, Nũez G, Schnurr M, et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature. 2010;464(7293):1357–61. doi:10.1038/nature08938.
- Bosmans JL, Holvoet P, Dauwe SEH, Ysebaert DK, Chapelle T, Jürgens A, Kovacic V, Van Marck EA, De Broe ME, Verpooten GA. Oxidative modification of low-density lipoproteins and the outcome of renal allografts at 1 1/2 years. Kidney Int. 2001;59(6):2346–56. doi:10.1046/j.1523-1755.2001.00752.x.
- Sheedy FJ, Grebe A, Rayner KJ, Kalantari P, Ramkhelawon B, Carpenter SB, Becker CE, Ediriweera HN, Mullick AE, Golenbock DT, et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat Immunol. 2013;14(8):812–20. doi:10.1038/ni.2639.
- Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–31. doi:10.1056/NEJMoa1707914.
- Chakraborty C, Sharma AR, Sharma G, Doss CGP, Lee SS. Therapeutic miRNA and siRNA: moving from bench to clinic as next generation medicine. Mol Ther Nucleic Acids. 2017;8:132–43. doi:10.1016/j.omtn.2017.06.005.
- Dvir T, Timko BP, Kohane DS, and Langer R. Nanotechnological strategies for engineering complex tissues. Nat. Nanotechnol. 2020;6(1):13–22.
- Netea MG, Schlitzer A, Placek K, Joosten LAB, Schultze JL. Innate and adaptive immune memory: an evolutionary continuum in the hostʻs response to pathogens. Cell Host Microbe. 2019;25(1):13–26. doi:10.1016/j.chom.2018.12.006.
- Tough DF, Lewis HD, Rioja I, Lindon MJ, Prinjha RK. Epigenetic pathway targets for the treatment of disease: accelerating progress in the development of pharmacological tools: IUPHAR Review 11. Br J Pharmacol. 2014;171(22):4981–5010. doi:10.1111/bph.12848.
- Grabiec AM, Krausz S, de Jager W, Burakowski T, Groot D, Sanders ME, Prakken BJ, Maslinski W, Eldering E, Tak PP, et al. Histone deacetylase inhibitors suppress inflammatory activation of rheumatoid arthritis patient synovial macrophages and tissue. J Immunol. 2010;184(5):2718–28. doi:10.4049/jimmunol.0901467.
- Municio C, Criado G. Therapies targeting trained immune cells in inflammatory and autoimmune diseases. Front Immunol. 2021;11:11. doi:10.3389/fimmu.2020.631743.
- Charlotte L, Mourits VP, Koeken VACM, Moorlag SJCFM, Janssen R, Folkman L, Barreca D, Krausgruber T, Fife-Gernedl V, Novakovic B, et al. Circadian rhythm influences induction of trained immunity by BCG vaccination. J Clin Invest. 2020;130(10):5603–17. doi:10.1172/JCI133934.
- Parihar A, Eubank TD, Doseff AI. Monocytes and macrophages regulate immunity through dynamic networks of survival and cell death. J Innate Immun. 2010;2(3):204–15. doi:10.1159/000296507.
- Chilvers ER, Cadwallader KA, Reed BJ, White JF, Condliffe AM. The function and fate of neutrophils at the inflamed site: prospects for therapeutic intervention. J R Coll Physicians Lond. 2000;34:68–74.
- Mackaness GB. The influence of immunologically committed lymphoid cells on macrophage activity in vivo. J Exp Med. 1969;129(5):973–92. doi:10.1084/jem.129.5.973.
- Chumakov K, Avidan MS, Benn CS, Bertozzi SM, Blatt L, Chang AY, Jamison DT, Khader SA, Kottilil S, Netea MG, et al. Old vaccines for new infections: exploiting innate immunity to control COVID-19 and prevent future pandemics. Proc Natl Acad Sci U S A. 2021;118(21):118. doi:10.1073/pnas.2101718118.
- Sánchez-Ramón S, Conejero L, Netea MG, Sancho D, Palomares Ó, Subiza JL. Trained immunity-based vaccines: a new paradigm for the development of broad-spectrum anti-infectious formulations. Front Immunol. 2018;9:2936. doi:10.3389/fimmu.2018.02936.
- Cirauqui C, Benito-Villalvilla C, Sánchez-Ramón S, Sirvent S, Diez-Rivero CM, Conejero L, Brandi P, Hernández-Cillero L, Ochoa JL, Pérez-Villamil B, et al. Human dendritic cells activated with MV130 induce Th1, Th17 and IL-10 responses via RIPK2 and MyD88 signalling pathways. Eur J Immunol. 2018;48(1):180–93. doi:10.1002/eji.201747024.
- Zanoni I, Tan Y, Di Gioia M, Broggi A, Ruan J, Shi J, Donado CA, Shao F, Wu H, Springstead JR, et al. An endogenous caspase-11 ligand elicits interleukin-1 release from living dendritic cells. Science (80). 2016;352:1232–36. doi:10.1126/science.aaf3036.
- Kleinnijenhuis J, Quintin J, Preijers F, Benn CS, Joosten LAB, Jacobs C, Van Loenhout J, Xavier RJ, Aaby P, Van Der Meer JWM, et al. Long-Lasting effects of bcg vaccination on both heterologous th1/th17 responses and innate trained immunity. J Innate Immun. 2014;6(2):152–58. doi:10.1159/000355628.
- Verrall AJ, Schneider M, Alisjahbana B, Apriani L, van Laarhoven A, Koeken VACM, van Dorp S, Diadani E, Utama F, Hannaway RF, et al. Early clearance of mycobacterium tuberculosis is associated with increased innate immune responses. J Infect Dis. 2020;221(8):1342–50. doi:10.1093/infdis/jiz147.
- Kleinnijenhuis J, Van Crevel R, Netea MG. Trained immunity: consequences for the heterologous effects of BCG vaccination. Trans R Soc Trop Med Hyg. 2014;109(1):29–35. doi:10.1093/trstmh/tru168.
- Covián C, Fernández-Fierro A, Retamal-Díaz A, Díaz FE, Vasquez AE, Lay MK, Riedcl CA, González PA, Bueno SM, Kalergis AM. BCG-Induced cross-protection and development of trained immunity: implication for vaccine design. Front Immunol. 2019;10:10. doi:10.3389/fimmu.2019.02806.
- Benito-Villalvilla C, Cirauqui C, Diez-Rivero CM, Casanovas M, Subiza JL, Palomares O. MV140, a sublingual polyvalent bacterial preparation to treat recurrent urinary tract infections, licenses human dendritic cells for generating Th1, Th17, and IL-10 responses via Syk and MyD88. Mucosal Immunol. 2017;10(4):924–35. doi:10.1038/mi.2016.112.
- Lorenzo-Gómez MF, Padilla-Fernández B, GarcíGarcíA-Cenador MB, Virseda-Rodríguez ÁJ, Martín-GarcíGarcíA I, Sánchez-Escudero A, Vicente-Arroyo MJ, Mirón-Canelo JA. Comparison of sublingual therapeutic vaccine with antibiotics for the prophylaxis of recurrent urinary tract infections. Front Cell Infect Microbiol. 2015;5:5. doi:10.3389/fcimb.2015.00050.
- Jensen ML, Dave S, van der Loeff MS, da Costa C, Vincent T, Leligdowicz A, Benn CS, Roth A, Ravn H, Lisse IM, et al. Vaccinia scars associated with improved survival among adults in rural Guinea-Bissau. PLoS One. 2006:1. doi:10.1371/journal.pone.0000101.
- Gillard GO, Bivas-Benita M, Hovav AH, Grandpre LE, Panas MW, Seaman MS, Haynes BF, Letvin NL. Correction: Thy1 + Nk cells from vaccinia virus-primed mice confer protection against vaccinia virus challenge in the absence of adaptive lymphocytes. PLoS Pathog. 2011;7(8). doi:10.1371/annotation/b29086ef-e08d-444c-8113-18a6dd429a7c.
- Piedra PA, Gaglani MJ, Kozinetz CA, Herschler GB, Fewlass C, Harvey D, Zimmerman N, Glezen WP. Trivalent live attenuated intranasal influenza vaccine administered during the 2003-2004 influenza type A (H3N2) outbreak provided immediate, direct, and indirect protection in children. Pediatrics. 2007;120(3):120. doi:10.1542/peds.2006-2836.
- Lee YJ, Lee JY, Jang YH, Seo SU, Chang J, and Seong BL. Non-Specific effect of vaccines: immediate protection against respiratory syncytial virus infection by a live attenuated influenza vaccine. Front Microbiol. 2018;9:83.
- Pasquali C, Salami O, Taneja M, Gollwitzer ES, Trompette A, Pattaroni C, Yadava K, Bauer J, Marsland BJ. Enhanced mucosal antibody production and protection against respiratory infections following an orally administered bacterial extract. Front Med. 2014;1:1. doi:10.3389/fmed.2014.00041.
- Roth M, Pasquali C, Stolz D, and Tamm M. Broncho Vaxom (OM-85) modulates rhinovirus docking proteins on human airway epithelial cells via Erk1/2 mitogen activated protein kinase and cAMP. PLoS One. 2017;12:e0188010.
- Tarancón R, Domínguez-Andrés J, Uranga S, Ferreira AV., Groh LA, Domenech M, González-Camacho F, Riksen NP, Aguilo N, Yuste J, et al. New live attenuated tuberculosis vaccine MTBVAC induces trained immunity and confers protection against experimental lethal pneumonia. PLoS Pathog. 2020;16(4):e1008404. doi:10.1371/journal.ppat.1008404.
- Libraty DH, Zhang L, Woda M, Acosta LP, Obcena A, Brion JD, Capeding RZ. Neonatal BCG vaccination is associated with enhanced T-helper 1 immune responses to heterologous infant vaccines. Trials Vaccinol. 2014;3:1–5. doi:10.1016/j.trivac.2013.11.004.
- Goodridge HS, Ahmed SS, Curtis N, Kollmann TR, Levy O, Netea MG, Pollard AJ, Van Crevel R, Wilson CB. Harnessing the beneficial heterologous effects of vaccination. Nat Rev Immunol. 2016;16(6):392–400. doi:10.1038/nri.2016.43.
- VANʻ-T Wout JW, Poell R, VAN Furth R. The role of BCG/PPD‐activated macrophages in resistance against systemic candidiasis in mice. Scand J Immunol. 1992;36(5):713–20. doi:10.1111/j.1365-3083.1992.tb03132.x.
- Buffen K, Oosting M, Quintin J, Ng A, Kleinnijenhuis J, Kumar V, van de Vosse E, Wijmenga C, van Crevel R, Oosterwijk E, et al. Autophagy controls BCG-induced trained immunity and the response to intravesical BCG therapy for bladder cancer. PLoS Pathog. 2014;10(10):10. doi:10.1371/journal.ppat.1004485.
- Saadatian-Elahi M, Aaby P, Shann F, Netea MG, Levy O, Louis J, Picot V, Greenberg M, Warren W. Heterologous vaccine effects. Vaccine. 2016;34:3923–30. doi:10.1016/j.vaccine.2016.06.020.
- Pennington SH, Thompson AL, Wright AKA, Ferreira DM, Jambo KC, Wright AD, Faragher B, Gilmour JW, Gordon SB, Gordon MA. Oral typhoid vaccination with live-attenuated Salmonella Typhi Strain Ty21a generates Ty21a-responsive and heterologous influenza virus–responsive CD4 + and CD8 + T cells at the huian intestinal mucosa. J Infect Dis. 2016;213(11):1809–19. doi:10.1093/infdis/jiw030.
- Di Luzio NR, Williams DL. Protective effect of glucan against systemic Staphylococcus aureus septicemia in normal and leukemic mice. Infect Immun. 1978;20(3):804–10. doi:10.1128/iai.20.3.804-810.1978.
- Krahenbuhl JL, Sharma SD, Ferraresi RW, Remington JS. Effects of muramyl dipeptide treatment on resistance to infection with Toxoplasma gondii in mice. Infect Immun. 1981;31(2):716–22. doi:10.1128/iai.31.2.716-722.1981.
- Ribes S, Meister T, Ott M, Redlich S, Janova H, Hanisch UK, Nessler S, and Nau R. Intraperitoneal prophylaxis with CpG oligodeoxynucleotides protects neutropenic mice against intracerebral Escherichia coli K1 infection. J Neuroinflammation. 2014;11(1):1–11.
- Khader SA, Divangahi M, Hanekom W, Hill PC, Maeurer M, Makar KW, Mayer-Barder KD, Mhlanga MM, Nemes E, Schlesinger LS, et al. Targeting innate immunity for tuberculosis vaccination. J Clin Invest. 2019;129(9):3482–91. doi:10.1172/JCI128877.
- Dhama K, Khan S, Tiwari R, Sircar S, Bhat S, Malik YS, Singh KP, Chaicumpa W, Bonilla-Aldana D-R-M. Coronavirus disease 2019–COVID-19. Clin Microbiol Rev. 2020;33(4): e00028-20. doi:10.1128/CMR.00028-20.
- WHO. WHO COVID-19 dashboard - up to date data on pandemic. WHO Heal Emerg Dasboard [Internet]. 2021. https://covid19.who.int/.
- Rabaan AA, Al-Ahmed SH, Sah R, Tiwari R, Yatoo MI, Patel SK, Pathak M, Malik YS, Dhama K, and Singh KP, et al. SARS-CoV-2/COVID-19 and advances in developing potential therapeutics and vaccines to counter this emerging pandemic. Ann Clin Microbiol Antimicrob. 2020;19(1):1–37.
- Chugh H, Awasthi A, Agarwal Y, Gaur RK, Dhawan G, Chandra R. A comprehensive review on potential therapeutics interventions for COVID-19. Eur J Pharmacol. 2021;890:890. doi:10.1016/j.ejphar.2020.173741.
- Izda V, Jeffries MA, Sawalha AH. COVID-19: a review of therapeutic strategies and vaccine candidates. Clin Immunol. 2021;222:222. doi:10.1016/j.clim.2020.108634.
- Iqbal Yatoo M, Hamid Z, Rather I, Nazir QUA, Bhat RA, Ul Haq A, Magray SN, Haq Z, Sah R, Tiwari R, et al. Immunotherapies and immunomodulatory approaches in clinical trials - a mini review. Hum Vaccines Immunother. 2021;17(7):1897–909. doi:10.1080/21645515.2020.1871295.
- Hyder Pottoo F, Abu-Izneid T, Mohammad Ibrahim A, Noushad Javed M, AlHajri N, Hamrouni AM. Immune system response during viral infections: immunomodulators, Cytokine Storm (CS) and immunotherapeutics in COVID-19. Saudi Pharm J. 2021;29(2):173–87. doi:10.1016/j.jsps.2020.12.018.
- Liu J, Liu Y, Xiang P, Pu L, Xiong H, Li C, Zhang M, Tan J, Xu Y, Song R, et al. Neutrophil-To-Lymphocyte ratio predicts severe illness patients with 2019 novel coronavirus in the early stage. medRxiv 2020.
- Malik YS, Ansari MI, Ganesh B, Sircar S, Bhat S, Pande T, Vinodhkumar OR, Kumar P, Iqbal Yatoo M, Tiwari R, et al. BCG vaccine: a hope to control COVID-19 pandemic amid crisis. Hum Vaccines Immunother. 2020;16(12):2954–62. doi:10.1080/21645515.2020.1818522.
- Covián C, Retamal-Díaz A, Bueno SM, and Kalergis AM. Could BCG vaccination induce protective trained immunity for SARS-CoV-2? Front Immunol. 2020;11:970.
- Nemes E, Geldenhuys H, Rozot V, Rutkowski KT, Ratangee F, Bilek N, Mabwe S, Makhethe L, Erasmus M, Toefy A, et al. Prevention of M. tuberculosis infection with H4:IC31 vaccine or BCG revaccination. N Engl J Med. 2018;379(2):138–49. doi:10.1056/NEJMoa1714021.
- Salem A, Nofal A, Hosny D. Treatment of common and plane warts in children with topical viable bacillus calmette-guerin. Pediatr Dermatol. 2013;30(1):60–63. doi:10.1111/j.1525-1470.2012.01848.x.
- Mukherjee S, Subramaniam R, Chen H, Smith A, Keshava S, and Shams H. Boosting efferocytosis in alveolar space using BCG vaccine to protect host against influenza pneumonia. PLoS One. 2017;12(7):e0180143.
- Giamarellos-Bourboulis EJ, Tsilika M, Moorlag S, Antonakos N, Kotsaki A, Domínguez-Andrés J, Kyriazopoulou E, Gkavogianni T, Adami ME, Damoraki G, et al. Activate: randomized clinical trial of BCG vaccination against infection in the elderly. Cell. 2020;183(2):315–23.e9. doi:10.1016/j.cell.2020.08.051.
- Moorlag SJCFM, van Deuren RC, van Werkhoven CH, Jaeger M, Debisarun P, Taks E, Mourits VP, Koeken VACM, de Bree LCJ, and ten Doesschate T, et al. Safety and COVID-19 symptoms in individuals recently vaccinated with BCG: a retrospective Cohort study. Cell Reports Med. 2020;1(5):100073.
- Dos Santos JC, Quixabeira VBL, Silva MVT, Damen MSMA, Schraa K, Jaeger M, Oosting M, Keating ST, Dorta ML, and Pinto SA, et al. Genetic variation in interleukin-32 influence the immune response against new world leishmania species and susceptibility to american tegumentary leishmaniasis. PLoS Negl Trop Dis. 2020;14(2):e0008029.
- Bagheri N, Montazeri H. On BCG vaccine protection from COVID-19: a review. SN Compr Clin Med. 2021;3(6):1261–71. doi:10.1007/s42399-021-00835-1.
- Schultze JL, Aschenbrenner AC. COVID-19 and the human innate immune system. Cell. 2021;184(7):1671–92. doi:10.1016/j.cell.2021.02.029.
- Netea MG, Giamarellos-Bourboulis EJ, Domínguez-Andrés J, Curtis N, van Crevel R, van de Veerdonk FL, Bonten M. Trained immunity: a tool for reducing susceptibility to and the severity of SARS-CoV-2 infection. Cell. 2020;181:969–77. doi:10.1016/j.cell.2020.04.042.
- de Chaisemartin C, de Chaisemartin L BCG vaccination in infancy does not protect against COVID‐19. Evidence from a natural experiment in Sweden. arXiv 2020.
- Escobar LE, Molina-Cruz A, Barillas-Mury C. BCG vaccine protection from severe coronavirus disease 2019 (COVID-19). Proc Natl Acad Sci U S A. 2020;117(30):17720–26. doi:10.1073/pnas.2008410117.
- Hamiel U, Kozer E, Youngster I. SARS-CoV-2 rates in BCG-raccinated and unvaccinated young adults. JAMA - J Am Med Assoc. 2020;323:2340–41. doi:10.1001/jama.2020.8189.
- Kinoshita M, Tanaka M. Impact of routine infant BCG vaccination on COVID-19. J Infect. 2020;81(4):625–33. doi:10.1016/j.jinf.2020.08.013.
- Arlehamn CSL, Sette A, Peters B. Lack of evidence for BCG vaccine protection from severe COVID-19. Proc Natl Acad Sci U S A. 2020;117(41):25203–04. doi:10.1073/pnas.2016733117.
- Singh S, Maurya RP, Singh RK. “Trained immunity” from Mycobacterium spp. exposure or BCG vaccination and COVID-19 outcomes. PLoS Pathog. 2020;16(10):e1008969. doi:10.1371/journal.ppat.1008969.
- Nowill AE, de Campos-Lima PO. Immune response resetting as a novel strategy to overcome SARS-CoV-2–induced Cytokine Storm. J Immunol. 2020;205(10):2566–75. doi:10.4049/jimmunol.2000892.
- Tomita Y, Sato R, Ikeda T, Sakagami T. BCG vaccine may generate cross-reactive T cells against SARS-CoV-2: in silico analyses and a hypothesis. Vaccine. 2020;38:6352–56. doi:10.1016/j.vaccine.2020.08.045.
- Myśliwska J, Trzonkowski P, Szmit E, Brydak LB, MachałMachałA M, Myśliwski A. Immunomodulating effect of influenza vaccination in the elderly differing in health status. Exp Gerontol. 2004;39(10):1447–58. doi:10.1016/j.exger.2004.08.005.
- Young A, Neumann B, Mendez RF, Reyahi A, Joannides A, Modis Y, Franklin RJM Homologous protein domains in SARS-CoV-2 and measles, mumps and rubella viruses: preliminary evidence that MMR vaccine might provide protection against COVID-19. medRxiv 2020.
- Hanker VS. Measles immunization: worth considering containment strategy for SARS-CoV-2 global outbreak. Indian Pediatr. 2020;57(4):380. doi:10.1007/s13312-020-1804-z.
- Sinaei R, Pezeshki S, Parvaresh S, Sinaei R. Why COVID-19 is less frequent and severe in children: a narrative review. World J Pediatr. 2021;17(1):10–20. doi:10.1007/s12519-020-00392-y.
- Valentini P, Sodero G, Buonsenso D. The relationship between COVID-19 and innate immunity in children: a review. Children. 2021;8:266. doi:10.3390/children8040266.
- Rivas MN, Ebinger JE, Wu M, Sun N, Braun J, Sobhani K, van Eyk JE, Cheng S, and Arditi M. BCG vaccination history associates with decreased SARS-CoV-2 seroprevalence across a diverse cohort of health care workers. J Clin Invest. 2021;2:131.
- Fu W, Ho PC, Liu CL, Tzeng KT, Nayeem N, Moore JS, Wang LS, and Chou SY. Reconcile the debate over protective effects of BCG vaccine against COVID-19. Sci Rep. 2021;11(1):1–9.
- Teijaro JR, Farber DL. COVID-19 vaccines: modes of immune activation and future challenges. Nat Rev Immunol. 2021;21(4):195–97. doi:10.1038/s41577-021-00526-x.
- Arts RJW, Joosten LAB, Netea MG. The potential role of trained immunity in autoimmune and autoinflammatory disorders. Front Immunol. 2018;9. doi:10.3389/fimmu.2018.00298.
- Van Der Meer JWM, Radl J, Meyer CJLM, Vossen JM, Van Nieuwkoop JA, Lobatto S, Van Furth R. Hyperimmunoglobulinaemia D and periodic fever: a new syndrome. Lancet. 1984;323(8386):1087–90. doi:10.1016/S0140-6736(84)92505-4.
- Walk J, de Bree LCJ, Graumans W, Stoter R, van Gemert GJ, van de Vegte-Bolmer M, Teelen K, Hermsen CC, Arts RJW, Behet MC, et al. Outcomes of controlled human malaria infection after BCG vaccination. Nat Commun. 2019;10(1):10. doi:10.1038/s41467-019-08659-3.
- Redelman-Sidi G, Glickman MS, Bochner BH. The mechanism of action of BCG therapy for bladder cancer—a current perspective. Nat Rev Urol. 2014;11(3):153–62. doi:10.1038/nrurol.2014.15.
- Tulic MK, Hodder M, Forsberg A, McCarthy S, Richman T, Dvaz N, Van Den Biggelaar AHJ, Thornton CA, and Prescott SL. Differences in innate immune function between allergic and nonallergic children: new insights into immune ontogeny. J Allergy Clin Immunol. 2011;127(2):470–478.
- Neeland MR, Koplin JJ, Dang TD, Dharmage SC, Tang ML, Prescott SL, Saffery R, Martino DJ, Allen KJ. Early life innate immune signatures of persistent food allergy. J Allergy Clin Immunol. 2018;142(3):857–64.e3. doi:10.1016/j.jaci.2017.10.024.
- Imran S, Neeland MR, Shepherd R, Messina N, Perrett KP, Netea MG, Curtis N, Saffery R, Novakovic B. A potential role for epigenetically mediated trained immunity in food allergy. iScience. 2020;23(6):23. doi:10.1016/j.isci.2020.101171.
- Zhang Y, Collier F, Naselli G, Saffery R, Tang ML, Allen KJ, Ponsonby AL, Harrison LC, Vuillermin P, Ranganathan S, et al. Cord blood monocyte–derived inflammatory cytokines suppress IL-2 and induce nonclassic “T H 2-type” immunity associated with development of food allergy. Sci Transl Med. 2016;8(321). doi:10.1126/scitranslmed.aad4322.
- Neeland MR, Andorf S, Manohar M, Dunham D, Lyu SC, Dang TD, Peters RL, Perrett KP, Tang MLK, and Saffery R, et al. Mass cytometry reveals cellular fingerprint associated with IgE+ peanut tolerance and allergy in early life. Nat Commun. 2020;11(1):1–10.
- Arnoldussen DL, Linehan M, and Sheikh A. BCG vaccination and allergy: a systematic review and meta-analysis. J Allergy Clin Immunol. 2011;127(1):246–253.
- Herz U, Gerhold K, Grüber C, Braun A, Wahn U, Renz H, and Paul K. BCG infection suppresses allergic sensitization and development of increased airway reactivity in an animal model. J Allergy Clin Immunol. 1998;102(5):867–74. doi:10.1016/S0091-6749(98)70030-2.
- Estcourt MJ, Campbell DE, Gold MS, Richmond P, Allen KJ, Quinn HE, Marsh JA, Peters RL, Valerio C, Dai D, et al. Whole-cell pertussis vaccination and decreased rsk of IgE-mediated food allergy: a nested case-control study. J Allergy Clin Immunol Pract. 2020;8(6):2004–14. doi:10.1016/j.jaip.2019.12.020.
- Messina NL, Gardiner K, Donath S, Flanagan K, Ponsonby AL, Shann F, Robins-Browne R, Freyne B, Abruzzo V, Morison C, et al. Study protocol for the Melbourne Infant Study: BCG for Allergy and Infection Reduction (MIS BAIR), a randomised controlled trial to determine the non-specific effects of neonatal BCG vaccination in a low-mortality setting. BMJ Open. 2019;9(12). doi:10.1136/bmjopen-2019-032844.
