Abstract
Objective: To estimate the prevalence of amyotrophic lateral sclerosis (ALS) in the United States for 2016 using data from the National ALS Registry (Registry). Established in 2009, the Registry collects data on ALS patients in the U.S. to better describe the epidemiology of ALS, examine risk factors such as environmental and occupational exposures, and characterize the demographics of those living with the disease. Methods: To identify adult prevalent cases of ALS, the Registry compiles data from three national administrative databases (maintained by the Centers for Medicare and Medicaid Services, the Veterans Health Administration, and the Veterans Benefits Administration). To ascertain cases not necessarily included in these databases and to better understand risk-factors associated with ALS and disease progression, the Registry also includes data collected from patients who voluntarily enroll via a web portal to complete online surveys. Results: In 2016, the Registry conservatively identified 16,424 adult persons who met the Registry definition of ALS for an age-adjusted prevalence rate of 5.2 per 100,000 U.S. population. The pattern of patient characteristics (e.g., age, sex, and race/ethnicity) has not changed from previous Registry reports. Overall, ALS was more common among whites, males, and persons aged 60–69 years. The age groups with the lowest number of ALS cases were persons aged 18–39 years. Males had a higher prevalence rate of ALS than females overall and across all data sources. Conclusions: Data collected by the National ALS Registry are being used to better describe the epidemiology and demographics of ALS in the U.S.
Introduction
In 2008, the U.S. Congress passed the amyotrophic lateral sclerosis (ALS) Registry Act, which authorized the creation and maintenance of the National ALS Registry (Registry) (Citation1). The main goals of this multi-faceted research platform are to better describe the incidence and prevalence of ALS in the U.S., examine risk factors such as environmental and occupational factors, and characterize the demographics of persons living with ALS (Citation1).
ALS in the U.S., like most non-communicable diseases apart from cancer, is not a nationally notifiable condition and, therefore, is not reported to state and local health officials and federal health agencies such as the Centers for Disease Control and Prevention/Agency for Toxic Substances Disease Registry (CDC/ATSDR) (Citation2). In the U.S., mostly infectious diseases are notifiable to CDC (Citation3). The only state where ALS is reportable is Massachusetts (Citation4). In addition, identifiable patient data from private health insurance providers are not available for identifying ALS cases. These providers include preferred-provider organization (PPO), health maintenance organization (HMO), high-deductible health plans (HDHP), point of service (POS), and exclusive provider organizations (EPO), among others. As of 2017, there were 907 health insurance companies in the U.S. and its territories (Citation5).
In order to best estimate prevalent ALS cases, the Registry employs an innovative use of administrative and self-reported data for case ascertainment, whereas traditional non-communicable disease registries (e.g. cancer) largely depend upon state laws that mandate health care providers to identify cases (Citation6).
This analysis provides estimated prevalence rates and case counts in the U.S. for 2016, the most recent year for which national data are available.
Methods
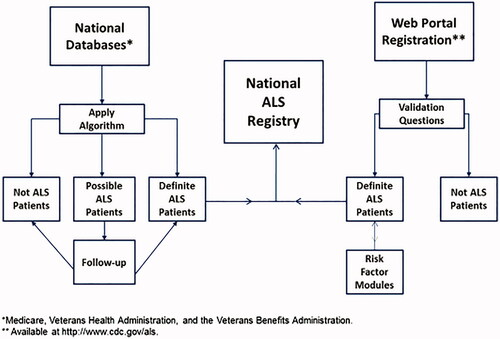
All activities involving human subjects were reviewed and approved by CDC’s Institutional Review Board (IRB). The National ALS Registry, established in 2009, uses a two-pronged approach to identify prevalent cases of ALS in the U.S. The first approach relies on existing administrative data (from the Centers for Medicare and Medicaid Services, the Veterans Heath Administration [VHA], and the Veterans Benefits Administration [VBA]). A pilot tested algorithm is applied to the administrative data that identifies persons with ALS on the basis of encounter codes such as having ALS listed as a code in the visit record or having such a code and having seen a neurologist, a death certificate listing ALS as a cause or contributing cause of death, and prescription for Riluzole () (Citation6).
The second approach, which was launched to the public on 19 October 2010, uses a secure web portal (https://www.cdc.gov/als) to identify cases that are not necessarily included in the national administrative databases (Citation7). This approach allows patients to self-identify and enroll in the National ALS Registry if specific screening criteria are met. An additional advantage of this approach is that those who self-enroll in the Registry can take brief online surveys that are used to evaluate possible risk factors for ALS (e.g. occupational, employment histories).
Information from both approaches is merged into a single record for each person. Merging records for persons identified as having ALS creates a unique record after data are de-duplicated by using a combination of the last five digits of the person’s Social Security number, sex, month and year of birth, and first and last name. This process ensures that persons who are identified in both the administrative databases and the web portal, and those who have records in multiple years, are not counted twice. Once an ALS case is identified, the patient remains a case until confirmed as deceased through the U.S. National Death Index (Citation8). This is referred to as cumulative prevalence of ALS and is calculated from the Registry by using the de-duplicated total number of persons with ALS identified through the two-pronged approach for the numerator. The Registry categorizes each case as “definite ALS,” “possible ALS,” and “not ALS”. Only “definite ALS” cases are entered into the Registry (). This model for identifying ALS cases has been previously published and validated and used for previous prevalent estimates (2010–2015) (Citation9–12).
Figure 1 Algorithm used to identify cases of amyotrophic lateral sclerosis (ALS) in the United States.

Age-adjusted rates were estimated using the 2000 U.S. standard population with the direct method (Citation13). Population estimates by age, gender, race, and census regions were determined using the U.S. Census Bureau (Citation14). Starting at the age of 18, we calculated the age-adjusted rates. Ages were grouped into intervals as stated in the Table. Ninety-five percent confidence intervals were calculated for all age groups, sex, race, and Census regions (Citation15).
Results
During 2016, a total of 16,424 persons were identified as “definite ALS” across all databases. The estimated prevalence rate for 2016 was 5.2 per 100,000 population. The age group 18–39 years had the lowest prevalence rate (0.2 per 100,000 population) and the age group 70–79 had the highest prevalence rate (17.2). Males continued to have a higher overall prevalence rate (7.3 per 100,000 population) than females (3.6). The prevalence rate for whites was almost 1.5 times that of blacks; whites had a prevalence rate of 5.0 compared with 3.4 for blacks. Information on race was unknown for 2,241 (13.6%) of the patients largely attributable to missing race fields from Medicare data. When all the data sources were combined, whites accounted for the majority of cases (74.4%) followed by unknown race, blacks, and other race ().
Table 1 Number and percentage of amyotrophic lateral sclerosis (ALS) cases (N = 16,424) and estimated prevalence, by age group, sex, race, and geographic region per 100,000 population — National ALS Registry, United States, 2016.
Prevalence rates were also calculated for the four major U.S. Census regions (Northeast, South, Midwest, and West). Rates were highest in the Midwest (8.8 per 100,000 population), followed by the Northeast (5.5), West (4.8), and the South (3.2) ().
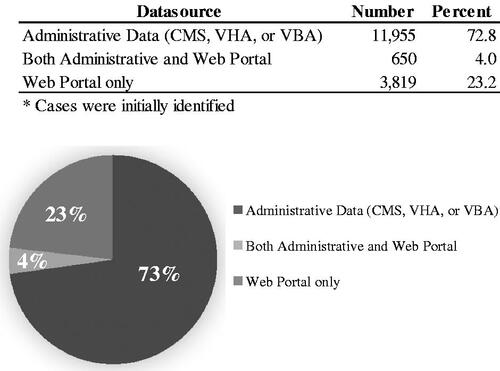
To determine the data source of patients, we collected the source for the first time the patient entered the Registry (). Data from Medicare, VHA, and VBA were combined and considered to be national administrative data. For the prevalent cases in 2016, almost 73% of the patients first came in through national administrative data, while 4% of the patients came through both the national administrative data and the web portal. Some 23% of patients came into the Registry solely from the web portal.
Discussion
This analysis presents updated ALS adult prevalence estimates for the United States for 2016. Data sources for the Registry remain unchanged: national administrative databases and the online web portal. The Registry’s novel approach of using national administrative databases is the cornerstone in identifying ALS cases as most cases came from this approach. The novel case-finding methods utilized by the Registry could potentially serve as a model for estimating the prevalence of other non-notifiable diseases such as Parkinson’s or Alzheimer’s (Citation16).
In the U.S., ALS remains more prevalent in whites, males, and persons aged ≥60 years; current patterns are similar to those identified previously from 2010 to 2015 (Citation9–12). The age-adjusted standardized prevalence rate of ALS cases for 2016, 5.2 per 100,000, is identical to that of 2015 which was also 5.2. Standardized rates for 2014 and 2015 were calculated and shown to be similar to the crude rates when using the entire population as the denominator for the respective year (data not shown). These findings are consistent with published research from well-established European ALS registries (Citation17–23).
Prevalence rates by U.S. Census regions are consistent with ALS demographics. Overall, for all years, whites have a higher prevalence of ALS than blacks. The higher ALS prevalence in the Midwest and Northeast likely reflects the higher proportion of whites, compared with the South and West (Citation24). The lowest prevalence in the South and West Census regions are most likely related to the population diversity in these states. These findings are also consistent with previous ALS mortality analyses showing that states in more northern latitudes have higher age-adjusted mortality rates than states in southern latitudes (Citation25,Citation26).
Missing ALS cases in the United States
In the U.S., ALS patients can receive their care from a number of sources such as government-funded (Medicare, VHA, and VBA) and through private insurance. Patients may also receive their insurance through Medicare Advantage (Part C), which includes private insurance companies approved by Medicare (Citation27). Part C data were requested by the Registry but not received from Medicare and are not included in this analysis. Those who receive their care from employee-sponsored private insurance are not captured by the Registry. These data are fragmented between datasets and do not have personal identifiable information (PII). While private insurance companies capture PII data, the federal government typically is not granted access to these data, especially on a national scale. Without PII such as name, date of birth, or social security numbers, private insurance cases cannot be matched with cases in the Registry. Eventually, some of these patients may move away from private insurance and seek care solely through government sponsored care, increasing the chances of capturing the case. Regardless, due to the variability and fragmented health care delivery model in the U.S., when compared to countries with a single-payer system, determining complete case counts is not possible as it is may be with ALS registries in Europe (Citation28–30).
The Registry is also likely missing cases from under-represented minority populations such as blacks, Hispanics, and Asians. To increase representation of these populations, the Registry will be undertaking future outreach efforts in states such as California, Texas, Florida, and New York which have a higher proportion of minorities. This outreach will be done by targeted media campaigns as well as collaboration with existing partners who directly serve those with ALS (e.g. ALS Association, Muscular Dystrophy Association, and the Les Turner ALS Foundation).
To help determine missing ALS cases, the Registry is utilizing capture-recapture statistical analyses. Capture-recapture is a statistical method that estimates the number of missing cases from different data sources (Citation31,Citation32). It was the intent of the authors to incorporate capture-recapture methodology in this analysis, however, the most recent Registry capture-recapture analysis was not yet published or citable at the time of this paper’s publication. Future prevalence estimates will utilize capture-recapture statistics, as well as prevalence ranges, in order to better describe the epidemiological picture of ALS in the U.S.
Limitations
The findings in this report are subject to several limitations. First, because ALS is a non-notifiable disease, it is challenging to ensure that all newly diagnosed and prevalent ALS cases in the U.S. are captured in the Registry. Therefore, the possibility of under-ascertainment exists. Even with notifiable conditions such as communicable infections (e.g. human immunodeficiency virus, tuberculosis, and shigellosis), the likelihood of under-ascertainment exists. Even the best surveillance systems in general (e.g. cancer registries) are not able to identify all cases. Second, although every attempt was made to de-duplicate the files using the established algorithm, differences in fields collected by the various sources, misspellings of names, and data entry errors could have prevented records from merging correctly. However, it is unlikely that this occurred in numbers sufficient to affect the overall conclusions or in a differential manner that affected conclusions. Finally, without PII such as name, date of birth, age, or sex, the Registry is currently unable to match cases from private insurance with national administrative datasets.
Conclusions
The National ALS Registry estimates that over 16,000 people in the U.S. lived with ALS during 2016. While case under-ascertainment is likely, largely due to a fragmented U.S. healthcare system, the Registry fills an important scientific gap by providing needed epidemiological estimates of ALS. We continue to improve case-ascertainment methods by adding new data sources and evaluating Registry completeness through capture-recapture analysis. ATSDR is committed to monitoring trends of ALS prevalence in the U.S. and advancing research.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention/Agency for Toxic Substances and Disease Registry.
Acknowledgements
The authors are grateful to those living with ALS who give their valuable time to contribute important health data to researchers. Without their help, these findings, and countless others, would not be possible.
Declaration of interest
The CDC/ATSDR authors have no declarations of interest.
Additional information
Funding
References
- US Public Health Service. ALS Registry Act. Washington, DC: 110th Congress 2008.
- Centers for Disease Control and Prevention. National Notifiable Diseases Surveillance System, Weekly Tables of Infectious Disease Data. Available at: https://www.cdc.gov/nndss/infectious-tables.html. Atlanta, GA [Available from: https://www.cdc.gov/nndss/infectious-tables.html.
- Centers for Disease Control and Prevention. 2021 National Notifiable Infectious Diseases. Available at https://wwwn.cdc.gov/nndss/conditions/notifiable/2021/infectious-diseases/.
- Massachusetts Department of Public Health. Massachusetts ALS Registry. Available at: https://www.mass.gov/als-registry. [Available from: https://www.mass.gov/als-registry.
- Insurance Information Institute. Facts + Statistics: Industry Overview. Available at: https://www.iii.org/fact-statistic/facts-statistics-industry-overview. [Available from: https://www.iii.org/fact-statistic/facts-statistics-industry-overview.
- Kaye WE, Sanchez M, Wu J. Feasibility of creating a National ALS Registry using administrative data in the United States. Amyotroph Lateral Scler Frontotemporal Degener. 2014;15:433–9.
- Centers for Disease Control and Prevention. National ALS Registry. Available at: https://www.cdc.gov/als/.
- Centers for Disease Control and Prevention. National Death Index. Available at: https://www.cdc.gov/nchs/ndi/index.htm
- Mehta P, Antao V, Kaye W, Sanchez M, Williamson D, Bryan L, Centers for Disease Control and Prevention (CDC), et al. Prevalence of amyotrophic lateral sclerosis - United States, 2010-2011. MMWR Suppl. 2014;63:1–14.
- Mehta P, Kaye W, Bryan L, Larson T, Copeland T, Wu J, et al. Prevalence of amyotrophic lateral sclerosis - United States, 2012-2013. MMWR Surveill Summ. 2016;65:1–12.
- Mehta P, Kaye W, Raymond J, Punjani R, Larson T, Cohen J, et al. Prevalence of amyotrophic lateral sclerosis - United States, 2015. MMWR Morb Mortal Wkly Rep. 2018;67:1285–9.
- Mehta P, Kaye W, Raymond J, Wu R, Larson T, Punjani R, et al. Prevalence of amyotrophic lateral sclerosis - United States, 2014. MMWR Morb Mortal Wkly Rep. 2018;67:216–8.
- Klein RJ, Schoenborn CA. Age adjustment using the 2000 projected U.S. population. Healthy People 2010 Stat Notes. 2001 Jan;(20):1–10.
- U.S. Census Bureau. ACS Demographic and Housing Estimates. Available at: https://data.census.gov/cedsci/table?d=ACS%205-Year%20Estimates%20Data%20Profiles&tid=ACSDP5Y2016.DP05
- Keyfitz N. 3. Sampling variance of standardized mortality rates. Hum Biol. 1966;38:309–17.
- Horton DK, Mehta P, Antao VC. Quantifying a nonnotifiable disease in the United States: the National amyotrophic lateral sclerosis registry model. JAMA 2014;312:1097–8.
- Nakken O, Lindstrøm JC, Tysnes OB, Holmøy T. Assessing amyotrophic lateral sclerosis prevalence in Norway from 2009 to 2015 from compulsory nationwide health registers. Amyotroph Lateral Scler Frontotemporal Degener. 2018;19:303–10.
- Mitchell JD, Gibson HN, Gatrell A. Amyotrophic lateral sclerosis in Lancashire and South Cumbria, England, 1976-1986: a geographical study. Arch Neurol. 1990;47:875–80.
- Huber S, Henn V. Unchanged incidence and prevalence of amyotrophic lateral sclerosis in the canton of Zürich. Schweiz Arch Neurol Psychiatr (1985) 1995;146:52–4.
- Bettoni L, Bazzani M, Bortone E, Dascola I, Pisani E, Mancia D. Steadiness of amyotrophic lateral sclerosis in the province of Parma, Italy, 1960-1990. Acta Neurol Scand. 2009;90:276–80.
- Traynor BJ, Codd MB, Corr B, Forde C, Frost E, Hardiman O. Incidence and prevalence of ALS in Ireland, 1995-1997: a population-based study. Neurology 1999;52:504–9.
- Chio A, Logroscino G, Traynor BJ, Collins J, Simeone JC, Goldstein LA, et al. Global epidemiology of amyotrophic lateral sclerosis: a systematic review of the published literature. Neuroepidemiology 2013;41:118–30.
- Dupré N, Korngut L. A real-world glimpse into the epidemiology of ALS in Canada. Can J Neurol Sci. 2013;40:625.
- US Census Bureau. Total population 2014 American community survey 1-year. Washington, DC: US Department of Commerce, US Census Bureau; 2014. Available at: https://www.census.gov/acs/www/data/data-tables-and-tools/data-profiles/2014/
- Larson TC, Kaye W, Mehta P, Horton DK. Amyotrophic lateral sclerosis mortality in the United States, 2011-2014. Neuroepidemiology 2018;51:96–103.
- Noonan CW, White MC, Thurman D, Wong LY. Temporal and geographic variation in United States motor neuron disease mortality, 1969-1998. Neurology 2005;64:1215–21.
- U.S. Department of Health & Human Services. What is Medicare Part C?Available at: https://www.hhs.gov/answers/medicare-and-medicaid/what-is-medicare-part-c/index.html.
- Irish Motor Neurone Disease Register. Available at: https://www.hiqa.ie/areas-we-work/health-information/data-collections/irish-motor-neurone-disease-register. [Available from: https://www.hiqa.ie/areas-we-work/health-information/data-collections/irish-motor-neurone-disease-register.
- Chiò A, Cucatto A, Calvo A, Terreni AA, Magnani C, Schiffer D. Amyotrophic lateral sclerosis among the migrant population to Piemonte, northwestern Italy. J Neurol. 1999;246:175–80.
- Hardiman O, Al-Chalabi A, Brayne C, Beghi E, van den Berg LH, Chio A, et al. The changing picture of amyotrophic lateral sclerosis: lessons from European registers. J Neurol Neurosurg Psychiatry. 2017;88:557–63.
- Wittes JT, Colton T, Sidel VW. Capture-recapture methods for assessing the completeness of case ascertainment when using multiple information sources. J Chronic Dis. 1974;27:25–36.
- Hook EB, Regal RR. The value of capture-recapture methods even for apparent exhaustive surveys. The need for adjustment for source of ascertainment intersection in attempted complete prevalence studies. Am J Epidemiol. 1992;135:1060–7.