Abstract
Background
Organized PSA testing for asymptomatic men aged 50–74 years will be implemented in Sweden to reduce opportunistic testing in groups who will not benefit. The aim of this study was to describe the opportunistic PSA testing patterns in a Swedish region before the implementation of organized PSA testing programs.
Method
We included all men in the Uppsala-Örebro health care region of Sweden who were PSA tested between 1 July 2012 and 30 June 2014. Information regarding previous PSA testing, prostate cancer diagnosis, socioeconomic situation, surgical procedures and prescribed medications were collected from population-wide registries to create the Uppsala–Örebro PSA cohort (UPSAC). The cohort was divided into repeat and single PSA testers. The background population used for comparison consisted of men 40 years or older, living in the Uppsala–Örebro region during this time period.
Results
Of the adult male population in the region, 18.1% had undergone PSA testing. Among men over 85 years old 21% where PSA tested. In our cohort, 62.1% were repeat PSA testers. Of men with a PSA level ≤1µg/l 53.8% had undergone repeat testing. Prostate cancer was found in 2.7% and 4.8% of the repeat and single testers, respectively.
Conclusion
Every fifth man in the male background population was PSA tested. Repeated PSA testing was common despite low PSA values. As repeated PSA testing was common, especially among older men who will not be included in organized testing, special measures to change the testing patterns in this group may be required.
Introduction
Screening for prostate cancer (PCa) with prostate-specific antigen (PSA) testing has been extensively debated but remains controversial. Using PSA testing to screen for PCa offers a modest survival benefit but it comes at the cost of over-diagnosis and over-treatment [Citation1–3]. Nevertheless, opportunistic PSA testing is increasing in many countries [Citation4–7]. In Sweden, where there is no organized screening program for PCa, PSA testing in the Stockholm region has increased dramatically from 2003 to 2011 and is apparent also in older men [Citation8].
Organized PSA screening has only been implemented in a small number of countries. In Sweden, the National Board of Health and Welfare decided against organized PSA screening due to a lack of evidence that the benefits outweigh the harm. However, in an attempt to reduce the opportunistic PSA testing organized PSA testing is now being implemented in parts of Sweden [Citation9]. The results of such a program will be affected by current PSA testing patterns in Sweden, which are only partly known.
The aim of this study is to describe the opportunistic PSA testing patterns in a large Swedish cohort with regard to frequency, socioeconomic factors, comorbidity and age before the implementation of organized PSA testing programs.
Method
Uppsala–Örebro PSA Cohort
The basis for this study was the newly created Uppsala–Örebro PSA Cohort (UPSAC) which includes men living in the Uppsala-Örebro health care region, that has had at least one PSA test taken between 2005 and 2014. Additional information on the men in UPSAC was added by linking the cohort to a number of Swedish registries; the National Prostate Cancer Registry (NPCR) and the Cancer Registry for data regarding cancer diagnosis and cancer characteristics (including International Society of Urological Pathology (ISUP) grade groups [Citation10]), the Prescribed Drug Register and the National Patient Register for information on prescribed medications, comorbidity and surgical procedures, the Longitudinal Integration Database for Health Insurance and Labour Market Studies (Swedish acronym LISA) for socioeconomic data as well as the Register of the Total Population and The Cause of Death Register (Supplementary text).
Study design
In this study, all men in UPPSAC who had a PSA test registered at the clinical chemistry laboratories in the Uppsala–Örebro health care region in Sweden during a two-year period, between 1 July 2012 and 30 June 2014, were included. The first PSA test during this period was chosen as the index PSA. Data on PSA testing between 2005 and 2014 (from 2005 to 2014 for Uppsala county, Gävleborg county and Värmland county and from 2007 to 2014 for Örebro county) was collected. Since data were available only from 2007 in Örebro county, a sensitivity analysis was performed were men from the Örebro county were excluded. The study population was divided into repeat testers and single testers. Men were considered to be single testers if they had not taken a PSA test for at least six years prior to the index PSA. Age for the men included in the study was defined as age at the time of the index PSA test.
All men that had relocated to outside the region were excluded as well as men who had been diagnosed with prostate cancer more than 28 days prior to the index PSA test. LUTS was considered to be present in men who had been prescribed medications or had undergone surgical procedures that would indicate LUTS within one year before and after the index PSA (Supplementary text).
Educational level of the population is reported annually from all Swedish schools. Highest education achieved is categorized into seven levels according to the Swedish Educational Nomenclature (SUN2000). In this study, these levels have been transcribed and categorized as short (0–9 years of education), medium length (9–12 years of education) and long (> 12 years of education) education.
From data retrieved from the National Patient Registry and the Cancer Registry, Charlson Comorbidity Index (CCI) [Citation11] was calculated to gain information on concomitant disease burden and men included in the study were divided into four categories based on their comorbidity index: CCI 0, CCI 1, CCI 2 and CCI 3+.
Data regarding the male background population in the Uppsala–Örebro region was retrieved from Statistics Sweden. When comparing the study population with the general population, only men aged 40 years or older in the general population were included to better match the age distribution in the study population.
Linking of registers and creation of dataset was done using SAS software (SAS Institute, Cary, NC, USA). All analyses were made using R Open Source Edition version 3.3.3. The regional research ethics board at Uppsala University approved the study [#2014/552].
Results
Study group characteristics
A total of 55,685 PSA tested men were included in the study among whom 37.9% were single testers and 62.1% were repeat testers (). The median age was 66.0 years for all men in the study, 62.2 years among single testers and 68.4 among repeat testers. Married men made up 56.3% and 64.8% of single and repeat testers, respectively. Among single testers, 18.9% had LUTS as compared to 34.2% among repeat testers.
Table 1. Characteristics of a cohort of 55,685 men with a PSA test registered at the clinical chemistry laboratories in the Uppsala–Örebro health care region, Sweden, between 1 July 2012 and 30 June 2014.
Within one year of the index PSA test, 3.5% of the cohort were diagnosed with prostate cancer: 4.8% and 2.7% of the single and repeat testers, respectively. Single testers had a higher incidence of ISUP grade group 5 cancers compared to repeat testers ().
Comparison with background population
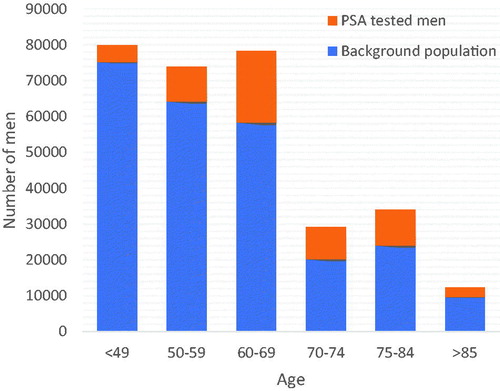
There were 307,633 men 40 years old or older, available for PSA testing in the included regions during the chosen time period. The 55,685 men included in the study comprised 18.1% of these men. The largest proportion of PSA tested men were among 70–74-year-old men of which 30.6% were PSA tested within the study period. Among the oldest men in the population (85 years old or older), 21.0% had a PSA test taken during the study period (). In the study population, 30.7%, 43.8% and 24.9% had a short, medium length and long education, respectively, as compared to the background population where 26.2%, 47.4% and 25.5% had a short, medium length and long education.
PSA testing patterns
Among repeat testers, 29.7% had an index PSA level <1 µg/l and 5.6% had a PSA that was >10 µg/l (). Among repeat testers with PSA <1 µg/l, 74.8% had undergone 1–3 previous PSA. The proportion of repeat testers having undergone four or more previous PSA tests increased with increasing index PSA level; 25.2%, 33.7%, 52.8% and 62.5% of men with PSA <1 µg/l, 1–2.9 µg/l, 3–9.9 µg/l and >10 µg/l, respectively. Among repeat testers, men with higher levels of PSA were older than men with lower levels of PSA; median age for repeat testers with PSA <1µg/l was 64.9 years and 75.8 years for men with PSA >10µg/l. Repeat testers with higher PSA levels were more likely to have LUTS: 23.8% of the men with a PSA level <1 µg/l and 51.6% of the men with PSA >10 µg/l had LUTS. Higher CCI was more common among men with higher PSA levels; 4.9% of repeat testers with PSA <1µg/l had CCI 3+ and 8.4% of repeat testers with PSA >10µg/l had CCI3+. Previous PSA testing was common in all age groups ().
Table 2. Number of previous PSA tests, median number of previous PSA tests (range within parenthesis), prevalence of LUTS and CCI among repeat PSA testers in a cohort of 55,685 men who were PSA tested in the Uppsala–Örebro health care region between 1 July 2012 and 30 June 2014.
Table 3. Number of previous PSA tests in different age groups in a cohort of 55,685 men who were PSA tested in the Uppsala–Örebro health care region between 1 July 2012 and 30 June 2014.
Sensitivity analysis
Since data from Örebro region was available only from the year 2007, a sensitivity analysis was performed, where the men from the Örebro region were excluded. A total number of 12,215 men included in our study were living in the Örebro region. Out of these men, 5508 men had only 5.5 years where their PSA testing was known. When the Örebro men were excluded from the study, the results did not change significantly (data not shown).
Discussion
In this population-based study, less than a fifth of the male population over the age of 40 had undergone PSA testing, but the proportion of PSA tested men varied between age groups with PSA testing being more common among older men. Of the men included in the study, a majority had undergone repeat PSA testing. Repeat PSA testing was common even among men with a PSA level of 1 µg/l or less. Marital status but not educational level affected the PSA testing patterns. Single testers had a higher incidence of prostate cancer and were more often diagnosed with high-risk cancers.
PSA testing patterns
Of the available male population, 18.1% had undergone PSA testing during the study period. The proportion of PSA tested men was largest among men over 60 years of age and particularly among men over 70 years of age, where approximately 30% of the men had undergone PSA testing. Notably, PSA testing was common even among the oldest men; over 20% of men over 85 years had been PSA tested during the study period. Previous studies on the prevalence of PSA testing have shown varying results. A British study estimated the overall annual rate of testing to be 6% [Citation5] and an Italian study showed that 34% of the male population aged over 50 years had undergone PSA testing at least once during a two-year period [Citation12]. A Swedish study on the PSA testing patterns of the Stockholm region showed that approximately 40-60% of all men aged 50-69 years had at least one PSA test during a five-year period [Citation8]. PSA testing patterns have previously been shown to vary across different regions of Sweden [Citation13]. Nonetheless, our results indicate a relatively low prevalence of PSA tested men in a large health care region which will most likely impact the results of an organized PSA testing program. It is plausible that the implementation of such a program will increase the number of men undergoing PSA testing. If so, a large proportion of men in the organized PSA testing programs will be men that are not previously PSA tested or not PSA tested in many years which might lead to a considerable increase in the detection of prostate cancers.
Proportion repeat testers and single testers
A majority of the study group were repeat testers (62.1%) and repeat PSA testing was common in spite of low PSA levels. Multiple, frequent testing in men with low PSA levels is incoherent with national guidelines, and in this case, organized PSA testing might lead to reduced over-testing. More than 20% of the study group were 75 years old or older. A large proportion of the older men in the study group had undergone multiple PSA testing; over 70% of men aged 75 years or older in the study group were repeat testers. There is limited evidence that PSA testing older men without symptoms of prostate cancer affects prostate cancer morbidity or prostate cancer mortality [Citation14]. Organized PSA testing will not include these men and their testing patterns may therefore not change unless an organized PSA testing program leads to increased awareness among health professionals and in the general population.
Socioeconomic factors affecting PSA testing
Being married was more common among repeat testers than among single testers (64.8% vs 56.3%). Other studies have shown that being married increases the likelihood of participating in cancer screening programs [Citation15,Citation16]. In a Swedish study, it was shown that married men had a lower risk of being diagnosed with advanced prostate cancer and a higher chance for health checkup as a reason for prostate cancer investigation than unmarried men [Citation17]. These results indicate that married men are more prone to partake in the preventive examination, such as PSA testing. It is not known if the implementation of an organized PSA testing program for prostate cancer can affect such inequalities.
Previous studies have shown that educational level corresponds to the level of diagnostic activity, treatment choice and outcome in prostate cancer as well as in other forms of cancer [Citation18–20]. Men with a longer education have been shown to be more likely to undergo PSA testing and as a corollary had higher incidence rates of localized cancer [Citation21]. In our study, no large differences could be seen in educational level when comparing single testers with repeat testers as well as when comparing the study group with the background population. Tomic et al. have shown that Swedish men with higher income are more often diagnosed with prostate cancer as a result of a health checkup compared to men with a lower income [Citation17]. The same connection could not be shown for educational level which can to some extent correspond with the results of our study. This indicates that other aspects than educational level needs to be taken into consideration regarding health care seeking behavior to fully understand how socioeconomic factors influence PSA testing patterns in Sweden.
Prostate cancer and PSA testing
Being diagnosed with prostate cancer within one year of the index PSA was more common among single testers than among repeat testers. Single testers were also more often diagnosed with a prostate cancer of higher ISUP grade group. It is possible that single testers seek health care not primarily for PSA testing but for other symptoms such as skeletal pain or fatigue and a PSA test is taken, leading to the detection of a more advanced prostate cancer. The higher proportion of prostate cancer among single testers may also, at least in part, be due to the fact that men with prevalent prostate cancer were excluded from the study.
Strengths and limitations
The main strengths of this study include the large number of men in the study and that there is virtually no loss to follow up. Information on the men included in the study was collected from multiple Swedish registries, which are all of high quality and near-complete coverage. The regions included in the study consist of both smaller communities and larger cities with university hospital care, which should give a fair representation of the Swedish ‘PSA population’.
Limitations include the lack of data regarding PSA testing before 2005 (2007 for the Örebro region), possibly leading to a misclassification of repeat testers as single testers. Another limitation relates to the definition of LUTS, as the use of surgical procedures and prescribed medications, inevitably will cause some misclassification.
Conclusion
This study shows that less than 20% of the male population in the Uppsala–Örebro health care region underwent PSA testing between 2012 and 2014, although the proportion was higher among older men. Repeat testing was more common than single testing and repeat PSA testing was common also in men with low PSA levels. Our results suggest that implementation of organized PSA testing could help optimize the use of health care system resources but special efforts may be required to address the testing patterns among older men that are not targeted by organized testing programs.
Supplemental Material
Download MS Word (49.9 KB)Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate cancer mortality: results of the European Randomised Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet. 2014;384(9959):2027–2035.
- Pinsky PF, Prorok PC, Yu K, et al. Extended mortality results for prostate cancer screening in the PLCO trial with median follow-up of 15 years. Cancer. 2017;123(4):592–599.
- Arnsrud Godtman R, Holmberg E, Lilja H, et al. Opportunistic testing versus organized prostate-specific antigen screening: outcome after 18 years in the Göteborg randomized population-based prostate cancer screening trial. Eur Urol. 2015;68(3):354–360.
- Farwell WR, Linder JA, Jha AK. Trends in prostate-specific antigen testing from 1995 through 2004. Arch Intern Med. 2007;167(22):2497–2502.
- Melia J, Moss S, Johns L. Rates of prostate-specific antigen testing in general practice in England and Wales in asymptomatic and symptomatic patients: a cross-sectional study. BJU Int. 2004;94(1):51–56.
- Smith DP, Supramaniam R, Marshall VR, et al. Prostate cancer and prostate-specific antigen testing in New South Wales. Med J Aust. 2008;189(6):315–318.
- Drummond FJ, Carsin AE, Sharp L, et al. Trends in prostate specific antigen testing in Ireland: lessons from a country without guidelines. Ir J Med Sci. 2010;179(1):43–49.
- Nordstrom T, Aly M, Clements MS, et al. Prostate-specific antigen (PSA) testing is prevalent and increasing in Stockholm County, Sweden, Despite no recommendations for PSA screening: results from a population-based study, 2003–2011. Eur Urol. 2013;63(3):419–425.
- Regionala cancercentrum i samverkan. Rekommendationer om organiserad prostatacancertestning [accessed 2019 Oct 2]. Available from https://www.cancercentrum.se/samverkan/vara-uppdrag/prevention-och-tidig-upptackt/prostatacancertestning/2018.
- Epstein JI, Egevad L, Amin MB, et al. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason grading of prostatic carcinoma: definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40(2):244–252.
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- Russo A, Autelitano M, Bellini A, et al. Estimate of population coverage with the prostate specific antigen (PSA) test to screen for prostate cancer in a metropolitan area of northern Italy. J Med Screen. 2002;9(4):179–180.
- Jonsson H, Holmström B, Duffy SW, et al. Uptake of prostate-specific antigen testing for early prostate cancer detection in Sweden. Int J Cancer. 2011;129(8):1881–1888.
- Fenton JJ, Weyrich MS, Durbin S, et al. Prostate-specific antigen-based screening for prostate cancer: evidence report and systematic review for the US preventive services task force. JAMA. 2018;319(18):1914–1931.
- Hanske J, Meyer CP, Sammon JD, et al. The influence of marital status on the use of breast, cervical, and colorectal cancer screening. Prev Med. 2016;89:140–145.
- El-Haddad B, Dong F, Kallail KJ, et al. Association of marital status and colorectal cancer screening participation in the USA. Colorectal Dis. 2015;17(5):O108–O114.
- Tomic K, Ventimiglia E, Robinson D, et al. Socioeconomic status and diagnosis, treatment, and mortality in men with prostate cancer. Nationwide population-based study. Int J Cancer. 2018;142(12):2478–2484.
- Cavalli-Bjorkman N, Lambe M, Eaker S, et al. Differences according to educational level in the management and survival of colorectal cancer in Sweden. Eur J Cancer. 2011;47(9):1398–1406.
- Eaker S, Halmin M, Bellocco R, et al. Social differences in breast cancer survival in relation to patient management within a National Health Care System (Sweden). Int J Cancer. 2009;124(1):180–187.
- Berglund A, Garmo H, Robinson D, et al. Differences according to socioeconomic status in the management and mortality in men with high risk prostate cancer. Eur J Cancer. 2012;48(1):75–84.
- Steenland K, Rodriguez C, Mondul A, et al. Prostate cancer incidence and survival in relation to education (United States). Cancer Causes Control. 2004;15(9):939–945.