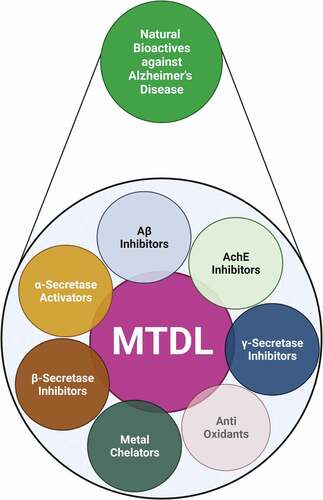
ABSTRACT
Alzheimer’s disease (AD) is a common form of dementia affecting the elderly worldwide. It is a multifactorial neurodegenerative disorder with no known preventive therapy. Many of the drugs used in the treatment of AD, such as galantamine, rivastigmine, and donepezil, have unpleasant side effects, and hence physicians are keen to find alternatives. Research has shown that plants and their phytochemicals can alleviate AD. These plant products can act through various modes, such as inhibition of amyloid β, acetylcholine, and γ-secretase, modulation of antioxidants, and α-secretase activation, which are known to involve in the improvement of brain functions. A recent approach that has garnered the attention of many researchers in designing a drug against AD is the multi-target-directed ligand (MTDL), wherein the same molecule act on multiple targets. Many studies have reported the potential of herbs to act on multiple targets and display biological properties. The current review summarizes the ongoing evidence on the use of herbs and their derived bioactive molecules in the treatment of AD and in relieving disease-associated pathological events. Currently available plant-derived MTDLs for the treatment or slowing down of the progression of AD are also discussed.
Introduction
Alzheimer’s disease (AD) is an irreversible, chronic neurodegenerative disorder characterized by deterioration of cognitive functions and behavioral disturbances [Citation1]. Globally, AD is the most common cause of dementia, affecting approximately 46.8 million people and expected to increase up to 131.5 million by 2050 [Citation2]. The probability of AD aggressively increases with age, more particularly after the age of 65. Thus, age is the primary risk factor for AD development [Citation3]. AD developed after 65 years of age is referred to as ‘sporadic’ (or late-onset), whereas AD developed before 65 is classified as ‘familial’ (or early-onset). Several complex pathogenic pathways are involved in the progression and development of the disease, including plaque formation, inflammatory cascade, oxidative stress, and cholinergic deficit [Citation4]. These cognitive deficits lead to memory-related clinical symptoms, such as loss of episodic and newly learned memories [Citation5] .
Acetylcholine- and glutamate-producing neurons are known to be damaged during AD, thereby affecting the synapses associated with them. This is in agreement with the early cognitive symptoms observed in AD [Citation6]. The main factor for the degeneration of neurons is due to the increased activity of cholinesterases (ChEs), which leads to a decrease in acetylcholine (ACh) levels, which in turn stops the neuronal transmission signals [Citation7]. Studies have also established that acetylcholinesterase (AChE) promote Aβ aggregation and a notable increase in the cortical levels of butyrylcholinesterase (BuChE), which is related to the formation of Aβ plaques and neurofibrillary tangles (NFTs) [Citation8–10].
Many natural compounds are known to have neuroprotective effects during AD. A large family of plant isolates has proven to be a modality for treatment by their inhibitory effect on Aβ, cholinesterase, beta, and gamma secretases. Potent activation of alpha secretases by plant products also substantiates the neuroprotective effect. This review gives a detailed insight into the list of plants and their isolates as neuroprotective agents during AD.
Molecular mechanism of AD
The formation of NFTs and senile plaques are the main histopathological hallmarks of AD [Citation11]. The senile plaques contain amyloid-beta (Aβ) peptide, which consists of 37–49 amino acid residues and are formed by the extracellular and transmembrane domains of amyloid precursor protein (APP) [Citation12]. In plaques, the oligomers might be trapped in fibrillar aggregates. Oligomers may be the hazardous Aβ species that contribute to signaling pathway deregulation (Fyn, FAK, GSK3b, and CDK5), causing changes in cytoskeletal and synaptic proteins, as well as synaptic and neural damage [Citation13] (). During sporadic AD, APP is cleaved by gamma and beta secretases to form 4 kDa Aβ peptide. The cleavage product has a strong tendency to form aggregates. Aβ accumulation has been one of the major pathological events resulting from an imbalance between production and clearance [Citation14]. The Aβ aggregation process initiates by self-assembling of Aβ monomers into low molecular weight oligomers, which in turn results in the formation of high molecular weight oligomers known as soluble aggregation intermediates. These further aggregate to form fibrils and accumulate in the brain [Citation15,Citation16]. It is believed that microglia and astrocytes then mount an inflammatory response to clear the amyloid aggregates, and this inflammation likely causes the destruction of adjacent neurons and their neurites.
Figure 1. Formation of neurofibrillary tangles (NFTs) and senile plaques. The senile plaques contain amyloid beta (Aβ) peptide, which consists of 37–49 amino acid residues and are formed by the extracellular and transmembrane domains of amyloid precursor protein (APP). Oligomers may be the hazardous Aβ species that contribute to signaling pathway deregulation (Fyn, FAK, GSK3b, and CDK5), causing changes in cytoskeletal and synaptic proteins, as well as synaptic and neural damage.
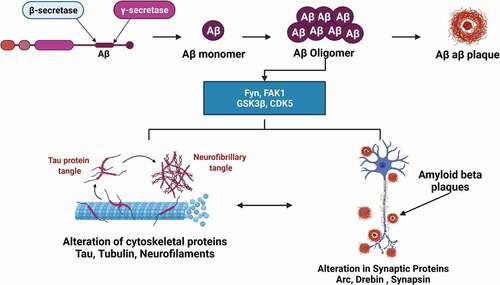
Other than plaques, the presence of NFTs is considered another characteristic feature in the neuropathological event of AD [Citation17]. These NFTs are insoluble twisted fibers formed by abnormal hyperphosphorylation of a microtubule-associated protein called ‘tau’. NTF in normal form serves as a microtubule-stabilizing protein and plays a role in intracellular (axonal and vesicular) transport [Citation18]. NFT may interfere with the regular axonal transport of components necessary for proper neuronal function and survival, eventually causing neurons to die. In addition, Aβ is thought to trigger neuronal cell death via controlling apoptosis inducers, generating oxidative stress, and increasing free radical-mediated pathways[Citation11].
Methodology
A detailed literature survey was performed using both offline and online resources. Data were mainly collected from various journal publishers, including Elsevier, Springer Nature, Taylor & Francis, Cambridge University Press, Oxford University Press, BioMed Central, and PLOS (Public Library of Science). The online databases such as Google Scholar, Pubget, Medline, PubMed, EMBASE, Mendeley, ScienceDirect, Scopus, and SpringerLink were also used to retrieve literature. The results were then cross-referred to generate the list of references (up to 2018) cited in this review. The Current review methodically summarizes the neuroprotective effects of phytochemicals in various models. Herbal extracts, bioactive constituents, and herbal formulations were included to provide references in the future.
Natural products in AD
Since time immemorial, natural products have been used as medicine for many ailments. Natural products are molecules with diverse functions and have been the source of most active constituents in medicine [Citation19,Citation20]. They are said to be the most successful basis of drug leads with lesser toxicity [Citation21,Citation22]. Natural products may be derived from plants, animals, or microorganisms. Most herbal medicines are complex and constitute many chemical components, which possess diverse biological and pharmacological activities.
Medicinal plants are nature’s gift that remains unexplored. The active component present in herbal medicine may serve as the basis for preparing synthetic drugs [Citation22]. Plants can synthesize chemical compounds involved in preventing or curing various diseases, including memory dysfunction and age-related disorders. In modern medicine, plants occupy a very significant place as a source of raw material for synthetic drugs [Citation23]
Cholinesterase inhibitors
The important etiological factor in the pathogenesis of memory deficit in AD is the impairment in cholinergic transmission [Citation24]. The inhibition of AChE increases the levels of acetylcholine in the brain and thus improves the cholinergic functions in AD patients Citation25. Hence, cholinesterase inhibitors are currently used as standard drugs for treating AD. Tacrine was the first AChE inhibitor drug approved for AD treatment [Citation26]. Later, many other AChE inhibitors such as rivastigmine, galantamine, and donepezil were also developed and approved by the FDA. These drugs alleviate the symptoms but are associated with side effects when used for an extended period [Citation27]. As AD has reached a state of public health burden, the ever-increasing reports of side effects from these synthetic and hybrid drugs have driven the research for a novel and safe AchE inhibitors from plant sources.
Plants continue to be the unvaryingly abundant source of therapeutic drugs for AD treatment because of their AChE inhibitory activity. Several plant extracts of various solvents have been reported to show anticholinesterase activity. Aqueous and methanolic root extracts of Acacia nilotica and Withania somnifera possessed moderate anticholinesterase activity (IC50 values of 0.079 and 33.38 µg/ml, respectively) [Citation28,Citation29]. Much lesser inhibitory activity was observed in hydroethanolic seed extracts of Myristica fragrans, which showed 50% enzyme inhibition at a concentration range between 100 and 150 µg/mL [Citation30]. Also, Pinus nigra was used to extracting essential oils possessing 94.4 µg/mL activity [Citation31]. Similarly, different extracts of plants belonging to varied plant families have shown considerable cholinesterase inhibitory activity, which is listed in ().
Table 1. Plants with potential AChE inhibitory activity
Alkaloids derived from various plant extracts show immense potential for AChE inhibitory activity. However, significantly few isolated compounds have been utilized for research and therapeutic purposes. Many isolated compounds from different classes of alkaloids have been considered and tabulated in ().
Table 2. Isolated compounds from plants with potential AChE inhibitory activity
γ- and β-secretase inhibitors
Many plant extracts and their derived compounds are found to influence the Aβ production pathways, mainly by interacting with brain enzymes like β- and γ-secretases [Citation112,Citation113]. As explained earlier, both β- and γ-secretases are involved in the synthesis of Aβ. β-secretase cleaves the APP to form a transmembrane C-99 fragment with the N-terminus of the Aβ peptide () followed by the action of γ-secretase, which cleaves C-99 fragment in the transmembrane domain to make the C-terminus of Aβ [Citation112].
Figure 2. β- and γ-secretases are involved in the synthesis of Aβ. β-secretase cleaves the APP to form transmembrane C-99 fragment with the N-terminus of the Aβ peptide. This is followed by the action of γ-secretase, which cleaves C-99 fragment in the transmembrane domain to make the C-terminus of Aβ.
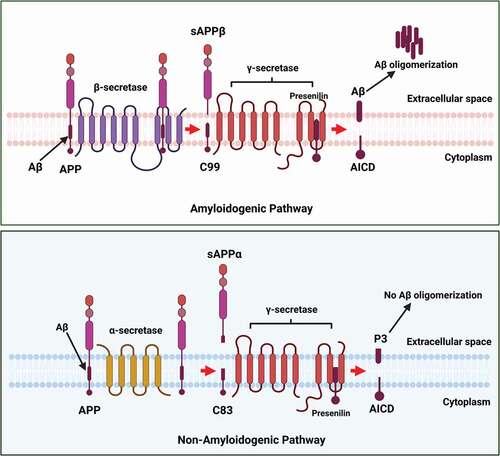
In addition to β-APP processing, γ-secretase also plays a vital role in the cleavage of the Notch family of cell-surface receptors, a protein mainly required for transcriptional regulation during neuron development [Citation114]. As a result, the use of γ-secretase inhibitors has provided insights into proteolytic activity and suggests that such inhibition might be a useful strategy for AD therapeutics [Citation115]. A triterpene isolated from Actaea racemosa reduced the formation of Aβ toxicity through the modulation of γ-secretase activity. Thus, it suggests that the isolated compound may bind to γ-secretase APP complex, modulating the cleavage of APP and hence lowering the formation of Aβ peptides. Citation116,demonstrated that the use of green tea polyphenol epigallocatechin-3-gallate (EGCG) inhibited LPS-induced Aβ elevation levels through the suppression of LPS-induced β- and γ-secretase activities [Citation116]. However, the inhibition of Notch protein by γ-Secretase inhibitors affects neuronal development, as Notch has multiple substrates that are involved in neuronal development [Citation117]. Hence, β-secretase, also referred to as β-site APP cleaving enzyme 1 (BACE-1), a transmembrane aspartic protease secreted in almost all tissues but present in higher amounts in neurons of the brain [Citation115], is considered as the most promising target for pharmaceutical research on AD, compared to γ-secretase.
α- secretase activators
α-secretase enzyme proteolytically cleaves the APP via the non-amyloidogenic pathway at L688 residue located within the Aβ sequence and thereby preventing the formation of Aβ (). The first enzyme for α-secretase was proposed in 1998, when ADAM17, also known as tumor necrosis factor-converting enzyme (TACE), was reported to possess α-secretase activity [Citation118]. Later, ADAM9 and ADAM10 were also shown to have α secretase activity [Citation119]. These three proteins belong to the ADAM (a disintegrin and metalloprotease) family. It is reported that mutations in ADAM10 alter the processing of APP and lead to AD by increasing Aβ levels [Citation120]. Thus, a promising yet underestimated approach to overcome AD would be, activating α-secretase processing of βAPP.
Moderate overexpression of ADAM10 in an APP mouse model showed a decreased level of Aβ, and prevented its accumulation. Such decreased levels of Aβ are found to alleviate cognitive deficits [Citation121,Citation122]. Various studies have corroborated that several drugs currently used in the treatment of AD promote α-secretase activity by activating associated signaling cascades. Thus, it has been considered as one of the best therapeutic approaches in AD [Citation123–125].
Aβ inhibitors
Bioactivity-guided isolation has led to the discovery of novel bioactive compounds from plants, which are useful in preventing Aβ-induced neuronal cells [Citation126]. In vitro assays were widely used to assess the activity of isolated compounds. It is observed that phenolic compounds, alkaloids, and glycosides comprise the major part of the isolated compounds with Aβ inhibitory activity. Their antioxidant activity and lipophilicity make it easy for them to cross the blood-brain barrier [Citation126]. A list of compounds with Aβ inhibitory activity is provided in ().
Table 3. Plants with inhibitory activity against Aβ
Table 4. Natural Compounds with inhibitory activity against Aβ
Antioxidants in AD
Oxidative stress is a process of ROS generation, which plays a central role in cellular injuries and various clinical disorders, including neurodegenerative diseases [Citation215]. The brain cells are continuously exposed to a surplus of free radicals, which leads to oxidative stress. Thus, ROS-induced oxidative stress in the brain is one of the most common etiologies of neurodegenerative disorders, including AD [Citation216,Citation217]. The oxidative stress not only mediates neurotoxicity induced by Aβ, but also enhances the production of Aβ [Citation218]. Thus, oxidative stress is a prime contributing factor for AD development, and antioxidants can be considered therapeutic approaches.
MTDL: A new therapeutic approach for AD
For 15 years, AD had been treated symptomatically, and the therapeutic approaches are of modest efficacy [Citation219]. The approved drugs fall into two categories: AChE inhibitors and N-methyl D-aspartate (NMDA) receptor antagonists, with four and one drug in each group, respectively [Citation220,Citation221]. These cholinergic drugs increased cholinergic system deficiency by inhibiting the AChE enzyme, which degrades acetylcholine. One of the important drugs belonging to this class is donepezil. Many evidence infers that AChE inhibition reinstates the cholinergic system and mediates the disease progression [Citation222].
On the other hand, the excessive NMDA glutamate receptor activity observed in AD was inhibited by a low-affinity, non-competitive and open channel blocker, memantine, which is frequently used with AChE inhibitors [Citation219]. These drugs are insufficient for AD therapy, and this warrants more research towards finding drugs against AD. Citation223,suggested that identification of Aβ or tau proteins as a target in AD created two groups of researchers, referred to as ‘baptists’ and ‘tauists’ [Citation223]. However, both these groups failed to develop the potential drugs which can cure the disease. Moreover, along with Aβ, antagonistic AChE also targets other aspects of AD.
Over the past nine decades, researchers have been targeting one factor at a time, which did not result in any drug to cure AD. Efficient pharmacotherapy may require simultaneous action on several targets involved in its pathogenesis due to the complexity of AD. Such effects may be achieved by administering a drug cocktail or a multicomponent drug. Besides AD, other neurological disorders such as depression, allergies, hypertension, schizophrenia, inflammation, and metabolic diseases can also be treated by this combination of drugs [Citation224]. But, this approach carries the risk of potentially hazardous drug-drug interactions caused by specific pharmacokinetic and pharmacodynamic properties of individual components. It would be ideal if a single molecule could simultaneously act on multiple targets with greater efficacy and safety profile. In 2005, Morphy and Rankovic proposed this innovative strategy to develop MTDLs as potential drug candidates. This approach can be more relevant and practical since AD is a complex neurological disorder with multiple causative factors.
Further, to reduce the side effects, many reports suggest using herbal alternatives to enhance the efficacy of the therapy in the future [Citation225]. Thus, identifying novel pharmacological neuroprotective MTDLs from plants is the new hope for treating AD. These natural products can simultaneously act on multiple targets associated with AD (enhance α-secretase activity; decrease β- and γ- secretase activity; inhibit Aβ; prevent oxidative stress and inflammation). Some plant products possessing multiple targets against AD are presented in tabulated in . The summary of the role of plant extracts and their phytochemicals in circumventing AD is represented in .
Table 5. Plant products with multiple targets against AD
Conclusion and prospects
Natural products have tremendous potential to act against AD and have given hope to the scientific fraternity as sources of drugs. Though the cause of AD is not clearly understood, natural products with multiple activities like AChE inhibition, NMDAR antagonist, antioxidant ability, amyloid inhibition, and anti-inflammation have the potential to be used as drugs. The healing power of culinary herbs and medicinal plants has attracted the researcher’s attention to study natural products as a potentially valuable resource for drug discovery against AD. Several natural products are used alone or in combination with some other neuroprotective agents to enhance memory and cognitive dysfunction and prevent AD.
Theoretically, phytochemical-based treatments against cognitive deficit could prove beneficial in clinical trials on humans due to their low toxicity and high bioavailability. The use of recent pharmaceutical technologies and developments in medicinal chemistry is to design novel drugs based on natural templates, which act on multiple targets, opens up a new window to using natural products in therapeutics against AD.
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- Christidi F, Migliaccio R, Santamaría-García H, et al. Social cognition dysfunctions in neurodegenerative diseases: neuroanatomical correlates and clinical implications. Behav Neurol. 2018;2018:1849794.
- Więckowska A, Kołaczkowski M, Bucki A, et al. Novel multi-target-directed ligands for Alzheimer’s disease: combining cholinesterase inhibitors and 5-HT6 receptor antagonists. Design, synthesis and biological evaluation. Eur J Med Chem. 2016;124:63–81.
- Guerreiro R, Bras J. The age factor in Alzheimer’s disease. Genome Med. 2015;7(1):106.
- Münch G, Schinzel R, Loske C, et al. Alzheimer’s disease – synergistic effects of glucose deficit, oxidative stress and advanced glycation endproducts. J Neural Transm. 1998;105(4):439–461. DOI:10.1007/s007020050069.
- LaFerla FM, Green KN, Oddo S. Intracellular amyloid-beta in Alzheimer’s disease. Nat Rev Neurosci. 2007;8(7):499–509.
- Kandimalla R, Reddy PH. Therapeutics of neurotransmitters in Alzheimer’s Disease. J Alzheimers Dis. 2017;57(4):1049–1069.
- Colovic MB, Krstic DZ, Lazarevic-Pasti TD, et al. Acetylcholinesterase inhibitors: pharmacology and toxicology. Curr Neuropharmacol. 2013;11(3):315–335. DOI:10.2174/1570159X11311030006.
- Alvarez A, Opazo C, Alarcón R, et al. Acetylcholinesterase promotes the aggregation of amyloid-beta-peptide fragments by forming a complex with the growing fibrils. J Mol Biol. 1997;272(3):348–361. DOI:10.1006/jmbi.1997.1245.
- Darvesh S, Cash MK, Reid GA, et al. Butyrylcholinesterase is associated with β-amyloid plaques in the transgenic APPSWE/PSEN1dE9 mouse model of Alzheimer disease. J Neuropathol Exp Neurol. 2012;71(1):2–14. DOI:10.1097/NEN.0b013e31823cc7a6.
- Inestrosa NC, Alvarez A, Pérez CA, et al. Acetylcholinesterase accelerates assembly of amyloid-beta-peptides into Alzheimer’s fibrils: possible role of the peripheral site of the enzyme. Neuron. 1996;16(4):881–891. DOI:10.1016/S0896-6273(00)80108-7.
- Ahn JY, Kim S, Jung SE, et al. Effect of licorice (Glycyrrhiza uralensis fisch) on amyloid-β-induced neurotoxicity in PC12 cells. Food Sci Biotechnol. 2010;19(5):1391–1395. DOI:10.1007/s10068-010-0198-4.
- Chen GF, Xu TH, Yan Y, et al. Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin. 2017;38(9):1205–1235. DOI:10.1038/aps.2017.28.
- Crews L, Masliah E. Molecular mechanisms of neurodegeneration in Alzheimer’s disease. Hum Mol Genet. 2010;19(R1):R12–20.
- Selkoe DJ, Hardy J, Hardy J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol Med. 2016;8(6):595–608.
- Morphy R, Rankovic Z. Designed multiple ligands. An emerging drug discovery paradigm. J Med Chem. 2005;48(21):6523–6543.
- Moreira PI, Carvalho C, Zhu X, et al. Mitochondrial dysfunction is a trigger of Alzheimer’s disease pathophysiology. Biochim Biophys Acta. 2010;1802(1):2–10. DOI:10.1016/j.bbadis.2009.10.006.
- Ittner LM, Ke YD, Delerue F, et al. Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer’s disease mouse models. Cell. 2010;142(3):387–397. DOI:10.1016/j.cell.2010.06.036.
- Mietelska-Porowska A, Wasik U, Goras M, et al. Protein modifications and interactions: their role in function and dysfunction. Int J Mol Sci. 2014;15(3):4671–4713. DOI:10.3390/ijms15034671.
- Butler MS. Natural products to drugs: natural product-derived compounds in clinical trials. Nat Prod Rep. 2008;25(3):475–516.
- Sengupta T, Vinayagam J, Singh R, et al. Plant-derived natural products for Parkinson’s disease therapy. Adv Neurobiol. 2016;12:415–496.
- Harvey A. Natural products in drug discovery. Drug Discov Today. 2008;13(19–20):894–901.
- Newman DJ, Cragg GM. Natural products as sources of new drugs over the last 25 years. J Nat Prod. 2007;70(3):461–477.
- Prakash S, Jain A. Antifungal activity and preliminay phytochemical studies of leaf extract of Solanum nigrum Linn. Int J Pharm Pharm Sci. 2011;3:352–355.
- Nampoothiri M, John J, Kumar N, et al. Modulatory role of Simvastatin against aluminium chloride-induced behavioural and biochemical changes in rats. Behav Neurol. 2015;2015:1–9.
- Murray AP, Faraoni MB, Castro MJ, Alza NP, and Cavallaro V. Natural AChE inhibitors from plants and their contribution to Alzheimer’s disease therapy. Curr. Neuropharmacol. 2013 Jul 1;11(4):388–413.
- Ved HS, Best JM, and Dave JR. Comparative inhibition of acetylcholinesterase by Tacrine, Physostigmine and Huperzine in the adult rat brain. In Enzymes of the Cholinesterase Family, 1995 pp. 477–478. Boston, MA: Springer.
- Howes MJR, Perry NSL, Houghton PJ. Plants with traditional uses and activities, relevant to the management of Alzheimer’s disease and other cognitive disorders. Phytother Res PTR. 2003;17(1):1–18.
- Crowch CM, Okello EJ. Kinetics of acetylcholinesterase inhibitory activities by aqueous extracts of Acacia nilotica (L.) and Rhamnus prinoides (L’Her.). Afr J Pharm Pharmacol. 2009. [accessed May 4, 2020]. https://eprint.ncl.ac.uk/153908.
- Vinutha B, Prashanth D, Salma K, et al. Screening of selected Indian medicinal plants for acetylcholinesterase inhibitory activity. J Ethnopharmacol. 2007;109(2):359–363. DOI:10.1016/j.jep.2006.06.014.
- Mukherjee PK, Kumar V, Houghton PJ. Screening of Indian medicinal plants for acetylcholinesterase inhibitory activity. Phytother Res. 2007;21(12):1142–1145.
- Bonesi M, Menichini F, Tundis R, et al. Acetylcholinesterase and butyrylcholinesterase inhibitory activity of Pinus species essential oils and their constituents. J Enzyme Inhib Med Chem. 2010;25(5):622–628. DOI:10.3109/14756360903389856.
- Ingkaninan K, Temkitthawon P, Chuenchom K, et al. Screening for acetylcholinesterase inhibitory activity in plants used in Thai traditional rejuvenating and neurotonic remedies. J Ethnopharmacol. 2003;89(2–3):261–264. DOI:10.1016/j.jep.2003.08.008.
- Tundis R, Menichini F, Conforti F, et al. A potential role of alkaloid extracts from Salsola species (Chenopodiaceae) in the treatment of Alzheimer’s disease. J Enzyme Inhib Med Chem. 2009;24(3):818–824. DOI:10.1080/14756360802399662.
- Elufioye TO, Obuotor EM, Sennuga AT, et al. Acetylcholinesterase and butyrylcholinesterase inhibitory activity of some selected Nigerian medicinal plants. Rev Bras Farmacogn. 2010;20(4):472–477. DOI:10.1590/S0102-695X2010000400002.
- Cortes N, Posada-Duque RA, and Alvarez R, et al. Neuroprotective activity and acetylcholinesterase inhibition of five Amaryllidaceae species: a comparative study. Life Sci. 2015 Feb 1;122:42–50.
- Fawole OA, Amoo SO, Ndhlala AR, et al. Anti-inflammatory, anticholinesterase, antioxidant and phytochemical properties of medicinal plants used for pain-related ailments in South Africa. J Ethnopharmacol. 2010;127(2):235–241. DOI:10.1016/j.jep.2009.11.015.
- Haznedaroglu MZ, Gokce G. Comparison of anti-acetylcholinesterase activity of bulb and leaf extracts of Sternbergia candida Mathew & T. Baytop. Acta Biol Hung. 2014;65(4):396–404.
- Moyo M, Ndhlala AR, Finnie JF, et al. Phenolic composition, antioxidant and acetylcholinesterase inhibitory activities of Sclerocarya birrea and Harpephyllum caffrum (Anacardiaceae) extracts. Food Chem. 2010;123(1):69–76. DOI:10.1016/j.foodchem.2010.03.130.
- Benamar H, Tomassini L, Venditti A, et al. Acetylcholinesterase inhibitory activity of pyrrolizidine alkaloids from Echium confusum Coincy. Nat Prod Res. 2017;31(11):1277–1285.
- Zhang XD, Liu XQ, Kim YH, et al. Chemical constituents and their acetyl cholinesterase inhibitory and antioxidant activities from leaves of Acanthopanax henryi: potential complementary source against Alzheimer’s disease. Arch Pharm Res. 2014;37(5):606–616. DOI:10.1007/s12272-013-0252-x.
- Załuski D, Kuźniewski R. In vitro Anti-AChE, Anti-BuChE, and Antioxidant Activity of 12 extracts of Eleutherococcus species. Oxid Med Cell Longev. 2016;2016:4135135.
- Wszelaki N, Kuciun A, Kiss AK. Screening of traditional European herbal medicines for acetylcholinesterase and butyrylcholinesterase inhibitory activity. Acta Pharm Zagreb Croat. 2010;60(1):119–128.
- Chougouo RDK, Nguekeu YMM, Dzoyem JP, et al. Anti-inflammatory and acetylcholinesterase activity of extract, fractions and five compounds isolated from the leaves and twigs of Artemisia annua growing in Cameroon. SpringerPlus. 2016;5(1):1525.
- Niño J, Hernández JA, Correa YM, et al. In vitro inhibition of acetylcholinesterase by crude plant extracts from Colombian flora. Mem Inst Oswaldo Cruz. 2006;101(7):783–785. DOI:10.1590/S0074-02762006000700013.
- Bakthira H, Awadh Ali NA, Arnold N, et al. Anticholinesterase activity of endemic plant extracts from Soqotra. Afr J Tradit Complement Altern Med. 2011;8(3):296–299. DOI:10.4314/ajtcam.v8i3.65292.
- Besbes Hlila M, Omri A, Ben Jannet H, et al. Phenolic composition, antioxidant and anti-acetylcholinesterase activities of the Tunisian Scabiosa arenaria. Pharm Biol. 2013;51(5):525–532. DOI:10.3109/13880209.2012.746713.
- Kamal Z, Ullah F, and Ayaz M, et al. Anticholinesterse and antioxidant investigations of crude extracts, subsequent fractions, saponins and flavonoids of atriplex laciniata L.: potential effectiveness in Alzheimer’s and other neurological disorders. Biol Res. 2015 Dec;48(1):21. DOI:10.1186/s40659-015-0011-1.
- Feitosa CM, Freitas RM, Luz NNN, et al. Acetylcholinesterase inhibition by somes promising Brazilian medicinal plants. Braz J Biol Rev Brasleira Biol. 2011;71(3):783–789.
- Cespedes CL, Balbontin C, Avila JG, et al. Inhibition on cholinesterase and tyrosinase by alkaloids and phenolics from Aristotelia chilensis leaves. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc. 2017;109(Pt 2):984–995. DOI:10.1016/j.fct.2017.05.009.
- Pisano MB, Cosentino S, Viale S, et al. Biological Activities of aerial parts Extracts of Euphorbia characias. BioMed Res Int. 2016;2016:1538703.
- Saleem H, Ahmad I, Shahid MN, et al. In vitro acetylcholinesterase and butyrylcholinesterase inhibitory potentials of jatropha gossypifolia plant extracts. Acta Pol Pharm. 2016;73(2):419–423.
- Orhan I, Sener B, Choudhary MI, et al. Acetylcholinesterase and butyrylcholinesterase inhibitory activity of some Turkish medicinal plants. J Ethnopharmacol. 2004;91(1):57–60. DOI:10.1016/j.jep.2003.11.016.
- Sonibare MA, Ayoola IO, Elufioye TO. Antioxidant and acetylcholinesterase inhibitory activities of leaf extract and fractions of Albizia adianthifolia (Schumach) W.F. Wright J Basic Clin Physiol Pharmacol. 2017;28(2):143–148.
- Kim JS, Kuk E, Yu KN, et al. Antimicrobial effects of silver nanoparticles. Nanomed Nanotechnol Biol Med. 2007;3(1):95–101. DOI:10.1016/j.nano.2006.12.001.
- Rauter AP, Martins A, Lopes R, et al. Bioactivity studies and chemical profile of the antidiabetic plant Genista tenera. J Ethnopharmacol. 2009;122(2):384–393. DOI:10.1016/j.jep.2008.10.011.
- Llorent-Martínez EJ, Ortega-Barrales P, Zengin G, et al. Evaluation of antioxidant potential, enzyme inhibition activity and phenolic profile of Lathyrus cicera and Lathyrus digitatus: potential sources of bioactive compounds for the food industry. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc. 2017;107(Pt B):609–619. DOI:10.1016/j.fct.2017.03.002.
- Satheeshkumar N, Mukherjee PK, Bhadra S, et al. Acetylcholinesterase enzyme inhibitory potential of standardized extract of Trigonella foenum graecum L and its constituents. Phytomedicine Int J Phytother Phytopharm. 2010;17(3–4):292–295.
- Keskin C. Antioxidant, anticancer and anticholinesterase activities of flower, fruit and seed extracts of Hypericum amblysepalum HOCHST. Asian Pac J Cancer Prev APJCP. 2015;16(7):2763–2769.
- Béjaoui A, Ben Salem I, Rokbeni N, et al. Bioactive compounds from Hypericum humifusum and Hypericum perfoliatum: inhibition potential of polyphenols with acetylcholinesterase and key enzymes linked to type-2 diabetes. Pharm Biol. 2017;55(1):906–911. DOI:10.1080/13880209.2016.1270973.
- Zachow LL, Ávila JM, Saldanha GA, et al. Chemical composition and evaluation of prolyl oligopeptidase and acetylcholinesterase inhibitory activities of Leonurus Sibiricus L. from Brazil. Nat Prod Res. 2017;31(12):1459–1463. DOI:10.1080/14786419.2016.1255890.
- Vladimir-Knezevic S, Blazekovic B, Kindl M, et al. Acetylcholinesterase inhibitory, antioxidant and phytochemical properties of selected medicinal plants of the Lamiaceae family. Mol Basel Switz. 2014;19(1):767–782.
- Loizzo MR, Menichini F, Conforti F, et al. Chemical analysis, antioxidant, antiinflammatory and anticholinesterase activities of Origanum ehrenbergii Boiss and Origanum syriacum L. essential oils. Food Chem. 2009;117(1):174–180. DOI:10.1016/j.foodchem.2009.03.095.
- Şenol FS, Orhan I, Celep F, et al. Survey of 55 Turkish Salvia taxa for their acetylcholinesterase inhibitory and antioxidant activities. Food Chem. 2010;120(1):34–43. DOI:10.1016/j.foodchem.2009.09.066.
- Lin HQ, Ho MT, Lau LS, et al. Anti-acetylcholinesterase activities of traditional Chinese medicine for treating Alzheimer’s disease. Chem Biol Interact. 2008;175(1–3):352–354. DOI:10.1016/j.cbi.2008.05.030.
- Ferhat M, Erol E, Beladjila KA, et al. Antioxidant, anticholinesterase and antibacterial activities of Stachys guyoniana and Mentha aquatica. Pharm Biol. 2017;55(1):324–329. DOI:10.1080/13880209.2016.1238488.
- Jung HA, Ali MY, Jung HJ, et al. Inhibitory activities of major anthraquinones and other constituents from Cassia obtusifolia against β-secretase and cholinesterases. J Ethnopharmacol. 2016;191:152–160.
- Cavallaro V, Alza NP, Murray MG, et al. Alkaloids from Habranthus tubispathus and H. jamesonii, two amaryllidaceae with acetyl- and butyrylcholinesterase inhibition activity. Nat Prod Commun. 2014;9(2):159–162.
- Tung BT, Hai NT, Thu DK. Antioxidant and acetylcholinesterase inhibitory activities in vitro of different fraction of Huperzia squarrosa (Forst.) Trevis extract and attenuation of scopolamine-induced cognitive impairment in mice. J Ethnopharmacol. 2017;198:24–32.
- Tappayuthpijarn P, Itharat A, Makchuchit S. Acetylcholinesterase inhibitory activity of Thai traditional nootropic remedy and its herbal ingredients. J Med Assoc Thail Chotmaihet Thangphaet. 2011;94(7):S183–189.
- Kuk EB, Jo AR, Oh SI, et al. Anti-Alzheimer’s disease activity of compounds from the root bark of Morus alba L. Arch Pharm Res. 2017;40(3):338–349. DOI:10.1007/s12272-017-0891-4.
- Gasca CA, Castillo WO, Takahashi CS, et al. Assessment of anti-cholinesterase activity and cytotoxicity of cagaita (Eugenia dysenterica) leaves. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc. 2017;109(2):996–1002. DOI:10.1016/j.fct.2017.02.032.
- Uddin MN, Afrin R, Uddin MJ, et al. Vanda roxburghii chloroform extract as a potential source of polyphenols with antioxidant and cholinesterase inhibitory activities: identification of a strong phenolic antioxidant. BMC Complement Altern Med. 2015;15(1):195. DOI:10.1186/s12906-015-0728-y.
- Adsersen A, Gauguin B, Gudiksen L, et al. Screening of plants used in Danish folk medicine to treat memory dysfunction for acetylcholinesterase inhibitory activity. J Ethnopharmacol. 2006;104(3):418–422. DOI:10.1016/j.jep.2005.09.032.
- Georgiev MI, Alipieva K, Orhan IE. Cholinesterases inhibitory and antioxidant activities of Harpagophytum procumbens from in vitro systems. Phytother Res PTR. 2012;26(2):313–316.
- Ustun O, Senol FS, Kurkcuoglu M, et al. Investigation on chemical composition, anticholinesterase and antioxidant activities of extracts and essential oils of Turkish Pinus species and pycnogenol. Ind Crops Prod. 2012;38:115–123.
- Khadri A, Neffati M, Smiti S, et al. Antioxidant, antiacetylcholinesterase and antimicrobial activities of Cymbopogon schoenanthus L. Spreng (lemon grass) from Tunisia. LWT - Food Sci Technol. 2010;43(2):331–336. DOI:10.1016/j.lwt.2009.08.004.
- Saliu JA, Olabiyi AA. Aqueous extract of Securidaca longipendunculata Oliv. and Olax subscropioidea inhibits key enzymes (acetylcholinesterase and butyrylcholinesterase) linked with Alzheimer’s disease in vitro. Pharm Biol. 2017;55(1):252–257.
- Carpinella MC, Andrione DG, Ruiz G, et al. Screening for acetylcholinesterase inhibitory activity in plant extracts from Argentina. Phytother Res PTR. 2010;24(2):259–263. DOI:10.1002/ptr.2923.
- Wu P, Li F, Zhang J. Phytochemical compositions of extract from peel of hawthorn fruit, and its antioxidant capacity, cell growth inhibition, and acetylcholinesterase inhibitory activity. BMC Complement Altern Med. 2017;17(1):151.
- Kim CR, Choi SJ, Oh SS, et al. Rubus coreanus Miquel inhibits acetylcholinesterase activity and prevents cognitive impairment in a mouse model of dementia. J Med Food. 2013;16(9):785–792. DOI:10.1089/jmf.2012.2663.
- Osama A, Awadelkarim S, Ali A. Antioxidant activity, acetylcholinesterase inhibitory potential and phytochemical analysis of Sarcocephalus latifolius Sm. bark used in traditional medicine in Sudan. BMC Complement Altern Med. 2017;17(1):270.
- Das A, Shanker G, Nath C, et al. A comparative study in rodents of standardized extracts of Bacopa monniera and Ginkgo biloba: anticholinesterase and cognitive enhancing activities. Pharmacol Biochem Behav. 2002;73(4):893–900. DOI:10.1016/S0091-3057(02)00940-1.
- Liu YC, Su J, Wu XD, et al. Five new Lycopodium alkaloids from the aerial parts of Phlegmariurus henryi. Fitoterapia. 2016;115:148–154.
- Lam CW, Wakeman A, James A, et al. Bioactive steroidal alkaloids from Buxus macowanii Oliv. Steroids. 2015;95:73–79.
- -Rahman A-U, Khalid A, Sultana N, et al. New natural cholinesterase inhibiting and calcium channel blocking quinoline alkaloids. J Enzyme Inhib Med Chem. 2006;21(6):703–710. DOI:10.1080/14756360600889708.
- Zhao H, Zhou S, Zhang M, et al. An in vitro AChE inhibition assay combined with UF-HPLC-ESI-Q-TOF/MS approach for screening and characterizing of AChE inhibitors from roots of Coptis chinensis. Franch J Pharm Biomed Anal. 2016;120:235–240.
- Andrade MT, Lima JA, Pinto AC, et al. Indole alkaloids from Tabernaemontana australis (Muell. Arg) Miers that inhibit acetylcholinesterase enzyme. Bioorg Med Chem. 2005;13(12):4092–4095. DOI:10.1016/j.bmc.2005.03.045.
- Shin KY, Kim KY, Suh YH. Dehydroevodiamine·HCl enhances cognitive function in memory-impaired rat models. Korean J Physiol Pharmacol Off J Korean Physiol Soc Korean Soc Pharmacol. 2017;21(1):55–64.
- Chlebek J, Novák Z, Kassemová D, et al. Isoquinoline Alkaloids from Fumaria officinalis L. and their biological activities related to Alzheimer’s disease. Chem Biodivers. 2016;13(1):91–99. DOI:10.1002/cbdv.201500033.
- Lima JA, Costa RS, Epifânio RA, et al. Geissospermum vellosii stembark: anticholinesterase activity and improvement of scopolamine-induced memory deficits. Pharmacol Biochem Behav. 2009;92(3):508–513. DOI:10.1016/j.pbb.2009.01.024.
- Khalid A, Anjum S, Khan , MR, et al. Kinetics and structure-activity relationship studies on pregnane-type steroidal alkaloids that inhibit cholinesterases. Bioorg Med Chem. 2004 May;12(9):1995–2003. DOI:10.1016/j.bmc.2004.03.002.
- Ahmad H, Ahmad S, Khan E, et al. Isolation, crystal structure determination and cholinesterase inhibitory potential of isotalatizidine hydrate from Delphinium denudatum. Pharm Biol. 2017;55(1):680–686. DOI:10.1080/13880209.2016.1240207.
- Choudhary MI, Nawaz SA, Azim , MK, et al. Juliflorine: a potent natural peripheral anionic-site-binding inhibitor of acetylcholinesterase with calcium-channel blocking potential, a leading candidate for Alzheimer’s disease therapy. Biochem Biophys Res Commun. 2005 Jul;332(4):1171–1177.
- Sichaem J, Rojpitikul T, Sawasdee P, et al. Furoquinoline Alkaloids from the Leaves of Evodia lepta as potential Cholinesterase inhibitors and their molecular docking. Nat Prod Commun. 2015;10(8):1359–1362.
- Dall’Igna OP, Fett P, Gomes MW, et al. Caffeine and adenosine A(2a) receptor antagonists prevent beta-amyloid (25–35)-induced cognitive deficits in mice. Exp Neurol. 2007;203(1):241–245. DOI:10.1016/j.expneurol.2006.08.008.
- Triggle DJ, Mitchell JM, Filler RB. The Pharmacology of Physostigmine.1998. [accessed May 10, 2020]. https://www.semanticscholar.org/paper/The-Pharmacology-of-Physostigmine-Triggle-Mitchell/
- Fadaeinasab M, Basiri A, Kia Y, et al. New Indole Alkaloids from the Bark of Rauvolfia Reflexa and their Cholinesterase Inhibitory Activity. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol. 2015;37(5):1997–2011. DOI:10.1159/000438560.
- Sichaem J, Ingkaninan K, Tip-Pyang S. A novel pyrrole alkaloid from the fruit peels of Strychnos nux-blanda. Nat Prod Res. 2017;31(2):149–154.
- Rollinger JM, Schuster D, Baier E, et al. Taspine: bioactivity-guided isolation and molecular ligand-target insight of a potent acetylcholinesterase inhibitor from Magnolia x soulangiana. J Nat Prod. 2006;69(9):1341–1346. DOI:10.1021/np060268p.
- Nguyen DH, Seo UM, Zhao BT, et al. Ellagitannin and flavonoid constituents from Agrimonia pilosa Ledeb. with their protein tyrosine phosphatase and acetylcholinesterase inhibitory activities. Bioorg Chem. 2017;72:293–300.
- Labed A, Ferhat M, Labed-Zouad I, et al. Compounds from the pods of Astragalus armatus with antioxidant, anticholinesterase, antibacterial and phagocytic activities. Pharm Biol. 2016;54(12):3026–3032. DOI:10.1080/13880209.2016.1200632.
- Heo HJ, Kim MJ, Lee JM, et al. Naringenin from Citrus junos has an inhibitory effect on acetylcholinesterase and a mitigating effect on amnesia. Dement Geriatr Cogn Disord. 2004;17(3):151–157. DOI:10.1159/000076349.
- Pereira VV, Duarte LP, Silva RR, et al. New jacaranone glucoside from Jacaranda oxyphylla leaves. Nat Prod Res. 2016;30(21):2421–2428. DOI:10.1080/14786419.2016.1195378.
- Ren Y, Houghton PJ, Hider RC, et al. Novel diterpenoid acetylcholinesterase inhibitors from Salvia miltiorhiza. Planta Med. 2004;70(3):201–204.
- Kang YQ, Zhou JC, Fan PH, et al. Scapaundulin C, a novel labdane diterpenoid isolated from Chinese liverwort Scapania undulate, inhibits acetylcholinesterase activity. Chin J Nat Med. 2015;13(12):933–936. DOI:10.1016/S1875-5364(15)30100-X.
- Chung YK, Heo HJ, Kim EK, et al. Inhibitory effect of ursolic acid purified from Origanum majorana L on the acetylcholinesterase. Mol Cells. 2001;11(2):137–143.
- Chen HW, He XH, Yuan R, et al. Sesquiterpenes and a monoterpenoid with acetylcholinesterase (AchE) inhibitory activity from Valeriana officinalis var. latiofolia in vitro and in vivo. Fitoterapia. 2016;110:142–149.
- Miyazawa M, Watanabe H, Kameoka H. Inhibition of Acetylcholinesterase activity by Monoterpenoids with a p-Menthane skeleton. J Agric Food Chem. 1997;45(3):677–679.
- Dzoyem JP, Tsamo AT, Melong R, et al. Cytotoxicity, nitric oxide and acetylcholinesterase inhibitory activity of three limonoids isolated from Trichilia welwitschii (Meliaceae). Biol Res. 2015;48(1):57. DOI:10.1186/s40659-015-0049-0.
- Alves CQ, Lima LS, David JM, et al. In vitro acetylcholinesterase activity of peptide derivatives isolated from two species of Leguminosae. Pharm Biol. 2013;51(7):936–939. DOI:10.3109/13880209.2013.770536.
- Hou YC, Chao PD, Chen SY. Honokiol and magnolol increased hippocampal acetylcholine release in freely-moving rats. Am J Chin Med. 2000;28(3–4):379–384.
- Kwak HM, Jeon SY, Sohng BH, et al. beta-Secretase (BACE1) inhibitors from pomegranate (Punica granatum) husk. Arch Pharm Res. 2005;28(12):1328–1332. DOI:10.1007/BF02977896.
- O M, Mp L, B A, et al. Plants and their chemical compounds affecting betha-amyloid and secretase activity as potential sources of neuroprotective herbal medicinal products. Part 1. Herba Pol. 2010. [(accessed 2020 May 10]. http://agro.icm.edu.pl/agro/element-bwmeta1-element-agro/.
- Okello EJ, Savelev SU, Perry EK. In vitro anti-beta-secretase and dual anti-cholinesterase activities of Camellia sinensis L. (tea) relevant to treatment of dementia. Phytother Res PTR. 2004;18(8):624–627.
- Hardy J. Amyloid, the presenilins and Alzheimer’s disease. Trends Neurosci. 1997;20(4):154–159.
- Lee YK, Yuk DY, Lee JW, et al. (-)-Epigallocatechin-3-gallate prevents lipopolysaccharide-induced elevation of beta-amyloid generation and memory deficiency. Brain Res. 2009;1250:164–174.
- Descamps O, Spilman P, Zhang Q, et al. AβPP-selective BACE inhibitors (ASBI): novel class of therapeutic agents for Alzheimer’s disease. J Alzheimers Dis JAD. 2013;37(2):343–355. DOI:10.3233/JAD-130578.
- Buxbaum JD, Liu KN, Luo Y, et al. Evidence that tumor necrosis factor alpha converting enzyme is involved in regulated alpha-secretase cleavage of the Alzheimer amyloid protein precursor. J Biol Chem. 1998;273(43):27765–27767. DOI:10.1074/jbc.273.43.27765.
- Allinson TMJ, Parkin ET, Turner AJ, et al. ADAMs family members as amyloid precursor protein α-secretases. J Neurosci Res. 2003;74(3):342–352. DOI:10.1002/jnr.10737.
- Suh J, Choi SH, Romano DM, et al. ADAM10 missense mutations potentiate β-amyloid accumulation by impairing prodomain chaperone function. Neuron. 2013;80(2):385–401. DOI:10.1016/j.neuron.2013.08.035.
- Lichtenthaler SF. α-secretase in Alzheimer’s disease: molecular identity, regulation and therapeutic potential. J Neurochem. 2011;116(1):10–21.
- Postina R, Schroeder A, Dewachter I, et al. A disintegrin-metalloproteinase prevents amyloid plaque formation and hippocampal defects in an Alzheimer disease mouse model. J Clin Invest. 2004;113(10):1456–1464. DOI:10.1172/JCI20864.
- Bandyopadhyay S, Goldstein LE, Lahiri DK, et al. Role of the APP non-amyloidogenic signaling pathway and targeting alpha-secretase as an alternative drug target for treatment of Alzheimer’s disease. Curr Med Chem. 2007;14(27):2848–2864. DOI:10.2174/092986707782360060.
- De Strooper B, Vassar R, Golde T. The secretases: enzymes with therapeutic potential in Alzheimer disease. Nat Rev Neurol. 2010;6(2):99–107.
- Hong-Qi Y, Zhi-Kun S, Sheng-Di C. Current advances in the treatment of Alzheimer’s disease: focused on considerations targeting Aβ and tau. Transl Neurodegener. 2012;1(1):21.
- Adewusi EA, Steenkamp V. Medicinal plants and their derivatives with amyloid beta inhibitory activity as potential targets for drug discovery. Asian Pac J Trop Dis. 2015;5(6):430–440.
- Hanish Singh JC, Alagarsamy V, Diwan PV, et al. Neuroprotective effect of Alpinia galanga (L.) fractions on Aβ(25–35) induced amnesia in mice. J Ethnopharmacol. 2011;138(1):85–91. DOI:10.1016/j.jep.2011.08.048.
- Yan JJ, Kim DH, Moon YS, et al. Protection against beta-amyloid peptide-induced memory impairment with long-term administration of extract of Angelica gigas or decursinol in mice. Prog Neuropsychopharmacol Biol Psychiatry. 2004;28(1):25–30. DOI:10.1016/S0278-5846(03)00168-4.
- Limpeanchob N, Jaipan S, Rattanakaruna S, et al. Neuroprotective effect of Bacopa monnieri on beta-amyloid-induced cell death in primary cortical culture. J Ethnopharmacol. 2008;120(1):112–117. DOI:10.1016/j.jep.2008.07.039.
- Jeong JC, Yoon CH, Lee WH, et al. Effects of Bambusae concretio Salicea (Chunchukhwang) on amyloid beta-induced cell toxicity and antioxidative enzymes in cultured rat neuronal astrocytes. J Ethnopharmacol. 2005;98(3):259–266. DOI:10.1016/j.jep.2004.12.034.
- Ramesh BN, Indi SS, Rao KSJ. Anti-amyloidogenic property of leaf aqueous extract of Caesalpinia crista. Neurosci Lett. 2010;475(2):110–114.
- Ogunruku OO, Oboh G, Passamonti S, et al. Capsicum annuum var. grossum (Bell Pepper) Inhibits β-Secretase Activity and β-Amyloid1–40 Aggregation. J Med Food. 2017;20(2):124–130. DOI:10.1089/jmf.2016.0077.
- Gray NE, Sampath H, Zweig JA, et al. Centella asiatica Attenuates Amyloid-β-Induced oxidative stress and mitochondrial dysfunction. J Alzheimers Dis JAD. 2015;45(3):933–946. DOI:10.3233/JAD-142217.
- Frydman-Marom A, Levin A, Farfara D, et al. Orally administrated cinnamon extract reduces β-amyloid oligomerization and corrects cognitive impairment in Alzheimer’s disease animal models. PloS One. 2011;6(1):e16564. DOI:10.1371/journal.pone.0016564.
- Papandreou MA, Kanakis CD, Polissiou MG, et al. Inhibitory activity on amyloid-beta aggregation and antioxidant properties of Crocus sativus stigmas extract and its crocin constituents. J Agric Food Chem. 2006;54(23):8762–8768. DOI:10.1021/jf061932a.
- Batarseh YS, Bharate SS, Kumar V, et al. Crocus sativus extract tightens the blood-brain barrier, reduces amyloid β Load and related toxicity in 5XFAD Mice. ACS Chem Neurosci. 2017;8(8):1756–1766. DOI:10.1021/acschemneuro.7b00101.
- Kang IJ, Jeon YE, Yin XF, et al. Butanol extract of Ecklonia cava prevents production and aggregation of beta-amyloid, and reduces beta-amyloid mediated neuronal death. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc. 2011;49(9):2252–2259. DOI:10.1016/j.fct.2011.06.023.
- Bai Y, Tohda C, Zhu S, et al. Active components from Siberian ginseng (Eleutherococcus senticosus) for protection of amyloid β(25–35)-induced neuritic atrophy in cultured rat cortical neurons. J Nat Med. 2011;65(3–4):417–423. DOI:10.1007/s11418-011-0509-y.
- Lin YL, Tsay HJ, and Liao YF, et al. The components of Flemingia macrophylla attenuate amyloid β-protein accumulation by regulating amyloid β-protein metabolic pathway. Evid-Based Compl Altern Med ECAM. 2012;795843. DOI:10.1155/2012/795843.
- Lai CSW, Yu MS, Yuen WH, et al. Antagonizing beta-amyloid peptide neurotoxicity of the anti-aging fungus Ganoderma lucidum. Brain Res. 2008;1190:215–224.
- Bastianetto S, Ramassamy C, Doré S, et al. The Ginkgo biloba extract (EGb 761) protects hippocampal neurons against cell death induced by beta-amyloid. Eur J Neurosci. 2000;12(6):1882–1890. DOI:10.1046/j.1460-9568.2000.00069.x.
- Sheeja Malar D, Beema Shafreen R, Karutha Pandian S, et al. Cholinesterase inhibitory, anti-amyloidogenic and neuroprotective effect of the medicinal plant Grewia tiliaefolia - An in vitro and in silico study. Pharm Biol. 2017;55(1):381–393. DOI:10.1080/13880209.2016.1241811.
- Park H, Oh MS. Houttuyniae Herba protects rat primary cortical cells from Aβ(25–35)-induced neurotoxicity via regulation of calcium influx and mitochondria-mediated apoptosis. Hum Exp Toxicol. 2012;31(7):698–709.
- Park TS, Ryu YK, Park HY, et al. Humulus japonicus inhibits the progression of Alzheimer’s disease in a APP/PS1 transgenic mouse model. Int J Mol Med. 2017;39(1):21–30. DOI:10.3892/ijmm.2016.2804.
- Cao Z, Wang F, Xiu C, et al. Hypericum perforatum extract attenuates behavioral, biochemical, and neurochemical abnormalities in Aluminum chloride-induced Alzheimer’s disease rats. Biomed Pharmacother Biomedecine Pharmacother. 2017;91:931–937.
- Kraus B, Wolff H, Heilmann J, et al. Influence of Hypericum perforatum extract and its single compounds on amyloid-beta mediated toxicity in microglial cells. Life Sci. 2007;81(11):884–894. DOI:10.1016/j.lfs.2007.07.020.
- Ho YS, Yu MS, Lai CSW, et al. Characterizing the neuroprotective effects of alkaline extract of Lycium barbarum on beta-amyloid peptide neurotoxicity. Brain Res. 2007;1158:123–134.
- Lee JW, Lee YK, Lee BJ, et al. Inhibitory effect of ethanol extract of Magnolia officinalis and 4-O-methylhonokiol on memory impairment and neuronal toxicity induced by beta-amyloid. Pharmacol Biochem Behav. 2010;95(1):31–40. DOI:10.1016/j.pbb.2009.12.003.
- Chun YS, Kim J, Chung S, et al. Protective roles of Monsonia angustifolia and its active compounds in experimental models of Alzheimer’s disease. J Agric Food Chem. 2017;65(15):3133–3140. DOI:10.1021/acs.jafc.6b04451.
- Fujiwara H, Tabuchi M, Yamaguchi T, et al. A traditional medicinal herb Paeonia suffruticosa and its active constituent 1,2,3,4,6-penta-O-galloyl-beta-D-glucopyranose have potent anti-aggregation effects on Alzheimer’s amyloid beta proteins in vitro and in vivo. J Neurochem. 2009;109(6):1648–1657. DOI:10.1111/j.1471-4159.2009.06069.x.
- Hritcu L, Noumedem JA, Cioanca O, et al. Methanolic extract of Piper nigrum fruits improves memory impairment by decreasing brain oxidative stress in amyloid beta(1–42) rat model of Alzheimer’s disease. Cell Mol Neurobiol. 2014;34(3):437–449. DOI:10.1007/s10571-014-0028-y.
- Hage S, Kienlen-Campard P, Octave JN, et al. In vitro screening on β-amyloid peptide production of plants used in traditional medicine for cognitive disorders. J Ethnopharmacol. 2010;131(3):585–591. DOI:10.1016/j.jep.2010.07.044.
- Figueiró M, Ilha J, Linck VM, et al. The Amazonian herbal Marapuama attenuates cognitive impairment and neuroglial degeneration in a mouse Alzheimer model. Phytomedicine Int J Phytother Phytopharm. 2011;18(4):327–333.
- Irie Y, Keung WM. Rhizoma acori graminei and its active principles protect PC-12 cells from the toxic effect of amyloid-beta peptide. Brain Res. 2003;963(1–2):282–289.
- Khodagholi F, Ashabi G. Dietary supplementation with Salvia sahendica attenuates memory deficits, modulates CREB and its down-stream molecules and decreases apoptosis in amyloid beta-injected rats. Behav Brain Res. 2013;241:62–69.
- Soodi M, Saeidnia S, Sharifzadeh M, et al. Satureja bachtiarica ameliorate beta-amyloid induced memory impairment, oxidative stress and cholinergic deficit in animal model of Alzheimer’s disease. Metab Brain Dis. 2016;31(2):395–404. DOI:10.1007/s11011-015-9773-y.
- Jeong EJ, Lee HK, Lee KY, et al. The effects of lignan-riched extract of Shisandra chinensis on amyloid-β-induced cognitive impairment and neurotoxicity in the cortex and hippocampus of mouse. J Ethnopharmacol. 2013;146(1):347–354. DOI:10.1016/j.jep.2013.01.003.
- Ban JY, Cho SO, Koh SB, et al. Protection of amyloid beta protein (25–35)-induced neurotoxicity by methanol extract of Smilacis chinae rhizome in cultured rat cortical neurons. J Ethnopharmacol. 2006;106(2):230–237. DOI:10.1016/j.jep.2005.12.034.
- Prema A, Thenmozhi AJ, Manivasagam T, et al. Fenugreek seed powder nullified aluminium chloride induced memory loss, biochemical changes, Aβ burden and apoptosis via regulating Akt/GSK3β signaling pathway. PloS One. 2016;11(11):e0165955. DOI:10.1371/journal.pone.0165955.
- Fujiwara H, Iwasaki K, Furukawa K, et al. Uncaria rhynchophylla, a Chinese medicinal herb, has potent antiaggregation effects on Alzheimer’s beta-amyloid proteins. J Neurosci Res. 2006;84(2):427–433. DOI:10.1002/jnr.20891.
- Mathew M, Subramanian S. In vitro evaluation of anti-Alzheimer effects of dry ginger (Zingiber officinale Roscoe) extract. Indian J Exp Biol. 2014;52(6):606–612.
- Adewusi EA, Fouche G, Steenkamp V. Effect of four medicinal plants on amyloid-β induced neurotoxicity in SH-SY5Y cells. Afr J Tradit Complement Altern Med. 2013;10(4):6–11.
- Durairajan SSK, Yuan Q, Xie L, et al. Salvianolic acid B inhibits Abeta fibril formation and disaggregates preformed fibrils and protects against Abeta-induced cytotoxicty. Neurochem Int. 2008;52(4–5):741–750. DOI:10.1016/j.neuint.2007.09.006.
- Chu YF, Chang WH, Black RM, et al. Crude caffeine reduces memory impairment and amyloid β(1–42) levels in an Alzheimer’s mouse model. Food Chem. 2012;135(3):2095–2102. DOI:10.1016/j.foodchem.2012.04.148.
- Zhang HY, Yan H, Tang XC. Huperzine A enhances the level of secretory amyloid precursor protein and protein kinase C-alpha in intracerebroventricular beta-amyloid-(1–40) infused rats and human embryonic kidney 293 Swedish mutant cells. Neurosci Lett. 2004;360(1–2):21–24.
- Shim SB, Lee SH, Chae KR, et al. Nicotine leads to improvements in behavioral impairment and an increase in the nicotine acetylcholine receptor in transgenic mice. Neurochem Res. 2008;33(9):1783–1788. DOI:10.1007/s11064-008-9629-5.
- Xian YF, Lin ZX, and Mao QQ, et al. Bioassay-guided isolation of neuroprotective compounds from Uncaria rhynchophylla against beta-amyloid-induced neurotoxicity. Evid-Based Compl Altern Med ECAM. 2012 Jan 1;802625. DOI:10.1155/2012/802625.
- Xian YF, Lin ZX, Mao QQ, et al. Protective effect of isorhynchophylline against β-amyloid-induced neurotoxicity in PC12 cells. Cell Mol Neurobiol. 2012;32(3):353–360. DOI:10.1007/s10571-011-9763-5.
- He FQ, Qiu BY, Zhang XH, et al. Tetrandrine attenuates spatial memory impairment and hippocampal neuroinflammation via inhibiting NF-κB activation in a rat model of Alzheimer’s disease induced by amyloid-β(1–42). Brain Res. 2011;1384:89–96.
- Han J, Qu Q, Qiao J, et al. Vincamine alleviates amyloid-β 25–35 Peptides-induced cytotoxicity in PC12 cells. Pharmacogn Mag. 2017;13(49):123–128. DOI:10.4103/0973-1296.196309.
- Xu W, Yang L, Li J. Protection against β-amyloid-induced neurotoxicity by naturally occurring Z-ligustilide through the concurrent regulation of p38 and PI3-K/Akt pathways. Neurochem Int. 2016;100:44–51.
- Kim TI, Lee YK, Park SG, et al. l-Theanine, an amino acid in green tea, attenuates beta-amyloid-induced cognitive dysfunction and neurotoxicity: reduction in oxidative damage and inactivation of ERK/p38 kinase and NF-kappaB pathways. Free Radic Biol Med. 2009;47(11):1601–1610. DOI:10.1016/j.freeradbiomed.2009.09.008.
- Esposito G, De Filippis D, Maiuri MC, et al. Cannabidiol inhibits inducible nitric oxide synthase protein expression and nitric oxide production in beta-amyloid stimulated PC12 neurons through p38 MAP kinase and NF-kappaB involvement. Neurosci Lett. 2006;399(1–2):91–95. DOI:10.1016/j.neulet.2006.01.047.
- Xiang S, Liu F, Lin J, et al. Fucoxanthin inhibits β-amyloid assembly and attenuates β-amyloid oligomer-induced cognitive impairments. J Agric Food Chem. 2017;65(20):4092–4102. DOI:10.1021/acs.jafc.7b00805.
- Ban JY, Cho SO, Jeon SY. 3,4-dihydroxybenzoic acid from Smilacis chinae rhizome protects amyloid beta protein (25–35)-induced neurotoxicity in cultured rat cortical neurons. Neurosci Lett. 2007;420(2):184–188.
- Na CS, Hong SS, Choi YH, et al. Neuroprotective effects of constituents of Eragrostis ferruginea against Aβ-induced toxicity in PC12 cells. Arch Pharm Res. 2010;33(7):999–1003. DOI:10.1007/s12272-010-0704-5.
- Lee DS, Cha BY, Woo JT, et al. Acerogenin A from acer nikoense maxim prevents oxidative stress-induced neuronal cell death through Nrf2-mediated heme oxygenase-1 expression in mouse hippocampal HT22 cell line. Mol Basel Switz. 2015;20(7):12545–12557.
- Zhao L, Wang JL, Wang YR, et al. Apigenin attenuates copper-mediated β-amyloid neurotoxicity through antioxidation, mitochondrion protection and MAPK signal inactivation in an AD cell model. Brain Res. 2013;1492:33–45.
- Tan JW, Kim MK. Neuroprotective effects of biochanin A against β-amyloid-induced neurotoxicity in PC12 cells via a mitochondrial-dependent apoptosis pathway. Mol Basel Switz. 2016;21(5):548.
- Sul D, Kim HS, Lee D, et al. Protective effect of caffeic acid against beta-amyloid-induced neurotoxicity by the inhibition of calcium influx and tau phosphorylation. Life Sci. 2009;84(9–10):257–262. DOI:10.1016/j.lfs.2008.12.001.
- Lee C, Park GH, Kim C-Y, et al. [6]-Gingerol attenuates β-amyloid-induced oxidative cell death via fortifying cellular antioxidant defense system. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc. 2011;49(6):1261–1269.
- Feng HL, Fan H, Dang HZ, et al. Neuroprotective effect of curcumin to Aβ of double transgenic mice with Alzheimer’s disease. J Chin Mater Medica. 2014;39(19):3846–3849.
- Li L, Du J, Zou L, et al. The Neuroprotective effects of decursin isolated from Angelica gigas Nakai Against Amyloid β-protein-induced apoptosis in PC 12 Cells via a mitochondria-related caspase pathway. Neurochem Res. 2015;40(8):1555–1562. DOI:10.1007/s11064-015-1623-0.
- Kiasalari Z, Heydarifard R, Khalili M, et al. Ellagic acid ameliorates learning and memory deficits in a rat model of Alzheimer’s disease: an exploration of underlying mechanisms. Psychopharmacology (Berl). 2017;234(12):1841–1852. DOI:10.1007/s00213-017-4589-6.
- Richard T, Poupard P, Nassra M, et al. Protective effect of ε-viniferin on β-amyloid peptide aggregation investigated by electrospray ionization mass spectrometry. Bioorg Med Chem. 2011;19(10):3152–3155. DOI:10.1016/j.bmc.2011.04.001.
- Mori T, Koyama N, Guillot-Sestier MV, et al. Ferulic acid is a nutraceutical β-secretase modulator that improves behavioral impairment and Alzheimer-like pathology in transgenic mice. PloS One. 2013;8(2):e55774. DOI:10.1371/journal.pone.0055774.
- Ban JY, Nguyen HTT, Lee HJ, et al. Neuroprotective properties of gallic acid from Sanguisorbae radix on amyloid beta protein (25–35) - induced toxicity in cultured rat cortical neurons. Biol Pharm Bull. 2008;31(1):149–153. DOI:10.1248/bpb.31.149.
- Liu R, Meng F, Zhang L, et al. Luteolin isolated from the medicinal plant Elsholtzia rugulosa (Labiatae) prevents copper-mediated toxicity in β-amyloid precursor protein Swedish mutation overexpressing SH-SY5Y cells. Mol Basel Switz. 2011;16(3):2084–2096.
- Cai L, Wang LF, Pan JP, et al. Neuroprotective effects of methyl 3,4-Dihydroxybenzoate against TBHP-induced oxidative damage in SH-SY5Y cells. Mol Basel Switz. 2016;21(8):1071.
- Onozuka H, Nakajima A, Matsuzaki K, et al. Nobiletin, a citrus flavonoid, improves memory impairment and Abeta pathology in a transgenic mouse model of Alzheimer’s disease. J Pharmacol Exp Ther. 2008;326(3):739–744. DOI:10.1124/jpet.108.140293.
- Kim DH, Kim S, Jeon SJ, et al. The effects of acute and repeated oroxylin A treatments on Abeta(25–35)-induced memory impairment in mice. Neuropharmacology. 2008;55(5):639–647. DOI:10.1016/j.neuropharm.2008.05.019.
- Li J, Ji X, Zhang J, et al. Paeoniflorin attenuates Aβ25–35-induced neurotoxicity in PC12 cells by preventing mitochondrial dysfunction. Folia Neuropathol. 2014;52(3):285–290. DOI:10.5114/fn.2014.45569.
- Yoon JH, Youn K, Ho CT. p-Coumaric acid and ursolic acid from Corni fructus attenuated β-amyloid(25–35)-induced toxicity through regulation of the NF-κB signaling pathway in PC12 cells. J Agric Food Chem. 2014;62(21):4911–4916.
- Zhang L, Yu H, Zhao X, et al. Neuroprotective effects of salidroside against beta-amyloid-induced oxidative stress in SH-SY5Y human neuroblastoma cells. Neurochem Int. 2010;57(5):547–555. DOI:10.1016/j.neuint.2010.06.021.
- Iuvone T, De Filippis D, Esposito G, et al. The spice sage and its active ingredient rosmarinic acid protect PC12 cells from amyloid-beta peptide-induced neurotoxicity. J Pharmacol Exp Ther. 2006;317(3):1143–1149. DOI:10.1124/jpet.105.099317.
- Wang S, Wang YJ, Su Y, et al. Rutin inhibits β-amyloid aggregation and cytotoxicity, attenuates oxidative stress, and decreases the production of nitric oxide and proinflammatory cytokines. Neurotoxicology. 2012;33(3):482–490. DOI:10.1016/j.neuro.2012.03.003.
- Lee YW, Kim DH, Jeon SJ, et al. Neuroprotective effects of salvianolic acid B on an Aβ25–35 peptide-induced mouse model of Alzheimer’s disease. Eur J Pharmacol. 2013;704(1–3):70–77. DOI:10.1016/j.ejphar.2013.02.015.
- Lu P, Mamiya T, Lu LL, et al. Silibinin attenuates amyloid beta(25–35) peptide-induced memory impairments: implication of inducible nitric-oxide synthase and tumor necrosis factor-alpha in mice. J Pharmacol Exp Ther. 2009;331(1):319–326. DOI:10.1124/jpet.109.155069.
- Yin F, Liu J, Ji X, et al. Silibinin: a novel inhibitor of Aβ aggregation. Neurochem Int. 2011;58(3):399–403. DOI:10.1016/j.neuint.2010.12.017.
- Kwon SH, Ma SX, Hwang JY, et al. Involvement of the Nrf2/HO-1 signaling pathway in sulfuretin-induced protection against amyloid beta 25–35 neurotoxicity. Neuroscience. 2015;304:14–28.
- Zhao LX, Wang Y, Liu T, et al. α-Mangostin decreases β-amyloid peptides production via modulation of amyloidogenic pathway. CNS Neurosci Ther. 2017;23(6):526–534. DOI:10.1111/cns.12699.
- Zhou YQ, Yang ZL, Xu L, et al. Akebia saponin D, a saponin component from Dipsacus asper Wall, protects PC 12 cells against amyloid-beta induced cytotoxicity. Cell Biol Int. 2009;33(10):1102–1110. DOI:10.1016/j.cellbi.2009.06.028.
- Fujihara K, Koike S, Ogasawara Y, et al. Inhibition of amyloid β aggregation and protective effect on SH-SY5Y cells by triterpenoid saponins from the cactus Polaskia chichipe. Bioorg Med Chem. 2017;25(13):3377–3383. DOI:10.1016/j.bmc.2017.04.023.
- Chen DL, Zhang P, Lin L, et al. Protective effect of Bajijiasu against β-amyloid-induced neurotoxicity in PC12 cells. Cell Mol Neurobiol. 2013;33(6):837–850. DOI:10.1007/s10571-013-9950-7.
- Gao Y, Li C, Yin J, et al. Fucoidan, a sulfated polysaccharide from brown algae, improves cognitive impairment induced by infusion of Aβ peptide in rats. Environ Toxicol Pharmacol. 2012;33(2):304–311. DOI:10.1016/j.etap.2011.12.022.
- Wang P, Liao W, Fang J, et al. A glucan isolated from flowers of Lonicera japonica Thunb. inhibits aggregation and neurotoxicity of Aβ42. Carbohydr Polym. 2014;110:142–147.
- Wang H, Xu Y, Yan J, et al. Acteoside protects human neuroblastoma SH-SY5Y cells against beta-amyloid-induced cell injury. Brain Res. 2009;1283:139–147.
- Ji ZN, Dong TTX, Ye WC, et al. Ginsenoside Re attenuate beta-amyloid and serum-free induced neurotoxicity in PC12 cells. J Ethnopharmacol. 2006;107(1):48–52. DOI:10.1016/j.jep.2006.02.004.
- Misiti F, Sampaolese B, Mezzogori D, et al. Protective effect of rhubarb derivatives on amyloid beta (1–42) peptide-induced apoptosis in IMR-32 cells: a case of nutrigenomic. Brain Res Bull. 2006;71(1–3):29–36. DOI:10.1016/j.brainresbull.2006.07.012.
- Jayaprakasam B, Padmanabhan K, Nair MG. Withanamides in Withania somnifera fruit protect PC-12 cells from beta-amyloid responsible for Alzheimer’s disease. Phytother Res PTR. 2010;24(6):859–863.
- Yu Y, Zhou L, Sun M, et al. Xylocoside G reduces amyloid-β induced neurotoxicity by inhibiting NF-κB signaling pathway in neuronal cells. J Alzheimers Dis JAD. 2012;30(2):263–275. DOI:10.3233/JAD-2012-110779.
- Seo JY, Lim SS, Kim J, et al. Alantolactone and Isoalantolactone prevent amyloid β25–35 -induced toxicity in mouse cortical neurons and scopolamine-induced cognitive impairment in mice. Phytother Res PTR. 2017;31(5):801–811. DOI:10.1002/ptr.5804.
- Rasoolijazi H, Azad N, and Joghataei MT, et al. The protective role of carnosic acid against beta-amyloid toxicity in rats. Sci World J. 2013;917082. DOI:10.1155/2013/917082.
- Mei Z, Zhang F, Tao L, et al. Cryptotanshinone, a compound from Salvia miltiorrhiza modulates amyloid precursor protein metabolism and attenuates beta-amyloid deposition through upregulating alpha-secretase in vivo and in vitro. Neurosci Lett. 2009;452(2):90–95. DOI:10.1016/j.neulet.2009.01.013.
- Azadmehr A, Oghyanous KA, Hajiaghaee R, et al. Antioxidant and neuroprotective effects of Scrophularia striata extract against oxidative stress-induced neurotoxicity. Cell Mol Neurobiol. 2013;33(8):1135–1141. DOI:10.1007/s10571-013-9979-7.
- Chen X, Guo C, Kong J. Oxidative stress in neurodegenerative diseases. Neural Regen Res. 2012;7(5):376–385.
- Liu Z, Zhou T, and Ziegler AC, et al. Oxidative stress in neurodegenerative diseases: from molecular mechanisms to clinical applications. Oxid Med Cell Longev. 2017 Oct;2525967. DOI:10.1155/2017/2525967.
- Paola D, Domenicotti C, Nitti M, et al. Oxidative stress induces increase in intracellular amyloid beta-protein production and selective activation of betaI and betaII PKCs in NT2 cells. Biochem Biophys Res Commun. 2000;268(2):642–646. DOI:10.1006/bbrc.2000.2164.
- Hampel H, Prvulovic D, Teipel S, et al. The future of Alzheimer’s disease: the next 10 years. Prog Neurobiol. 2011;95(4):718–728. DOI:10.1016/j.pneurobio.2011.11.008.
- Wilkinson D. Pharmacotherapy of Alzheimer’s disease. Psychiatry. 2005;4(1):43–47.
- Witt A, Macdonald N, Kirkpatrick P. Memantine hydrochloride. Nat Rev Drug Discov. 2004;3(2):109–110.
- Lleo A, Greenberg SM, Growdon JH. Current pharmacotherapy for Alzheimer’s disease. Annu Rev Med. 2006;57(1):513–533.
- Mudher A, Lovestone S. Alzheimer’s disease-do tauists and baptists finally shake hands? Trends Neurosci. 2002;25(1):22–26.
- Costantino L, Barlocco D. Designed multiple ligands: basic research vs clinical outcomes. Curr Med Chem. 2012;19(20):3353–3387.
- Cavalli A, Bolognesi ML, Minarini A, et al. Multi-target-directed ligands to combat neurodegenerative diseases. J Med Chem. 2008;51(3):347–372. DOI:10.1021/jm7009364.
- Nasir MN, Abdullah J, Habsah M, et al. Inhibitory effect of asiatic acid on acetylcholinesterase, excitatory post synapticpotential and locomotor activity. Phytomedicine. 2012;19(3–4):311–316. DOI:10.1016/j.phymed.2011.10.004.
- Patil SP, Maki S, Khedkar SA, et al. Withanolide A and asiatic acid modulate multiple targets associated with amyloid-beta precursor protein processing and amyloid-beta protein clearance. J Nat Prod. 2010;73(7):1196–1202. DOI:10.1021/np900633j.
- Zhang X, Wu J, Dou Y, et al. Asiatic acid protects primary neurons against C2-ceramide-induced apoptosis. Eur J Pharmacol. 2012;679(1–3):51–59. DOI:10.1016/j.ejphar.2012.01.006.
- Jia L, Liu J, Song Z, et al. Berberine suppresses amyloid-beta-induced inflammatory response in microglia by inhibiting nuclear factor-kappaB and mitogen-activated protein kinase signalling pathways. J Pharm Pharmacol. 2012;64(10):1510–1521. DOI:10.1111/j.2042-7158.2012.01529.x.
- Ji HF, Shen L. Berberine: a potential multipotent natural product to combat Alzheimer’s disease. Mol Basel Switz. 2011;16(8):6732–6740.
- Baum L, Ng A. Curcumin interaction with copper and iron suggests one possible mechanism of action in Alzheimer’s disease animal models. J Alzheimers Dis JAD. 2004;6(4):367–377–-449.
- Hamaguchi T, Ono K, Yamada M. REVIEW: curcumin and Alzheimer’s disease. CNS Neurosci Ther. 2010;16(5):285–297.
- Jaques JADS, Rezer JFP, Carvalho FB, et al. Curcumin protects against cigarette smoke-induced cognitive impairment and increased acetylcholinesterase activity in rats. Physiol Behav. 2012;106(5):664–669. DOI:10.1016/j.physbeh.2012.05.001.
- Mutsuga M, Chambers JK, Uchida K, et al. Binding of curcumin to senile plaques and cerebral amyloid angiopathy in the aged brain of various animals and to neurofibrillary tangles in Alzheimer’s brain. J Vet Med Sci. 2012;74(1):51–57. DOI:10.1292/jvms.11-0307.
- Narasingapa RB, Jargaval MR, Pullabhatla S, et al. Activation of α-secretase by curcumin-aminoacid conjugates. Biochem Biophys Res Commun. 2012;424(4):691–696. DOI:10.1016/j.bbrc.2012.07.010.
- Villaflores OB, Chen YJ, Chen CP, et al. Curcuminoids and resveratrol as anti-Alzheimer agents. Taiwan J Obstet Gynecol. 2012;51(4):515–525. DOI:10.1016/j.tjog.2012.09.005.
- Hoerr R, Noeldner M. Ensaculin (KA-672 HCl): a multitransmitter approach to dementia treatment. CNS Drug Rev. 2002;8(2):143–158.
- Teismann P, Ferger B. Effects of ensaculin on dopamine metabolite levels and K(+)-induced glutamate release. Eur J Pharmacol. 2000;398(2):247–250.
- Bieschke J, Russ J, Friedrich RP, et al. EGCG remodels mature alpha-synuclein and amyloid-beta fibrils and reduces cellular toxicity. Proc Natl Acad Sci U S A. 2010;107(17):7710–7715. DOI:10.1073/pnas.0910723107.
- Jeon SY, Bae K, Seong YH, et al. Green tea catechins as a BACE1 (beta-secretase) inhibitor. Bioorg Med Chem Lett. 2003;13(22):3905–3908. DOI:10.1016/j.bmcl.2003.09.018.
- Mandel SA, Amit T, Weinreb O, et al. Simultaneous manipulation of multiple brain targets by green tea catechins: a potential neuroprotective strategy for Alzheimer and Parkinson diseases. CNS Neurosci Ther. 2008;14(4):352–365. DOI:10.1111/j.1755-5949.2008.00060.x.
- Smith A, Giunta B, Bickford PC, et al. Nanolipidic particles improve the bioavailability and alpha-secretase inducing ability of epigallocatechin-3-gallate (EGCG) for the treatment of Alzheimer’s disease. Int J Pharm. 2010;389(1–2):207–212. DOI:10.1016/j.ijpharm.2010.01.012.
- Jiang H, Wang X, Huang L, et al. Benzenediol-berberine hybrids: multifunctional agents for Alzheimer’s disease. Bioorg Med Chem. 2011;19(23):7228–7235. DOI:10.1016/j.bmc.2011.09.040.
- Yan JJ, Jung JS, Kim TK, et al. Protective effects of ferulic acid in amyloid precursor protein plus presenilin-1 transgenic mouse model of Alzheimer disease. Biol Pharm Bull. 2013;36(1):140–143. DOI:10.1248/bpb.b12-00798.
- Hoi CP, Ho YP, Baum L, et al. Neuroprotective effect of honokiol and magnolol, compounds from Magnolia officinalis, on beta-amyloid-induced toxicity in PC12 cells. Phytother Res PTR. 2010;24(10):1538–1542. DOI:10.1002/ptr.3178.
- Ono K, Li L, Takamura Y, et al. Phenolic compounds prevent amyloid β-protein oligomerization and synaptic dysfunction by site-specific binding. J Biol Chem. 2012;287(18):14631–14643. DOI:10.1074/jbc.M111.325456.
- Shimmyo Y, Kihara T, Akaike A. Three distinct neuroprotective functions of myricetin against glutamate-induced neuronal cell death: involvement of direct inhibition of caspase-3. J Neurosci Res. 2008;86(8):1836–1845.
- Ali T, Yoon GH, Shah SA, et al. Osmotin attenuates amyloid beta-induced memory impairment, tau phosphorylation and neurodegeneration in the mouse hippocampus. Sci Rep. [Internet]. 2015;5(1). 10.1038/srep11708.
- Shah SA, Yoon GH, Chung SS, et al. Osmotin reduced amyloid beta (Aβ) burden by inhibiting SREBP2 expression in APP/PS1 mice. Mol Psychiatry. 2017;22(3):323. DOI:10.1038/mp.2017.12.
- Kim J, Lee HJ, Lee KW. Naturally occurring phytochemicals for the prevention of Alzheimer’s disease. J Neurochem. 2010;112(6):1415–1430.
- Choi CW, Choi YH, Cha MR, et al. In vitro BACE-1 inhibitory activity of resveratrol oligomers from the seed extract of Paeonia lactiflora. Planta Med. 2011;77(4):374–376. DOI:10.1055/s-0030-1250370.
- Feng X, Liang N, Zhu D, et al. Resveratrol inhibits β-amyloid-induced neuronal apoptosis through regulation of SIRT1-ROCK1 signaling pathway. PloS One. 2013;8(3):e59888. DOI:10.1371/journal.pone.0059888.
- Frozza RL, Bernardi A, Hoppe JB, et al. Neuroprotective effects of resveratrol against Aβ administration in rats are improved by lipid-core nanocapsules. Mol Neurobiol. 2013;47(3):1066–1080. DOI:10.1007/s12035-013-8401-2.
- Vingtdeux V, Dreses-Werringloer U, and Zhao H, et al. Therapeutic potential of resveratrol in Alzheimer’s disease. BMC Neurosci. 2008 Dec;9(2):S6. DOI:10.1186/1471-2202-9-S2-S6.
- Braidy N, Jugder B-E, Poljak A, et al. Molecular targets of tannic acid in Alzheimer’s Disease. Curr Alzheimer Res. [Internet]. 2017;14(8). 10.2174/1567205014666170206163158.