Abstract
The effect of microwave cooking, steaming, stir-frying and boiling on the antioxidant potential of bell peppers (red and green), jalapeno, cayenne, serrano and habanero peppers was investigated. Antioxidant potential was measured employing two techniques: FRAP and DPPH, and the values reported based on dry matter content. Microwaving increased the antioxidant potential of all peppers studied 2 to 10 times above corresponding raw samples. Stir-frying and steaming resulted in enhancement of the antioxidant concentrations of peppers (1.4–3 and 1.6 − 7.3 times, respectively) above raw samples except steamed red bell peppers and stir-fried habanero peppers which yielded values below raw samples. Boiling resulted in a decrease in the antioxidant concentration of most peppers except those with medium pungency (jalapeno and serrano), which yielded values two and four times above raw samples, respectively. All cooked peppers showed an increase in antiradical activity (1.1–8.9 times) above raw samples, except boiled bell peppers (green) and cayenne peppers, which showed antiradical activities of 1.0 and 0.7 respectively below the raw samples. Our findings suggest that cooking most peppers with techniques such as microwave cooking and stir-frying that involve no addition of water may be a better way to prepare them as this generally increases the antioxidant potential of the peppers.
PUBLIC INTEREST STATEMENT
Peppers are widely consumed globally due to the health benefits that are associated with the substances that they contain. Even though in some instances, the peppers are consumed raw such as in salads, a majority of consumption involves some form of heat treatment. The effect of heat on the beneficial substances in the peppers is a matter of great interest. The present study aims to evaluate the effect of various types of heat applications on the beneficial substances in various types of peppers. Microwave cooking, stir-frying and other heat application methods that do not involve adding much water like boiling are presented as better ways of preparing the peppers for consumption with minimal loss of the beneficial substances.
1. Introduction
Peppers are gaining popularity in recent years due to their numerous health-promoting benefits. These benefits coupled with their attractive colors, aroma and taste have greatly increased the use of peppers. Healthful benefits range from reduction in gastric ulcers, cancer and cardiovascular diseases to the easing of pains associated with arthritis, headaches and muscle strains (Srinivasan, Citation2005). These healthful benefits are believed to be due to the large amounts of bioactive compounds (antioxidants) in the peppers, such as phenolic acids, flavonoids, carotenoids and vitamins. Capsaicinoids and more primarily the capsaicin molecule are responsible for the burning sensation in peppers and their pungent smell (Surh & Lee, Citation1996). Peppers may be consumed raw as in salads or cooked. Studies on the effect of cooking on the antioxidant potential of peppers are quite varied, with emerging arguments for an increase (Erguder et al., Citation2007; Hamed et al., Citation2019; Ornelas-Paz et al., Citation2010; Shobana & Naidu, Citation2000; Turkmen et al., Citation2005), a decrease (Chuah et al., Citation2008; Hwang et al., Citation2012; Jimenes-Monreal et al., Citation2009; Loizzo et al., Citation2013; Ornelas-Paz et al., Citation2010) or no effect (Jimenes-Monreal et al., Citation2009; Shobana & Naidu, Citation2000) in the antioxidant potential of peppers when cooked.
Indeed, studies by Turkmen and others (Turkmen et al., Citation2005) found the total phenolic content, and antioxidant activity of green peppers to increase when these peppers were boiled, steamed or microwaved. Similar findings were registered by Shobana and others (Shobana & Naidu, Citation2000) who showed that boiling of pepper extracts at 100°C for 30 minutes retained or increased their antioxidant activity leading them to conclude that the spice constituents were stable to thermal denaturation. Recent studies (Ornelas-Paz et al., Citation2010) on the other hand found that whereas boiling and grilling increased the total phenolic content of pungent peppers, these cooking techniques decreased the total phenolic content of non-pungent peppers. Thermal treatment of black pepper was found to cause a significant increase in the content of phenolic substances in the peppers (Horvathova et al., Citation2007) whereas, microwave drying was found (Arslan & Ozcan, Citation2011) to cause a higher increase in the antioxidant activity of red pepper compared to sun drying. Yet, studies by Choi (Choi, Citation1996) using red pepper extract (capsaicin) showed moderate antioxidant activity at 100°C and no activity at 150°C, an effect that they attributed to the possible degradation of capsaicin.
Studies with green pepper found them to lose their Free radical scavenging ability when either boiled, pressure cooked, microwaved, fried or even grilled but not when baked, yet, insignificant losses were registered with the ABTS radical anion (Jimenes-Monreal et al., Citation2009), agreeing with those of Loizzo et al. (Loizzo et al., Citation2013) that showed that frying decreased the antioxidant potential of bell peppers. Other recent studies (Chuah et al., Citation2008) with colored bell peppers; (green, red and yellow), showed that there was only a slight decrease in the radical scavenging activity in these peppers when boiled, microwaved or stir fried for 5 minutes, and a significant decrease if boiled for more than 5 minutes. Studies by Hwang (Hwang et al., Citation2012) and others found that boiling, steaming, stir-frying or roasting reduced the antioxidant properties of red peppers, agreeing with those of Erguder et al. (Erguder et al., Citation2007) who showed that boiling, microwave cooking or baking decreased the antioxidant activity of peppers. Recent studies by Hamed et al. equally showed that the antioxidant potential of various peppers especially at the mature stages decreased after roasting (Hamed et al., Citation2019). These studies roughly map those of Schweiggert and others (Schweiggert et al., Citation2006) who studied the effect of blanching and storage on the capsaicin activity of chili peppers and found that heating and drying resulted in a degradation of the initial capsaicin content.
The FRAP method (Benzie & Strain, Citation1996) measures the ability of the reductant (antioxidants in peppers) to reduce the ferric to the ferrous complex whose magnitude grows with time and the DPPH method (Brand-Williams et al., Citation1995) measures the ability of the antioxidants in the peppers to reduce the free radical DPPH whose magnitude decreases with time. The peppers used in this study were those commercially available in the Southeastern United States of America. To our knowledge, there is a scarcity of published reports on the effect of these cooking methods on the antioxidant potential of peppers commercially available and largely consumed in Southeastern United States. It is believed that such an understanding could suggest better ways to prepare the peppers in order to reap maximum healthful benefits.
The foregoing statements indicate a continual need for more information on the effect of cooking (or heat) on the antioxidant potential of various peppers. These arguments may stem from variations in the type of peppers used, the method of cooking of the peppers, and the type of analytical techniques employed. Addressing this effect is a rather rigorous undertaking given the wide range of varying parameters; and how the pepper cultivars, total phenolics, or even target molecules such as capsaicinoids may govern this. The objective of this study is to shed more light on this topic by employing two comparable techniques (DPPH and FRAP), to address the effect of an array of cooking methods (boiling, steaming, stir-frying and microwave cooking), on a broad spectrum of peppers (jalapeno, serrano, cayenne and habanero and bell peppers), of varying pungency.
2. Experimental methods
2.1. Materials
2.1.1. Chemicals
Iron (II) sulfate heptahydrate (FeSO4.7H2O, >99.99%), Iron (III) chloride hexahydrate (FeCl3.6H2O, >99%), hydrochloric acid (ACS plus), acetic acid (ACS plus), methanol and ethanol (both HPLC grade) were all obtained from Fisher Scientific, Pittsburg, PA. The free radical 2, 2-diphenyl-1-picrylhydrazyl (DPPH) was obtained from Sigma, St. Louis, MO and 4, 6-tri-(2-pyridyl)-s-triazine (TPTZ, 99%) was obtained from Acros organics, West Chester, PA.
2.1.2. Equipment
The Daigger 22685D vortex-mixer (Vernon Hills, Illinois, USA) was used in methanol extraction and A Sorvall Legend X1 centrifuge (Thermo Scientific, Waltham, Massachusetts, USA) was used to separate sample extracts before filtration. A commercial 700 W microwave, oven (Sunbeam SBM 7700 W, Sunbeam, Boca Raton FL, USA) was used to microwave the samples, whereas steaming was done on an Ester Inspire 4751, Boca Raton, FL, USA. Spectrophotometric measurements for both the FRAP reaction and the DPPH experiments were performed on a Cintra 6 Double Beam UV-Visible spectrometer (GBC Scientific Equipment, Victoria, Australia).
2.1.3. Plant material
Fresh peppers were obtained from local vendors and markets throughout northeast Alabama. The fruits were carefully selected to ensure only the ones without damage or blemishes were used. The peppers were washed carefully with tap water, rinsed with distilled de-ionized water, dried on paper towels and cut carefully to remove the inedible parts, before they were well mixed and then sampled out for cooking. The peppers were classified according to their pungency (degree of hotness), which was generally linked to the presence of heat-producing molecules (capsaicinoids), of which capsaicin is the primary one, with the assumption that the greater the concentration of these compounds the hotter the pepper tastes. Accordingly, therefore, based on the Scoville chart the bell peppers (green and red peppers) were classified as non-pungent (or sweet peppers), serrano and jalapeno, as peppers with mild pungency and cayenne and habanero as peppers with medium pungency.
2.2. Cooking and preparation of pepper samples
Randomly selected peppers were used in this study. For each cultivar, the peppers were cut into small pieces and mixed. These were then divided into six portions (75 g each), for each of the cooking processes. The peppers were cooked in triplicates to ensure reproducibility.
2.2.1. Cooking procedures
In the microwave cooking process, samples were placed in a microwave dish and cooked on medium for 2 minute, without scarring. For boiling, the samples were added to 250 mL in a stainless steel pan cooked for 2 minutes, drained and cooled, whereas in the steaming process the samples were placed on a steaming tray above boiling water at 100°C, covered and steamed for 5 minutes. Stir-frying of samples was effected by placing them on a hot pan at 300°C with 2.89 g of vegetable cooking oil (great value) and sautéed for 5 minutes. Cooked Samples from all these methods underwent the extraction process immediately.
2.2.2. Extraction process
All the pepper samples (raw and cooked) were processed by dipping in liquid nitrogen and crushing into powder using a mortar and pestle and extracted immediately. In the Extraction process, 5-g samples in 20 mL solution mixture 90% methanol and 10% acetic acid were vortexed on high for 5 minutes and centrifuged (403 RCF) for 10 minutes. The extraction process was repeated 3 times and the extracts combined before analysis.
2.2.3. Dry weight analysis
Dry weight analysis was done using a conventional oven at 70°C. In this case, triplicate 5-g portions of the peppers were dried to constant mass for 24–36 hours. This was done to determine the moisture content of which was used to calculate the dry weight of the peppers for all measurements.
2.3. Determination of antioxidant activity
The antioxidant potential of the pepper samples was assayed by the Ferric Antioxidant reducing Power (FRAP) method described elsewhere (Benzie & Strain, Citation1996). Briefly, acetate buffer (pH 3.6), 0.02 M FeCl3, and 2, 4, 6,-tripyridyl-s-triazine (TPTZ) in 0.04 M of HCl were used to prepare the FRAP reagent. The FRAP reagent was prepared by mixing 2.5 mL of Iron (III) chloride, 2.5 mL of TPTZ and 25 mL of acetate buffer. For UV-vis analysis, 100 µL of sample was added to 2600 µL of FRAP reagent, 300 µL of distilled de-ionized water and the reaction (standard FRAP reaction) absorbance monitored at 595 nm, on a spectrum ranging from 200 to 700 nm. Ferrous ion solutions of varying concentration measured under similar conditions were used to calibrate the UV-vis spectra. The ferrous ion solutions yielded a linear relationship (with a regression, R2 = 0.998 and n = 6), this was thereby used in the spectra calibration yielding a ferrous Antioxidant Equivalent Value (AEV).
The antiradical activity measurements were done using the free radical DPPH and the method of Brand-Williams and others (Brand-Williams et al., Citation1995). In this case, the sample was extracted in the ethanol, vortexed and centrifuged before analysis. In the UV-vis analysis, a 1:3 DPPH to sample ratio in a 3 mL total volume was used and the loss of the free radical DPPH monitored at 516 nm. In both processes (FRAP and DPPH), the standard sample measurements were compared against blank determinations performed under similar conditions without pepper sample extracts.
2.4. Statistical analysis
The cooking of peppers by any given method, the dry weight analysis of the samples and the extraction and analytical procedures were repeated three times. Statistical analysis of the data was done using Sigma Plot Graph and Data Analysis software 11.2 for windows (SYSTAT Software Inc, San Jose, CA, USA). Differences between variables were duly tested for significant differences using analysis of variance, and significance was set at p ≤ 0.05.
3. Results and discussion
3.1. Total antioxidant concentrations
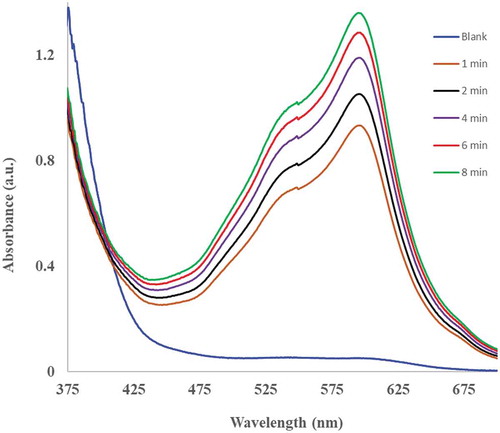
The antioxidant potential of all pepper sample extracts cooked or prepared in various ways was measured using the FRAP method. Figure shows such typical absorbance spectra (from such measurements) of the habanero pepper sample extract obtained in a standard FRAP reaction containing 500 µL of the extract with 2.2 mL of FRAP reagent and 300 µL of water. The signal corresponded to the colored complex obtained by the reduction of the ferric Fe (III)-TPTZ complex to the ferrous Fe (II)-TPTZ complex by the reductant (habanero pepper extract). Similar results (absorbance measurements) were obtained when other pepper sample extracts were assayed with FRAP reagent, and the results were recorded based on a dry matter content (Figure ). The microwaved samples from the non-pungent bell peppers (green and red) were found to contain the highest concentration of antioxidants compared to all the other non-pungent pepper samples, Figure . The concentration of antioxidants in these samples followed the order; microwaved> stir-fried > steamed >raw > boiled (Figure ). In this case, boiling the peppers resulted in a 64% and 90% reduction in antioxidant concentration in the green and red bell peppers, respectively, compared to raw non-pungent peppers. The registered higher concentration of antioxidant in the green bell pepper samples agrees with the findings of others (Lin & Tang, Citation2007; Ornelas-Paz et al., Citation2010) and is likely due to the fact, raw green peppers have been found to contain higher phenolic content than raw-red bell peppers (Chuah et al., Citation2008), giving the possibility of the green peppers registering high antioxidant yields even when cooked. Similarly, even though the capsaicin content of the peppers within the same cultivar increases with ripening (Cisneros-Pineda et al., Citation2007), heat treatment can cause loss of capsaicinoids in the red peppers more than the green ones (Suresh et al., Citation2007).
Figure 1. UV-vis spectra showing the signal generation and increase over time when 500 µL of habanero pepper was run with 2.2 mL of FRAP reagent and 300 µL of water were run in a standard FRAP reaction and the reaction followed at 595 nm over time
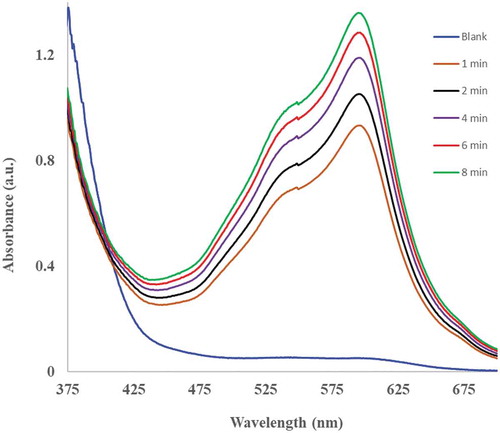
Figure 2. Effect of various cooking methods on the antioxidant potential, Antioxidant Equivalent Values (AEV) of red and green bell peppers, serrano and jalapeno and cayenne and habanero peppers. The AEV values are a measure of the ferrous iron II equivalent reducing power obtained using Fe (II) standards in place of the peppers in a standard reaction consisting of 500 µL of pepper sample with 2.2 mL FRAP reagent and 300-µL water. The values are means ± standard deviation of triplicate determinations, at p < 0.05
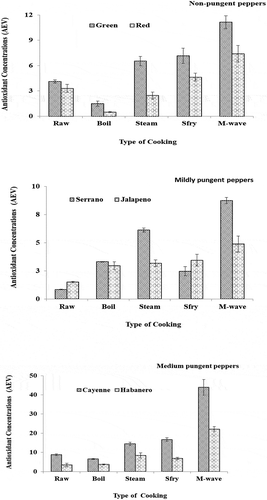
Serrano and jalapeno peppers represented the peppers with medium pungency. In the case of serrano peppers, microwaved samples equally yielded the highest concentration of antioxidants and the antioxidant concentration in the samples followed the order; microwaved > stir-fried > steamed > boiled > raw (Figure ). In this case, the microwaved serrano samples had antioxidant concentrations ten times higher than the raw samples whereas the boiled samples had concentrations 4 times more than the raw samples (Figure , Table ). On the other hand, even though the jalapeno microwaved pepper samples had the highest antioxidant concentrations, the concentrations in the various cooked jalapeno samples were considerably lower and varied widely in content from those in the serrano peppers. These reductions followed the order; microwaved > stir-fried ≥ steamed ≥ boiled > raw. In the case of jalapeno peppers, the antioxidant concentration in the stir-fry, steam and boiled samples was roughly equal and much higher than those in the raw samples (Figure ), Table ). The cooked serrano pepper samples registered higher antioxidant concentrations than the cooked jalapeno pepper samples, this agrees with the findings of (Ornelas-Paz et al., Citation2010), who found this to be due to the higher capsaicinoids content in the raw serrano more than in the jalapeno, thereby raising the possibility of the serrano peppers registering higher antioxidant concentrations even when cooked.
Table 1. Effect of various cooking methods on the antioxidant potential of peppers measured by FRAP technique
In the case of the mild pungent peppers (cayenne and habanero); cayenne pepper samples, were found to have much higher concentration of antioxidants in all samples compared to the habanero peppers (Figure ). These findings roughly agrees with those of others (Ornelas-Paz et al., Citation2010), who found raw habanero pepper samples to contain the lowest amount of raw phenolic content in a group with serrano, bell peppers and others. In this study, the antioxidant in the cooked habanero and cayenne samples followed an order that exactly matched that of the non-pungent peppers. However, the antioxidant concentration in the boiled cayenne peppers was only 26% lower than those found in the raw cayenne peppers, whereas the levels in their microwaved samples were five times more than those in the raw samples (Figure , Table). Similarly, microwaved habanero peppers were found to contain the highest concentration of antioxidants and the levels to decrease in the order; microwaved > stir-fried > steamed > raw > boiled. It was found that whereas the microwaved habanero samples had antioxidant levels about 4 times those of the raw samples, the boiled fractions had levels only slightly below (5%) those of the raw samples (Figure , Table ).
3.2. Antiradical activity
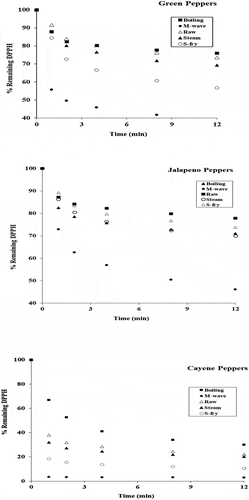
All cooked bell pepper (green) samples indicated a high ability to reduce the free radical DPPH in the order; microwaved > stir-fried > steamed > raw > boiled samples, thereby mapping and confirming the general order of total antioxidants found in these peppers using the FRAP method (Figure , Table ). All the sample values in the antiradical study just like those in the antioxidant study were based on dry matter content. Microwave cooked jalapeno peppers had the highest radical reducing activity, much higher than all the samples from other cooking techniques. The boiled and steamed jalapeno samples had very comparable antioxidant activity and the activity of the jalapeno samples followed the order; microwaved > boiled > stir-fried > raw, which agreed roughly with that measured by the FRAP technique. When the free radical DPPH was employed, microwaved cayenne samples exhibited the highest antioxidant activity and boiled samples the lowest. The radical reducing activity of the cayenne samples varied in the order; microwaved > stir-fried > steamed > raw > boiled, directly mapping and confirming the results obtained from the FRAP technique measurements for the cayenne pepper samples.
Table 2. Effect of various cooking methods on the antioxidant activity of peppers as measured by the DPPH method
Figure 3. Effect of various cooking methods on the free radical scavenging activity of green, jalapeno and cayenne peppers in a 3 mL (total volume) standard reaction with a 1:3 DPPH to sample ratio and monitored at 516 nm on a UV—Vis spectrophotometer. Values are means of triplicate determinations at p < 0.05.
In most of the findings in this study (for both measurement techniques), microwave samples were found to contain the highest concentration of antioxidant or antiradical activity compared to all the other cooking methods. During microwave cooking, the electric field component of the microwave induces the rotation of dipoles in foods and the heat is generated by the friction of molecules (Aguilar-Reynosa et al., Citation2017). Even though some studies have suggested that this process may lead to the formation of free radicals especially if the foods are microwaved for longer times (Alam & Waheed, Citation2012; Eke et al., Citation2017), it is generally agreed to lead to changes in foods associated with antioxidant activity, bioactive components and anti-nutritional components (trypsin inhibitor, tannins, saponins and phytic acid) (Chang et al., Citation2011). Accordingly, studies have registered minimal loss of phenolics or vitamin C in vegetables during microwave cooking (Tian et al., Citation2016; Xu et al., Citation2014), this has largely been attributed to the retention of the bioactive components of the vegetables microwaved compared to those stir-fried or even boiled due to the shorter cooking time or presoaking treatment involved. Such studies indicate the need to eliminate the addition of water in microwave cooking as the water may cause a softening and rapture of the lignocellulosic structure, thereby enabling soluble bioactive substances to be released from the food matrix increasing loss due to leaching and thermal liability (Guo et al., Citation2017). Studies also found microwave cooking to effectively decrease the anti-nutritional factors (trypsin inhibitor, tannins, saponins and phytic acid) and increase the protein digestively of foods (Hefnawy, Citation2011).
Most of the cooked peppers in this study were found to have higher antioxidant potential compared to the raw peppers except for boiled bell peppers (green, red) and cayenne peppers, steamed red bell peppers and stir-fried habanero peppers. The increase in total antioxidant concentrations by microwave cooking, stir-frying and steaming and other cooking methods has been found by others. Indeed, Turkmen and others (Turkmen et al., Citation2005) showed that several cooking methods caused an increase in the phenolic content of peppers. In general, the increase in the antioxidant amounts may be attributed to a loosening of antioxidant moieties, dehydration of the food matrix and improved extractability of the antioxidant compounds such as phenols from the peppers (Cisneros-Pineda et al., Citation2007; Lin & Tang, Citation2007; Ornelas-Paz et al., Citation2010). Dehydration is expected to be more pronounced with processes such as microwave cooking and stir-frying which involve the addition of minimal or no water. This may roughly explain why higher antioxidant concentrations and radical reducing activity were registered for these two cooking methods for most of the peppers in this study (Tables and ). Boiling substantially decreased the antioxidant concentration in all peppers except peppers with mild pungency (jalapeno and serrano) (Table); these findings agree with those of others (Chuah et al., Citation2008) who showed that boiling reduces the capsaicinoid content of various peppers. The losses due to boiling may be attributed to the dissolution of phenolic compounds into the cooking water (Chuah et al., Citation2008). On the other hand, the ability of jalapeno peppers to increase the antioxidant concentration with boiling agrees with the observations recorded by Martinez and others (Ornelas-Paz et al., Citation2010).
Cayenne peppers registered the highest antioxidant concentration of all the peppers used in this study (Table ). These values were higher than those of habanero, which is believed to have more pungency than cayenne, and the differences may be due to variations in the cultivars and ripening stages (Lin & Tang, Citation2007) especially for the habanero peppers. The non-pungent bell peppers (green and red) had higher or comparable total antioxidant concentrations than those of peppers with mild pungency (jalapeno and serrano). This may be due to the presence in higher concentrations of other antioxidant potential compounds besides capsaicin (which is mainly responsible for the pungency in peppers) in the non-pungent peppers compared to the levels in the peppers with mild or medium pungency.
4. Conclusions
Cooking was found to generally increase the antioxidant concentration of peppers and improve their free radical reducing ability. Specifically, microwave cooking increased the antioxidant and free radical reducing ability of all peppers studied. Stir-frying and steaming equally increased the antioxidant concentration of all peppers, except for steamed red bell peppers and stir-fried habanero peppers, whose concentrations were significantly reduced. Boiling resulted in a reduction of the antioxidant concentration of all peppers except the two with mid-pungency, jalapeno and serrano. In summary, it is recommended that cooking methods such as microwave cooking or stir-frying with minimal or no water be used in the preparation and consumption of peppers, and boiling peppers before consumption should be avoided except for peppers with medium pungency, such as jalapeno and serrano.
Competing Interests
The authors declare no competing interests.
Additional information
Funding
Notes on contributors
Nixon O. Mwebi
Nixon Mwebi is a professor of Chemistry and the Chair of the chemistry department at the University of LaVerne since 2018. Before that, he was professor of Chemistry at Jacksonville State University for 13 years. He obtained his PhD in chemistry from University of Maryland College Park, Masters in Chemistry from Hampton University, a bachelor’s degree in Chemistry from Kenyatta University in Nairobi Kenya. He teaches environmental, analytical and food chemistry and conducts research in analytical, environmental, natural products, free radical research, and food chemistry. His food chemistry research focusses on the quality of food from pre to post-harvesting and more especially the impact of preparation and processing on food quality.
References
- Aguilar-Reynosa, A., Romaní, A., Rodríguez-Jasso, R. M., Aguilar, C. N., Garrote, G., & Ruiz, H. A. (2017). Microwave heating processing as alternative of pretreatment in second-generation biorefinery: An overview. Energy Conversion and Management, 136(March 15), 50–14. https://doi.org/10.1016/j.enconman.2017.01.004
- Alam, Z., & Waheed, R. (2012). Effects of oxidized tallow on the rabbit serum lipids and antioxidant activity of the In-vitro lipids. Toxicology Research, 28(3), 151–157. https://doi.org/10.5487/TR.2012.28.3.151
- Arslan, D., & Ozcan, M. M. (2011). Dehydration of red bell –pepper (capsicum annum L): Change in drying behavior, color and antioxidant content. Food Bioprod Process, 89(4), 504–513. https://doi.org/10.1016/j.fbp.2010.09.009
- Benzie, I. F. F., & Strain, J. J. (1996). The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Analytical Biochemistry, 239(1), 70–76. https://doi.org/10.1006/abio.1996.0292
- Brand-Williams, W., Cuvelier, M. E., & Berset, C. (1995). Use of a free radical method to evaluate antioxidant activity. Lebensmittel-Wissenschaft und -Technologie, 28(1), 25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
- Chang, H. J., Xu, X. L., Li, C. B., Huang, M., Liu, D. Y., & Zhou, G. H. (2011). A comparison of heat-induced changes of intramuscular connective tissue and collagen of beef semitendinosus muscle during water bath and microwave heating. Journal of Food Process Engineering, 34(6), 2233–2250. https://doi.org/10.1111/j.1745-4530.2009.00568.x
- Choi, O. S. (1996). Emulsification stability of olerosin red pepper and changes in antioxidant activity during cooking. Journal of the Korean Society of Food Science and Nutrition, 25(1), 104–109.
- Chuah, A. M., Lee, Y.-C., Yamaguchi, T., Takamura, H., Yin, L.-J., & Matoba, T. (2008). Effect of cooking on the antioxidant properties of colored peppers. Food Chemistry, 111(1), 20–28. https://doi.org/10.1016/j.foodchem.2008.03.022
- Cisneros-Pineda, O., Torres-Tapia, L. W., Gutiérrez-Pacheco, L. C., Contreras-Martín, F., González-Estrada, T., & Peraza-Sánchez, S. R. (2007). Capsaicinoids quantification in chili peppers cultivated in the state of Yucatan. Mexico Food Chemistry, 104(4), 1755–1760. https://doi.org/10.1016/j.foodchem.2006.10.076
- Eke, B. C., Jibiri, N. N., Bede, E. N., Anusionwu, B. C., Orji, C. E., & Alisi, C. S. (2017). Effect of ingestion of microwaved foods on serum anti-oxidant enzymes and vitamins of albino rats. Journal of Radiation Research and Applied Sciences, 10(2), 148–151. https://doi.org/10.1016/j.jrras.2017.03.001
- Erguder, B. I., Avci, A., Devrim, E., & Durak, L. (2007). Effects of cooking techniques on antioxidant enzyme activities of some fruits and vegetables. Turkish Journal of Medical Sciences, 37(93), 151–156.
- Guo, Q., Sun, D., Cheng, J. H., & Han, Z. (2017). Microwave processing techniques and their recent applications in the food industry. Trends in Food Science and Technology, 67(26), 236–247. https://doi.org/10.1016/j.tifs.2017.07.007
- Hamed, M., Kalita, D., Bartolo, M. E., & Jayanty, S. S. (2019). Capsaicinoids, polyphenols and antioxidant activities of capsicum annum: Comparative study of the effect of ripening stage and cooking methods. Antioxidants, 8(9), 364. https://doi.org/10.3390/antiox8090364
- Hefnawy, T. H. (2011). Effect of processing methods on nutritional composition and anti-nutritional factors in lentils (Lens culinaris). Annals of Agricultural Sciences, 56(2), 57–61. https://doi.org/10.1016/j.aoas.2011.07.001
- Horvathova, J., Suhaj, M., & Sumko, P. (2007). Effect of thermal treatment and storage on antioxidant activity of some spices. Journal of Food and Nutrition Research, 46(1), 20–27.
- Hwang, I. G., Shin, Y. J., Lee, S., Lee, J., & Yoo, S. M. (2012). Effects of different cooking methods on the antioxidant properties of red peppers (capsicum annum L.). Preventive Nutrition and Food Science, 17(4), 286–292. https://doi.org/10.3746/pnf.2012.17.4.286
- Jimenes-Monreal, A. M., Garcia-Diz, L., Martinez-Tome, M., Mariscal, M., & Murcia, M. A. (2009). Influence of cooking methods on antioxidant activity of vegetables. Journal of Food Science, 74(3), H97–103. https://doi.org/10.1111/j.1750-3841.2009.01091.x
- Lee, Y., & Howard, L. (1999). Firmness and phytochemical losses in pasteurized yellow banana peppers (Capsicum annum) as affected by calcium chloride and storage. Journal of Agricultural and Food Chemistry, 47(2), 700–703. https://doi.org/10.1021/jf980921h
- Lin, J. Y., & Tang, C. Y. (2007). Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chemistry, 101(1), 140–147. https://doi.org/10.1016/j.foodchem.2006.01.014
- Loizzo, M. R., Pugliese, A., Bonesi, M., De Luka, D., O’Brien, N., Menichini, F., & Tundis, R. (2013). Influence of drying and cooking process on the phytochemical content, antioxidant and hypoglycemic properties of two bell Capsicum annum L cultivars. Food Chemical Toxicology, 53(March 2013), 393–401. https://doi.org/10.1016/j.fct.2012.12.011
- Ornelas-Paz, J. J., Martinez-Burrola, J. M., Ruiz-Cruz, S., Santana-Rodríguez, V., Ibarra-Junquera, V., Olivas, G. I., & Pérez-Martínez, J. D. (2010). Effect of cooking on the capsaicinoids and phenolics contents of Mexican peppers. Food Chemistry, 119(4), 1619–1625. https://doi.org/10.1016/j.foodchem.2009.09.054
- Pino, J., Gonzalez, M., Ceballos, L., Centurion-Yah, A. R., Trujillo-Aguirre, J., Latournerie-Moreno, L., & Sauri-Duch, E. (2007). Characterization of total capsaicinoids, color and volatile compounds of habanero chilli pepper (Capsicum chinense Jack.) cultivars grown in Yucatan. Food Chemistry, 104(4), 1682–1688. https://doi.org/10.1016/j.foodchem.2006.12.067
- Schweiggert, U., Schieber, A., & Carle, R. (2006). Effects of blanching and storage on capsaicinoid stability and peroxidase activity of hot chili peppers (capsicum frutescens L.). Innovative Food Sci Emerging Technol, 7(3), 217–224. https://doi.org/10.1016/j.ifset.2006.03.003
- Shobana, S., & Naidu, K. A. (2000). Antioxidant activity of selected Indian spices. Prostaglandins Leukotrienes and Essential, 62(2), 107–110. https://doi.org/10.1054/plef.1999.0128
- Srinivasan, K. (2005). Spices as influencers of body metabolism: An overview of three decades of research. Food Research International (Ottawa, Ont.), 38(1), 77–86. https://doi.org/10.1016/j.foodres.2004.09.001
- Suresh, D., Manjunatha, H., & Srinivasan, K. (2007). Effect of heat processing of spices on the concentrations of their bioactive principles: Turmeric (Curcuma longa), red pepper (Capsicum annuum) and black pepper (Piper nigrum). Journal of Food Composition and Analysis: An Official Publication of the United Nations University, International Network of Food Data Systems, 20(3–4), 346–351. https://doi.org/10.1016/j.jfca.2006.10.002
- Surh, Y.-J., & Lee, S. S. (1996). Capsaicin in hot chili pepper: Carcinogen, co-carcinogen or anticarcinogen. Food and Chemical Toxicology : An International Journal Published for the British Industrial Biological Research Association, 34(3), 313–316. https://doi.org/10.1016/0278-6915(95)00108-5
- Tian, J., Chen, J., Lv, F., Chen, S., Chen, J., Liu, D., & Ye, X. (2016). Domestic cooking methods affect the phytochemical composition and antioxidant activity of purple-fleshed potatoes. Food Chemistry, 197(April), 1264–1270. https://doi.org/10.1016/j.foodchem.2015.11.049
- Turkmen, N., Sari, F., & Velioglu, Y. S. (2005). The effect of cooking methods on total phenolics and antioxidant activity of selected vegetables. Food Chemistry, 93(4), 713–718. https://doi.org/10.1016/j.foodchem.2004.12.038
- Xu, F., Zheng, Y., Yang, Z., Cao, S., Shao, X., & Wang, H. (2014). Domestic cooking methods affect the nutritional quality of red cabbage. Food Chemistry, 161(October), 162–167. https://doi.org/10.1016/j.foodchem.2014.04.025