Abstract
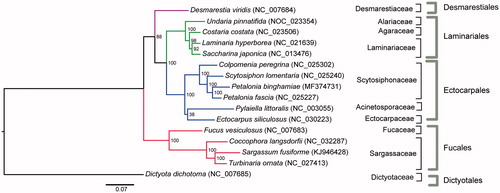
Here, the complete mitogenome of Petalonia binghamiae (J. Agardh) K. L. Vinogradova was determined and analyzed. The length of the complete P. binghamiae mitogenome was determined to be 37,460 bp. The mitogenome contains a set of 68 genes, including 35 protein-coding genes, 3 rRNA genes, 24 tRNA genes, 1 tRNA pseudogene, and 5 unidentified open reading frames (ORFs). The noncoding sequence constitutes 7.03% of the P. binghamiae mtDNA. Twenty-seven of the 35 (77.14%) protein-coding genes ended with a TAA stop codon, 6 (17.14%) with TAG, and 2 (5.71%) with TGA. All protein-coding genes in P. binghamiae were inferred to use the start codon ATG, except for nad11. The mitogenome phylogenetic analysis, based on 35 protein-coding genes, reveals that P. binghamia showed that P. binghamiae was clustered together with P. fascia. The complete mitochondrial genome sequence provided here would be useful for further understanding the evolution of Petalonia.
Petalonia binghamiae (J. Agardh) K. L. Vinogradova is an edible, economically important marine brown alga belonging to the family Scytosiphonaceae (Abbott and Hollenberg Citation1976). This species is found in the upper to mid-tidal zones of warm to temperate coastal areas throughout the Pacific Ocean and Indian Ocean, including along the coasts of North America, Korea, Japan, Pakistan, South Africa, Taiwan, and mainland China. The life history (Brophy and Murray Citation1989), morphological variability (Rhew and Boo Citation1991; Brophy Citation1995), chemical composition (Bano et al. Citation1987), and photosynthetic characteristics (Zou and Gao Citation2010) of this species have been studied both in the field and in culture, but only limited information is available about its genetics and systematics.
Here, we determined the complete mitogenome sequence of P. binghamiae. The genomic DNA of one P. binghamiae individual taken from a population located in eastern China (Nanji Island, Zhengjiang Province, 27°28′1′′N, 121°3′12′′E) was used for the genome sequencing. Specimen (accession number: 2016050053) was deposited in the Culture Collection of Seaweed at the Ocean University of China. Paired-end reads were sequenced using a Illumina’s HiSeq × Ten system (Illumina, San Diego, CA, USA). The genomic library was assembled using SOAPdenovo2 software (Luo et al. Citation2012). Transfer RNA genes were identified using the tRNAscan-SE Search Server (Schattner et al. Citation2005). Other regions of the mitogenome were annotated from the mitogenome of P. fascia using Geneious R10 (Biomatters Ltd., Auckland, New Zealand). Phylogenetic analysis of a set of 35 conserved protein-coding genes present in the 16 Phaeophyceae mitogenome was conducted. Maximum likelihood (ML) tree search and ML bootstrap analysis were performed using RaxML (Stamatakis Citation2006). Bootstrap probability values were run with 1000 replicates under the CpREV + G + I + F model.
The complete mitogenome of P. binghamiae (MF374731) consists of a circular DNA molecule with a length of 37,460 bp. The overall A + T content of the compete mitogenome is 65.6%. The mitogenome contains a set of 68 genes, including 35 protein-coding genes, 3 rRNA genes, 24 tRNA genes, 1 tRNA pseudogene, and 5 unidentified open reading frames (ORFs). No intron was observed in the mitochondrial coding regions. The lengths of three ribosomal RNA genes are 2717 bp (rnl rRNA), 1530 bp (rns rRNA), and 129 bp (rrn5 rRNA). A total of 2481 bp of noncoding sequences (intergenic spacers) were present in the mitogenome. Thirty-five protein-coding genes were identified in P. binghamiae. Twenty-seven of the 35 (77.14%) protein-coding genes ended with a TAA stop codon, 6 (17.14%) with TAG, and 2 (5.71%) with TGA. All protein-coding genes in P. binghamiae were inferred to use the start codon ATG, except for nad11.
Maximum likelihood (ML) analyses showed that P. binghamiae was clustered together with P. fascia (); moreover, five clades in the ML tree were identified, representing five different orders (Desmarestiales, Dictyotales, Ectocarpales, Fucales, and Laminariales) (). The complete mitochondrial genome sequence provided here would be useful for further understanding the evolution of Petalonia.
Disclosure statement
The authors report no conflicts of interest.
Additional information
Funding
References
- Abbott IA, Hollenberg GJ. 1976. Marine algae of California. Stanford (CA): Stanford University Press.
- Bano S, Perveen S, Ahmad VU, Bano N, Shameel M. 1987. Chemical constituents of Endarachne binghamiae (Scytosiphonales, Phaeophyta) from the Karachi Coast. Bot Mar. 30:371.
- Brophy TC. 1995. Seasonality of the rocky intertidal brown alga endarachne binghamiae (Scytosiphonales: Phaeophyta) in southern California. Fullerton: California State University.
- Brophy TC, Murray SN. 1989. Field and culture studies of a population of Endarachne binghamiae (phaeophyta) from southern california. J Phycol. 25:6–15.
- Luo R, Liu B, Xie Y, Li Z, Huang W, Yuan J, He G, Chen Y, Pan Q, Liu Y, et al. 2012. SOAPdenovo2: an empirically improved memory-efficient short-read de novo assembler. GigaScience. 1:18.
- Rhew KS, Boo SM. 1991. Morphological variability in Endarachne binghamiae J. Agardh (Phaeophyta) from the east coast of Korea. Korean J Bot. 34:45–51.
- Schattner P, Brooks AN, Lowe TM. 2005. The tRNAscan-SE, snoscan and snoGPS web servers for the detection of tRNAs and snoRNAs. Nucleic Acids Res. 33:W686–W689.
- Stamatakis A. 2006. RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics (Oxford, England). 22:2688–2690.
- Zou D, Gao K. 2010. Acquisition of inorganic carbon by Endarachne binghamiae (Scytosiphonales, Phaeophyceae). Eur J Phycol. 45:117–126.