ABSTRACT
BACKGROUND: Repeated cigarette use in individuals generally occurs due to the craving for smoking. Orexin-leptin hormones and temperament character traits are thought to be important factors affecting nicotine craving.
OBJECTIVE: The aims of this study were to determine orexin-leptin blood levels, which appear in nicotine deprivation, in health professionals who were diagnosed as having tobacco use disorder in accordance with the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-V) and to assess the temperament-character traits of the same group and to provide biologic and psychotherapeutic data for the treatment of tobacco abuse.
METHODS: The study sample consisted of faculty of medicine students and resident physicians who were smokers (n = 40). The control group comprised medical students and resident physicians who were non-smokers (n = 40). The Sociodemographic and Clinical Information Form, DSM-IV SCID-I Clinical Version, and Temperament and Character Inventory (TCI) were applied to both groups. Substance Craving Scale (SCS) and Fagerstrom Test for Nicotine Dependence were applied only smoking group. Blood samples were taken for plasma orexin and serum leptin levels for both groups.
RESULTS: The plasma orexin levels were lower in the smokers group (p < .001). No statistically significant relations were determined between the SCS and plasma orexin and serum leptin levels in the smoking group. No significant differences were determined between the leptin (U = 119.5, p = .33) and orexin (U = 99, p = .11) levels of the heavy and very heavy smokers. The total points of novelty seeking (NS), and NS3 and NS4 subscale points in the TCI of the smoking group (p = .003, p = .003, p = .002), and the self-directedness (SD) SD2 and SD5 (p = .02, p = .01) subscale points, and total cooperativeness points (TC), and C4 and C5 subscale points (p = .001, p = .002, p = .001) of the non-smoking group were found as high.
CONCLUSIONS/IMPORTANCE: Differences were detected between the smokers and non-smokers in TCI scale subgroups and in terms of orexin levels. These results will greatly assist in the fight against craving that appears as a result of smoking cessation. Orexin appears to be more specific for nicotine craving than leptin. It seems more likely that the quest for the treatment of craving will continue through orexin. Temperament and personality traits are important for determining psychotherapeutic and supportive approaches to the release of tobacco and tobacco products. A number of studies investigating monoaminergic mechanisms indirectly related to orexin and leptin are required to research temperament, which has a greater genetic burden.
Introduction
Craving is recognized as a prominent feature of nicotine dependence [Citation1,Citation2]. It is temporarily suppressed by the administration of nicotine, but, over time, nicotine produces craving. It is commonly the first symptom of dependence and can occur within days of initiating smoking in individuals who have no tolerance and who report no symptoms of nicotine withdrawal [Citation3]. It intensifies during nicotine withdrawal but can persist for years after other withdrawal symptoms have resolved [Citation1].
Brain structures related to craving can be classified as orbitofrontal cortex, anterior cingulate cortex, prelimbic cortex, basolateral amygdala, nucleus accumbens, ventral tegmental area, and hypothalamic–pituitary–adrenal axis. It is claimed that the craving inhibitory system includes the mesolimbic system beginning from the ventral tegmental area and terminating in the nucleus accumbens [Citation4].
Dopaminergic neurons in the VTF have a key role in the rewards system, and the release of dopamine in the nucleus accumbens is related with the use of many substances (ethanol, opioids, cannabinoids, phencyclidine, toluene, psychostimulants, and nicotine) [Citation5–7].
Nicotinic acetylcholine receptors (nAChRs) exist extensively in the reward pathway. Through these nicotinic receptors, nicotine affects the activity of the dopamine reward pathway and increases extra-cellular dopamine levels in the nucleus accumbens, striatum, and frontal cortex, which is an expected result [Citation8]. Thus, craving is inhibited by nicotine-induced dopamine release.
Research conducted to date has shown that orexin neurons are spread throughout a wide area in the brain. One of these areas is the dopaminergic neurons in the midbrain and VTF [Citation9]. Orexin causes an excitatory effect in the VTF and directly stimulates cells that contain dopamine [Citation10].
Several studies were conducted previously for the purpose of assessing the effects of nicotine on orexin and its receptors. In these studies, increases were reported in prepro-OX mRNA, OX receptor mRNA, and OX peptide expression after chronic and acute nicotine use [Citation11]. It has been shown that the increase of orexin synthesis following nicotine uptake [Citation12] and the increased levels of orexin increase dopamine release in the reward pathway [Citation13]. As a consequence of the lack of nicotine, there is a decrease in the levels of orexin and dopamine, and it has been suggested that this begins the craving process [Citation14].
Researchers also conducted studies to investigate the mesolimbic dopaminergic modulation of leptin and reported that leptin receptors were expressed in dopaminergic neurons in the VTF [Citation15]. As a result of examinations of brain samples to assess the VTF, it was shown that leptin hyperpolarizes dopamine neurons and decrease the frequency of action potential formation [Citation15]. It was also reported that extra-cellular dopamine levels decreased in the nucleus accumbens as a result of central infusion of leptin [Citation16].
The psychobiologic personality hypothesis of Cloninger defines a four-dimensional temperament consisting of novelty seeking (NS), harm avoidance (HA), reward dependence (RD), and persistence (P). Existing evidence suggests that variation in each of these dimensions is highly correlated with activity in a specific monoaminergic pathway: NS with low basal dopaminergic activity, HA with high serotonergic activity, RD with low basal noradrenergic activity, and P with glutaminergic activity [Citation17]. In many studies, NS has been associated with a variety of smoking behaviors such as tobacco use, smoking status, and nicotine addiction severity [Citation18–21]. HA and RD were moderately related to the initiation of cigarette smoking and severity of nicotine addiction [Citation18–20].
The aims of this study were to determine orexin-leptin blood levels, which appear in nicotine deprivation, in health professionals who were diagnosed as having tobacco use disorder in accordance with the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-V) [Citation22] and to assess the temperament-character traits of the same group and to provide biologic and psychotherapeutic data for the treatment of tobacco abuse.
We hypothesize that the decreased levels of serum orexin and the increased plasma leptin levels cause to increase the nicotine craving in smoking group. Also, we hypothesize that the smoking group has higher NS and HA subscales points than the non-smoking group, and that these higher points in these scales cause to increase in nicotine craving.
Method and material
Sampling of the study
The study population consisted of 1038 (1st grade, n = 227; 2nd grade, n = 185; 3rd grade, n = 208; 4th grade, n = 147; 5th grade, n = 152; 6th grade, n = 119) faculty of medicine students of a university, and 230 resident physicians of the hospital of the same university. The sample size was calculated using power analysis (G*Power 3.0.10 programme). A sample size of 39 participants per group was determined based on a power of 70%, one-tailed and an effect size d = 0.50 (d = effect size) of α2 = 0.05. The study was conducted on 40 participants in each group.
Inclusion criteria
The inclusion criteria for the smoking group were as follows:
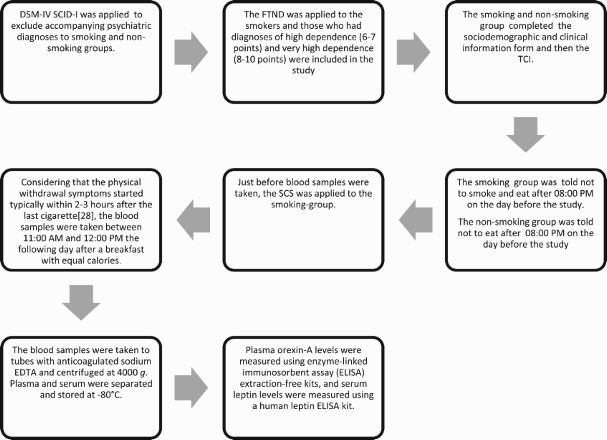
To comply with the DSM-V tobacco use disorder diagnosis criteria, being aged between 18–65 years, having high-addiction according to the Fagerstrom Test for Nicotine Dependence (FTND) [Citation23] criteria (6–7 points) and very high level addiction (8–10 points), agreeing to participate in the study, and providing written consent for the study. In order to exclude accompanying psychiatric diagnoses, the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) [Citation24] clinical version was applied.
The inclusion criteria for the non-smoking group:
The control (non-smoking) group consisted of participants who matched the smoking group in terms of number, age, sex, body mass index (BMI), and educational status, and who studied at the same medicine faculty and hospital.
Exclusion criteria
The exclusion criteria for the smoking group were as follows:
Alcohol or substance use disorder (according to DSM-V criteria), having one of the other DSM-V diagnoses within the past 6 months, pregnancy, having received psychotropic medication within the past 6 months, having any of the life-long neurologic diseases, sleep-wake cycle disorders.
The exclusion criteria for the non-smoking group were as follows:
In addition to the exclusion criteria of the smoking group, individuals who have any history of smoking in their lifetime were excluded.
Clinical evaluation tools
Sociodemographic and Clinical Information Form
This form was prepared to capture the participants’ sociodemographic data, resume, and family history. This form was formulated by the researcher and questioned: sex, marital status, age, body weight, height, duration of education, place of birth, place of growing up, duration of smoking, parental cigarette use characteristics, night total sleep duration, and presence of psychiatric illness.
Substance Craving Scale
The Substance Craving Scale (SCS) is a 5-item question form based on self-declaration [Citation25]. The Turkish version of the scale was prepared by Evren et al. [Citation26] both for alcohol and for other substances. Each item is assessed with points between 0 and 6. In this way, the total craving point has a maximum of 30. The Cronbach alpha value was found as 0.84 for the whole craving scale. It was determined that the corrected item-total correlation values for each item ranged between 0.75 and 0.82.
 The flow chart of the study process.
The flow chart of the study process.
Temperament and Character Inventory
The Temperament and Character Inventory (TCI) consists of 240 items and is a self-assessment scale in which the answers are given as “true” or “false.” The validity and reliability of the scale were performed by Köse et al. [Citation27]. Cronbach alpha values were found as 0.60 and 0.85 in the temperament dimension, and 0.82 and 0.83 in the character dimension. The lowest Cronbach alpha coefficients were determined in RD (0.60) and perseverance (0.62).
Data collection
The local ethics board was contacted and permission was received for the study. The local ethics board approval date: 14.01.2015 and number: 20478486-19. The study process was completed between March 2015 and May 2016.
Statistical analysis
The SPSS for Windows version 17.0 statistical package program was used in the statistical analyses. First, descriptive statistics were made in analyses of the data. The Chi-square test was used for categorical variables, t-test and variance analysis were used for independent groups in numeric variables (when normal distribution conditions were not met, the Mann–Whitney U and Kruskal–Wallis tests were used).
Dependent and independent variables are listed below:
Dependent Variable: Smoking status (smokers/non-smokers).
Independent Variables: Sociodemographic and Clinical Characteristics, Temperament and Character Traits, Plasma Leptin and Orexin Levels.
Results
The sociodemographic and clinical features of the study groups’ characteristics are provided in .
Table 1. Sociodemographic and clinical characteristics of the study groups.
shows the statistically significant differences between the smokers and non-smokers in the TCI subscales. There was a moderate positive correlation between smoking duration and NS3 (r = 0.33, p = .003) and a low correlation between NS-total (r= 0.29, p = .009).
Table 2. Temperament and character traits of the study groups.
The mean plasma orexin and serum leptin levels of the smokers and non-smokers are given in .
Table 3. Plasma leptin and orexin levels of the groups.
The serum leptin levels were higher in the female participants. The serum leptin levels differed significantly in the non-smoking group (U = 50, p < .001) and in the smoking group (U = 72, p = .002) between men and women (U = 50, p < .001).
The relations between plasma orexin and serum leptin levels and SCS in the smoking group
No statistically significant relations were determined between the SCS and plasma orexin and serum leptin levels in the smoking group.
The relation between TCI and craving
The distribution of the heavy and very heavy smokers (as per the FTND) according to the orexin and leptin levels is given in . No significant differences were determined between the leptin (U = 119.5, p = .33) and orexin (U = 99, p = .11) levels of the heavy and very heavy smokers.
Table 4. The mean orexin and leptin values according to heavy and very heavy smoking status in the Fagerstrom Test for Nicotine Dependence.
Discussion
Sociodemographic and clinical features
This was the first study to evaluate the orexin-leptin levels, which are affected by smoking status, and the TCI among smokers and non-smokers.
It was shown that there were decreases in the number of orexin neurons and in the amount of the OX-A receptors in cerebrospinal fluid during the ageing process [Citation29]. There were no statistical differences in terms of age among the groups, which is important in terms of the comparison of orexin-A levels obtained from the blood samples. Again, it was reported that there were no differences between the sexes in terms of orexin-A levels [Citation30]. In several studies conducted on leptin levels, it was considered that leptin levels were higher in women and the reason for this was estrogen [Citation31]. The fact that there were no differences between the groups in terms of sex has increased the reliability of the statistics on leptin.
No significant differences were determined between the groups in terms of BMI. When one considers that leptin is especially related with BMI and body fat rate, the lack of significant difference between the groups becomes more important.
It is well known that orexin is the main regulator that ensures sleep-wake balance [Citation32]. The lack of difference between the groups in terms of sleep duration increases the reliability of the study.
Plasma orexin and serum leptin levels
In the present study, the mean plasma orexin level values obtained as a result of the nicotine deprivation of the smokers were significantly low when compared with the non-smokers. There is only one similar study in the literature, and the mean orexin level in the smoking group was found to be low; however, this was not found to be statistically significant [Citation14]. It was claimed that orexin levels decreased after the deprivation of nicotine and dopamine release decreased, thus starting the craving process [Citation14].
Plaza-Zabala et al. [Citation33] conducted a study on animal models and showed that rats that had orexin gene deletion and were treated with the Orexin-A receptor antagonist SB-334867 had less nicotine withdrawal symptoms. In the same study, increases were observed in nicotine search behaviors as a result of intracerebroventricular orexin application, and this increase was blocked with SB-334867. In another study, a significant decrease was determined in ethanol consumption as a result of administering the Orexin-A receptor antagonist, GSK1059865, in ethanol-dependent rats [Citation34].
In light of these data, it seems that the importance of future studies on orexin-A receptor antagonists for search and withdrawal symptoms for nicotine and other substances will increase. When all the literature data are considered, our study resulted with a decrease of orexin levels in smokers in the craving period, which was an expected result.
The average serum leptin level obtained as a result of nicotine deprivation in the smoker group was lower than in the non-smoker group. However, no statistically significant differences were determined. Only four studies assessed the relation of serum leptin and craving among smokers [Citation12,Citation14,Citation35,Citation36]. Only in the study conducted by Von der Goltz et al. [Citation14] was a control group used, and although serum leptin levels were found to be lower than in the smokers group, the difference was not found to be statistically significant. In addition to the addiction process, leptin is an important risk factor for relapse in smokers during the critical cigarette smoking cessation process [Citation37].
As it was reported and discussed in other studies, the sample group in this study consisted of a small number of individuals [Citation14,Citation38–40] However, increasing leptin levels were found to be related with relapse in studies that were conducted with more than one hundred participants [Citation41,Citation42], and it was claimed that leptin levels could be biologic markers for relapse in women [Citation41].
In the study, the lower level of leptin in the control group during the withdrawal period may be related to the presence of many other factors affecting the metabolism of peripheral leptin. For this reason, unlike previous studies, it seems important to reexamine leptin in terms of the presence of biologic markers for relapse in patients with deprivation.
Temperament and character traits
A significant difference was determined between the smokers and non-smokers in terms of NS-total points. This result is consistent with previous studies [Citation18,Citation19,Citation43] This result is also in agreement with the findings of smokers’ high cigarette seeking sense score [Citation44,Citation45], substance abusers with high NS scores, and type 2 alcohol-dependents [Citation43,Citation46]. Individuals seeking novelty are more likely to encounter tobacco in their surroundings than individuals who seek less stimulus. In addition, they may find smoking more rewarding, they respond more to the effects of nicotine, which enhances temperament and ensures wakefulness.
The HA2 subscale points were found lower in smokers. Similar results were shown in some other studies in which substance use in teenagers was assessed [Citation43,Citation47], and in several other studies [Citation18,Citation19], non-smokers in the present study may have been displaying risk behaviors without considering their health. These results suggest that the behavioral inhibition system and serotonin are the place in the etiology and treatment of abuse of tobacco and tobacco products.
Although studies on smoking and the TCI have generally studied temperament traits, character traits have not been studied extensively [Citation20,Citation48]. In these studies, it was revealed that SD subscale points were higher in non-smokers. In the present study, the SD2 and SD5 sub-scale points were determined to be lower in smokers. These results indicate to physicians that individuals with low scores on this subscale require more support for coping with withdrawal symptoms.
In the present study, total C, and C1, C4, and C5 scores were higher in the control group than in the smoking group. In the study conducted by Etter et al. [Citation20], it was reported that those who had stopped smoking previously had higher C points when compared with those who just stopped smoking. This result showed how important social support was for the efforts to quit smoking. The smoking group in our study consisted of heavy and very heavy smokers, and one of the underlying causes of their heavy smoking maybe being less prone to cooperativeness.
The relation between plasma orexin-serum leptin levels and nicotine craving
We found no statistically significant result between orexin-leptin levels and the substance abstinence scale. The SCS used in our study. There are a total of 5 items on the scale of substance abuse. Nicotine dependence may not have been sufficiently evaluated. For this reason, this study may not have any relationship between orexin and leptin levels and nicotine craving.
There is only one study in the literature that examined the relationship between nicotine craving and orexin. In a study by Von der Goltz et al. [Citation14], there was a negative correlation between plasma orexin levels and nicotine craving, and a positive correlation with serum leptin levels. In that study, the Questionnaire of Smoking Urges was used. There are 32 items in this questionnaire and 2 subscales; one evaluates intention and desire to smoke, and anticipation of pleasure from smoking, and the other primarily evaluates the anticipation of relief from the negative effect and nicotine withdrawal, and the urgent and overwhelming desire to smoke.
Limitations
The study has some limitations. First, dopamine levels were not quantitatively demonstrated simultaneously with plasma orexin-leptin levels. It is also thought that variations in dopaminergic genes may have an effect on craving. For this reason, genetic and in vivo studies will become even more important in the future. Another limitation may be the evaluation of peripheral blood for the orexin produced in the central nervous system. However, it is known that orexin can pass the blood–brain barrier through passive diffusion. Among other limitations medium reliability of FTND, non-treatment seeking population, reporting and recall bias, floor effects, desirability bias, single-center and cross-sectional design, shared method variance can be considered.
Conclusions
Orexin and leptin are claimed to be effective in the craving stage in many substance use disorders, as well as tobacco use disorders. Differences were detected between the smokers and non-smokers in TCI scale subgroups and in terms of orexin levels. These results will greatly assist in the fight against craving that appears as a result of smoking cessation. Orexin appears to be more specific for nicotine craving than leptin. It seems more likely that the quest for the treatment of craving will continue through orexin. Temperament and personality traits are important for determining psychotherapeutic and supportive approaches to the release of tobacco and tobacco products. A number of studies investigating monoaminergic mechanisms indirectly related to orexin and leptin are required to increase temperament, which has a greater genetic burden.
Disclosure statement
No potential conflict of interest was reported by the authors.
ORCID
Kadir Aşçibaşi http://orcid.org/0000-0002-5295-512X
Additional information
Funding
References
- US Department of Health and Human Services. The health consequences of smoking: Nicotine addiction: A report of the Surgeon General. Washington (DC): US Government Printing Office; 1988. (DHHS Publication No. CDC 88-8406); 14.
- Shadel WG, Shiffman S, Niaura R, et al. Current models of nicotine dependence: what is known and what is needed to advance understanding of tobacco etiology among youth. Drug Alcohol Depend. 2000;59:9–22. DOI: https://doi.org/10.1016/S0376-8716(99)00162-3.
- Franza JR D, Rigotti NA, McNeill AD, et al. Initial symptoms of nicotine dependence in adolescents. Tob Control. 2000;9(3):313–319. http://doi.org/10.1136/tc.9.3.313.
- Di Franza JR, Wellman RJ. A sensitization-homeostasis model of nicotine craving, withdrawal, and tolerance: integrating the clinical and basic science literature. Nicotine Tob Res. 2005;7(1):9–26. doi:10.1080/14622200412331328538.
- Pierce RC, Kumaresan V. The mesolimbic dopamine system: the final common pathway for the reinforcing effect of drugs of abuse? Neurosci Biobehav Rev. 2006;30(2):215–238. https://doi.org/10.1016/j.neubiorev.2005.04.016.
- Adinoff B. Neurobiologic processes in drug reward and addiction. Harv Rev Psychiatry. 2004;12(6):305–320. doi:10.1080/10673220490910844.
- Melis M, Spiga S, Diana M. The dopamine hypothesis of drug addiction: hypodopaminergic state. Int Rev Neurobiol. 2005;63:101–154. doi:10.1016/S0074-7742(05)63005-X.
- Wonnacott S, Sidhpura N, Balfour DJ. Nicotine: from molecular mechanisms to behaviour. Curr Opin Pharmacol. 2005;5(1):53–59. doi:10.1016/j.coph.2004.12.002.
- Fadel J, Deutch AY. Anatomical substrates of orexin–dopamine interactions: lateral hypothalamic projections to the ventral tegmental area. Neuroscience. 2002;111(2):379–387. https://doi.org/10.1016/S0306-4522(02)00017-9.
- Korotkova TM, Sergeeva OA, Eriksson KS, et al. Excitation of ventral tegmental area dopaminergic and nondopaminergic neurons by orexins/hypocretins. J Neurosci. 2003;23(1):7–11. http://www.jneurosci.org/content/23/1/7.full.
- Pasumarthi RK, Reznikov LR, Fadel J. Activation of orexin neurons by acute nicotine. Eur J Pharmacol. 2006;535(1):172–176. https://doi.org/10.1016/j.ejphar.2006.02.021.
- Kane JK, Parker SL, Matta SG, et al. Nicotine up-regulates expression of orexin and Its receptors in Rat brain. Endocrinology. 2000;141(10):3623–3629. doi:10.1210/endo.141.10.7707.
- Narita M, Nagumo Y, Hashimoto S, et al. Direct involvement of orexinergic systems in the activation of the mesolimbic dopamine pathway and related behaviors induced by morphine. J Neurosci. 2006;26(2):398–405. doi:10.1523/JNEUROSCI.2761-05.2006.
- Von der Goltz C, Koopmann A, Dinter C, et al. Orexin and leptin are associated with nicotine craving: a link between smoking, appetite and reward. Psychoneuroendocrinology. 2010;35(4):570–577. https://doi.org/10.1016/j.psyneuen.2009.09.005.
- Hommel JD, Trinko R, Sears RM, et al. Leptin receptor signaling in midbrain dopamine neurons regulates feeding. Neuron. 2006;51(6):801–810. doi:10.1016/j.neuron.2006.08.023.
- Krügel U, Schraft T, Kittner H, et al. Basal and feeding-evoked dopamine release in the rat nucleus accumbens is depressed by leptin. Eur J Pharmacol 2003;482(1):185–187. https://doi.org/10.1016/j.ejphar.2003.09.047.
- Cloninger CR, Svrakic DM, Przybeck TR. A psychobiological model of temperament and character. Arch Gen Psychiatry. 1993;50(12):975–990. doi:10.1001/archpsyc.1993.01820240059008.
- Pomerleau CS, Pomerleau OF, Flessland KA, et al. Relationship of Tridimensional Personality Questionnaire scores and smoking variables in female and male smokers. J Subst Abuse. 1992;4(2):143–154. https://doi.org/10.1016/0899-3289(92)90014-O.
- Heath AC, Madden PAF. Genetic influences on smoking behavior. In: Turner JR, Cardon LR, Hewitt JK, editors. Behavior genetic approaches in behavioral medicine. New York: Plenum Press; 1995. p. 45–66.
- Etter JF, Pélissolo A, Pomerleau CS, et al. Associations between smoking and heritable temperament traits. Nicotine Tob Res. 2003;5(3):401–409. https://doi.org/10.1080/1462220031000094240.
- Elovainio M, Kivimäki M, Viikari J, et al. The mediating role of novelty seeking in the association between the type 4 dopamine receptor gene polymorphism and cigarette-smoking behavior. Pers Individ Dif. 2005;38(3):639–645. doi:10.1097/01.PSY.0000041547.31072.25 doi: 10.1016/j.paid.2004.05.018
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th ed. Arlington (VA): American Psychiatric Pub; 2013.
- Uysal MA, Kadakal F, Karşidağ C, et al. Fagerstrom Test for Nicotine Dependence: reliability in a Turkish sample and factor analysis. Tuberk Toraks. 2004;52(2):115–121. http://www.tuberktoraks.org/managete/fu_folder/2004-02/2004-52-2-115-121.pdf.
- First MB, Spitzer RL, Gibbon M, et al. Structured clinical interview for DSM-IV axis I disorders. New York (NY): New York State Psychiatric Institute; 1995.
- Flannery BA, Volpicelli JR, Pettinati HM. Psychometric features of the Penn Alcohol Craving Scale. Alcohol Clin Exp Res. 1999;23(8):1289–1295. doi:10.1111/j.1530-0277.1999.tb04349.x.
- Evren C, Gürol DT, Ögel K. Reliability and validity of the Penn Alcohol Craving Scale (PACS) revised version for substance craving in male substance dependent inpatients. Turk Psikiyatri Derg. 2011;22(1):70.
- Köse S, Sayar K, Kalelioglu Ü, et al. Turkish version of the Temperament and Character Inventory (TCI): reliability, validity, and factorial structure. Bulletin of Clinical Psychopharmacology. 2004;14(3):107–131.
- Hughes JR. The nicotine withdrawal syndrome: a brief review and update. Int J Smoking Cessation. 1992;1:21–26.
- Hunt NJ, Rodriguez ML, Waters KA, et al. Changes in orexin (hypocretin) neuronal expression with normal aging in the human hypothalamus. Neurobiol Aging 2015;36(1):292–300. doi:10.1016/j.neurobiolaging.2014.08.010.
- Iwasa T, Matsuzaki T, Munkhzaya M, et al. Developmental changes in the hypothalamic mRNA levels of prepro-orexin and orexin receptors and their sensitivity to fasting in male and female rats. Int J Dev Neurosci. 2015;46:51–54. doi:10.1016/j.ijdevneu.2015.07.005.
- Casabiell X, Piñeiro V, Vega F, et al. Leptin, reproduction and sex steroids. Pituitary. 2001;4(1):93–99. doi:10.1023/A:1012999130035.
- Saper CB, Chou TC, Scammell TE. The sleep switch: hypothalamic control of sleep and wakefulness. Trends Neurosci. 2001;24(12):726–731. https://doi.org/10.1016/S0166-2236(00)02002-6.
- Plaza-Zabala A, Flores Á, Martín-García E, et al. A role for hypocretin/orexin receptor-1 in cue-induced reinstatement of nicotine-seeking behavior. Neuropsychopharmacology. 2013;38(9):1724–1736. doi:10.1038/npp.2013.72.
- Lopez MF, Moorman DE, Aston-Jones G, et al. The highly selective orexin/hypocretin 1 receptor antagonist GSK1059865 potently reduces ethanol drinking in ethanol dependent mice. Brain Res. 2016;1636:74–80. doi:10.1016/j.brainres.2016.01.049.
- Potretzke S, Nakajima M, Cragin T, et al. Changes in circulating leptin levels during acute stress and associations with craving in abstinent smokers: a preliminary investigation. Psychoneuroendocrinology. 2014;47:232–240. doi:10.1016/j.psyneuen.2014.05.008.
- Da Silva Gomes A, Toffolo MCF, van Keulen HV, et al. Influence of the leptin and cortisol levels on craving and smoking cessation. Psychiatry Res. 2015;229(1):126–132. doi:10.1016/j.psychres.2015.07.060.
- Aguiar-Nemer AS, Toffolo MC, da Silva CJ, et al. Leptin influence in craving and relapse of alcoholics and smokers. J Clin Med Res. 2013;5(3):164–167. http://doi.org/10.4021/jocmr1159w.
- Jahn H, Kellner M, Naber D, et al. Leptin as a possible modulator of craving for alcohol. Arch Gen Psyhiatry. 2001;58(5):509–510. doi:10.1001/archpsyc.58.5.509.
- Kiefer F, Jahn H, Schick M, et al. Alcohol intake, tumour necrosis factor-α, leptin and craving: factors of a possibly vicious circle? Alcohol Alcohol. 2002;37(4):401–404. https://doi.org/10.1093/alcalc/37.4.401.
- Al’Absi M, Hooker S, Fujiwara K, et al. Circulating leptin levels are associated with increased craving to smoke in abstinent smokers. Pharmacol Biochem Behav. 2011;97(3):509–513. doi:10.1016/j.pbb.2010.10.004.
- Kraus T, Reulbach U, Bayerlein K, et al. Leptin is associated with craving in females with alcoholism. Addict Biol. 2004;9(3-4):213–219. doi:10.1111/j.1369-1600.2004.tb00535.x.
- Hillemacher T, Bleich S, Frieling H, et al. Evidence of an association of leptin serum levels and craving in alcohol dependence. Psychoneuroendocrinology. 2007;32(1):87–90. https://doi.org/10.1016/j.psyneuen.2006.09.013.
- Wills TA, Vaccaro D, McNamara G. Novelty seeking, risk taking, and related constructs as predictors of adolescent substance use: an application of Cloninger’s theory. J Subst Abuse. 1994;6(1):1–20. https://doi.org/10.1016/S0899-3289(94)90039-6.
- Kassel JD, Shiffman S, Gnys MA, et al. Psychosocial and personality differences in chippers and regular smokers. Addict Behav. 1994;19(5):565–575. https://doi.org/10.1016/0306-4603(94)90012-4.
- Gilbert DG. Smoking: individual difference, psychopathology, and emotion. Washington (DC): Taylor & Francis; 1995.
- Cloninger CR. A systematic method for clinical description and classification of personality variants: A proposal. Arch Gen Psychiatry. 1987;44(6):573–588. doi:10.1001/archpsyc.1987.01800180093014.
- Masse LC, Tremblay RE. Behavior of boys in kindergarten and the onset of substance use during adolescence. Arch Gen Psychiatry. 1997;54(1):62–68. doi:10.1001/archpsyc.1997.01830130068014.
- Gurpegui M, Jurado D, Luna JD, et al. Personality traits associated with caffeine intake and smoking. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(5):997–1005. https://doi.org/10.1016/j.pnpbp.2007.02.006.
