Abstract
This study aimed to confirm the use of microRNA (miR)-30c-5p and miR-373 as the potential biomarkers for Parkinson's disease (PD). In this study, 148 patients with PD admitted to our hospital were enrolled as the research group (RG) and 126 physically examined healthy individuals as the control group (CG) between December 2017 and June 2018. Serum miR-30c-5p and miR-373 expressions in both groups were determined by quantitative real-time polymerase chain reaction. Furthermore, the correlation of their expressions with PD severity was determined by Spearman's correlation analysis. Serum miR-30c-5p and miR-373 expressions were higher in RG than in CG (both P < 0.001). Their expressions were not related to smoking history and diabetes mellitus history in RG (both P>0.001) but were related to sex, age, coronary heart disease history, depression history, and Hoehn–Yahr score (all P < 0.05). Moreover, the relative expressions of serum miR-30c-5p and miR-373 were positively correlated with PD severity (P < 0.05). MiR-30c-5p and miR-373 may be involved in PD development and progression and can be adopted as potential biomarkers for PD.
Background
Parkinson's disease (PD) is a disabling disease characterized by degenerative changes in the central nervous system, and it is primarily caused by the loss of dopaminergic neurons in the substantia nigra pars compacta (Alessi and Sammler Citation2018). PD can cause cognitive disorder and limb disability, leading to the development of psychosomatic diseases. Therefore, PD has become a crucial challenge in modern medicine and the modern society (Demirci et al. Citation2017; Kouchaki et al. Citation2018). Despite the continuous advancement in diagnosis and treatment technology, the clinical efficacy of treatments for PD remains unsatisfactory. Therefore, the detection and treatment of PD as early as possible are necessary to improve its prognosis and ease social burden (Wang et al. Citation2016; Yang et al. Citation2017). PD can be classified into the following stages according to its severity: early, midterm, and advanced. The higher its severity, the worse its prognosis; hence, identifying appropriate evaluation indexes to judge PD severity for the determination prognosis is crucial (Gao et al. Citation2018). According to the growing understanding of PD pathogenesis, a good biomarker is beneficial to diagnose and evaluate PD (Wei et al. Citation2018).
MicroRNAs (miRs) are a class of endogenous, noncoding, single-stranded RNAs (Mehta and Baltimore Citation2016; Peng and Croce Citation2016). MiRs are related to inflammatory response, endocrine changes, and pathological process of tumor and other diseases, and relevant miRs regulate the expressions of other messenger RNAs (mRNAs) and proteins by binding to their target gene mRNA (Alarcón et al. Citation2015; Chadly et al. Citation2018). According to the study by Patil et al. (Patil et al. Citation2019), because the methods currently used for the clinical diagnosis of PD may be inaccurate, the identification and verification of molecular biomarkers such as circulating miRs will improve the accuracy of PD diagnosis. They found that miRs can supplement and improve the accuracy of the existing methods used for PD diagnosis and can help improve clinical diagnosis and treatment of PD. Researchers have confirmed that miR-30c-5p is closely related to tumor progression, bacterial infection, and inflammatory response, and recent studies have suggested that it is a potential biomarker for multisystem atrophy (Zhong et al. Citation2014; Marques et al. Citation2017). Related studies have confirmed that miR-373 can serve as a tumor suppressor in brain tumor (Wei et al. Citation2016). However, at present, the expression and clinical value of miR-30c-5p and miR-373 in patients with PD remain poorly understood. Therefore, we analyzed serum miR-30c-5p and miR-373 expressions in patients with PD to determine their clinical value and short-term prognosis, thereby providing a new theoretical basis for the diagnosis and treatment of PD in molecular biology.
Material and methods
Data collection
In this study, we enrolled a total of 148 patients with PD admitted to our hospital as the research group (RG) and 126 physically examined healthy individuals as the control group (CG) from December 2017 and June 2018. RG comprised 86 males and 62 females, with a mean age of 71.36 ± 3.61 years, whereas CG comprised 74 males and 52 females, with a mean age of 65.45 ± 3.42 years. We included patients who were diagnosed with PD according to the criteria for PD diagnosis (Berg et al. Citation2013) and who had normal hepatic and renal function but no malignant tumor. Those who were undergoing or had undergone treatment were excluded. This study was approved by the Ethics Committee of the China–Japan Friendship Hospital, and all patients signed informed consent forms before the start of the study.
Main reagents and instruments
The following reagents and instruments were used: TRIzol reagent (GENMED Co., United States), quantitative real-time polymerase chain reaction (qRT-PCR) kit and minScript reverse transcription kit, HBS-1096A enzyme microplate reader (Shanghai Precision Instruments Co., Ltd., China), and qRT-PCR instrument (Thermo Fisher Company, United States). The primer sequences of miR-30c-5p, miR-373, and internal reference (U6) as well as miR negative control were all designed by Shanghai GenePharma Company (Table ).
Table 1. Primer sequences of miR-30c-5p, miR-373, and internal reference (U6).
Determination of serum miR-30c-5p and miR-373 expressions
Within 24 h after patient admission, we extracted an elbow venous blood sample (5 ml) from each subject and collected it in anticoagulant-free vacuum blood collection tubes. The sampled blood was centrifuged at 3500 g for 15 min to collect serum, which was then stored in an Eppendorf tube in a low-temperature refrigerator at −80°C for subsequent analysis. Serum miR-30c-5p and miR-373 expressions in both groups were determined using qRT-PCR. Total RNA was extracted from the collected serum according to the operating instructions of the TRIzol reagent and dissolved in 20 µl diethyl pyrocarbonate water. Next, analysis was completed by reverse transcription using a reverse transcription kit using a total reaction volume of 15 µl. Relative quantification of the target genes was performed using the 2−△△Ct method.
Statistical analysis
Data were analyzed statistically using SPSS 17.0. Enumeration data are expressed as the number of cases/percentage [n (%)] and were compared using the χ2 test between the groups. Measurement data are expressed as mean ± standard deviation, and they were compared using t-test or F test between the groups. Furthermore, receiver operating characteristic curves were constructed, and their correlation was analyzed using Spearman's correlation analysis. In addition, P-values of <0.05 indicated statistically significant difference.
Results
General clinical data of patients
General clinical data such as age, sex, body mass index, smoking history, drinking history, hypertension history, diabetes mellitus history, and depression history were compared between the two groups, and no significant differences were observed (all P > 0.05; Table ).
Table 2. General clinical data of patients.
miR-30c-5p and miR-373 expressions
Serum miR-30c-5p and miR-373 expressions were higher in RG than in CG (1.95 ± 0.24 vs. 0.42 ± 0.15 and 1.82 ± 0.33 vs. 1.01 ± 0.29, respectively; both P < 0.001; Table and Figures and ).
Figure 1. The relative expression of serum miR-30c-5p was significantly higher in the research group than in the control group (P < 0.001).
Note: *** indicates P < 0.001.
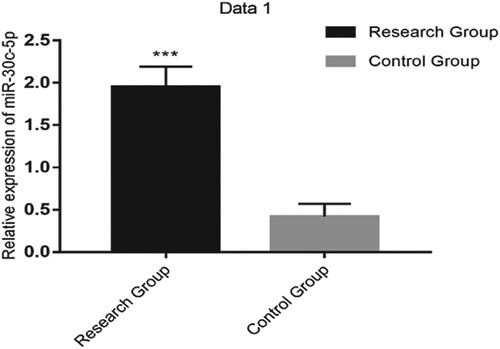
Figure 2. The relative expression of serum miR-373 was significantly higher in the research group than in the control group (P < 0.001).
Note: *** indicates P < 0.001.
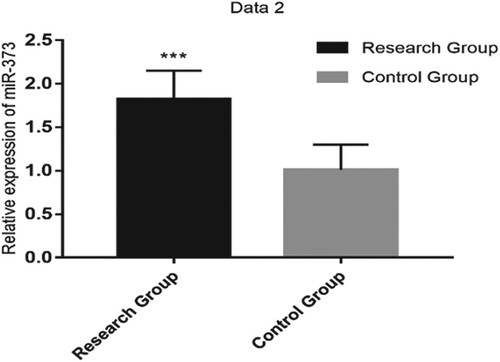
Table 3. Serum miR-30c-5p and miR-373 expressions in the two groups.
Serum miR-30c-5p expression and clinicopathological characteristics of patients with PD
Serum miR-30c-5p expression was not related to smoking history and diabetes mellitus history in RG (both P > 0.001), but it was associated with sex; age; history of coronary heart disease, tardive dyskinesia, postural instability, or gait disorder; and Hoehn–Yahr score, mini-mental state examination (MMSE), and Montgomery–Åsberg Depression Rating Scale (MADRS) scores (all P < 0.05; Table ).
Table 4 Analysis of serum miR-30c-5p expression and clinicopathological characteristics of patients with PD.
Serum miR-373 expression and clinicopathological characteristics of patients with PD
Serum miR-373 expression was not related to smoking history and diabetes mellitus history in RG (both P > 0.001), but it was associated with sex; age; history of coronary heart disease, tardive dyskinesia, postural instability, or gait disorder; and Hoehn–Yahr, MMSE, and MADRS scores (all P < 0.05; Table ).
Table 5. Serum miR-373 expression and clinicopathological characteristics of patients with PD.
Correlation of serum miR-30c-5p expression with PD severity
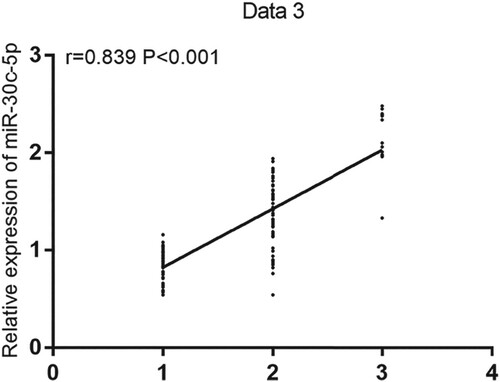
The mean serum miR-30c-5p expression in patients with early, midterm, or advanced PD was 0.81 ± 0.26, 1.54 ± 0.37, and 2.26 ± 0.23, respectively. Serum miR-30c-5p expression was significantly upregulated in patients with midterm or advanced PD compared with that in patients with early PD (both P < 0.05), and the relative expression of serum miR-30c-5p increased as PD aggravated. Early, midterm, and advanced PD stages were set to 1, 2, and 3, respectively. Then, the correlation of the relative expression of serum miR-30c-5p with PD severity was analyzed by Spearman's correlation analysis; subsequently, a positive correlation was found (r = 0.839, P < 0.001; Figure ).
Correlation of serum miR-373 expression with PD severity
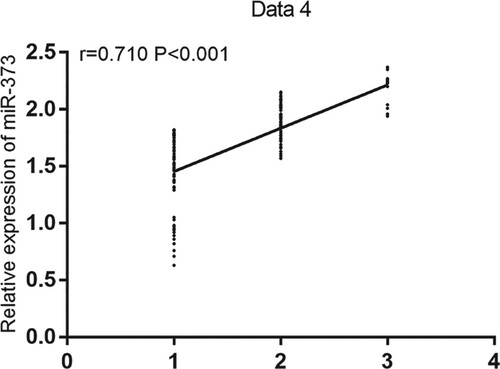
Serum miR-373 expression in patients with early, midterm, or advanced PD was 1.56 ± 0.27, 1.75 ± 0.31, or 2.08 ± 0.29, respectively. Compared with that in patients with early PD, serum miR-373 expression in patients with midterm or advanced PD was significantly upregulated (both P < 0.05), and the relative expression of serum miR-373 increased as PD aggravated. Likewise, early, midterm, and advanced PD were set to 1, 2, and 3, respectively. Using Spearman's correlation analysis, we found a positive correlation between the relative expression of serum miR-373 and PD severity (r = 0.710, P < 0.001; Figure ).
Discussion
PD is a degenerative disease of the nervous system, and its incidence continues to increase globally, posing a severe threat to human health (Torra et al. Citation2018; Uysal-Cantürk et al. Citation2018; van Wijk et al. Citation2018). Early intervention and control of PD are the key to improving the quality of life of affected patients; therefore, researchers are constantly searching for more sensitive and accurate diagnostic methods for PD (Kam et al. Citation2018; Lionnet et al. Citation2018). The clinical manifestation of early PD lacks specificity, and the traditional diagnostic and treatment methods for PD are defective; hence, its prognosis is not optimistic. Therefore, investigating biomarkers that are strongly linked to the diagnosis and prognosis of PD is crucial (Gupta et al. Citation2018).
MiRs are a class of endogenous, noncoding, single-stranded RNAs with tumor-promoting or tumor-suppressing function (Valentino et al. Citation2017). Abnormal miR expression is closely related to immunoregulation, and miRs play an important role in regulating the biological functions of the cells of the immune system (Turchinovich et al. Citation2016). According to some studies, miRs are related to changes in the substantia nigra pars compacta in patients with PD, and miRs are closely related to the mechanisms of neuroinflammation and nutrition (Wang et al. Citation2015; Chen et al. Citation2016; Yao et al. Citation2018). Some studies have reported that serum miR-30c-5p expression in patients with PD was significantly upregulated compared with that in normal individuals (Vallelunga et al. Citation2019). In the present study, we first analyzed the differences in serum miR-30c-5p and miR-373 expressions between patients with PD and healthy individuals, and we found that serum miR-30c-5p and miR-373 expression were significantly higher in patients with PD than in healthy individuals. By analyzing the relationship between serum miR-30c-5p and miR-373 expressions and the clinicopathological characteristics of patients with PD, we found that their expressions were related to history of tardive dyskinesia, postural instability, and gait disorder as well as Hoehn–Yahr, MMSE, and MADRS scores. However, only few studies have focused on the effects and clinical significance of the changes in serum miR-30c-5p and miR-373 expressions on PD progression and prognosis. Therefore, further studies and attention are required. In a follow-up study, the correlation of serum miR-30c-5p and miR-373 expressions in patients with PD can be further explored to improve the study results. According to our results, we hypothesized that miR-30c-5p and miR-373 play important roles in evaluating PD severity.
In this study, we investigated serum miR-30c-5p and miR-373 expressions in patients with PD and their expression in those with different PD severities; however, certain deficiencies exist. For example, the regulatory effect of changes in serum miR-30c-5p and miR-373 expressions on related leukocytes in patients with PD is not analyzed more specifically, and the biological function of the two factors is not explained further. Moreover, the clinical routine inflammatory factors of miR-30c-5p, miR-373, and PD were not analyzed, which had a certain influence on the improvement of the study design. Therefore, we will refer to the latest research in real time in the later stage, add corresponding research schemes, conduct more in-depth analysis and research, and make up for the design defects to constantly improve related research.
In conclusion, serum miR-30c-5p and miR-373 expressions were upregulated in patients with PD, and they may be involved in the occurrence and development of PD. Therefore, they can be used as potential serum biomarkers for the assessment of progressive brain changes in patients with PD.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
Additional information
Funding
References
- Alarcón CR, Lee H, Goodarzi H, Halberg N, Tavazoie SF. 2015. N6-methyladenosine marks primary microRNAs for processing. Nature. 519(7544):482–485. doi: https://doi.org/10.1038/nature14281
- Alessi DR, Sammler E. 2018. LRRK2 kinase in Parkinson’s disease. Science. 360(6384):36–37. doi: https://doi.org/10.1126/science.aar5683
- Berg D, Lang AE, Postuma RB, Maetzler W, Deuschl G, Gasser T, Siderowf A, Schapira AH, Oertel W, Obeso JA, et al. 2013. Changing the research criteria for the diagnosis of Parkinson’s disease: obstacles and opportunities. Lancet Neurol. 12(5):514–524. doi: https://doi.org/10.1016/S1474-4422(13)70047-4
- Chadly DM, Best J, Ran C, Bruska M, Woźniak W, Kempisty B, Schwartz M, LaFleur B, Kerns B, Kessler JA, et al. 2018. Developmental profiling of microRNAs in the human embryonic inner ear. PLoS One. 13(1):e0191452. doi: https://doi.org/10.1371/journal.pone.0191452
- Chen L, Mo M, Li G, Cen L, Wei L, Xiao Y, Chen X, Li S, Yang X, Qu S, Xu P. 2016. The biomarkers of immune dysregulation and inflammation response in Parkinson disease. Transl Neurodegener. 5(1):16. doi: https://doi.org/10.1186/s40035-016-0063-3
- Demirci S, Aynalı A, Demirci K, Demirci S, Arıdoğan BC. 2017. The serum levels of resistin and its relationship with other proinflammatory cytokines in patients with Alzheimer’s disease. Clin Psychopharmacol Neurosci. 15(1):59–63. doi: https://doi.org/10.9758/cpn.2017.15.1.59
- Gao Y, Nie K, Mei M, Guo M, Huang Z, Wang L, Zhao J, Huang B, Zhang Y, Wang L. 2018. Changes in cortical thickness in patients with early Parkinson’s disease at different Hoehn and Yahr stages. Front Hum Neurosci. 12:469. doi: https://doi.org/10.3389/fnhum.2018.00469
- Gupta D, Julka A, Jain S, Aggarwal T, Khanna A, Arunkumar N, de Albuquerque VHC. 2018. Optimized cuttlefish algorithm for diagnosis of Parkinson’s disease. Cogn Syst Res. 52:36–48. doi: https://doi.org/10.1016/j.cogsys.2018.06.006
- Kam T-I, Mao X, Park H, Chou S-C, Karuppagounder SS, Umanah GE, Yun SP, Brahmachari S, Panicker N, Chen R, et al. 2018. Poly (ADP-ribose) drives pathologic α-synuclein neurodegeneration in Parkinson’s disease. Science. 362(6414):eaat8407. doi: https://doi.org/10.1126/science.aat8407
- Kouchaki E, Kakhaki RD, Tamtaji OR, Dadgostar E, Behnam M, Nikoueinejad H, Akbari H. 2018. Increased serum levels of TNF-α and decreased serum levels of IL-27 in patients with Parkinson disease and their correlation with disease severity. Clin Neurol Neurosurg. 166:76–79. doi: https://doi.org/10.1016/j.clineuro.2018.01.022
- Lionnet A, Leclair-Visonneau L, Neunlist M, Murayama S, Takao M, Adler CH, Derkinderen P, Beach TG. 2018. Does Parkinson’s disease start in the gut? Acta Neuropathol. 135(1):1–12. doi: https://doi.org/10.1007/s00401-017-1777-8
- Marques TM, Kuiperij HB, Bruinsma IB, van Rumund A, Aerts MB, Esselink RA, Bloem BR, Verbeek MM. 2017. MicroRNAs in cerebrospinal fluid as potential biomarkers for Parkinson’s disease and multiple system atrophy. Mol Neurobiol. 54(10):7736–7745. doi: https://doi.org/10.1007/s12035-016-0253-0
- Mehta A, Baltimore D. 2016. MicroRNAs as regulatory elements in immune system logic. Nat Rev Immunol. 16(5):279–294. doi: https://doi.org/10.1038/nri.2016.40
- Patil KS, Basak I, Dalen I, Hoedt E, Lange J, Lunde KA, Liu Y, Tysnes O-B, Forsgren L, Aarsland D, et al. 2019. Combinatory microRNA serum signatures as classifiers of Parkinson’s disease. Parkinsonism Relat Disord. 64:202–210. doi: https://doi.org/10.1016/j.parkreldis.2019.04.010
- Peng Y, Croce CM. 2016. The role of MicroRNAs in human cancer. Signal Transduct Target Ther. 1:15004. doi: https://doi.org/10.1038/sigtrans.2015.4
- Torra A, Parent A, Cuadros T, Rodríguez-Galván B, Ruiz-Bronchal E, Ballabio A, Bortolozzi A, Vila M, Bové J. 2018. Overexpression of TFEB drives a pleiotropic neurotrophic effect and prevents Parkinson’s disease-related neurodegeneration. Mol Ther. 26(6):1552–1567. doi: https://doi.org/10.1016/j.ymthe.2018.02.022
- Turchinovich A, Tonevitsky AG, Burwinkel B. 2016. Extracellular miRNA: A collision of two paradigms. Trends Biochem Sci. 41(10):883–892. doi: https://doi.org/10.1016/j.tibs.2016.08.004
- Uysal-Cantürk P, Hanağası HA, Bilgiç B, Gürvit H, Emre M. 2018. An assessment of Movement Disorder Society Task Force diagnostic criteria for mild cognitive impairment in Parkinson’s disease. Eur J Neurol. 25(1):148–153. doi: https://doi.org/10.1111/ene.13467
- Valentino A, Reclusa P, Sirera R, Giallombardo M, Camps C, Pauwels P, Crispi S, Rolfo C. 2017. Exosomal microRNAs in liquid biopsies: future biomarkers for prostate cancer. Clin Transl Oncol. 19(6):651–657. doi: https://doi.org/10.1007/s12094-016-1599-5
- Vallelunga A, Iannitti T, Dati G, Capece S, Maugeri M, Tocci E, Picillo M, Volpe G, Cozzolino A, Squillante M. 2019. Serum miR-30c-5p is a potential biomarker for multiple system atrophy. Mol Biol Rep. 46(2):1661–1666. doi: https://doi.org/10.1007/s11033-019-04614-z
- van Wijk BC, Cagnan H, Litvak V, Kühn AA, Friston KJ. 2018. Generic dynamic causal modelling: an illustrative application to Parkinson’s disease. Neuroimage. 181:818–830. doi: https://doi.org/10.1016/j.neuroimage.2018.08.039
- Wang Q, Liu Y, Zhou J. 2015. Neuroinflammation in Parkinson’s disease and its potential as therapeutic target. Transl Neurodegener. 4(1):19. doi: https://doi.org/10.1186/s40035-015-0042-0
- Wang W, Wang X, Fujioka H, Hoppel C, Whone A, Caldwell M, Cullen P, Liu J, Zhu X. 2016. Parkinson’s disease-associated mutant VPS35 causes mitochondrial dysfunction by recycling DLP1 complexes. Nat Med. 22(1):54–63. doi: https://doi.org/10.1038/nm.3983
- Wei F, Wang Q, Su Q, Huang H, Luan J, Xu X, Wang J. 2016. miR-373 inhibits glioma cell U251 migration and invasion by down-regulating CD44 and TGFBR2. Cell Mol Neurobiol. 36(8):1389–1397. doi: https://doi.org/10.1007/s10571-016-0338-3
- Wei H, Xu Y, Xu W, Zhou Q, Chen Q, Yang M, Feng F, Liu Y, Zhu X, Yu M, Li Y. 2018. Serum Exosomal miR-223 serves as a potential diagnostic and prognostic biomarker for dementia. Neuroscience. 379:167–176. doi: https://doi.org/10.1016/j.neuroscience.2018.03.016
- Yang X, Lou Y, Liu G, Wang X, Qian Y, Ding J, Chen S, Xiao Q. 2017. Microglia P2Y6 receptor is related to Parkinson’s disease through neuroinflammatory process. J Neuroinflammation. 14(1):38. doi: https://doi.org/10.1186/s12974-017-0795-8
- Yao L, Ye Y, Mao H, Lu F, He X, Lu G, Zhang S. 2018. MicroRNA-124 regulates the expression of MEKK3 in the inflammatory pathogenesis of Parkinson’s disease. J Neuroinflammation. 15(1):13. doi: https://doi.org/10.1186/s12974-018-1053-4
- Zhong K, Chen K, Han L, Li B. 2014. MicroRNA-30b/c inhibits non-small cell lung cancer cell proliferation by targeting Rab18. BMC Cancer. 14(1):703. doi: https://doi.org/10.1186/1471-2407-14-703