ABSTRACT
The purpose of the study was to investigate the expressions of VEGF and pAkt protein, and to evaluate their clinicopathological implications in oral cancer. A total of 55 cases of oral squamous cell carcinoma (OSCC) and 15 cases of inflammatory lesion as a control were included in the study. Expression pattern of pAkt and VEGF protein was evaluated through immunohistochemistry. In addition, apoptosis was evaluated using TUNEL assay. pAkt protein showed 43.63% expression in oral cancer, whereas undetectable expression was noticed in inflammatory lesion (p 0.05). High expression of VEGF (58.18%) was noticed in OSCC cases and inflammatory lesion did not show any expression. The expression pattern of VEGF increased with increasing the grade of tumour and difference in expression pattern among grades was statistically significant (p < 0.05). Moreover, VEGF protein expression was associated with stages of the cancer). The positive rate of apoptosis was 70.9%. The percentage of apoptotic cells varied from 0.3% to 20.3% and the median was 9.5. These findings advocate that overexpression of VEGF and upregulation of Akt may play a role in the development and progression of oral squamous cell carcinoma.
Introduction
Oral cancer is one of the most common types of cancer worldwide and holds a major health problem in developing countries, representing the leading cause of death (Mehrotra et al. Citation2006). Besides, oral squamous cell carcinoma (OSCC) represents 80–90% of all oral malignancies (Massano et al. Citation2006; Sambandam et al. Citation2016). In spite of improvements in cancer treatment, oral cancer remains the substantial cause of morbidity and mortality worldwide. The 5-year survival rate for oral cancers is lowest among other cancers including skin melanomas and cancer of the testis, breast, colon, rectum and kidneys (Conway et al. Citation2009). The exact mechanism behind the development and progression of oral cancer is not completely known. Although some of the genetic and epigenetic events underlying this complex disease have been recognised (Agrawal et al. Citation2011; Stransky et al. Citation2011), specific biomarkers are lacking along with costly therapeutic alternatives for this dreaded disease. Various markers are in use to diagnose oral cancer. But, new treatment strategies are needed (Mao and Hong Citation2004) and evaluation of specific biomarkers will be essential towards the diagnosis and treatment of oral cancer. Angiogenesis, the development of new blood vessels from the pre-existing ones, is one of the vital phenotypes of tumour formation and also significant in the normal physiological processes such as growth and development, wound healing and reproduction (Hasina and Lingen Citation2001). Angiogenesis plays an important role in the development and progression of tumour. Growth of tumours needs a large amount of blood supply in order to reach sufficient oxygen and nutrients to the tissues. In various experimental models, it has been established that carcinogenesis is linked with new vessel formation (Carmeliet Citation2000). Vascular endothelial growth factor (VEGF) is a significant angiogenic factor that plays a role in cancer development and progression. Previous studies have confirmed that over-expression of VEGF is linked with poor survival in cancer patients including ovarian cancer, lung cancer, colorectal cancer and other types of tumours (Cheng et al. Citation2013; Tsai et al. Citation2013; Gkiozos et al. Citation2015; Hou et al. Citation2015). Anti-angiogenesis therapy is an essential step in the cancer prevention and a previous study confirmed that 153Sm-Bevasesomab and Salvia Officinalis act as anti-angiogenesis (Yavari Citation2018; Zare Citation2019).
The serine/threonine kinase AKT shows critical roles in several features of cancer pathophysiology, containing cell survival, growth, metabolism as well as metastasis (Brazil et al. Citation2004; Hennessy et al. Citation2005; Manning and Cantley Citation2007). There are three subtypes of the serine/threonine kinase AKT in mammals, AKT1, AKT2 and AKT3, which are main molecules in the PI3K signal transduction pathway (Toulany et al. Citation2017). AKT appears to be a sensible target for cancer therapy on the grounds that the PI3K/AKT signalling pathway is commonly dysregulated in up to 70% of human breast cancer (Lopez-Knowles et al. Citation2010) and upregulation of AKT in cancer is linked with overall poor prognosis (Pérez-Tenorio and Stål Citation2002). The significance of AKT in human cancer is principally inferred from commonly happening mutations in the enzymes that regulate the activity of these second messenger phospholipids (PtdIns(3,4,5)P3, PtdIns(3,4)P2) and finally cause the activation of AKT through membrane recruitment (Kumar and Purohit Citation2013). Moreover, Akt has further effects on tumour-induced angiogenesis that are mediated, in part, via hypoxia inducible factor 1α and vascular endothelial growth factor (VEGF) (Vivanco and Sawyers Citation2002).
Apoptosis is a very tightly programmed cell death with distinct biochemical and genetic pathways that shows an important role in the development and homeostasis in normal tissues (Lockshin and Williams Citation1965). There are four main factors which are involved in activating and prompting the apoptotic process. These are the caspases, Bcl-2 family of proteins, tumour necrosis factor receptor (TNF-R) superfamily, as well as adaptor proteins (Strasser et al. Citation2000). It is commonly recognized that apoptosis is vital in the death of normal and neoplastic cells (Nakano Citation1997). Moreover, Bcl-2, Bcl-xL and Mcl-1 proteins appear to inhibit apoptosis, while Bax, Bad and Bcl-xS proteins seemingly play a role in the promotion of apoptosis (Oltvai et al. Citation1993; Hockenbery Citation1994). Previous findings reported that the over-expression of Bax, Bcl-2 and Bcl-XL was linked with better overall survival, lower recurrence rates and disease-specific survival in oral squamous cell carcinoma (Camisasca et al. Citation2009). The current study was performed to explore the expression pattern of phosphorylated protein kinase B (pAkt) and VEGF protein in oral cancer and correlation of expression profiles with clinicopathological features of the tumour. Apoptotic cells were detected in samples of oral tissues using terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labelling (TUNEL) assay kit and the result was interpreted accordingly.
Materials and methods
Study design and patient selection
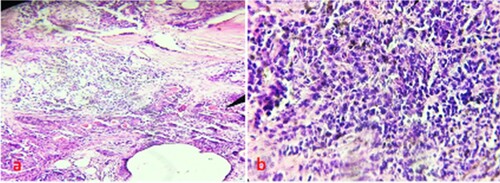
A total of 55 cases of OSCC including 40 males and 15 females, aged 48 years on average were included in the study. A total of 15 cases of inflammatory lesion of oral were used as a control group. The ethical approval and consent was taken from patients and haematoxylin and eosin staining was performed to confirm the cancer and inflammatory lesion Figure a,b. A total of 25 cancer cases were of good differentiation, 17 cases were of moderate differentiation and 13 cases of poor differentiation. Moreover, 34 cases in I-II stage and 21 cases in III-IV stage.
Expressional evaluation of pAkt and VEGF protein
A 4–5 μm-thick sections were made from paraffin-embedded block of OSCC and inflammatory lesion. Expression pattern of pAkt and VEGF protein was evaluated through immunohistochemistry staining as done in previously explained methods (Rahmani et al. Citation2015; Babiker et al. Citation2018)[Flow diagram]. Briefly, tissue sections were deparaffinized using xylene and antigen retrieval was performed in citrate buffer to unmask the antigen site. Endogenous peroxidase activity was blocked as per the manufacture’s instruction. Sections were incubated overnight at 4°C with mouse monoclonal anti-human pAkt and VEGF antibody. Subsequently, the tissue was incubated with biotinylated rabbit anti-mouse antibody, followed by incubation with streptavidin–biotin enzyme complex. Diaminobenzidine (DAB) chromogen was used and then the tissue was counterstained with haematoxylin.
Scoring method
pAkt and VEGF protein exhibited less than or equal to 05% of cells that displayed positivity is considered as a negative case. If more than 05% of cells were positive for pAkt and VEGF was considered as a positive case. The expression positivity was applied, +1 for 5–20% considered as a weak expression, +2 for 21–60% as moderate positivity and +3 for more than 60% considered as a strong expression. A total of 5 fields from each tissue were selected, and 100 cells from each field were counted and the result was interpreted accordingly and photographed to analyse the expression pattern.
In-situ apoptosis detection
Apoptotically fragmented cellular DNA was identified by the TUNEL assay (Apoptosis Detection Kit). Apoptotic activity was quantified by the apoptotic index, which represented the percentage of apoptotic epithelial cells in each tissue. The result was analysed and photograph was taken.
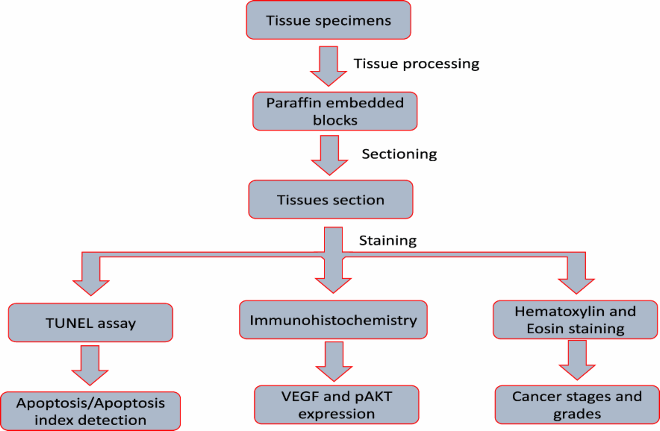
Flow diagram of methodology
Statistical analysis
Expression of markers was correlated with clinicopathological features of the patients. Chi-square (λ)2 test was used to make the correlation of marker with age, sex, grade and stage of the tumour. Statistical significance was defined as p < 0.05.
Result
Expression pattern of pAkt protein
Expression pattern of pAkt was examined through immunohistochemistry and it was noticed that pAkt showed cytoplasmic expression in 24/55 (43.63%) of oral cancer cases (Figure a, b, c) and 31/55 (56.36%) cases did not show any expression. But, pAkt protein did not show any expression in inflammatory oral lesion. Moreover, pAkt expression was further analysed based on age, sex, histological grade and stage of the tumour. The expression of pAkt was noticed in different grades of the tumour as 8/19 cases (42.10%), 9/20 cases (45%) and 7/16 cases (43.75%) in Grade I, Grade II and Grade III, respectively (Table ) and difference in expression pattern among the different grade was statically insignificant (p>0.05). In addition, phosphorylated Akt was not associated with different stages of oral cancer (Stage I/II vs III/IV) and this was statically significant (p<0.05).
Figure 2. Immunohistochemical staining a: pAkt showed cytoplasmic expression in oral cancer b:pAkt expression was negative in oral cancer c: Inflammatory lesion did not express pAkt.
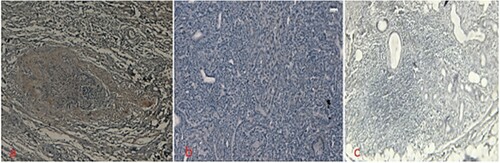
Table 1. pAkt expression pattern and its association with clinicopathological findings.
The oral cancer cases were categorized into two groups according to age of the patients as <50 years (21 cases, 38.18%) and ≥50 years (34 cases, 61.81%), and expression pattern among different age group was statically insignificant (p>0.05) Table . Also, expression pattern was analysed according to gender and it was found insignificant (p>0.05).
Expression of VEGF protein
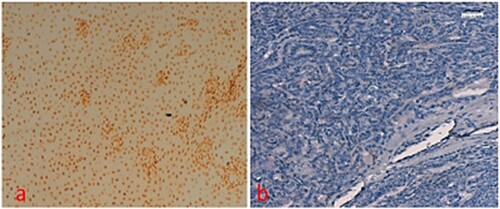
VEGF expressions were noticed in 58.18% (32/55) in oral cancer and positivity was cytoplasmic (Table ), whereas expression was not seen in inflammatory oral lesion (Figure a, b, c). The difference in expression pattern in oral cancer and inflammatory lesions of oral was statistically significant (p < 0.05). Expression of VEGF was analysed as per the gender, age, grade and stage of the tumour. The expression pattern was noticed in different grades of the tumour as (9 cases, 47.36%), (12 cases, 60%) and (11 cases, 68.75%) in Grade I, Grade II and Grade III, respectively (Table ) and VEGF positivity was statistically significant among the different grades. Furthermore, VEGF expression was associated with stages of cancer and the expression pattern was statically significant (p < 0.05). The oral cancer cases were categorized into two groups according to age of the patients as<50 years and ≥ 50 years, and the expression pattern among different age groups was statically insignificant (p>0.05) Table . Also, expression pattern was analysed according to male/female patients and it was found to be insignificant (p>0.05).
Figure 3. Immunohistochemical staining a: VEGF showed cytoplasmic expression in oral cancer b: VEGF expression was negative in oral cancer c: Inflammatory lesion did not show any expression.
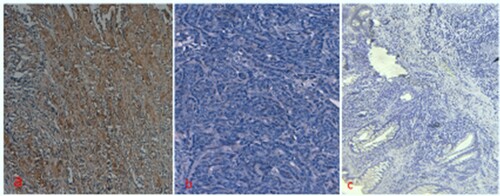
Table 2. VEGF expression pattern and its association with clinical parameters of oral cancer cases.
The association between pAkt and VEGF expression
pAkt expression was positively correlated with VEGF expression as the expression of both markers was high in oral cancer cases and inflammatory lesion of oral tissue did not show any expression for pAkt and VEGF proteins. Expression pattern of both marker pAkt and VEGF was statically insignificant in age, sex and stage/ grades of the cancer.
Evaluation of apoptosis
Apoptotic cells were detected and brown staining was considered as TUNEL staining positive. Cells were examined in five random high-power fields (>500 cells). The apoptotic index was measured by counting the percentage of positive cells from at least 500 cells. The positive rate of apoptosis was seen 70.9% (39/55) Figure a-b. The percentage of apoptotic cells varied from 0.3 to 20.3% and median was 9.5. Positivity of apoptosis according to the tumour grade was analysed and it was found insignificant. Apoptosis was not detected in inflammatory lesion of oral tissues.
Discussion
Cancer is a multi-factorial disease including alterations of various cell-signalling pathways. Moreover, inactivation of tumour suppressor genes, activation of angiogenesis process, inhibition of apoptosis, inactivation of various growth factor and transcription factor have been noticed in the cancer. Akt is a serine threonine protein kinase with oncogenic and anti-apoptotic activities (Gasinska et al. Citation2004). Phosphorylated AKT (p-AKT), the active form of AKT that affects a variety of cellular functions, comprising differentiation, motility, cell growth, proliferation, survival and intracellular trafficking (Vivanco and Sawyers Citation2002). The class I phosphoinositide 3 kinase/protein kinase B (PI3 K/AKT) signalling pathway is one of the most frequently dysregulated pathways in cancer (Yuan and Cantley Citation2008; Courtney et al. Citation2010). Phosphorylation of AKT modulates signals from phosphatase and tensin homologue deleted on chromosome 10 (PTEN) and the mammalian target of rapamycin (mTOR), causing varied effects of AKT on cells (Cheung and Testa Citation2013) and AKT1 is accepted as an apoptotic inhibitor that contributes to cancer progression (Cheung and Testa Citation2013).
This study reported that pAkt showed expression in 43.63% oral cancers cases, whereas pAKT expression was not seen in inflammatory oral lesion. Expression of pAkt was noticed in different grades of the tumour as (8/19 cases, 42.10%), (9/20 cases, 45%) and (16/7 cases, 43.75%) in Grade I, Grade II and Grade III, respectively and difference in expression pattern among the different grade was statically insignificant. In addition, phosphorylated Akt was not associated with stages of cancer and this finding was statically insignificant (p > 0.05).
Our observations are in consistent with the study of Lim et al. Citation2005, which demonstrated high pAKT such as 34.52% expression in oral cancer (Lim et al. Citation2005 Nov) and pAkt expression was as high as 68.5% in OSCC and more often expressed in late stages of tumour (Alyasiri et al. Citation2012). Another study based on oral squamous cell carcinoma was performed and the finding revealed that patients that showed 38.8% had high pAKT expression and lower pAKT expressions were associated with pathological remission (Lyu et al. Citation2016). In addition to this, the expression of pan-Akt and its phosphorylated form (p-Akt), Akt1 and Akt2 in oral squamous cell carcinoma (OSCC) specimens was measured to investigate its role in oral cancer. Finding of the study revealed that pan-Akt was expressed in 80% of oral squamous cell carcinoma cases, whereas Akt1, Akt2 and p-Akt were expressed in all oral squamous cell carcinoma cases (Lamaroon and Krisanaprakornkit Citation2009).
Angiogenesis is the formation of new blood vessels and it plays a vital role in tumour growth as tumours were required to form blood vessels to supply oxygen as well as nutrients for growth and spread. Vascular endothelial growth factor (VEGF) is considered as a main mediator of angiogenesis and VEGF is required for normal embryonic vasculogenesis and angiogenesis was shown by two separate landmark studies (Carmeliet and Jain Citation2000; Ferrara Citation2001). Activation or upregulation of angiogenesis is one of the important processes in the tumour development and progression. In this context, VEGF plays an important role in the tumour growth and its upregulation or overexpression of VEGF has been recognized in many tumours. In the current study, it was observed that VEGF expression was high in oral cancer, whereas expression was not seen in inflammatory lesion and difference in expression pattern among cancer and control cases was statistically significant (p < 0.05). Moreover, the expression pattern among different grades of the cancer was statistically significant (p < 0.05). Besides, VEGF expression was associated with stages of cancer.
Our observations are consistent with the study of Lim et al. Citation2005, VEGF showed high expression in oral cancer cases as 67.85% cancer cases showed expression (Lim et al. Citation2005 Nov). There was no significant correlation between VEGF expression and clinicopathological factors (Lim et al. Citation2005 Nov). It was noticed that expression pattern increased with increasing tumour grade and difference in expression pattern among grade was statistically significant and VEGF expression was also associated with stages of cancer. This result is consistent with the previous study, and it was reported that poorly differentiated and invasive oral squamous cell carcinoma showed high-level staining of VEG. A significant correlation was observed between immunohistochemical VEGF expression and histological differentiation, tumour size of specimens (Kim et al. Citation2015 Feb). Another study finding was in accordance with our result as all evaluated cases were VEGF-positive. Moreover, positivity of VEGF was increased with the grade of tumours as 10% had Grade 1 VEGF positivity, 25% had Grade 2 VEGF positivity and 65% had Grade 3 VEGF positivity. Moreover, VEGF expression was more common in the cancerous tissue than the normal surrounding mucosa taken as control. Thus, VEGF is overexpressed in cancerous mucosa than non-cancerous mucosa (Singhal et al. Citation2016). Besides, another study showed the role of VEGF in oral carcinomas and it was VEGF-A and -C expression in 76 out of 120 SCCs (63%) and 81 out of 120 SCCs (67.5%), respectively, while their expression was not seen in any normal tissue samples. Hence, the study suggests that VEGF-A and -C expression is linked with oral carcinoma (Naruse et al. Citation2011). VEGF plays a significant role in the development and progression of cancer and high expression of VEGF was seen high grade and stages of tumour. In this regard, a pioneer study result demonstrated that vascular endothelial growth factor and microvessel density associated statistically significant with the clinical stage, tumours located in the oral cavity and larynx more often expressed high VEGF immunostaining compared with tumours located in the lower lip. High VEGF expression was associated with higher clinical stage and worse overall survival in the cohort of patients (Kyzas et al. Citation2005). Previous study based on urinary bladder was in accordance with the current study as VEGF was highly expressed in cancer cases and inflammatory lesion did not show any expression. Expression of VEGF increased with tumour progression [Rahmani et al. Citation2012].
Various types of stimuli play a role in the induction of apoptosis in a cell type-dependent manner (Yang and Korsmeyer Citation1996; Rahmani et al. Citation2012). The terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate (dUTP)-biotin nick end labelling (TUNEL) method that identifies apoptosis and programmed cell death has been used to recognize apoptotic cells at the single-cell level (Gavrieli et al. Citation1992). In the current study, apoptosis was seen in 70.9% (39/55) in oral cancer cases and apoptosis was not seen in inflammatory lesion. The percentage of apoptotic cells varied from 0.3 to –20.3% and the median was 9. 5 and a pioneer finding result revealed that positive staining for apoptosis was observed in 92% of the slides, with apoptotic index from 0 to 83 and mean AI was 27.4 (Watanabe et al. Citation1999). This finding was in agreement with earlier finding, TUNEL immunostaining was found to be positive in most of the tongue cancer specimens analysed as compared with healthy controls (in which it was always negative=0), advising TUNEL as a possible indicator in the carcinogenic process (Ben-Izhak et al. Citation2008). In this content, a previous finding study demonstrated that the apoptosis index (AI) was significantly higher in the pelvic lymph nodes and primary tumours from patients in the neoadjuvant chemotherapy group compared with the control group (Thompson Citation1995; Cheung and Testa Citation2013). Another study based on urinary bladder cancer reported that apoptosis was seen in cancer cases, whereas inflammatory lesion did not show apoptosis (Zhang et al. Citation2014 Jun; Vivanco and Sawyers Citation2002).
Conclusion
Angiogenesis is the process which forms new blood vessels and VEGF is one of the features of tumour dedifferentiation and plays a role in the development and progression of oral cancer. Our finding advocates that expressions of VEGF protein are linked with cancer grades and stages. VEGF protein is overexpressed in oral cancer cases and its expression in inflammatory lesion of oral was undetectable. The results of the present study designated that pAkt is overexpressed in oral cancer tumours and its expression in inflammatory lesion of oral undetectable. The results of this study confirm the role of pAKt and VEGF in the development and progression of oral cancer. Overexpression of VEGF and upregulation of Akt is an important prognostic factor for oral squamous cell carcinoma.
Acknowledgement
Researchers would like to thank the Deanship of Scientific Research, Qassim University, Saudi Arabia for funding publication of this project.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Agrawal N, Frederick MJ, Pickering CR, Bettegowda C, Chang K, Li RJ, Fakhry C, Xie T-X, Zhang J, Wang J, et al. 2011. Exome sequencing of headand neck squamous cell carcinoma reveals inactivating mutations inNOTCH1. Science. 6046:1154–1157. doi: https://doi.org/10.1126/science.1206923
- Alyasiri NS, Mehdi SJ, Alam MS, Ali A, Mandal AK, Gupta S, Singh I, Rizvi MM. 2012. PTEN-mediated AKT activation contributes to the reduced apoptosis among Indian oral squamous cell carcinoma patients. J Cancer Res Clin Oncol. 138(1):103–109. doi: https://doi.org/10.1007/s00432-011-1077-y
- Babiker AY, Almatroudi A, Allemailem KS, Husain NEOS, Alsammani MA, Alsahli MA, Rahmani AH. 2018. Clinicopathologic aspects of squamous cell carcinoma of the uterine cervix: role of PTEN, BCL2 and P53. Appl. Sci. 8:2124. doi: https://doi.org/10.3390/app8112124
- Ben-Izhak O, Laster Z, Akrish S, Cohen G. 2008. Nagler RM: TUNEL as a tumor marker of tongue cancer. Anticancer Res. 28:2981–2986.
- Brazil DP, Yang ZZ, Hemmings BA. 2004. Advances in protein kinase B signalling: AKTion on multiple fronts. Trends Biochem Sci. 29(5):233–242. doi: https://doi.org/10.1016/j.tibs.2004.03.006
- Camisasca DR, Honorato J, Bernardo V, da Silva LE, da Fonseca EC, de Faria PA, Dias FL. 2009. Lourenco SQ: expression of Bcl-2 family proteins and associated clinicopathologic factors predict survival outcome in patients with oral squamous cell carcinoma. Oral Oncol. 45:225–233. doi: https://doi.org/10.1016/j.oraloncology.2008.05.021
- Carmeliet P. 2000. Mechanisms of angiogenesis and arteriogenesis. Nat Med. 6(4):389–395. doi: https://doi.org/10.1038/74651
- Carmeliet P, Jain RK. 2000. Angiogenesis in cancer and other diseases. Nature. 407:249–257. doi: https://doi.org/10.1038/35025220
- Cheng D, Liang B, Li Y. 2013. Serum vascular endothelial growth factor (VEGF-C) as a diagnostic and prognostic marker in patients with ovarian cancer. PLoS One. 8:e55309. doi: https://doi.org/10.1371/journal.pone.0055309
- Cheung M, Testa JR. 2013. Diverse mechanisms of AKT pathway activation in human malignancy. Curr Cancer Drug Targets. 13(3):234-244. doi: https://doi.org/10.2174/1568009611313030002
- Conway DI, Hashibe M, Boffetta P, Wunsch-Filho V, Muscat J, La Vecchia C, Winn DM. 2009. Enhancing epidemiologic research on head and neck cancer: INHANCE – The international head and neck cancer epidemiology consortium. Oral Oncol. 45:743–746. doi: https://doi.org/10.1016/j.oraloncology.2009.02.007
- Courtney KD, Corcoran RB, Engelman JA. 2010. The PI3K pathway as drug target in human cancer. J ClinOncol. 28( ):1075–1083. doi: https://doi.org/10.1200/JCO.2009.25.3641
- Ferrara N. 2001. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am J Physiol Cell Physiol. 280:C1358–C1366. doi: https://doi.org/10.1152/ajpcell.2001.280.6.C1358
- Gasinska A, Fowler JF, Lind BK. 2004. Influence of overall treatment time and radiobiological parameters on biologically effective doses in cervical cancer patients treated with radiation therapy alone. ActaOncol. 43:657–666.
- Gavrieli Y, Sherman Y, Ben-Sasson SA. 1992. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J. Cell Biol. 119:493–501. doi: https://doi.org/10.1083/jcb.119.3.493
- Gkiozos I, Tsagouli S, Charpidou A, Grapsa D, Kainis E, Gratziou C, Syrigos K. 2015. Levels of vascular endothelial growth factor in serum and pleural fluid are independent predictors of survival in advanced non-small cell lung cancer: results of a prospective study. Anticancer Res. 35:1129–1137.
- Hasina R, Lingen MW. 2001. Angiogenesis in oral cancer. J Dent Educ. 65:1282–1290. doi: https://doi.org/10.1002/j.0022-0337.2001.65.11.tb03489.x
- Hennessy BT, Smith DL, Ram PT, Lu Y, Mills GB. 2005. Exploiting the PI3K/AKT pathway for cancer drug discovery. Nat Rev Drug Discov. 4(12):988–1004. doi: https://doi.org/10.1038/nrd1902
- Hockenbery DM. 1994. Bcl-2 in cancer, development and apoptosis. J Cell Sci. 18:51–55. doi: https://doi.org/10.1242/jcs.1994.Supplement_18.7
- Hou X, Wei JC, Fu JH, Wang X, Luo RZ, He JH, Zhang LJ, Lin P, Yang HX. 2015. Vascular endothelial growth factor is a Useful Predictor of Postoperative Distant metastasis and survival prognosis in Esophageal squamous cell carcinoma. Ann SurgOncol. 22:3666–3673.
- Kim S-K, Park S-G, Kim K-W. 2015 Feb. Expression of vascular endothelial growth factor in oral squamous cell carcinoma. J Korean Assoc Oral Maxillofac Surg. 41(1):11–18. doi: https://doi.org/10.5125/jkaoms.2015.41.1.11
- Kumar A, Purohit R. 2013. Cancer associated E17K mutation causes rapid conformational drift in AKT1 Pleckstrin Homology (PH) Domain. PLoS ONE. 8(5). Article ID e64364. doi: https://doi.org/10.1371/annotation/7c19edf3-4648-45de-8132-16092fc4bf40
- Kyzas PA, Stefanou D, Batistatou A, Agnantis NJ. 2005. Prognostic significance of VEGF immunohistochemical expression and tumor angiogenesis in head and neck squamous cell carcinoma. J Cancer Res Clin Oncol. 131(9):624–630. doi: https://doi.org/10.1007/s00432-005-0003-6
- Lamaroon A, Krisanaprakornkit S. 2009. Overexpression and activation of Akt2 protein in oral squamous cell carcinoma. Oral Oncol. 45:e175–e179. doi: https://doi.org/10.1016/j.oraloncology.2009.06.003
- Lim J, Kim J-H, Paeng J-Y, Kim M-J, Hong S-D, Lee J-I, Hong S-P. 2005 Nov. Prognostic value of activated Akt expression in oral squamous cell carcinoma. J ClinPathol. 58(11):1199–1205.
- Lockshin RA, Williams CM. 1965. Programmed cell death-I. Cytology of degeneration in the intersegmental muscles of the Pernyi silkmoth. J Insect Physiol. 11(2):123–133. doi: https://doi.org/10.1016/0022-1910(65)90099-5
- Lopez-Knowles E, O'Toole SA, McNeil CM, Millar EK, Qiu MR, Crea P, Daly RJ, Musgrove EA, Sutherland RL. 2010. PI3K pathway activation in breast cancer is associated with the basal-like phenotype and cancer-specific mortality. Int J Cancer. 126(5):1121–1131. doi: https://doi.org/10.1002/ijc.24831
- Lyu J, Song H, Tian Z, Miao Y, Ren G, Guo W. 2016. Predictive value of pAKT/PTEN expression in oral squamous cell carcinoma treated with cetuximab-based chemotherapy. Oral Surg Oral Med Oral Pathol Oral Radiol. 121(1):67–72. doi: https://doi.org/10.1016/j.oooo.2015.09.002
- Manning BD, Cantley LC. 2007. AKT/PKB signaling: navigating downstream. Cell. 129(7):1261–1274. doi: https://doi.org/10.1016/j.cell.2007.06.009
- Mao L, Hong WK. 2004. Papadimitrakopoulou VA: Focus on head and neck cancer. Cancer Cell. 5:311–316. doi: https://doi.org/10.1016/S1535-6108(04)00090-X
- Massano J, Regateiro FS, Januário G, Ferreira A. 2006. Oral squamous cell carcinoma: review of prognostic and predictive factors. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontol. 102(1):67–76. doi: https://doi.org/10.1016/j.tripleo.2005.07.038
- Mehrotra R, Gupta A, Singh M, Ibrahim R. 2006. Application of cytology and molecular biology in diagnosing premalignant or malignant oral lesions. Mol Cancer. 5:11. doi: https://doi.org/10.1186/1476-4598-5-11
- Nakano R. 1997. Apoptosis: gene-directed cell death. An Overview. Horm. Res. 48(Suppl.3):2–4. doi: https://doi.org/10.1159/000191292
- Naruse T, Kawasaki G, Yanamoto S, Mizuno A, Umeda M. 2011. Immunohistochemical study of VEGF expression in oral squamous cell carcinomas: correlation with the mTOR-HIF-1α pathway. Anticancer Res. 31:4429–4437.
- Oltvai ZN, Milliman CL, Korsmeyer SJ. 1993. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell. 74:609–619. doi: https://doi.org/10.1016/0092-8674(93)90509-O
- Pérez-Tenorio G, Stål O. 2002. Activation of AKT/PKB in breast cancer predicts a worse outcome among endocrine treated patients. Br J Cancer. 86(4):540–545. doi: https://doi.org/10.1038/sj.bjc.6600126
- Rahmani A, Alzohairy M, Khadri H, Mandal AK, Rizvi MA. 2012. Expressional evaluation of vascular endothelial growth factor (VEGF) protein in urinary bladder carcinoma patients exposed to cigarette smoke. Int J ClinExpPathol. 5:195–202.
- Rahmani AH, Babiker AY, AlWanian WM, Elsiddig SA, Faragalla HE, Aly SM. 2015. Association of cytokeratin and vimentin protein in the genesis of transitional cell carcinoma of urinary bladder patients. Dis Markers. 2015:204759. doi: https://doi.org/10.1155/2015/204759
- Sambandam Y, Sakamuri S, Balasubramanian S, Haque A. 2016. RANK ligand modulation of autophagy in oral squamous cell carcinoma tumor cells. J Cell Biochem. 117(1):118–125. doi: https://doi.org/10.1002/jcb.25255
- Singhal A, Hadi R, Chaturvedi A, Sharma ID, Misra S, Husain N. 2016. Vascular endothelial growth factor expression in oral cancer and its role as a predictive marker: a prospective study. Saudi Surg J. 4(2):52–56. doi: https://doi.org/10.4103/2320-3846.183673
- Stransky N, Egloff AM, Tward AD, Kostic AD, Cibulskis K, Sivachenko A, Kryukov GV, Lawrence MS, Sougnez C, McKenna A, et al. 2011. The mutational landscape of head and neck squamous cell carcinoma. Science. 6046:1157–1160. doi: https://doi.org/10.1126/science.1208130
- Strasser A, O'Connor L, Dixit VM. 2000. Apoptosis signaling. Annu Rev Biochem. 69:217–245. doi: https://doi.org/10.1146/annurev.biochem.69.1.217
- Thompson CB. 1995. Apoptosis in the pathogenesis and treatment of disease. Science. 267:1456–1462. doi: https://doi.org/10.1126/science.7878464
- Toulany M, Maier J, Iida M, Rebholz S, Holler M, Grottke A, Jüker M, Wheeler DL, Rothbauer U, Rodemann HP. 2017. Akt1 and Akt3 but not Akt2 through interaction with DNA-PKcs stimulate proliferation and post-irradiation cell survival of K-RAS-mutated cancer cells. Cell Death Discov. 3:170722017. doi: https://doi.org/10.1038/cddiscovery.2017.72
- Tsai HL, Yang IP, Lin CH, Chai CY, Huang YH, Chen CF, Hou MF, Kuo CH, Juo SH, Wang JY. 2013. Predictive value of vascular endothelial growth factor overexpression in early relapse of colorectal cancer patients after curative resection. Int J Colorectal Dis. 28:415–424. doi: https://doi.org/10.1007/s00384-012-1570-z
- Vivanco I, Sawyers CL. 2002. The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer. 2:489–501. doi: https://doi.org/10.1038/nrc839
- Watanabe I, Toyoda M, Okuda J, Tenjo T, Tanaka K, Yamamoto T, Kawasaki H, Sugiyama T, Kawarada Y, Tanigawa N. 1999. Detection of apoptotic cells in human colorectal cancer by Two different in situ methods: antibody against single-stranded DNA and terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick End-labeling (TUNEL) methods. Jpn J Cancer Res. 90(2):188–193. doi: https://doi.org/10.1111/j.1349-7006.1999.tb00732.x
- Yang E, Korsmeyer SJ. 1996. Molecular thanatopsis: a discourse on the BCL2 family and cell death. Blood. 88:386–401. doi: https://doi.org/10.1182/blood.V88.2.386.bloodjournal882386
- Yavari K. 2018. Anti-angiogenesis therapy of cancer cells using 153Sm-Bevasesomab. Emerg Sci J. 2(3):130–139. doi: https://doi.org/10.28991/esj-2018-01136
- Yuan TL, Cantley LC. 2008. PI3K pathway alterations in cancer: variations on a theme. Oncogene. 27:5497–5510. doi: https://doi.org/10.1038/onc.2008.245
- Zare H. 2019. Effects of Salvia Officinalis Extract on the breast cancer cell Line. SciMedicine Journal. 1:25–29. doi: https://doi.org/10.28991/SciMedJ-2019-0101-4
- Zhang H, Peng W, Zhang Y. 2014 Jun. Detection of cell apoptosis in pelvic lymph nodes of patients with cervical cancer after neoadjuvant chemotherapy. J Int Med Res. 42(3):641–650. doi: https://doi.org/10.1177/0300060513506328