Abstract
Background: Serum cystatin C (Scyst) has been suggested as an alternative index of glomerular filtration rate (GFR) and could be useful in renal transplant patients. Methods: In a 60‐subject cohort (40 ± 12 years old), we compared the simultaneous measurements of Scyst, serum creatinine (Screat), creatinine clearance (Ccreat), Cockcroft and Gault's estimated clearance (Ccg) and GFR measured using inulin clearance (Cin). Receiver operating characteristic (ROC) analysis was performed using two Cin cut‐off (60 and 90 mL/min/1.73 m2). Results: A significant correlation was found among Cin on one hand and 1/Scyst, Ccreat, 1/Screat and Ccg on the other hand. Best fits (sensitivity/specificity) at 90 mL/min/1.73 m2 were 1.18 mg/L (0.72/0.80) for Scyst, 1.32 mg/dL (0.67/0.90) for Screat, 77 mL/min (0.80/0.70) for Ccg and 104 mL/min (0.88/0.80) for Ccreat. The areas under the ROC curves were not significantly different. Conclusions: This study provides cut‐off values for Screat and Ccg for detection of renal failure in renal transplant patients. However, the results also suggest that Scyst is not a more sensitive marker than Screat or Ccg for detecting renal failure in renal transplant patients.
Introduction
Rapid and accurate knowledge of renal function, i.e., evaluation of glomerular filtration rate (GFR), is of major importance in the follow‐up of renal transplant patients, especially to detect rejection and to adapt drug dosage.
Inulin clearance remains one gold standardCitation[1] for accurate determination of GFR, although expensive and cumbersome, it requires exact timing of urine collection. In clinical routine, GFR is estimated from plasma concentration measurement of creatinine (Screat).
Despite its convenience and low cost, estimation derived from Screat is not a sensitive marker of renal failure and is unable to detect mild renal function impairment of 50% or less.Citation[2]
Since the introduction of automated immunoassays, Cystatin C (Scyst) has been suggested as an alternative indicator of GFR.
The physical properties (low molecular weight and positive charge at physiological pH) suggest that this substance is easily filtered through the glomerulus.Citation[3]
The plasma concentration of Scyst in adults is relatively constant as produced by all investigated nucleated cells, but a slight increase is seen after 50 years.Citation[4]
We found only 6 studies comparing cystatin C and GFR in transplant recipients, usually very small cohortCitation[5], Citation[6], Citation[7], Citation[8], Citation[9], Citation[10] with only oneCitation[6] comparing Scyst to Cin in more than 30 pediatric patients. The aims of this study were to evaluate the specificity and sensitivity of Scyst as compared with GFR (inulin clearance) for the determination of renal function in renal transplant patients and to compare its predictive value with Screat and Cin.
Materials and Methods
Patients and Samples
Informed consent was obtained from all patients in accordance with the local ethical committee.
Data (n = 103) were collected from 60 stable renal transplant patients who were referred for GFR estimation from August 1997 to April 2000.
Time since transplantation was 6.7 ± 3 months. Mean age was 40.4 ± 11.9 years. Sex ratio was 21/39.
Immunosuppressive treatment at the time of the evaluation included: cyclosporin A twice daily, glucocorticoids, and either mycophenolate mofetil or azathioprine.
According to Levey et al.,Citation[11] renal failure was defined as a Cin value below 90 mL/min. Two cut‐off values of inulin clearance were studied: 60 and 90 mL/min/1.73 m2.
Inulin Clearance (Cin)
All GFR measurements were performed in fasting patients. GFR was measured using inulin clearance with constant infusion and urinary collections as described in details elsewhere.Citation[12] All results were indexed to 1.73 m2 body surface.
Creatinine Clearance (Ccreat)
GFR was estimated by Ccg:Citation[13]with K = 1.23 for men and 1.05 for women.
Analytical Methods
Measurement of inulin. Concentrations of inulin were measured by using a fully enzymatic method. Briefly, inulin was hydrolyzed by inulinase (Fructozyme Nova Nordisk® Bioindustrie, Nanterre, France) to its fructose monomers, followed by enzymatic conversion of fructose to glucose‐6‐phosphate by G6PD. Plasma glucose was removed by oxidation before determination.
Measurement of cystatin C. Scyst was measured by latex particle‐enhanced immunonephelometry (N Latex Cystatin C Behring®) on a Dade Behring BNII Automat.
Measurement of creatinine. Screat concentrations were assayed by a kinetic colorimetric technique using picric acid (Creatinine Sigma Diagnostics) on a multiparameter DAX48 Bayer Automat.Citation[14]
CV. Intraassay coefficients of variation were 2.7% for inulin, ranging from 2.3% to 4.1% when Scyst varies from 0.6 to 4 mg/L, and ranging from 1% to 2.7% when Screat varies from 7.13 to 1.10 mg/dL. Interassay coefficient of variation is 3.6% for inulin.
Statistical Analysis
Receiver operating characteristics (ROC) plots and analysis were performed as described by Zweig et al.Citation[15] using MedCalc® v.4.20.014 (MedCalc Software, Mariakerke, Belgium), Microsoft EXCEL® 97 SR‐1 for Windows (Microsoft Corp, USA) and GraphPad PRISM v.2.01 (GraphPad Software, Inc., San Diego, USA). P less than 0.05 was considered statistically significant.
Results
Patients
Main patient characteristics and results of Scyst, Screat and Ccg in different ranges of Cin are presented in .
Table 1. Main Characteristics of Renal Transplant Patients and Results of Scyst, Screat and Ccg in Different Ranges of Cin (mean ± standard deviation) (n = 60 patients, 103 samples)
The cut‐off of GFR defining renal failure was chosen at 90 mL/min/1.73 m2. According to these criteria, Cin ≤ 90 mL/min/1.73 m2 was observed in 93 of the 103 samples. For subjects with normal GFR (Cin > 90 mL/min/1.73 m2), Scyst was 1.06 ± 0.22 mg/L (0.77–1.42), Screat was 1.11 ± 0.30 mg/dL (0.72–1.72) and Ccg was 87 ± 26 (61–142).
Correlation of Scyst, Screat and Ccg with Cin
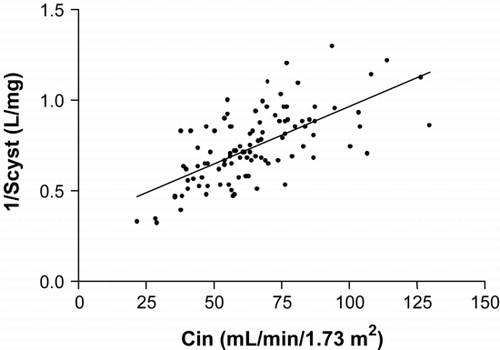
A significant correlation (p < 0.01) was found among Cin on one hand and 1/Scyst, Ccreat, 1/Screat and Ccg on the other hand [r2 = 0.43 ();0.60;0.40;0.44].
Validation of the Scyst Measurement for Detection of Renal Failure
The relationship between sensitivity and specificity at different cut‐off values for each parameter using ROC curves was performed. Best fits (sensitivity/specificity) at 90 mL/min/1.73 m2 were 1.18 mg/L (0.72/0.80) for Scyst, 1.32 mg/dL (0.67/0.90) for Screat, 77 mL/min (0.80/0.70) for Ccg and 104 mL/min (0.88/0.80) for Ccreat. Best fits (sensitivity/specificity) at 60 mL/min/1.73 m2 were 1.52 mg/L (0.60/0.87) for Scyst, 1.48 mg/dL (0.61/0.83) for Screat, 52 mL/min (0.54/0.91) for Ccg and 76 mL/min (0.83/0.92) for Ccreat.
No significant difference was found among area under the ROC curve (AURC) for Screat, Ccg or Ccreat and Scyst, for any cut‐off. Nevertheless, AURC was greater with Ccreat than with Scyst at 60 mL/min/1.73 m2. It should be noted that Ccg did as well as suitable as the three other markers in renal transplant patients.
Choosing another cut‐off threshold at 80 mL/min/1.73 m2 did not alter the results since ROC curves for Scyst or Screat vs. GFR were not statistically different.
Proportional Changes in Scyst and Screat (Figure not Published)
Experimental Scyst and Screat values were gathered according to their GFR values in several discrete ranges of 10 mL/min/1.73 m2. In each range, the mean concentration of the analyte at a GFR of 90 mL/min/1.73 m2 as previously described by Newmann.Citation[16]
Plotting the “proportional increase in analyte” calculated vs. GFR, the 2 curves for Screat and Scyst were similar in any range of GFR. We could not demonstrate that Scyst was a more sensible marker of renal function impairment than Screat.
Is Cystatin C an Ideal Filtration Marker?
The serum concentrations of cystatin C and creatinine at different GFR were compared with the theoretical curves obtained assuming glomerular filtration as their exclusive route of elimination (Figure not shown.
Discussion
As previously reported in this specific population, we found a good correlation between Scyst and Cin, though inferior to values previously found,Citation[7], Citation[8], Citation[9] suggesting that cystatin is indeed an index of renal function in renal transplant recipients. However, the correlation was higher between Cin and Ccreat or Ccg. Moreover, the correlation between Scyst and Cin was lower than that observed in nongrafted patients.Citation[12]
As a note of caution, we would emphasize that correlation does not mean identity and that it is inappropriate to state that Scyst is a better marker than Screat on the sole basis of the value of the correlation coefficient.
A ROC analysis was done to compare the diagnostic accuracy of Scyst and Screat vs. Cin.
In a relatively large cohort, including a wide range of GFR, the best fit for a 90 mL/min/1.73 m2 Cin cut‐off (sensitivity/specificity) was 0.72/0.80 for Scyst.
Our results indicate that Scyst has a reasonably good predictive value for detection of renal failure. However, in our cohort, Scyst was not significantly better than Screat or Ccg.
Our results are at variance with those reported by PlebaniCitation[9] in a small group of 12 renal transplant recipients but in agreement with the data of RischCitation[7] in a cohort of 30 renal transplant recipients or PaskalevCitation[17] in a 40‐patient cohort. However, the predictive values of all four parameters were not as good as those found in non‐transplanted patient population.Citation[12] However, due to the very small number of patients with Cin > 90 mL/min/1.73 m2 used for the ROC curves, these results should be confirmed in larger series.
Scyst did perform better than Screat and this may be of importance when it is cumbersome to obtain a timed urine sample to calculate Ccreat despite several studies outline mixed conclusions whether CysC offers a significant advantage in renal transplant patients.Citation[18]
Due to our study design, i.e., stable transplant recipients, we were not able to test whether Scyst can exhibit more rapid change than Screat in parallel to variation in GFR. However, Thervet et al. showed that cystatin C allows earlier diagnosis of renal function recovery than does plasma creatinine in patients with delayed graft function.Citation[19]
TenstadCitation[20] looked at renal clearance of cystatin C in rats using Cr‐EDTA as gold standard and suggests that there is an extra renal clearance of cystatin C. We have represented the tubular secretion of creatinine by plotting Screat and Scyst against GFR as compared to an ideal theoretical curve (Cf Chantrel Citation[12]). Most of the values of Screat lay under the theoretical curve, indicating the magnitude of tubular secretion (Figure not shown). This phenomenon was less marked than with Screat when applying this representation to Scyst. More studies are needed to confirm this observation and its meaning.
Conclusion
In this study, we provided some cut‐off values of Screat, Ccg and Scyst for detecting renal failure in renal transplant patients.
The specificity and sensitivity, as assessed by ROC curves, were similar for Screat, Ccg and Scyst and the measurement of Scyst did not confer any advantages over the “classical” marker to detect mild renal failure in stable renal kidney transplant patients. We showed further that Ccg is a suitable marker for the follow‐up of renal transplant patient.
As our study involved stable patients without complications, we cannot exclude that Cystatin C would provide a more precise assessment of GFR during inflammatory processes or other additional conditions, especially during acute rejection or infections, both common problems in patients with renal transplants.
Acknowledgments
For research support: C. Barth and C. Klein for their technical expertise.
| Abbreviations | ||
| GFR: | = | Glomerular filtration rate |
| Screat: | = | Serum creatinine level |
| Scyst: | = | Serum cystatin C level |
| Cin: | = | Inulin clearance |
| ROC: | = | Receiver operating characteristic |
| Ccg: | = | Glomerular filtration rate estimated by Cockcroft and Gault's formula |
| Ccreat: | = | Creatinine clearance |
| AURC: | = | Area under the ROC curve |
References
- Gaspari F., Perico N., Remuzzi G. Application of newer clearance techniques for the determination of glomerular filtration rate. Curr. Opin. Nephrol. Hypertens. 1998; 7: 675–680, [PUBMED], [INFOTRIEVE], [CSA]
- Perrone R. D., Madias N. E., Levey A. S. Serum creatinine as an index of renal function: new insights into old concepts. Clin. Chem. 1992; 38: 1933–1953, [PUBMED], [INFOTRIEVE]
- Grubb A. Diagnostic value of analysis of cystatin C and protein HC in biological fluids. Clin. Nephrol. 1992; 38: S20–S27, [PUBMED], [INFOTRIEVE]
- Norlund L., Fex G., Lanke J. Reference intervals for the glomerular filtration rate and cell‐proliferation markers: serum cystatin C and serum beta2‐microglobulin/cystatin C ratio. Scand. J. Clin. Lab. Invest. 1997; 57: 463–470, [PUBMED], [INFOTRIEVE]
- Herget‐Rosenthal S., Trabold S., Huesing J., Heemann U., Philipp T., Kribben A. Cystatin C—an accurate marker of glomerular filtration rate after renal transplantation?. Transpl. Int. 2000; 13: 285–289, [CSA], [CROSSREF]
- Bokenkamp A., Domanetzki M., Zinck R., Schumann G., Byrd D., Brodehl J. Cystatin C serum concentrations underestimate glomerular filtration rate in renal transplant recipients. Clin. Chem. 1999; 45: 1866–1868, [PUBMED], [INFOTRIEVE], [CSA]
- Risch L., Blumberg A., Huber A. Rapid and accurate assessment of glomerular filtration rate in patients with renal transplants using serum cystatin C. Nephrol. Dial. Transplant. 1999; 14: 1991–1996, [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Le Bricon T., Thervet E., Benlakehal M., Bousquet B., Legendre C., Erlich D. Changes in plasma cystatin C after renal transplantation and acute rejection in adults. Clin. Chem. 1999; 45: 2243–2249, [PUBMED], [INFOTRIEVE], [CSA]
- Plebani M., Dall'Amico R., Mussap M., Montini G., Ruzzante N., Marsilio R., Giordano G., Zacchello G. Is serum cystatin C a sensitive marker of glomerular filtration rate (GFR)? A preliminary study on renal transplant patients. Ren. Fail. 1998; 20: 303–309, [PUBMED], [INFOTRIEVE], [CSA]
- Le Bricon T., Thervet E., Froissart M., Benlakehal M., Bousquet B., Legendre C., Erlich D. Plasma cystatin C is superior to 24‐h creatinine clearance and plasma creatinine for estimation of glomerular filtration rate 3 months after kidney transplantation. Clin. Chem. 2000; 46: 1206–1207, [letter; comment][PUBMED], [INFOTRIEVE], [CSA]
- Levey A. S., Madaio M. P., Perrone R. D. Laboratory assessment of renal disease: clearance, urinalysis, and renal biopsy. The Kidney, B. M. Brenner, F. C. Rector. Saunders, Philadelphia 1991; 919–968
- Chantrel F., Agin A., Offner M., Koehl C., Moulin B., Hannedouche T. Comparison of cystatin C versus creatinine for detection of mild renal failure. Clin. Nephrol. 2000; 5: 374–381
- Cockcroft D. W., Gault M. H. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31–34, [PUBMED], [INFOTRIEVE]
- Larsen K. Creatinine assay by a reaction kinetic principle. Clin. Chem. Acta. 1972; 41: 209, [CROSSREF]
- Zweig M. H., Campbell G. Receiver‐operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin. Chem. 1993; 39: 561–577, [PUBMED], [INFOTRIEVE]
- Newman D. J., Thakkar H., Edwards R. G., Wilkie M., White T., Grubb A. O., Price C. P. Serum cystatin C measured by automated immunoassay: a more sensitive marker of changes in GFR than serum creatinine. Kidney Int. 1995; 47: 312–318, [PUBMED], [INFOTRIEVE]
- Paskalev E., Lambreva L., Simeonov P., Koicheva N., Beleva B., Genova M., Marcovska R., Nashkov A. Serum cystatin C in renal transplant patients. Clin. Chim. Acta 2001; 310(1)53–56, [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Laterza O. F., Price C. P., Scott M. G. Cyctatin C: an improved estimator of glomerular filtration rate?. Clin. Chem. 2002; 48(12)2297–2298, [CSA]
- Thervet E., Le Bricon T., Hugot V., Benlakehal M., Bedrossian J., Beuzard Y., Legendre C., Erlich D. Early diagnosis of renal function recovery by cystatin C in renal allograft recipients. Transplant. Proc. 2000; 32: 2779, [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Tenstad O., Roald A. B., Grubb A., Aukland K. Renal handling of radiolabeled human cystatin C in the rat. Scand. J. Clin. Lab. Invest. 1996; 56: 409–414, [PUBMED], [INFOTRIEVE]