Abstract
The purpose of this review is to examine vitamin D in the context of sport nutrition and its potential role in optimizing athletic performance. Vitamin D receptors (VDR) and vitamin D response elements (VDREs) are located in almost every tissue within the human body including skeletal muscle. The hormonally-active form of vitamin D, 1,25-dihydroxyvitamin D, has been shown to play critical roles in the human body and regulates over 900 gene variants. Based on the literature presented, it is plausible that vitamin D levels above the normal reference range (up to 100 nmol/L) might increase skeletal muscle function, decrease recovery time from training, increase both force and power production, and increase testosterone production, each of which could potentiate athletic performance. Therefore, maintaining higher levels of vitamin D could prove beneficial for athletic performance. Despite this situation, large portions of athletic populations are vitamin D deficient. Currently, the research is inconclusive with regards to the optimal intake of vitamin D, the specific forms of vitamin D one should ingest, and the distinct nutrient-nutrient interactions of vitamin D with vitamin K that affect arterial calcification and hypervitaminosis. Furthermore, it is possible that dosages exceeding the recommendations for vitamin D (i.e. dosages up to 4000-5000 IU/day), in combination with 50 to 1000 mcg/day of vitamin K1 and K2 could aid athletic performance. This review will investigate these topics, and specifically their relevance to athletic performance.
Introduction
Vitamin D, a fat-soluble vitamin, was first discovered in cod liver oil [Citation1] and has since been identified as an essential vitamin, acting as a precursor steroid to a host of metabolic and biological processes. Once converted into its biologically-active form, 1,25-dihydroxyvitamin D [Citation2], it regulates the expression of over 900 gene variants [Citation3]. These gene expressions have been shown to have significant impact on a wide variety of health and performance-related variables, such as exercise-induced inflammation, tumour suppressor genes, neurological function, cardiovascular health, glucose metabolism, bone health and skeletal muscle performance [Citation4–Citation10]. Surprisingly enough, 88.1 % of the world’s population has inadequate vitamin D levels [Citation11]. Deficiency has been shown to be linked to a variety of adverse psychological and health outcomes, such as suicidal thoughts [Citation12], depression [Citation13], cognitive decline and neurological impairment [Citation14], and an increased risk of cancer [Citation15]. Furthermore, individuals with inefficient vitamin D stores have an increased risk of bone disorders like spondyloarthritis [Citation16], rickets [Citation1, Citation17], and fractures due to higher bone resorption from an overproduction of parathyroid hormone (PTH) [Citation18, Citation19]. Lastly, deficiency has catabolic effects on muscle tissue [Citation20], causes muscle weakness [Citation21], and impairs cross-bridge formation [Citation22], all of which could impair athletic performance. Due to the increase in enzymatic activity of exercise [Citation23], athletes may be as susceptible, if not more susceptible to becoming vitamin D deficient when compared to the general population. A recent meta-analysis pooling 23 studies with 2313 athletes found that 56 % of athletes had inadequate vitamin D levels [Citation24]. Because of the high prevalence of vitamin D deficiency [Citation25] and its effects on human physiology, this review is aimed to identify the role of vitamin D in athletic performance (for health related aspects of vitamin D, see [Citation11, Citation18, Citation26, Citation27]). This review will cover how vitamin D is metabolized in the body, its potential roles in athletic performance, sources of vitamin D, differences between vitamin D2 and vitamin D3, optimal levels of vitamin D for athletes, and strategies to achieve these levels and prevent toxicity by nutrient-nutrient interactions.
Metabolism of vitamin D
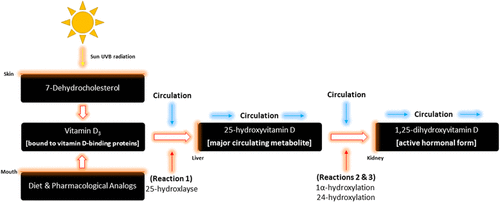
Vitamin D travels in the bloodstream bound to vitamin D-binding proteins [Citation28] and undergoes a three-stage process of key enzymatic reactions [Fig. ]: 25-hydroxylation, 1α-hydroxylation and 24-hydroxylation [Citation18, Citation29]. The steroid precursor vitamin D3 first travels to the liver where it is hydroxylated to 25-hydroxyvitamin D [25(OH)D] by 25-hydroxlayse, which is mediated by the cytochrome P450 enzymes, CYP27A1 (in the mitochondria) and CYP2R1 [Citation29]. This 25(OH)D is then hydroxylated by CYP27B1 (1α-hydroxylation) [Citation29]. This final step occurs primarily in the kidney [Citation18], but various other tissues, namely skeletal muscle, have also been shown to express CYP24A1 enzymes, where 25(OH)D becomes the active hormonal form, 1,25-dihydroxyvitamin D [Citation29]. 1,25-dihydroxyvitamin D then interacts with vitamin D receptors (VDR), which are located in almost every tissue in the body [Citation30, Citation31], and is then transcribed into the cell and binds to vitamin D response elements (VDREs) located in DNA [Citation18]. If 1,25-dihydroxyvitmain D does not interact with VDREs, it is further degraded by CYP24A1 (24-hydroxylase) to the inactive form, calcitroic acid [Citation29].
Vitamin D and performance
Vitamin D3 receptors exist in human skeletal muscle tissue [Citation30, Citation31], indicating that 1,25-dihydroxyvitamin D has a direct effect on skeletal muscle activity. Research on the muscle effects of vitamin D3 [Citation32] is limited to diseased populations [Citation20, Citation33], or healthy untrained adults [Citation34]. Until recently, a few reviews and meta-analyses have shown that increasing serum 25(OH)D levels in a given population have a positive effect on muscle strength, power and mass [Citation33–Citation35] but the only study that examined the effects in athletes [Citation36], had mixed results. In addition, von Hurst and Beck [Citation36] concluded that the optimal intake and serum concentration of 25(OH)D have yet to be identified in the athletic population.
Maximal oxygen uptake
Vitamin D receptors (VDR) are present in cardiac muscle and vascular tissue [Citation7], indicating that 1,25-dihydroxyvitamin D might influence maximal oxygen uptake (VO2max) via the ability to transport and utilize oxygen within the blood to various tissues. Multiple correlative studies showed a positive correlation between VO2max and serum 25(OH)D concentration in non-athletes [Table ] [Citation37–Citation39]. However many confounding variables were not addressed, such as concomitant multivitamin [Citation38] and supplement intake [Citation37, Citation39]. Studies performed in athletes are conflicting. Koundourakis and his colleagues [Citation40] found that there was a significant correlation between 25(OH)D levels and performance parameters in 67 Caucasian male professional soccer players (age 25.6 ± 6.2). A linear relationship was seen between pre- and post- offseason measurements of 25(OH)D and muscle strength indicated by squat jump (SJ), countermovement jump (CMJ), sprinting ability (10- and 20- m sprint) and VO2max [Citation40]. A more recent publication by Fitzgerald et al. [Citation41], concluded that there was no association between 25(OH)D levels and an individual’s VO2max in 52 Caucasian competitive ice hockey players [Citation41]. Additionally, the correlation commonly seen between the serum 25(OH)D concentrations and VO2max is inversely related to increases in physical activity and training status [Citation39]. Forney and her colleagues [Citation42] recently investigated the association between serum 25(OH)D, VO2max and training status in 39 physically-active college students (20 men, 19 women). They showed that the participants with higher (>35 ng-mL-1) serum 25(OH)D levels had a significantly higher VO2max (+20 %) than the low (<35 ng-mL-1) serum 25(OH)D group [Citation42]. However, this correlation was limited to males only.
Table 1 Vitamin D correlation and intervention studies on Maximal Oxygen Uptake (VO2max)
Intervention trials in the athletic population are scarce. To our knowledge, only one exists looking at the effects of vitamin D supplementation on VO2max. Jastrzebski [Citation43] performed a single-blinded trial of supplementation with 6000 IU/day of vitamin D3 versus a placebo during an 8-week training cycle in 14 elite lightweight rowers with sufficient 25(OH)D concentrations (>30 ng/mL). They demonstrated a significantly increase in VO2max (12.1 % and 10.3 %, respectively) and 25(OH)D concentrations by 400 % (~120 ng/mL). The authors concluded that supplementation of vitamin D3 during the 8-week training period significantly improved aerobic metabolism in the elite rowers [Citation43]. Further research is needed to test whether an ergogenic effect exists in athletes who are severely deficient in serum 25(OH)D, and if supraphysiological dosages of vitamin D3, such as that used by Jastrzebski, have an ergogenic effect in vitamin D replete athletes in other sport disciplines.
The specific mechanism by which increased levels of 25(OH)D affect VO2max remains unclear [Citation39], however this phenomenon could be due to the fact that the CYP enzymes that activate vitamin D3 into 1,25-dihydroxyvitamin D3 have heme-containing proteins [Citation44] and could potentially affect the binding affinity of oxygen to hemoglobin.
Recovery
The ability to recover rapidly is important for athletes to train at high intensities more frequently. Human skeletal muscle tissue responds to training stimuli and/or tissue damage through remodeling [Citation45–Citation47]. During recovery, 1,25-dihydroxyvitamin D increases the myogenic differentiation and proliferation [Citation48] and down-regulates myostatin, an inhibitory regulator of muscle synthesis of C2C12 myoblasts in culture [Citation49]. Stratos and colleagues [Citation50] showed this marked increase in skeletal muscle regeneration in a crushed soleus muscle (in vivo) of 56 male Wistar rats (300 to 325 g body weight), after a supraphysiological dose of ~100,000 IU of vitamin D. They [Citation50] separated rats into a high (332,000 IU/kg) and low (33,200 IU/kg) dose groups and examined recovery response times to the crushed soleus muscle. When compared to the low dose group, the high dose group had a significant attenuation of apoptosis four days post-injury, indicative of an increase in cellular matrix proteins [Citation50]; which is crucial for skeletal tissue repair [Citation51]. This increase in cellular turnover rate led to the enhanced recovery time, an increase in tetanic force production (only 10 % lower than the non-injured limb), and an increase in twitch force when compared to the control group [Citation50]. As murine models display regenerative capacities that exceed those of humans, it is important to note the limitations of extending the aforementioned findings to humans; however, the finding that vitamin D supplementation enhances the recovery in peak isometric force shortly after intense exercise was recently supported in much lower doses in modestly-active humans [Citation52].
In a randomized, double-blind, placebo-controlled study, Barker et al. [Citation52] demonstrated that 4000 IU/day for 35 days of vitamin D in healthy and moderately active adults attenuated the inflammatory biomarkers alanine (ALT) and aspartate (AST) immediately following 10 sets of 10 repetitions of peak isometric force eccentric-concentric jumps. Furthermore, although peak power output decreased in both the groups, the supplementation group only decreased by 6 %, while the placebo group’s power decreased by 32 % immediately post-exercise [Citation52]. This discrepancy persisted at 48 h [Citation52]. Further research examining higher dosages would be warranted to determine if recovery and power output are improved to a greater degree [Table ].
Table 2 Vitamin D in vitro, in vivo and intervention studies on recovery
Force and power production
Vitamin D3 has also been shown to increase force and power output of skeletal muscle tissue [Citation19], perhaps through the sensitization of calcium binding sites on the sarcoplasmic reticulum, leading to an enhanced cross-bridge cycling and muscular contraction [Citation53]. There is further evidence that vitamin D3 might also potentially increase both size and number of type II muscle fibers [Citation20, Citation54, Citation55]. These findings have only been supported in mobility-limited elderly (≥65 years old) women [Citation55], and have yet to be tested in the athletic population. On the other hand, increases in force and power production have been studied in athletes with positive results during a randomized placebo-controlled study in 10 male professional soccer players [Citation56]. After an 8-week long intervention of either receiving 5000 IU/day of vitamin D3 or a placebo, the vitamin D3 group had a significant increase in serum 25(OH)D levels and a significant improvement in both their 10-m sprint times and vertical jump when compared to the placebo group [Citation56]. Confounding variables were well-controlled, in that the authors instructed the athletes to maintain current nutritional intake, and excluded any athlete who was taking a multivitamin, vitamin D, fish oil and/or were regular sunbed users or who just returned from a vacation in a sunlight enriched climate. However, other studies have shown no significant benefit of vitamin D supplementation in athletes with moderately deficient or adequate levels [Citation10, Citation41, Citation42], indicating that these performance benefits might be limited to individuals with significant vitamin D deficiency [Table ].
Table 3 Vitamin D correlation and intervention studies on force & power production
Vitamin D and testosterone
Testosterone is an endogenous hormone important for muscular adaptations to training. Naturally low testosterone levels in young men have been linked to decreases in protein anabolism, strength, beta-oxidation, and an increase in adipose deposition [Citation57]. Thus, athletes endeavour to optimize natural androgenic production. A recent cross-sectional study done on 2299 older men (62 ± 11 years of age) showed that 25(OH)D levels correlated with testosterone and androgen levels in men [Citation58]. Low testosterone, or hypogonadism, was identified in 18 % of the participants, and these men had significantly lower mean 25(OH)D levels than the rest of the population. Furthermore, only 11.4 % of the sample had sufficient levels of vitamin D.
Additionally a 12-month, double-blind, randomized control trial in 54 non-diabetic males demonstrated that the group receiving 3332 IU/day of vitamin D had a significant increase in circulating 25-hydroxyvitamin D, total testosterone, bioactive testosterone, and free testosterone levels [Citation59]. These findings support the notion that elevating 25(OH)D levels may augment testosterone production in non-diabetic male subjects, which indicates that vitamin D supplementation might have ergogenic potential through the enhancement of endogenous testosterone production. More research is required in order to investigate this potential role of vitamin D and testosterone levels in various study populations [Table ].
Table 4 Vitamin D correlation, in vivo and intervention studies testosterone
The specific mechanism of action of 25(OH)D on testosterone in men could potentially be related to two processes: inhibited testosterone aromatization and enhanced androgen binding. Evidence for both of these mechanisms comes from animal models. Specifically, higher 25(OH)D levels inhibit gonadal aromatization of testosterone in VDR knockout mice [Citation60]. Secondly, VDR and vitamin D metabolizing enzymes have been located in human and rat testis and have been shown to enhance the affinity of androgen binding receptors [Citation57, Citation61, Citation62]. This effect increases the rate at which androgens can bind to testosterone-producing glands resulting in higher concentrations of steroid hormones, leading to an increase in skeletal muscle hypertrophy, strength and power output [Citation63, Citation64].
Sources
Sunlight
Humans acquire vitamin D from two different sources, endogenous production after sun exposure, or via the diet (from food or supplementation). Unlike the metabolism of dietary vitamin D, the synthesis of vitamin D3 by the skin is a non-enzymatic biological process [Citation65]. Once the skin is exposed to the Sun’s ultraviolet B (UVB) radiation, it then converts stored 7-dehydrocholesterol into circulating vitamin D3, 25(OH)D [Citation29] and other isomers [Citation66]. The amount of UVB exposure determines the amount and the specific isomers of vitamin D3 that will form [Citation66, Citation67]. The recommended dosage of sunlight exposure during the summer is five to 20 min per day to 5.0 % of exposed skin at a UVB radiation of 290–315 nm [Citation68, Citation69] two to three times a week [Citation70]. Additionally, it has been shown that 15 min of adequate (290–315 nm) UVB exposure during the summer months in a bathing suit can produce 10,000 to 20,000 IU of vitamin D3 [Citation71]. However, multiple factors can affect the rate and synthesis of vitamin D3 [Table ] [Citation25, Citation66, Citation72–Citation74].
Table 5 Factors affecting the rate and synthesis of endogenously produced vitamin D
Diet
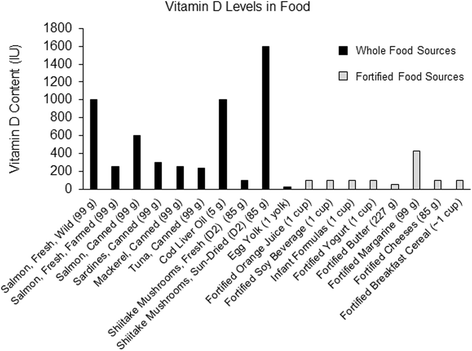
Vitamin D derived from diet and supplementation comes in two forms, the plant-based vitamin D2 (ergocalciferol), and the more bioavailable mammal and fish source, vitamin D3 (cholecalciferol) [Citation75]. Vitamin D can be found in various food products [Fig. ] [Citation76], such as fortified cereals and milk, natural foods like salmon, or through various vitamin D analogues produced synthetically in a laboratory [Table ] [Citation18, Citation76]. Both sources are considered prohormone compounds, capable of increasing circulating 25(OH)D, after they have been converted by the enzymatic reactions described earlier.
Table 6 Vitamin D sources from pharmacological analogs
Dosage for optimal performance
Both D2 and D3 are capable of increasing plasma 25(OH)D concentration, but vitamin D3 might be more effective than vitamin D2 [Citation75, Citation77, Citation78]. When compared to vitamin D3, vitamin D2 is less stable, less bioavailable with increasing age, and it has been shown in multiple clinical studies that the amount of vitamin D2 absorbed is significantly lower than with vitamin D3. Furthermore, vitamin D2 has a lower affinity to VDRs [Citation54, Citation75, Citation77–Citation79] and a higher rate of deactivation once hydroxylated in the kidney due to side-chain variations [Citation77]. Lastly, an epidemiological study during the winter months in Dunedin, New Zealand investigated the effects of 1000 IU/day of either vitamin D2 or vitamin D3 supplementation over a 25-week period in 95 healthy, adult participants (18–50 years old) [Citation78]. The participants who received the vitamin D2 supplement had a larger decrease in serum 25(OH)D (74 nmol/L to 50 nmol/L) levels than those who took vitamin D3 (80 nmol/L to 72 nmol/L) [Citation78]. However, both results show that 1000 IU/day of vitamin D was inadequate to increase serum 25(OH)D concentrations and actually caused a decline with both isoforms.
With vitamin D3 proving more efficacious, the optimal dosage varies depending on the individual and the institution providing the guidelines. The Institute of Medicine (IOM) recommends 400–800 IU/day for children, adults and individuals >70 years of age to maintain serum vitamin D at >50 nmol/L [Citation11, Citation80, Citation81]. Alternatively, the Endocrine Society (ES) recommends a slightly higher intake, with dosages of 400–1000 IU/day for infants, 600–1000 IU/day for children, and 1500-2000 IU/day for adults in order to maintain adequate serum vitamin D concentrations of 75 nmol/L [Citation82]. These recommendations correspond with a review in 2004 [Citation83], stating that 70 nmol/L is the lowest desirable serum concentration to prevent adverse health effects. Other recommendations have suggested optimal levels may be 90 to greater than 120 nmol/L [Citation86, Citation87], based on estimations made from that of levels seen in individuals inhabiting very sunlight-rich environments [Citation84] and/or have shown optimal lower-extremity function [Citation85].
The definitions of hypovitaminosis or hypervitaminosis are more controversial. The IOM defines inadequate stores of 25(OH)D as 30–50 nmol/L, and deficiency as 25(OH)D <30 nmol/L [Citation88], and sets the upper limit of dietary intake of vitamin D to 4000 IU/day [Citation69]. The ES on the other hand, defines vitamin D deficiency at levels of 25(OH)D <50 nmol/L, insufficiency as 25(OH)D between 51 to 74 nmol/L [Citation89], and sets the upper limit of dietary intake of vitamin D to 10,000 IU/day [Citation19]. However, recent reviews have suggested that this is more of a theoretical concern [Citation72, Citation83, Citation84, Citation90]. The optimal vitamin D dosage and level are clearly controversial [Citation88, Citation91]. Furthermore, the optimal levels needed for athletic performance have not yet been determined. Growing evidence has supported that 600–800 IU/day may not be sufficient for optimal levels of vitamin D, especially for the athletic population [Citation92], since serum 25(OH)D concentrations greater than 100 nmol/L have been proposed to be optimal for lower body skeletal muscle function [Citation85] and low vitamin D levels are linked to increased bone turnover, increasing the risk of stress fractures [Citation93]. It has been shown that it takes roughly 2000 to 5000 IU/day of vitamin D from all available sources in order to optimize bone health by maintaining serum 25(OH)D levels of 75 to 80 nmol/L [Citation84, Citation85, Citation94, Citation95]. Furthermore, this dosage would be unattainable from natural UVB exposure during the months of October to April when residing in latitudes of 42.2 to 52° N [Citation96] which is indicated by the high prevalence of vitamin D deficient indoor and outdoor athletes in a multitude of disciplines [Citation24, Citation97–Citation99]. Lastly, studies that have shown to improve athletic prowess utilize dosages higher than 3000 IU/day, but none have yet to reach levels greater than 100 nmol/L. Thus, it remains unclear, but athletes may benefit from 25(OH)D levels ≥100 nmol/L in order to increase skeletal muscle function and reduce the risk of stress fractures.
However, no study to date has looked at the effects of vitamin D supplementation and skeletal muscle function in the athletic population with 25(OH)D levels of ≥100 nmol/L [Citation36]. Additionally, the previous performance intervention studies presented in this review supplemented with dosages far greater than the recommended dosages of 600–2000 IU/day (e.g., 5000 IU/day of D3) and 1000 IU/day of vitamin D3 during the winter months is not enough to prevent a decline in serum 25(OH)D stores [Citation78].
Toxicity & hypercalcemia
Although it has been reported that vitamin D toxicity might occur with dosages of ≥10,000 IU/day for an extended period [Citation71, Citation84], producing adverse effects like hypercalcemia, the level of vitamin D causing toxicity is unclear [Citation71, Citation100], and due to ethical reasons, no prospective studies have analyzed the effect of vitamin D intoxication in humans. Recently, an accidental overdose of 2,000,000 IU of vitamin D3 in two elderly patients did not cause adverse effects and only elevated blood calcium levels slightly [Citation101]. More importantly, adverse effects have only been reported at serum concentrations of 25(OH)D above 200 nmol/L, which would take daily dosages of 40,000 IU or more of vitamin D to achieve [Citation84], and serum concentrations of 25(OH)D of <140 nmol/L have not been correlated with hypercalcemia. 1,25-dihydroxyvitamin D works synergistically with calcium and allows it to be absorbed from the gastrointestinal tract and stimulates mature osteoblasts to produce receptor activator nuclear factor-kB ligand (RANKL) [Citation102]. RANKL in turn stimulates mineralization and bone resorption via osteoclastogenesis. Increased levels of 25(OH)D can accelerate this process, causing a rise in calcium concentration in the blood, a higher absorption rate of calcium by the kidneys, and could potentially lead to kidney stones and/or potential vascular calcification [Citation103].
Vitamin K
Any discussion of vitamin D toxicity merits mention of vitamin K. As with calcium, vitamin K works synergistically with vitamin D to regulate bone resorption, activation and distribution [Citation104]. Vitamin K carboxylates the newly-formed ostecalcin proteins that are produced in mature bone cells and are tightly regulated by vitamin D [Citation105]. Once the protein is carboxylated, it interacts with calcium ions in bone tissue [Citation106] and has a significant effect on bone mineralization, formation, the prevention of bone loss, and potentially the stoppage of fractures in women [Citation105, Citation107–Citation111]. However, when levels of vitamin K are inadequate, the ostecalcin production is not suppressed [Citation109]. This situation facilitates a build-up of un-carboxylated (inactive) ostecalcin proteins in bone, leading to a potential increase in calcium release from bone and the deposition of calcium into soft tissues (causing arterial calcification) [Citation112, Citation113]. Thus, vitamin D3 toxicity might occur only in the absence of sufficient vitamin K stores.
Recommended dosages of vitamin K range from 50 mcg to 1000 mcg [Citation108]. However, these recommendations are controversial since vitamin K stores are rapidly depleted without constant supply [Citation114] and like vitamin D, vitamin K also has two variants: K1 and K2. Sources of vitamin K can be found in pharmacological analogues and naturally in the diet. Vitamin K1, the most abundant form found in an individual’s diet [Citation115], is high in green leafy cruciferous vegetables, fruits, various vegetable oils and beans [Citation114]. Vitamin K2, the more bioavailable form of vitamin K [Citation114], comes in a variety of fish, offal, meat, dairy products, fermented cheese (e.g., blue cheese), and fermented products like natto (fermented soybeans, a Japanese delicacy) [Citation116].
Both forms play different roles in the body [Citation117], but the IOM has set the recommended dietary intake only for the K1 isoform (90 mcg/day for women and 120 mcg/day for men), with no upper limit, and has yet to set any dietary recommendation for vitamin K2 [Citation114]. Specifically, vitamin K1 has a key role in the carboxylation of various blood clotting proteins, where vitamin K2 is essential for the carboxylation and activation of osteocalcin and matrix Gla protein (MGP) (an essential protein needed to prevent soft tissue calcification) [Citation118]. More importantly, one of the vitamin K2 variants, MK-4, is more effective at mitigating osteoclast formation and the negative health effects of vitamin D overdose [Citation114, Citation115]. Furthermore, 10 mg/day (10,000 mcg) of synthetic vitamin K1 (phytonadione, sold as Konakion® [Citation119]) has been shown to be beneficial for elite female marathon runners by increasing bone formation and preventing bone loss [Citation120] and mega-doses of 45 mg/day (45,000 mcg) of MK-4 in combination with vitamin D3 could prevent osteoporosis in postmenopausal women [Citation102, Citation109]. Thus, although MK-4 might have the greatest effect on carboxylation of osteocalcin, both vitamin K1 and K2 interact with each other in order to optimize bone health and are essential to the human body. Further research in athletic populations should focus on the optimal dosage for vitamin D3 in combination with vitamin K.
Conclusion
In summary, an interesting theme has emerged from animal studies that supraphysiological dosages of vitamin D3 have potential ergogenic effects on the human metabolic system and lead to multiple physiological enhancements. These dosages could increase aerobic capacity, muscle growth, force and power production, and a decreased recovery time from exercise. These dosages could also improve bone density. However, both deficiency (12.5 to 50 nmol/L) and high levels of vitamin D (>125 nmol/L) can have negative side effects, with the potential for an increased mortality [Citation121]. Thus, maintenance of optimal serum levels between 75 to 100 nmol/L [Citation11, Citation86] and ensuring adequate amounts of other essential nutrients including vitamin K are consumed, is key to health and performance. Coaches, medical practitioners, and athletic personnel should recommend their patients and athletes to have their plasma 25(OH)D measured, in order to determine if supplementation is needed. Based on the research presented on recovery, force and power production, 4000-5000 IU/day of vitamin D3 in conjunction with a mixture of 50 mcg/day to 1000 mcg/day of vitamin K1 and K2 seems to be a safe dose and has the potential to aid athletic performance. Lastly, no study in the athletic population has increased serum 25(OH)D levels past 100 nmol/L, (the optimal range for skeletal muscle function) using doses of 1000 to 5000 IU/day. Thus, future studies should test the physiological effects of higher dosages (5000 IU to 10,000 IU/day or more) of vitamin D3 in combination with varying dosages of vitamin K1 and vitamin K2 in the athletic population to determine optimal dosages needed to maximize performance.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
DTD formulated the idea for the literature review, acquired the research articles, interpreted the literature presented, and drafted the initial manuscript; BD and MSK helped advise the direction of the manuscript, the interpretation of literature presented, and thoroughly edited/rewrote the manuscript to prepare for final submission. Each author read and approved the final manuscript prior to submission.
References
- McCollumESimmondsNBeckerJShipleyPAn experimental demonstration of the existence of a vitamin which promotes calcium depositionJ Biol Chem19221922 293 298
- Jones G. Metabolism and biomarkers of vitamin D. Scand J Clin Lab Invest. 2012;72(Suppl 243):7–13.
- Wang T-T, Tavera-Mendoza LE, Laperriere D, Libby E, MacLeod NB, Nagai Y, et al. Large-scale in silico and microarray-based identification of direct 1,25-dihydroxyvitamin D3 target genes. Mol Endocrinol. 2005;19:2685–95.
- Smith DT, Broughton KS, Larson-meyer DE. Vitamin D status and biomarkers of inflammation in runners. 2012;3:35–42.
- Alvarez-DíazSValleNGarcíaJMPeñaCFreijeJMPQuesadaV et al Cystatin D is a candidate tumor suppressor gene induced by vitamin D in human colon cancer cellsJ Clin Invest2009119 2343 58 https://doi.org/10.1172/JCI37205 19662683
- DhesiJKJacksonSHDBearneLMMonizCHurleyMVSwiftCG et al Vitamin D supplementation improves neuromuscular function in older people who fallAge Ageing200433 589 95 https://doi.org/10.1093/ageing/afh209 15501836
- Reddy VangaSGoodMHowardPAVacekJLRole of vitamin D in cardiovascular healthAm J Cardiol2010106 798 805 https://doi.org/10.1016/j.amjcard.2010.04.042 20816120
- Sukumar D, Shapses SA, Schneider SH. Vitamin D supplementation during short-term caloric restriction in healthy overweight/obese older women: effect on glycemic indices and serum osteocalcin levels. Mol Cell Endocrinol. 2015;410:1–5.
- SchoenmakersIFrancisRMMcCollEChadwickTGoldbergGRHarleC et al Vitamin D supplementation in older people (VDOP): Study protocol for a randomised controlled intervention trial with monthly oral dosing with 12,000 IU, 24,000 IU or 48,000 IU of vitamin D3 Trials201314 299 3848647 https://doi.org/10.1186/1745-6215-14-299 24041337
- CloseGLLeckeyJPattersonMBradleyWOwensDJFraserWD et al The effects of vitamin D(3) supplementation on serum total 25[OH]D concentration and physical performance: a randomised dose–response studyBr J Sports Med201347 692 6 https://doi.org/10.1136/bjsports-2012-091735 23410885
- BendikIFriedelARoosFFWeberPEggersdorferMVitamin D: a critical and essential micronutrient for human healthFront Physiol20145 248 4092358 https://doi.org/10.3389/fphys.2014.00248 25071593
- GrudetCMalmJWestrinABrundinLSuicidal patients are deficient in vitamin D, associated with a pro-inflammatory status in the bloodPsychoneuroendocrinology201450C 210 9 https://doi.org/10.1016/j.psyneuen.2014.08.016
- PolakMAHoughtonLAReederAIHarperMJConnerTSSerum 25-hydroxyvitamin D concentrations and depressive symptoms among young adult men and womenNutrients20146 4720 30 4245559 https://doi.org/10.3390/nu6114720 25353666
- CheiCLRamanPYinZ-XShiX-MZengYMatcharDBVitamin D levels and cognition in elderly adults in ChinaJ Am Geriatr Soc201462 11 2125 9 https://doi.org/10.1111/jgs.13082 25283584
- HolickMFVitamin D: its role in cancer prevention and treatmentProg Biophys Mol Biol200692 49 59 1:CAS:528:DC%2BD28XlsFKrsbs%3D https://doi.org/10.1016/j.pbiomolbio.2006.02.014 16566961
- GuillotXPratiCWendlingDVitamin D and spondyloarthritisExpert Rev Clin Immunol201410 1581 9 1:CAS:528:DC%2BC2cXhvFCitL3P https://doi.org/10.1586/1744666X.2014.972944 25346170
- WelchTRBergstromWHTsangRCVitamin D-deficient rickets: the reemergence of a once-conquered diseaseJ Pediatr2000137 143 5 1:STN:280:DC%2BD3cvjvVWmtg%3D%3D https://doi.org/10.1067/mpd.2000.109008 10931400
- BikleDDVitamin D metabolism, mechanism of action, and clinical applicationsChem Biol201421 319 29 3968073 1:CAS:528:DC%2BC2cXisFehu7k%3D https://doi.org/10.1016/j.chembiol.2013.12.016 24529992
- OganDPritchettKVitamin D and the athlete: risks, recommendations, and benefitsNutrients20135 1856 68 3725481 1:CAS:528:DC%2BC3sXpsFent78%3D https://doi.org/10.3390/nu5061856 23760056
- SatoYIwamotoJKanokoTSatohKLow-dose vitamin D prevents muscular atrophy and reduces falls and hip fractures in women after stroke: a randomized controlled trialCerebrovasc Dis200520 187 92 1:CAS:528:DC%2BD2MXpslCmsrY%3D https://doi.org/10.1159/000087203 16088114
- GlerupHMikkelsenKPoulsenLHassEOverbeckSAndersenH et al Hypovitaminosis D myopathy without biochemical signs of osteomalacic bone involvementCalcif Tissue Int200066 419 24 1:CAS:528:DC%2BD3cXksVSjsLs%3D https://doi.org/10.1007/s002230010085 10821877
- PfeiferMBegerowBMinneHVitamin D and muscle functionOsteoporos Int20023 187 94 https://doi.org/10.1007/s001980200012
- Macdougall JD, Hicks AL, Macdonald JR, Mckelvie RS, Green HJ, Smith KM. Muscle performance and enzymatic adaptations to sprint interval training. J Appl Physiol. 1998;84:2138–142.
- Farrokhyar F, Tabasinejad R, Dao D, Peterson D, Ayeni O, Hadioonzadeh R, et al. Prevalence of vitamin D inadequacy in athletes: A systematic-review and meta-analysis. Sport Med. 2014;5:365–78.
- CalvoMWhitingSPrevalence of vitamin D insufficiency in Canada and the United States: importance to health status and efficacy of current food fortification and dietary supplement useNutr Rev200361 107 13 https://doi.org/10.1301/nr.2003.marr.107-113 12723644
- Hossein-nezhadAHolickMFVitamin D for health: a global perspectiveMayo Clin Proc201388 720 55 3761874 1:CAS:528:DC%2BC3sXpvVyku7c%3D https://doi.org/10.1016/j.mayocp.2013.05.011 23790560
- Schlögl M, Holick MF. Vitamin D and neurocognitive function. Clin Interv Aging. 2014;9:559–68.
- HolickMFVitamin D: evolutionary, physiological and health perspectivesCurr Drug Targets201112 4 18 1:CAS:528:DC%2BC3MXkslajtbg%3D https://doi.org/10.2174/138945011793591635 20795941
- ProsserDEJonesGEnzymes involved in the activation and inactivation of vitamin DTrends Biochem Sci200429 664 73 1:CAS:528:DC%2BD2cXpslGis7o%3D https://doi.org/10.1016/j.tibs.2004.10.005 15544953
- Bischoff H, Borchers M, Gudat F, Duermueller U, Theiler R, Stähelin H, et al. In situ detection of 1,25-dihydroxyvitamin D3 receptor in human skeletal muscle tissue. Histochem J. 2001;33:19–24.
- Simpson R, Thomas G, Arnold A. Identification of 1,25-dihydroxyvitamin D3 receptors and activities in muscle*. J Biol Chem. 1985;260:8882–891.
- HamiltonBVitamin d and athletic performance: the potential role of muscleAsian J Sports Med20112 211 19 3289217 22375241
- StocktonKAMengersenKParatzJDKandiahDBennellKLEffect of vitamin D supplementation on muscle strength: a systematic review and meta-analysisOsteoporos Int201022 859 71 20924748
- Tomlinson PB, Joseph C, Angioi M. Effects of vitamin D supplementation on upper and lower body muscle strength levels in healthy individuals. A systematic review with meta-analysis. J Sci Med Sport. 2014;18(5):575-80.
- Beaudart C, Buckinx F, Rabenda V, Gillain S, Cavalier E, Slomian J, et al. The effects of vitamin D on skeletal muscle strength, muscle mass and muscle power: A systematic review and meta-analysis of randomized controlled trials. J Clin Endocrinol Metab. 2014;99:4336–345.
- Von HurstPRBeckKLVitamin D and skeletal muscle function in athletesCurr Opin Clin Nutr Metab Care201417 539 45 https://doi.org/10.1097/MCO.0000000000000105
- GregorySMParkerBACapizziJAGrimaldiASClarksonPMMoeckel-ColeS et al Changes in vitamin D are not associated with changes in cardiorespiratory fitnessClin Med Res20132 68 https://doi.org/10.11648/j.cmr.20130204.16
- MowryDACostelloMMHeelanKAAssociation among cardiorespiratory fitness, body fat, and bone marker measurements in healthy young femalesJ Am Osteopath Assoc2009109 10 534 9 19861594
- ArdestaniAParkerBMathurSClarksonPPescatelloLSHoffmanHJ et al Relation of vitamin D level to maximal oxygen uptake in adultsAm J Cardiol2011107 1246 9 4460985 1:CAS:528:DC%2BC3MXkt1ahu7g%3D https://doi.org/10.1016/j.amjcard.2010.12.022 21349488
- Koundourakis NE, Androulakis NE, Malliaraki N, Margioris AN. Relation of vitamin D level to maximal oxygen uptake in adults. Am J Cardiol. 2011;107:1246–9.
- FitzgeraldJPetersonBWarpehaJWilsonPRhodesGIngrahamSVitamin D status and VO2peak during a skate treadmill graded exercise test in competitive ice hockey playersJ Strength Cond Res201428 3200 5 https://doi.org/10.1519/JSC.0000000000000523 24832977
- ForneyLEarnestCCHenaganTJohnsonLCastleberryTStewartLVitamin D Status, Body Composition, and Fitness Measures in College-Aged StudentsJ Strength Cond Res201428 814 24 https://doi.org/10.1519/JSC.0b013e3182a35ed0 23897020
- JastrzębskiZEffect of vitamin D supplementation on the level of physical fitness and blood parameters of rowers during the 8-week high intensity trainingFacicula Educ Fiz şi Sport20142 57 67
- SugimotoHShiroYDiversity and substrate specificity in the structures of steroidogenic cytochrome P450 enzymesBiol Pharm Bull201235 818 23 1:CAS:528:DC%2BC38Xht1Klt7vN https://doi.org/10.1248/bpb.35.818 22687469
- D’AntonaGLanfranconiFPellegrinoMABroccaLAdamiRRossiR et al Skeletal muscle hypertrophy and structure and function of skeletal muscle fibres in male body buildersJ Physiol2006570 Pt 3 611 27 1479884 https://doi.org/10.1113/jphysiol.2005.101642 16339176
- SchoenfeldBThe mechanisms of muscle hypertrophy and their application to resistance trainingJ Strength Cond Res201024 2857 72 https://doi.org/10.1519/JSC.0b013e3181e840f3 20847704
- SchoenfeldBDoes exercise-induced muscle damage play a role in skeletal muscle hypertrophy?J Strength Cond Res201226 1441 53 https://doi.org/10.1519/JSC.0b013e31824f207e 22344059
- GarciaLAFerriniMGNorrisKCArtazaJN1,25(OH)2vitamin D3 enhances myogenic differentiation by modulating the expression of key angiogenic growth factors and angiogenic inhibitors in C2C12 skeletal muscle cellsJ Steroid Biochem Mol Biol2013133 1 11 3513642 1:CAS:528:DC%2BC38XhslCrsL7P https://doi.org/10.1016/j.jsbmb.2012.09.004 22982629
- GarciaLAKingKKFerriniMGNorrisKCArtazaJN1,25(OH)2vitamin D3 stimulates myogenic differentiation by inhibiting cell proliferation and modulating the expression of promyogenic growth factors and myostatin in C2C12 skeletal muscle cellsEndocrinology2011152 2976 86 3138228 1:CAS:528:DC%2BC3MXhtVemt7nE https://doi.org/10.1210/en.2011-0159 21673099
- Stratos I, Li Z, Herlyn P, Rotter R, Behrendt A-K, Mittlmeier T, et al. Vitamin D increases cellular turnover and functionally restores the skeletal muscle after crush injury in rats. Am J Pathol. 2013;182:895–904.
- LundDCornelisonDEnter the matrix: shape, signal and superhighwayFEBS J2013280 4089 99 3659178 1:CAS:528:DC%2BC3sXht1ylsrzL https://doi.org/10.1111/febs.12171 23374506
- BarkerTSchneiderEDDixonBMHenriksenVTWeaverLKSupplemental vitamin D enhances the recovery in peak isometric force shortly after intense exerciseNutr Metab (Lond)201310 69 https://doi.org/10.1186/1743-7075-10-69
- Ainbinder A, Boncompagni S, Protasi F, Dirksen RT. Role of mitofusin-2 in mitochondrial localization and calcium uptake in skeletal muscle. Cell Calcium. 2015;57:14–24.
- Todd JJ, Pourshahidi KL, McSorley EM, Madigan SM, Magee PJ. Vitamin D: Recent advances and implications for athletes. Sport Med. 2015;45:213–29.
- Ceglia L, Niramitmahapanya S, da Silva MM, Rivas DA, Harris SS, Bischoff-Ferrari H, et al. A randomized study on the effect of vitamin D3 supplementation on skeletal muscle morphology and vitamin D receptor concentration in older women. J Clin Endocrinol Metab. 2013;98:E1927–35.
- Close GL, Russell J, Cobley JN, Owens DJ, Wilson G, Fraser WD, et al. Assessment of vitamin D concentration in non-supplemented professional athletes and healthy adults during the winter months in the UK: implications for skeletal muscle function. J Sports Sci. 2013;31:344–53.
- Mauras N, Hayes V, Welch S, Rini A, Helgeson K, Dokler M, Veldhuis JD, Urban RJ. Testosterone deficiency in young men: Marked alterations in whole body protein kinetics, strength, and adiposity*. J Clin Endocrinol Metab. 1998;83:1886–892.
- Wehr E, Pilz S, Boehm B, März W, Obermayer-Pietsch B. Association of vitamin D status with serum androgen levels in men. Clin Endocrinol (Oxf). 2010;73:243–8.
- Pilz S, Frisch S, Koertke H, Kuhn J, Dreier J, Obermayer-Pietsch B, et al. Effect of vitamin D supplementation on testosterone levels in men. Horm Metab Res. 2011;43:223–5.
- Kinuta K, Tanaka H, Moriwake T, Aya K, Kato S, Seino Y. Vitamin D is an important factor in estrogen biosynthesis of both female and male gonads*. Endocrinology 200AD, 141:1317–324.
- Blomberg Jensen M, Nielsen JE, Jørgensen A, Rajpert-De Meyts E, Kristensen DM, Jørgensen N, et al. Vitamin D receptor and vitamin D metabolizing enzymes are expressed in the human male reproductive tract. Hum Reprod. 2010;25:1303–11.
- Blomberg JensenMDissingSNon-genomic effects of vitamin D in human spermatozoaSteroids201277 903 9 1:CAS:528:DC%2BC38XksFems70%3D https://doi.org/10.1016/j.steroids.2012.02.020 22414629
- HerbstKLBhasinSTestosterone action on skeletal muscleCurr Opin Clin Nutr Metab Care20047 271 7 1:CAS:528:DC%2BD2cXkvVWiu74%3D https://doi.org/10.1097/00075197-200405000-00006 15075918
- Urban RJ. Growth hormone and testosterone: Anabolic effects on muscle. Horm Res Pædiatrics. 2011;76:81–3.
- Holick M, MacLaughlin J, Clark M, Holick S, Potts J, Anderson R, et al. Photosynthesis of previtamin D3 in human skin and the physiologic consequences. Am Assoc Adv Sci. 1980;210:203–05.
- WebbARWho, what, where and when-influences on cutaneous vitamin D synthesisProg Biophys Mol Biol200692 17 25 1:CAS:528:DC%2BD28XlsFKrsb4%3D https://doi.org/10.1016/j.pbiomolbio.2006.02.004 16766240
- GilchrestBSun exposure and vitamin D sufficiencyAm J Clin Nutr200888 570 7
- Lim HW, Gilchrest BA, Cooper KD, Bischoff-Ferrari HA, Rigel DS, Cyr WH, et al. Sunlight, tanning booths, and vitamin D. J Am Acad Dermatol. 2005;52:868–76.
- WolpowitzDGilchrestBAThe vitamin D questions: how much do you need and how should you get it?J Am Acad Dermatol200654 301 17 https://doi.org/10.1016/j.jaad.2005.11.1057 16443061
- Holick MF. Sunligth "D"ilemma: Risk of skin cancer or bone disease and muscle weakness. Lancet 2001:4–6.
- HeaneyRPVitamin D in health and diseaseClin J Am Soc Nephrol20083 1535 41 1:CAS:528:DC%2BD1cXhtFensrfM https://doi.org/10.2215/CJN.01160308 18525006
- Mithal A, Wahl DA, Bonjour J-P, Burckhardt P, Dawson-Hughes B, Eisman JA, et al. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos Int. 2009;20:1807–20.
- Walker N, Love TD, Baker DF, Healey PB, Haszard J, Edwards AS, et al. Knowledge and attitudes to vitamin D and sun exposure in elite New Zealand athletes: a cross-sectional study. J Int Soc Sports Nutr. 2014;11:47.
- MatsuokaLYWortsmanJHollisBWUse of topical sunscreen for the evaluation of regional synthesis of vitamin D3J Am Acad Dermatol199022 772 5 1:STN:280:DyaK3c3ntVGjtw%3D%3D https://doi.org/10.1016/0190-9622(90)70107-S 2161436
- Tripkovic L, Lambert H, Hart K, Smith CP, Bucca G, Penson S, et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: a systematic review and meta-analysis. Am J Clin Nutr. 2012;95:1357–64.
- HolickMVitamin D deficiencyN Engl J Med2007357 266 281 1:CAS:528:DC%2BD2sXotVejsbc%3D https://doi.org/10.1056/NEJMra070553 17634462
- HoughtonL,AViethRThe case against ergocalciferol (vitamin D2) as a vitamin supplementAm J Clin Nutr200684 694 7 1:CAS:528:DC%2BD28XhtFWgtr3N 17023693
- LoganVFGrayARPeddieMCHarperMJHoughtonLALong-term vitamin D3 supplementation is more effective than vitamin D2 in maintaining serum 25-hydroxyvitamin D status over the winter monthsBr J Nutr2013109 1082 8 1:CAS:528:DC%2BC3sXjvVakt7w%3D https://doi.org/10.1017/S0007114512002851 23168298
- HarrisSSDawson-HughesBPlasma vitamin D and 25OHD responses of young and old men to supplementation with vitamin D3J Am Coll Nutr200221 357 62 1:CAS:528:DC%2BD38XmsFejsro%3D https://doi.org/10.1080/07315724.2002.10719235 12166534
- Post JL, Ilich Ernst JZ. Controversies in vitamin D recommendations and its possible roles in nonskeletal health issues. J Nutr Food Sci. 2013;03:1–5.
- RossACTaylorCL Yaktine AL2011 Calcium Vitamin D Valle HB Del
- EndocrineS Evalutation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline2011
- ViethRWhy the optimal requirement for Vitamin D3 is probably much higher than what is officially recommended for adultsJ Steroid Biochem Mol Biol200489–90 575 9 https://doi.org/10.1016/j.jsbmb.2004.03.038 15225842
- CannellJHollisBUse of vitamin D in clinical practiceAltern Med Rev200813 6 20 18377099
- Heaney RP. Assessing vitamin D status. Curr Opin Clin Nutr Metab Care. 2011;14:440–44.
- Vieth R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr. 1999;69:842–56.
- Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr. 2006;84:18–28.
- PramyothinPHolickMFVitamin D supplementation: guidelines and evidence for subclinical deficiencyCurr Opin Gastroenterol201228 139 50 1:CAS:528:DC%2BC38XivV2hurY%3D https://doi.org/10.1097/MOG.0b013e32835004dc 22274617
- Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911–30.
- HollisBWCirculating 25-hydroxyvitamin D levels indicative of vitamin D sufficiency: implications for establishing a new effective dietary intake recommendation for vitamin DJ Nutr2005135 317 22 1:CAS:528:DC%2BD2MXhsVKju74%3D 15671234
- EngelmanCDVitamin D recommendations: the saga continuesJ Clin Endocrinol Metab201196 3065 6 1:CAS:528:DC%2BC3MXhtlyitbnN https://doi.org/10.1210/jc.2011-2355 21976747
- Barger-LuxMJHeaneyRPDowellSChenTCHolickMFInternational original article vitamin D and its major metabolites : Serum levels after graded oral dosing in healthy menOsteoporos Int199825 222 30 https://doi.org/10.1007/s001980050058
- LappeJCullenDHaynatzkiGReckerRAhlfRThompsonKCalcium and vitamin d supplementation decreases incidence of stress fractures in female navy recruitsJ Bone Miner Res200823 741 9 1:CAS:528:DC%2BD1cXmtFyrsbY%3D https://doi.org/10.1359/jbmr.080102 18433305
- Dawson-HughesBHeaneyRPHolickMFLipsPMeunierPJViethREstimates of optimal vitamin D statusOsteoporos Int200516 713 6 1:CAS:528:DC%2BD2MXls1ahurk%3D https://doi.org/10.1007/s00198-005-1867-7 15776217
- HeaneyRPDaviesKMChenTCHolickMFJanet Barger-LuxMHuman serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferolAm J Clin Nutr200377 204 10 1:CAS:528:DC%2BD3sXhtFymtA%3D%3D 12499343
- WebbARKlineLHolickMFInfluence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skinJ Clin Endocrinol Metab198867 373 8 1:CAS:528:DyaL1cXkvFWltbs%3D https://doi.org/10.1210/jcem-67-2-373 2839537
- ValtueñaJDominguezDTilLGonzález-GrossMDrobnicFHigh prevalence of vitamin D insufficiency among elite Spanish athletes the importance of outdoor training adaptationNutr Hosp201430 124 31 25137271
- Villacis D, Yi A, Jahn R, Kephart CJ, Charlton T, Gamradt SC, et al. Prevalence of Abnormal Vitamin D Levels Among Division I NCAA Athletes. Sports Health. 2014;6:340–7.
- ConstantiniNWArieliRChodickGDubnov-RazGHigh prevalence of vitamin D insufficiency in athletes and dancersClin J Sport Med201020 368 71 https://doi.org/10.1097/JSM.0b013e3181f207f2 20818195
- HolickMFThe vitamin D epidemic and its health consequencesJ Nutr2005135 2739S 48 1:CAS:528:DC%2BD2MXht1CgsbjJ 16251641
- Van den OuwelandJFleurenHDrabbeMVollaardHPharmacokinetics and safety issues of an accidental overdose of 2,000,000 IU of vitamin D3 in two nursing home patients: a case reportBMC Pharmacol Toxicol201415 57 4185191 https://doi.org/10.1186/2050-6511-15-57 25269374
- Suda T, Ueno Y, Fujii K, Shinki T. Vitamin D and bone. J Cell Biochem. 2003;88:259–66S.
- Zittermann A. Vitamin D and cardiovascular disease. Anticancer Res. 2014;34:4641–648.
- Kidd PM. Vitamins D and K as pleiotropic nutrients: clinical importance to the skeletal and cardiovascular systems and preliminary evidence for synergy. Altern Med Rev. 2010;15:199–222.
- GundbergCMLianJBBoothSLVitamin K-dependent carboxylation of osteocalcin: friend or foe?Adv Nutr An20123 149 157 1:CAS:528:DC%2BC38XmtVGrs7k%3D https://doi.org/10.3945/an.112.001834 22516722
- MizuguchiMFujisawaRNaraMNittaKKawanoKFourier-Transform infrared spectroscopic study of Ca2 + −binding to osteocalcinCalcif Tissue Int200169 337 42 1:CAS:528:DC%2BD38XpvFyktw%3D%3D https://doi.org/10.1007/s002230010042 11800230
- Fitzpatrick TB, Basset GJC, Borel P, Carrari F, DellaPenna D, Fraser PD, et al. Vitamin deficiencies in humans: can plant science help? Plant Cell. 2012;24:395–414.
- BinkleyNCKruegerDCKawaharaTNEngelkeJAChappellRJSuttieJWA high phylloquinone intake is required to achieve maximal osteocalcin γ-carboxylationAm J Clin Nutr200276 1055 60 1:CAS:528:DC%2BD38XosF2qs7s%3D 12399278
- AkiyamaYHaraKTajimaTMurotaSMoritaIEffect of vitamin K2 (menatetrenone) on osteoclast-like cell formation in mouse bone marrow culturesEur J Pharmacol1994263 181 5 1:CAS:528:DyaK2MXhtFensrk%3D https://doi.org/10.1016/0014-2999(94)90539-8 7821350
- AkiyamaYHaraKKobayashiMTomiugaTNakamuraTInhibitory Effect of Vitamin K2 (Menatetrenone) on Bone Resorption in Ovariectomized Rats. A Histomorphometric and Dual Energy X-Ray Absorptiometic StudyThe Japanese Journal of Pharmacology199980 1 67 74 1:CAS:528:DyaK1MXjtlGis70%3D https://doi.org/10.1254/jjp.80.67 10446758
- FeskanichDWeberPWillettWCRockettHBoothSLColditzGAVitamin K intake and hip fractures in women: a prospective studyAm J Clin Nutr199969 74 9 1:CAS:528:DyaK1MXjvFSmtA%3D%3D 9925126
- MasterjohnCVitamin D toxicity redefined: vitamin K and the molecular mechanismMed Hypotheses200768 1026 34 1:CAS:528:DC%2BD2sXit12murY%3D https://doi.org/10.1016/j.mehy.2006.09.051 17145139
- El AsmarMSNaoumJJArbidEJVitamin k dependent proteins and the role of vitamin k2 in the modulation of vascular calcification: a reviewOman Med J201429 172 7 4052396 https://doi.org/10.5001/omj.2014.44 24936265
- HamidiMSCheungAMVitamin K and musculoskeletal health in postmenopausal womenMol Nutr Food Res201458 1647 57 1:CAS:528:DC%2BC2cXhtVWlsrjN https://doi.org/10.1002/mnfr.201300950 24956598
- IwamotoJVitamin K2 therapy for postmenopausal osteoporosisNutrients20146 1971 80 4042573 https://doi.org/10.3390/nu6051971 24841104
- MurakamiKYamanakaNOhnishiKFukayamaMYoshinoMInhibition of angiotensin I converting enzyme by subtilisin NAT (nattokinase) in natto, a Japanese traditional fermented foodFood Funct20123 674 8 1:CAS:528:DC%2BC38XnvVOlt7s%3D https://doi.org/10.1039/c2fo10245e 22453301
- DowdPHershlineRHamSWNaganathanSMechanism of action of vitamin KNat Prod Rep199411 251 64 1:CAS:528:DyaK2MXosFOktg%3D%3D https://doi.org/10.1039/np9941100251 15200013
- Fusaro M, Crepaldi G, Maggi S, Galli F, D’Angelo A, Calò L, et al. Vitamin K, bone fractures, and vascular calcifications in chronic kidney disease: an important but poorly studied relationship. J Endocrinol Invest. 2011;34:317–23.
- Konakion® Konakion®2010 1 6
- CraciunAMWolfJKnapenMHBrounsFVermeerCImproved bone metabolism in female elite athletes after vitamin K supplementationInt J Sports Med199819 479 84 1:CAS:528:DyaK1cXntlGqs7w%3D https://doi.org/10.1055/s-2007-971948 9839845
- Visser M, Deeg DJ, Puts MT, Seidell JC, Lips P. Low serum concentrations of 25-hydroxyvitamin D in older persons and the risk of nursing home admission. Am J Clin Nutr. 2006;84:671–672.