Abstract
Most people are born with the ability to digest lactose, the major carbohydrate in milk and the main source of nutrition until weaning. Approximately 75% of the world’s population loses this ability at some point, while others can digest lactose into adulthood. This review discusses the lactase-persistence alleles that have arisen in different populations around the world, diagnosis of lactose intolerance, and its symptomatology and management.
Introduction
Lactose is a disaccharide that is abundant in mammalian milk and essential for the nourishment of newborn infants. It is hydrolyzed by the intestinal brush-border enzyme, lactase, into absorbable sugars, namely glucose and galactose. In most infants, intestinal lactase activity is maximal during the perinatal period; however, after 2–12 years of age, two distinct groups emerge, ie, a “lactase non-persistence” group with low lactase activity (hypolactasia) and a “lactase-persistence” group of individuals who retain their neonatal level of lactase activity into adulthood.Citation1–Citation3
Reduction in lactase activity causes primary maldigestion of lactose, a condition that is occasionally asymptomatic. When symptoms are present, lactose intolerance is diagnosed. It is important to distinguish between primary hypolactasia and secondary causes of maldigestion of lactose, including celiac disease, infectious enteritis, or Crohn’s disease, which have distinct pathogenic and therapeutic implications. Moreover, primary hypolactasia should be distinguished from congenital lactase deficiency, a rare autosomal recessive disease with unique molecular mechanisms that affects infants from birth.Citation4
Lactase-persistence alleles and polymorphisms for lactose tolerance
The LCT gene is 49.3 kb in length and located on the long (q) arm of chromosome 2 at position 21. It contains 17 exons and is translated into a 6 kb transcript (NCBI Reference Sequence NG_008104.1). Individuals with hypolactasia and lactase persistence have identical coding sequences, except for some silent mutations; thus, both lactases are identical.Citation5
Enattah et alCitation6 devised a brilliant strategy using polymorphic microsatellite markers flanking LCT, encompassing a region of 47 kb, in a haplotype linkage analysis of nine Finnish families with hypolactasia. Two variants were associated with lactase persistence. A polymorph variant, LCT-13910C>T, in intron 13 of the MCM6 gene that is 13,910 bp from the initiation codon of LCT, demonstrated a complete association, while the LCT-22018G>A variant in intron 9 of MCM6 gene upstream of the LCT locus 22,018 bp was strongly, but not completely, associated.Citation1,Citation2,Citation6 The functional role of MCM6 in vertebrates is unknown, but it has been implicated in “licensing” DNA replication during the cell cycle.Citation1 This association was confirmed in a study of DNA collected from subjects of Finnish, South Korean, Italian, German, French, or white or African North American descent.Citation1,Citation6
In subjects of European descent, the LCT-13910C>T variant completely associated with the lactase-persistence phenotype and presented different allelic frequencies in countries within Europe, Oceania, Asia, and the Americas, as shown in .
Table 1 Frequencies of the European variant LCT-13910C>T in countries within the Americas, Asia, Europe, and Oceania
Both genotypes of LCT-13910CT and LCT-13910TT were associated with the lactase-persistence phenotype, indicating that the presence of one single lactase-persistence allele in the heterozygous state has a dominant effect, rendering the person a lactose digester, whereas the genotype LCT- 13910CC, when the lactase-persistence allele LCT-13910T is absent, is consistent with lactose maldigestion.Citation2,Citation3
Despite the association of LCT-13910C>T with lactose digestion in Europeans, analysis of this variant in Africa demonstrated its restriction to populations with a high prevalence of the lactase-persistence phenotype (). This finding suggests the presence of other lactase-persistence alleles (). Thus, as shown in , different alleles have originated in various locations around the world over the course of human history after the emergence of modern man from Africa.Citation17
Figure 1 Tendency of lactase-persistence polymorphic variants in the world, based on the reports presented in , , and .
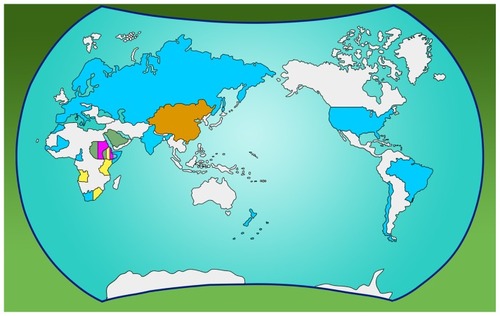
Table 2 Frequencies of the lactase persistence allele (LCT-13910C>T) reported in African countries
Table 3 Frequencies of other lactase persistence alleles in the MCM6 gene
Genotyping of LCT-13910C>T versus LCT-22018G>A has shown almost full agreement. Patients with LCT- 13910CC were also LCT-22018GG, while individuals with LCT-13910CT had the LCT-22018GA genotype. LCT-13910TT was associated with LCT-22018AA, except for a few cases in FinlandCitation6 and China,Citation32 and in Japanese Brazilians.Citation33
Functional in vitro studies of these polymorphic alleles have shown that LCT-13910T,Citation1,Citation34,Citation35 LCT-13907G, LCT- 13915G, and LCT-14010C act as enhancers of the LCT promoterCitation29 unlike in ancestral constructs (LCT-13910C, LCT-13907C, LCT-13915T, and LCT-14010G). These effects are most likely mediated by the Oct-1 transcriptional factor binding site in the variant enhancer and by HNF1α binding in the LCT promoter. However, further evaluation is required to determine whether these actions correspond to the situation in vivo.Citation34–Citation36
LCT gene regulation of lactase-persistence alleles occurs at the transcriptional level. LCT mRNA levels, which are distinguished by polymorphic markers in the coding region of LCT, were several times higher in individuals with LCT-13910T/-22018A alleles than in individuals with LCT-13910C/-22018G alleles.Citation1 After 5 years of age, an imbalance appears in the mRNA levels of LCT-13910C and LCT-13910T, with the LCT-13910T allele representing approximately 92% of LCT mRNA in children heterozygous for LCT-13910CT.Citation3
Several transcription factors (Cdx2, GATA-4, GATA-5, GATA-6, and HNF1α) activate the LCT promoter in intestinal cell culture at the −100 to −20 bp binding site regions of LCT which are repressed by PDX-1.Citation1 Mutation of the PDX-1 binding site does not prevent LCT promoter repression, which suggests that PDX-1 might function by binding to another DNA binding site or by inhibiting other transcriptional factors. PDX-1 overexpression resulted in strong repression of Cdx2 and HNF1α activation of the LCT promoter.Citation1 However, the exact mechanism for downregulation of LCT after weaning is unknown.
Haplotype conservation around lactase-persistence alleles indicates that these alleles emerged recently in different parts of the world and have been subject to strong positive selection in communities of high and perhaps intermittently exclusive consumers of fresh milk.Citation28 Nevertheless, the selective advantage provided by drinking fresh milk is not yet clear among populations reliant on agriculture with dairy farming as their main source of income, but has been discussed in detail elsewhere.Citation37 Gene-culture coevolution is a likely hypothesis in Africa, because high lactase-persistence allele frequencies are preferentially found in pastoral communities. In populations more likely to consume agricultural products, cheese and fermented milk, which have lower concentrations of lactose, the frequencies of lactase-persistence variants are possibly due to genetic drift.Citation38
It is estimated that the LCT-13910T allele initially originated on the background of a more common haplotype approximately 5000–12,000 years ago and re-emerged recently (1400–3000 years ago) on another haplotype background in restricted populations west of the Urals and north of the Caucasus. Citation7 The LCT-13907G and LCT-13910T alleles share the same ancestral lactase non-persistence haplotype, although they are on backgrounds of different lactase-persistence haplotypes. Citation25,Citation28,Citation35 LCT-13915G and LCT-14010C originated on different haplotype backgrounds,Citation25,Citation28,Citation29,Citation35 but age estimates are similar for both, at approximately 4095 ± 2045 years.Citation35
Diagnosis
Initially, the most accurate method available for the diagnosis of lactose maldigestion was direct biochemical assay of lactase activity from a jejunal sample. This assay is performed with a glucose oxidase reagent, which detects glucose liberated from lactose, with a cutoff value of 10 U/g protein.Citation1,Citation2 Due to the invasiveness of a jejunal biopsy, this method has been replaced by endoscopic duodenal biopsy.Citation39,Citation40 Mean lactase activity was about 40% lower in the duodenum compared with the jejunum,Citation39 but the Quick lactase test performed in samples taken from the postbulbar duodenum effectively identified patients with severe duodenal hypolactasia, with a sensitivity and specificity of 95% and 100%, respectively.Citation40
Lactose tolerance tests have been developed to confirm the ability of intestinal lactase to hydrolyze and absorb lactose, and to avoid intestinal biopsies. Blood glucose levels were measured before and after an oral load of lactose at prespecified time intervals, with a maximum rise of 20 mg/dL, indicating lactose tolerance.Citation41 Oral ethanol administration before lactose load is used to inhibit galactose metabolism for the determination of the blood maximum rise of glucose (at least 20 mg/dl) and galactose (at least 10 mg/dl), indicating lactose tolerance. Thus, galactose concentration in combination with glucose concentration improves the correlation with jejunal lactase activity than using only glucose maximum rise after lactose load.Citation42 Nonetheless, of all the indirect lactose tolerance tests currently available, breath hydrogen after ingestion of 50 g of lactose was considered the most suitable test for population screening for lactase deficiency.Citation43 Use of the 50 g lactose dose has been criticized, because it is equivalent to 4–5 cups of milk, an amount that is far more than an individual usually ingests at one time,Citation44 so an oral load of 25 g, ie, the mean quantity contained in 500 mL of semiskimmed milk, may be considered a more appropriate amount, with high sensitivity and specificity.Citation41,Citation44,Citation45
The lactose breath test is based on fermentation of undigested lactose by intestinal flora, producing hydrogen, carbon dioxide, and methane that are absorbed and eliminated via the lungs, but these gases also cause bloating, flatulence, abdominal pain, and diarrhea. Despite being widely used, the reliability of this test depends on the activity of bacterial flora. A false-negative result can occur if antibiotics have been taken within one month of being tested, if colonic pH is acidic enough to inhibit bacterial activity, or if there has been adaptation in the bacterial flora as a result of continuous lactose exposure.Citation41,Citation44,Citation45
The discovery of lactase-persistence alleles prompted use of genetic tests for diagnosis of lactase non-persistence by polymerase chain reaction restriction fragment length polymorphism, Citation45–Citation47 real-time polymerase chain reaction,Citation48–Citation50 and Pyrosequencing® technology.Citation51 Compared with the lactose hydrogen breath test, the genetic test is a simple, noninvasive, and more comfortable examination that does not provoke symptoms of lactose intolerance and is less cumbersome,Citation46,Citation51 with easy transfer of a venous blood sample to the laboratory. Citation45,Citation47 However, other polymorphic variants in Europeans (LCT-13914G>A)Citation50 and in African and Arab populations (LCT-13907C>G, LCT-13913T>C, and LCT-13915T>G, close to LCT-13910C>T, depicted in ) affect the diagnostic accuracy of LCT-13910C>T typing by altering the melting profiles of the real-time polymerase chain reaction kit.Citation50 The reverse-hybridization strip assay based on multiplex DNA amplification and ready-to-use membrane test strips that detect LCT polymorphic variants (-13907C>G, -13910C>T, -13913T>C, -13914G>A, -13915T>G, and -22018G>A) represents a reliable tool for genetic diagnosis of lactase non-persistence, overcoming the interference of different melting profiles of the real-time polymerase chain reaction kit by the other polymorphic variants.Citation12
The genetic test provides a more direct result, ie, a hypolactasia or lactase persistence genotype, whereas interpretation of the lactose breath test depends on the cutoff level, dose of lactose given, and duration of the test and age of the individual, among the other factors already discussed,Citation45–Citation47,Citation49,Citation51 and is costly.Citation47,Citation51 The discovery of other single nucleotide polymorphisms associated with lactase persistence (see ) implies that DNA genotyping should provide information on the DNA sequence around the polymorphic site of the MCM6 gene.Citation51 In addition to the reverse-hybridization strip assay,Citation12 Pyrosequencing technology may be a cost-effective option (€10 per test for polymerase chain reaction and Pyrosequencing reagents) for direct DNA sequencing, allowing genotyping of other single nucleotide polymorphisms.Citation51 The genetic test does not provide information on symptoms of lactose tolerance; however, measurement of lactase activity in intestinal biopsy does not provide it either.Citation47
Contribution of lactose ingestion to symptomatology
The age of onset of primary hypolactasia varies between different ethnic groups. Hypolactasia does not cause any disturbance or discomfort unless lactose-containing food is consumed. Colonic microflora ferment undigested lactose in the intestinal lumen, which leads to production of short-chain fatty acids, hydrogen, carbon dioxide, and methane. These byproducts cause bloating, flatulence, and abdominal pain. Undigested lactose acidifies the colon and increases the osmotic load, resulting in loose stools and diarrhea.Citation52 Stools are usually voluminous, foamy, and aqueous. Although hypolactasia- related diarrhea can become chronic, affected individuals typically do not lose weight. However, some patients can experience constipation due to decreased intestinal motility, possibly caused by production of methane.Citation52
Some authors have reported that the clinical presentation of lactose intolerance is not restricted to gut symptoms. Systemic complaints, such as headache, vertigo, memory impairment, lethargy, muscle and joint pains, allergy, cardiac arrhythmia, mouth ulcers, and sore throat, have been reported in less than 20% but up to 86% of these patients.Citation53,Citation54 Putative toxic metabolites, such as acetaldehyde, acetoin, ethanol, peptide, and protein toxins, can alter cell signaling mechanisms and are possibly responsible for these systemic symptoms. They are generated by lactose fermentation in colonic bacteria.Citation53,Citation55 When systemic complaints are present, it is important to assess whether they result from lactose intolerance, are coincidental, or emanate from an allergy to cow’s milk protein, which is present in up to 20% of patients with symptoms of lactose intolerance.Citation52 Minenna et al reported a possible association between gastroesophageal reflux disease and lactose malabsorption in 30 subjects; however, further studies are required to ascertain a causal relationship, given that both lactose intolerance and reflux are very common conditions.Citation56
There is considerable intraindividual and interindividual variability in the severity of symptoms, according to the amount of lactose ingested and the patient’s ability to digest it. Factors contributing to this variability include osmolality and the fat content of lactose-containing food, gastric emptying rate, ability of colonic microflora to ferment lactose, intestinal transit time, colonic water absorption capacity, and individual perception of abdominal pain and discomfort.Citation52,Citation57 Valid evidence is missing for a relationship between symptoms and amount of lactose ingested.Citation57 Most studies have included a small number of participants and/or subjects, with lactose maldigestion diagnosed by the breath hydrogen test but not always concomitant with lactose intolerance. In this regard, the available dataCitation58 demonstrate that a single dose of lactose (up to 12 g, equivalent to that contained in approximately one glass of milk) administered alone produces no or minor symptoms in persons with lactose intolerance or maldigestion. Lactose doses of 15–18 g are well tolerated when offered together with other nutrients. With doses larger than 18 g, intolerance becomes progressively more frequent, and quantities over 50 g elicit symptoms in most individuals.Citation58
Various reports indicate that symptoms typically considered secondary to lactose ingestion are not truly related to maldigestion.Citation59,Citation60 On self-report questionnaires, individuals commonly associate ingestion of lactose-containing products with onset of abdominal symptoms, even in the absence of objective evidence for lactose maldigestion, such as an altered lactose breath test.Citation61,Citation62 Symptoms frequently attributed to lactose maldigestion can be secondary to irritable bowel syndrome,Citation62 which shares a similar clinical presentation, or food allergy.Citation63 Even a “nocebo effect”, ie, occurrence of symptoms after ingestion of an inert substance when negative expectations about its content exist,Citation64 has been considered to be contributory to this exaggerated perception of lactose intolerance.Citation65 However, this concept requires more consistent evidence.Citation66 The misleading diagnosis of lactose intolerance and subsequent implementation of a dairy-restricted diet is not without consequences. The negative clinical impact of imposed restrictions, which mainly involve bone metabolism, is a topic that will be discussed in a following section.
Along with irritable bowel syndrome and cow’s milk protein allergy, the differential diagnosis of lactose intolerance includes bacterial overgrowth, celiac disease, and inflammatory bowel disease.Citation57,Citation67 When bloating and flatulence are the predominant symptoms, it is also advisable to rule out the possible contribution of other dietary sources of intestinal gas, such as beans, which contain two indigestible sugars, stachyose and raffinose.Citation57,Citation68
Management
The goal of treatment is to improve symptoms while maintaining an adequate intake of calcium, thus preventing secondary bone disease caused by a milk-restricted diet. Considerable efforts have been made to confirm whether decreased lactase enzyme activity can impair calcium absorption and prevent attainment of optimal peak bone mass. When evaluating peak bone mass and bone turnover rate in a young population with molecularly defined lactose maldigestion, Enattah et al showed that hypolactasia and lactose maldigestion do not alter calcium absorption or bone turnover rate, nor do they impair acquisition of peak bone mass. Moreover, the LCT- 13910CC genotype does not appear to be a risk factor for stress fractures in this population.Citation69 Although decreased calcium absorption, evaluated by the strontium absorption test in patients with the LCT-13910CC genotype, was reported by Obermayer-Pietsch et al,Citation70 the predominant idea in the literature is that low calcium intake, rather than deficient calcium absorption, is the major factor contributing to loss of bone mass.Citation57,Citation69 Several studies in patients with presumed or confirmed lactose intolerance have also reported lower calcium intake in this population.Citation57,Citation71,Citation72
Several reports have been published that address the relationship among the LCT-13910C>T genotype, lactose intolerance, bone mineral density, and fracture risk. Studies in postmenopausal womenCitation73 and elderly peopleCitation74 with the LCT-13910CC genotype have identified lower bone mineral density and a higher incidence of bone fractures in comparison with individuals with other lactase genotypes. However, these results have not been confirmed by other studiesCitation75,Citation76 or in younger subjects.Citation69,Citation77
Recently, Tolonen et al showed that young men with the LCT-13910TT genotype had the highest bone trabecular density at the distal radius and tibia, but other bone traits or low-energy fractures were not associated with the LCT-13910C>T genotype.Citation78 In addition to height and bone parameters, Koek et al assessed the correlation between vitamin D receptor polymorphisms and LCT-13910C>T genotypes in the elderly. This study found that the LCT- 13910CC genotype was associated with lower dietary calcium intake and lower serum calcium levels, but not with bone mineral density and fracture risk. No interaction was detected between LCT-13910C>T genotypes and vitamin D receptor polymorphisms.Citation10
The available data suggest that deficient calcium intake plays a major role in lactose intolerance that may be related to bone disease. Therefore, an objective diagnosis through either the hydrogen breath test or molecular detection of hypolactasia is key to the appropriate clinical management of patients with symptoms suggestive of lactose intolerance. This approach avoids inappropriate calcium-restricted diets and adverse consequences for bone health.
The initial recommendation for management of lactose intolerance is to aim for remission of symptoms by temporarily avoiding milk and dairy products. As mentioned earlier, most individuals with lactose malabsorption can tolerate up to 12 g of lactose without significant symptoms. After the initially restricted diet, lactose should be gradually reintroduced until the patient’s threshold for symptoms is reached.Citation79 At this point, several behavioral measures can be adopted to overcome possible symptoms, including having fermented and matured milk products in the diet, consuming lactose together with other foods, and distributing lactose intake over the day. Although lactose tablets have been cited as a potential trigger of symptoms of lactose intolerance, such a small amount of lactose cannot be blamed for provoking symptoms, even when differences in individual symptom thresholds are considered.Citation80
If the measures suggested here do not suffice in reducing symptoms, pharmacological strategies can be implemented. The main pharmacological measures in use include lactase supplements, lactose-hydrolyzed or lactose-reduced milk, probiotics, colonic adaptation, and rifaximin. Ingestion of probiotics containing lactase may have the potential to aid lactose digestion in intolerant patients, but studies that have investigated this have published conflicting results. Therefore, the role of probiotics in lactose intolerance management is currently uncertain.Citation79 Yoghurt containing live cultures providing endogenous beta galactosidase are an alternative source of calories and calcium, and are well tolerated by many lactose-intolerant patients. However, yoghurt containing milk or its derivatives added after fermentation can cause symptoms.Citation79 Overall, the available evidence-based data are insufficient to ascertain the efficacy of these interventions, as discussed at a recent National Institutes of Health conference.Citation58
Attention must be paid to daily ingestion of calcium and vitamin D, with supplementation as required. For adolescents and young adults, the dietary calcium recommendation is generally 1200–1500 mg. In adults, the amount varies according to gender and menopausal status. Calcium should be supplemented if there is not enough in the diet, and vitamin D should also be monitored and supplemented if necessary.Citation79 Well designed, randomized, placebo-controlled trials are still required before strong clinical recommendations can be made for the management of patients who are intolerant of lactose-hydrolyzed milk and yoghurt.
Conclusion
Random mutations have occurred in regions upstream of the LCT gene that have an enhancer effect on the LCT promoter, which enables carriers with the lactase-persistence phenotype to exist in populations all over the world. No “gold standard” test is available for the diagnosis of lactose intolerance. The lactose breath test, although considered the best method, may be influenced by confounding factors. Genetic testing has been a new tool for the diagnosis of hypolactasia/lactase persistence, but may not detect all the single nucleotide polymorphisms associated with this disorder. Symptoms of lactose intolerance might have been exaggerated, such that up to 12 g of lactose is possibly well tolerated by lactase non-persistence individuals, which negates the need for restrictions on lactose-hydrolyzed milk, as well as fermented and matured milk products, preventing any subsequent effects on bone mass density.
Acknowledgment
The authors thank Marcos Antonio Retzer for his contribution to .
Disclosure
The authors have no conflict of interests to declare in this work.
References
- TroelsenJTAdult-type hypolactasia and regulation of lactase expressionBiochim Biophys Acta20051723193215777735
- RasinperäHSavilahtiEEnattahNSA genetic test which can be used to diagnose adult-type hypolactasia in childrenGut2004531571157615479673
- RasinperäHKuokkanenMKolhoKLLindahlHEnattahNSSavilahtiETranscriptional down regulation of the lactase (LCT) gene during childhoodGut2005541660166116227366
- Robayo-TorresCCNicholsBLMolecular differentiation of congenital lactase deficiency from adult-type hypolactasiaNutr Rev200765959817345962
- BollWWagnerPManteiNStructure of the chromosomal gene and cDNAs coding for lactase-phlorizin hydrolase in humans with adult-type hypolactasia or persistence of lactaseAm J Hum Genet1991488898901902057
- EnattahNSSahiTSavilahtiETerwilligerJDPeltonenLJãverlãIIdentification of a variant associated with adult-type hypolactasiaNat Genet20023023323711788828
- EnattahNSTrudeauAPimenoffVEvidence of still-ongoing convergence evolution of the lactase persistence T-13910 alleles in humansAm J Hum Genet20078161562517701907
- AlmonREngfeldtPTyskCSjöströmMNilssonTKPrevalence and trends in adult-type hypolactasia in different age cohorts in Central Sweden diagnosed by genotyping for the adult-type hypolactasia-linked LCT-13910C>T mutationScand J Gastroenterol20074216517017327935
- UptonJGeorgePThe prevalence of lactose intolerance (adult hypolactasia) in a randomly selected New Zealand populationN Z Med J2010123117118
- KoekWNvan MeursJBvan der EerdenBCThe T-13910C polymorphism in the lactase phlorizin hydrolase gene is associated with differences in serum calcium levels and calcium intakeJ Bone Miner Res2010251980198720225268
- KuokkanenMButzowRRasinperäHLactase persistence and ovarian carcinoma risk in Finland, Poland and SwedenInt J Cancer2005117909415880573
- TagCGOberkaninsCKriegshäuserGEvaluation of a novel reverse-hybridization StripAssay for typing DNA variants useful in diagnosis of adult-type hypolactasiaClin Chim Acta2008392586218396155
- LemberMTorniainenSKullMLactase non-persistence and milk consumption in EstoniaWorld J Gastroenterol2006127329733117143950
- KozlovABorinskayaSVershubskyGGenes related to the metabolism of nutrients in the Kola Sami populationInt J Circumpolar Health200867566618468259
- KhabarovaYTorniainenSNurmiHJärveläIIsokoskiMMattilaKPrevalence of lactase persistent/non-persistent genotypes and milk consumption in a young population in north-west RussiaWorld J Gastroenterol2009151849185319370782
- NagyDTömöryGCsányiBComparison of lactase persistence polymorphism in ancient and present-day Hungarian populationsAm J Phys Anthropol201114526226921365615
- CoelhoMLuiselliDBertorelleGMicrosatellite variation and evolution of human lactase persistenceHum Genet200511732933915928901
- AlmonRÁlvarez-LeonEEEngfeldtPSerra-MajemLMagnusonANilssonTKAssociations between lactase persistence and the metabolic syndrome in a cross-sectional study in the Canary IslandsEur J Nutr20104914114619844753
- BabuJKumarSBabuPPrasadJHGhoshalUCFrequency of lactose malabsorption among healthy southern and northern Indian populations by genetic analysis and lactose hydrogen breath and tolerance testsAm J Clin Nutr20109114014619889824
- AnagnostouPBattaggiaCCoiaVTracing the distribution and evolution of lactase persistence in southern Europe through the study of the T-13910 variantAm J Hum Biol20092121721919107920
- MattarRMonteiroMSVillaresCASantosAFSilvaJMKCarrilhoFJFrequency of LCT-13910C>T single nucleotide polymorphism associated with adult-type hypolactasia/lactase persistence among Brazilians of different ethnic groupsNutr J200984619799794
- MoralesEAzocarLMaulXPerezCChianaleJMiqueJFThe European lactase persistence genotype determines the lactase persistence state and correlates with gastrointestinal symptoms in the Hispanic and Amerindian Chilean population: a case-control and population-based studyBMJ Open20111e000125
- HeyerEBrazierLSégurelLLactase persistence in Central Asia: phenotype, genotype, and evolutionHum Biol20118337939221740154
- MulcareCAWealeMEJonesALThe allele of a single-nucleotide polymorphism 13.9 kb upstream of the lactase gene (LCT) (C13.9kbT) does not predict or cause the lactase-persistence phenotype in AfricansAm J Hum Genet2004741102111015106124
- IngramCJEElaminFFMulcareCAA novel polymorphism associated with lactose tolerance in Africa: multiple causes for lactase persistence?Hum Genet200712077978817120047
- LokkiAIJärveläIIsraelssonELactase persistence genotypes and malaria susceptibility in Fulani of MaliMalar J201110921235777
- TorniainenSParkerMIHolmbergVLahtelaEDandaraCJärveläIScreening of variants for lactase persistence/non-persistence in populations from South Africa and GhanaBMC Genet2009103119575818
- IngramCJERagaTOTarekegnAMultiple rare variants as a cause of a common phenotype: several different lactase persistence associated alleles in a single ethnic groupJ Mol Evol20096957958819937006
- TishkoffSAReedFARanciaroAConvergent adaptation of human lactase persistence in Africa and EuropeNat Genet200739314017159977
- ImtiazFSavilahtiESarnestoAThe T/G_13915 variant upstream of the lactase gene (LCT) is the founder allele of lactase persistence in an urban Saudi populationJ Med Genet2007441416825431
- KhabarovaYTorniainenSSavilahtiEIsokoskiMMattilaKJärveläIThe -13914G>A variant upstream of the lactase gene (LCT) is associated with lactase persistence/non-persistenceScand J Clin Lab Invest20107035435720509822
- XuLSunHZhangXThe -22018A allele matches the lactase persistence phenotype in northern Chinese populationsScand J Gastroenterol20104516817419947896
- MattarRMonteiroMSSilvaJMKCarrilhoFJLCT-22018G>A single nucleotide polymorphism is a better predictor of adult-type hypolactasia/lactase persistence in Japanese-Brazilians than LCT- 13910C>TClinics (São Paulo)20106513991400
- LewinskyRHJensenTGKMøllerJStensballeAOlsenJTroelsenJTT-13910 DNA variant associated with lactase persistence interacts with Oct-1 and stimulates lactase promoter activity in vitroHum Mol Genet2005143945395316301215
- EnattahNSJensenTGKNielsenMIndependent introduction of two lactase-persistence alleles into human populations reflects different history of adaptation to milk cultureAm J Hum Genet200882577218179885
- OldsLCAhnJKSibleyE-13915*G DNA polymorphism associated with lactase persistence in Africa interacts with Oct-1Hum Genet201112911111320960210
- IngramCJEMulcareCAItanYThomasMGSwallowDMLactose digestion and the evolutionary genetics of lactase persistenceHum Genet200912457959119034520
- GerbaultPMoretCCurratMSanchez-MazasAImpact of selection and demography on the diffusion of lactase persistencePLoS One20094e636919629189
- LagmanJMRowlandRActivity of duodenal disaccharidases in relation to normal and abnormal mucosal morphologyJ Clin Pathol1990435375402116456
- KuokkanenMMyllyniemiMVauhkonenMA biopsy-based quick test in the diagnosis of duodenal hypolactasia in upper gastrointestinal endoscopyEndoscopy20063870871216761211
- LawDConklinJPimentelMLactose intolerance and the role of the lactose breath testAm J Gastroenterol20101051726172820686460
- JussilaJDiagnosis of lactose malabsorption by the lactose tolerance test with peroral ethanol administrationScand J Gastroenterol196943613685346680
- NewcomerADMcGillDBThomasPJHofmannAFProspective comparison of indirect methods for detecting lactase deficiencyN Engl J Med1975293123212361186802
- RomagnuoloJSchillerDBaileyRJUsing breath tests wisely in a gastroenterology practice: an evidence-based review of indications and pitfalls in interpretationAm J Gastroenterol2002971113112612014715
- MattarRMonteiroMSVillaresCASantosAFCarrilhoFJSingle nucleotide polymorphism C/T-13910, located upstream of the lactase gene, associated with adult-type hypolactasia: validation for clinical practiceClin Biochem20084162863018237552
- BüningCGenschelJJurgaJIntroducing genetic testing for adult-type hypolactasiaDigestion20057124525016024930
- HogenauerCHammerHFMellitzerKRennerWKrejsGJToplakHEvaluation of a new DNA test compared with the lactose hydrogen breath test for the diagnosis of lactase non-persistenceEur J Gastroenterol Hepatol20051737137615716664
- BodlajGStöcherMHufnaglPGenotyping of the lactase-phlorizin hydrolase –13910 polymorphism by lightCycler PCR and implications for the diagnosis of lactose intoleranceClin Chem20065214815116391332
- SzilagyiAMalolepszyPHamardEComparison of a real-time polymerase chain reaction assay for lactase genetic polymorphism with standard indirect tests for lactose maldigestionClin Gastroenterol Hepatol2007519219616876487
- TagCGSchifflersMCMohnenMGressnerAMWeiskirchenRA novel proximal _13914G>A base replacement in the vicinity of the common-13910T/C lactase gene variation results in an atypical light cycler melting curve in testing with the MutaREAL lactase testClin Chem20075314614817202502
- TorbjörnKOlssonLASimultaneous genotyping of the three lactose tolerance linked polymorphisms LCT-13907C>G, LCT-13910C>T and LCT-13915T>G with Pyrosequencing technologyClin Chem Lab Med200846808418034639
- LomerMCParkesGCSandersonJDReview article: lactose intolerance in clinical practice – myths and realitiesAliment Pharmacol Ther2008279310317956597
- MatthewsSBWaudJPRobertsAGCampbellAKSystemic lactose intolerance: a new perspective on an old problemPostgrad Med J20058116717315749792
- HarringtonLKMayberryJFA re-appraisal of lactose intoleranceInt J Clin Pract2008621541154618822024
- CampbellAKMatthewsSBVasselNBacterial metabolic ‘ toxins’: a new mechanism for lactose and food intolerance, and irritable bowel syndromeToxicology201027826827620851732
- MinennaMFPalieriAPanellaCIerardiEGastro-oesophageal reflux disease and lactose malabsorption: casual comorbidity or neglected association?Dig Liver Dis20063843743816574518
- SuchyFJBrannonPMCarpenterTONational Institutes of Health Consensus Development Conference: lactose intolerance and healthAnn Intern Med201015279279620404261
- ShaukatALevittMDTaylorBCSystematic review: effective management strategies for lactose intoleranceAnn Intern Med201015279780320404262
- JellemaPSchellevisFGvan der WindtDAKneepkensCMvan der HorstHELactose malabsorption and intolerance: a systematic review on the diagnostic value of gastrointestinal symptoms and self-reported milk intoleranceQ J Med2010103555572
- SavaianoDABousheyCJMcCabeGPLactose intolerance symptoms assessed by meta-analysis: a grain of truth that leads to exaggerationJ Nutr20061361107111316549489
- CasellasFVarelamEApariciACasausMRodríguezPDevelopment, validation, and applicability of a symptoms questionnaire for lactose malabsorption screeningDig Dis Sci2009541059106518716868
- CasellasFApariciACasausMRodríguezPMalageladaJRSubjective perception of lactose intolerance does not always indicate lactose malabsorptionClin Gastroenterol Hepatol2010858158620385250
- ErminiaRIlariaBTizianaMHRQoL questionnaire evaluation in lactose intolerant patients with adverse reactions to foodsIntern Emerg Med5262011 [Epub ahead of print.]
- CollocaLMillerFGThe nocebo effect and its relevance for clinical practicePsychosom Med20117359860321862825
- VerniaPDi CamilloMFogliettaTAvalloneVEDe CarolisADiagnosis of lactose intolerance and the “nocebo” effect: the role of negative expectationsDig Liver Dis20104261661920227928
- Di StefanoMCorazzaGRThe patient’s expectation during H2 breath testing: don’t underestimate the reader’s expectationDig Liver Dis2011438620620123
- NovilloAPeraltaDDimaGBesassoHSoiferLFrequency of bacterial overgrowth in patients with clinical lactose intoleranceActa Gastroenterol Latinoam20104022122421053480
- SuarezFLLevittMDAn understanding of excessive intestinal gasCurr Gastroenterol Rep2000241341910998670
- EnattahNVälimäkiVVVälimäkimMJLöyttyniemiESahiTJärveläIMolecularly defined lactose malabsorption, peak bone mass and bone turnover rate in young Finnish menCalcif Tissue Int20047548849315365657
- Obermayer-PietschBMGugatschkaMReitterSAdult-type hypolactasia and calcium availability: decreased calcium intake or impaired calcium absorption?Osteoporos Int20071844545117103297
- LovelaceHYBarrSIDiagnosis, symptoms, and calcium intakes of individuals with self-reported lactose intoleranceJ Am Coll Nutr200524515715670985
- NicklasTAQuHHughesSOSelf-perceived lactose intolerance results in lower intakes of calcium and dairy foods and is associated with hypertension and diabetes in adultsAm J Clin Nutr20119419119821525197
- BácsiKKósaJPLazáryALCT 13910 C/T polymorphism, serum calcium, and bone mineral density in postmenopausal womenOsteoporos Int20092063964518704543
- EnattahNSSulkavaRHalonenPKontulaKJärveläIGenetic variant of lactase-persistent C/T-13910 is associated with bone fractures in very old ageJ Am Geriatr Soc200553798215667380
- EnattahNPekkarinenTVälimäkiMJLöyttyniemiEJärveläIGenetically defined adult-type hypolactasia and self reported lactose intolerance as risk factors of osteoporosis in Finnish postmenopausal womenEur J Clin Nutr2005591105111116015262
- GugatschkaMHoellerAFahrleitner-PammerACalcium supply, bone mineral density and genetically defined lactose maldigestion in a cohort of elderly menJ Endocrinol Invest200730465117318022
- LaaksonenMMImpivaaraOSievänenHAssociations of genetic lactase non-persistance and sex with bone loss in young adulthoodBone2009441003100919168163
- TolonenSLaaksonenMMikkiläVLactase gene c/t(-13910) polymorphism, calcium intake, and pQCT bone traits in Finnish adultsCalcif Tissue Int20118815316121136048
- MontaltoMCuriglianoVSantoroLManagement and treatment of lactose malabsorptionWorld J Gastroenterol20061218719116482616
- MontaltoMGalloASantoroLLow-dose lactose in drugs neither increases breath hydrogen excretion nor causes gastrointestinal symptomsAliment Pharmacol Ther2008281003101218657134