Abstract
Human islet amyloid polypeptide (h-IAPP) is a peptide hormone that is synthesized and cosecreted with insulin from insulin-secreting pancreatic β-cells. Recently, h-IAPP was proposed to be the main component responsible for the cytotoxic pancreatic amyloid deposits in patients with type 2 diabetes mellitus (T2DM). Since the causative factors of IAPP (or amylin) oligomer aggregation are not fully understood, this review will discuss the various forms of h-IAPP aggregation. Not all forms of IAPP aggregates trigger the destruction of β-cell function and loss of β-cell mass; however, toxic oligomers do trigger these events. Once these toxic oligomers form under abnormal metabolic conditions in T2DM, they can lead to cell disruption by inducing cell membrane destabilization. In this review, the various factors that have been shown to induce toxic IAPP oligomer formation will be presented, as well as the potential mechanism of oligomer and fibril formation from pro-IAPPs. Initially, pro-IAPPs undergo enzymatic reactions to produce the IAPP monomers, which can then develop into oligomers and fibrils. By this mechanism, toxic oligomers could be generated by diverse pathway components. Thus, the interconnections between factors that influence amyloid aggregation (eg, absence of PC2 enzyme, deamidation, reduction of disulfide bonds, environmental factors in the cell, genetic mutations, copper metal ions, and heparin) will be presented. Hence, this review will aid in understanding the fundamental causative factors contributing to IAPP oligomer formation and support studies for investigating novel T2DM therapeutic approaches, such as the development of inhibitory agents for preventing oligomerization at the early stages of diabetic pathology.
Introduction
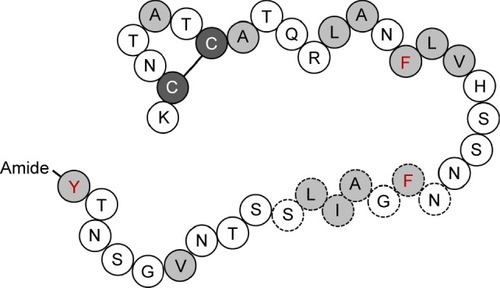
Type 2 diabetes mellitus (T2DM) is caused by the reduction of β-cell mass and accumulation of islet amyloid polypeptide (IAPP, or amylin) in the pancreatic islets, leading to insulin resistance.Citation1 Islet amyloid deposition could affect and occur in up to 80% of diabetic patients.Citation2 IAPP, a 4 kDa peptide hormone composed of 37 amino acids (), is synthesized and cosecreted with insulin from pancreatic β-cells. As a glucomodulatory hormone, IAPP is responsible for slowing down gastric emptying and regulating glucose levels. It inhibits the effects of insulin as well as arginine-stimulated glucagon release by α-cells.Citation3,Citation4 In addition, IAPP is responsible for other physiological functions such as sleep and appetite regulation via the gut–brain axis, and it functions as a growth factor to maintain β-cell mass.Citation5 The incidence of T2DM is considered to increase with age because of the reduced proliferative capacity and increased apoptosis rate of β-cells, which can also lead to higher rates of IAPP aggregation.Citation6 The IAPP monomer appears to show normal biological activity in healthy β-cells. Moreover, oligomers can regularly form and degrade in a normal cell system. However, unlike normal monomers and oligomers, toxic oligomers can cause membrane-perforating toxicity, which is a decisive factor that is indicative of the cell cytotoxicity in diabetes models. IAPP oligomers can be generated via the amyloid fibril off-pathway in the fibril assembly process. However, the exact structure and role of toxic IAPP oligomers have been difficult to establish thus far. The toxic amyloid-β oligomer has a cross-β structure,Citation7 and it is considered that the IAPP oligomer that is ultimately cytotoxic to pancreatic β-cells would share similar structural elements.Citation8 On the bright side, rifampicin has been reported to prevent the formation of human-IAPP (h-IAPP) fibrils. However, rifampicin did not seem to be able to control the production of toxic h-IAPP oligomers from the off-pathway and could therefore not prevent its cytotoxic effects.Citation9 Currently, the causative factors of the conversion of soluble IAPP into insoluble fibrils are unknown. However, it is known that these factors lead to the destruction of β-cell function to the point where they cannot properly respond to insulin.Citation2
Figure 1 Schematic diagram of the primary sequence of IAPP.
Abbreviation: IAPP, islet amyloid polypeptide.
The final stage of cell disruption involves membrane destabilization and disruption. Hence, it is important to understand how amyloidogenic peptides cause cell membrane disruption. The “carpet” and “barrel-stave” models are used to represent the mechanisms of cell membrane disruption. In the carpet model, monomers interrupt the membrane through nonspecific binding, by dissolving into the membrane in a detergent-like manner and exceeding the concentration threshold value of the membrane.Citation10 In the barrel-stave model, after assembling on the membrane surface, peptides insert themselves into the membrane, forming channels or pores.Citation11 Thus, membrane disruption from barrier deformation is observed in these two models. In particular, both models reveal how high cytotoxicity can result from cell disruption induced by a toxic IAPP oligomer. Further in-depth studies with these models are needed to characterize the pore generation induced by h-IAPP, identify the cytotoxic mechanisms, screen for the causative factors that initiate toxic oligomer formation, and ultimately identify how these causative factors can be inhibited.
Factors that cause oligomer aggregation
Environmental condition of the cell
Amyloidogenic protein aggregation can be induced by several factors within the cellular environment. IAPP or amylin, as a potential amyloidogenic protein, was shown to interact with the lipid membrane in one of the pathogenic cytometabolic pathways.Citation12 IAPP fibrillization was found to play a role in the cytotoxicity in vitro by disrupting the negatively charged phospholipid membrane.Citation13 In the same context, it was suggested that a high-fat diet could increase the lipid level, thereby increasing the possibility of oligomer penetration or membrane disruption through increased oligomer–lipid binding. As another possibility, the hydrophobic region of IAPP could be exposed in protein misfolding, which would implicate it in the fibrillation process.Citation14 Changes in pH were reported to affect the conformation and aggregation patterns of IAPP, where the alkaline pH of 8.8 was suggested to be more favorable for aggregate formation than the acidic condition of pH 4.0.Citation15,Citation16 Likewise, chemical modification, salt concentrations, natural ligands, racemization, isomerization, deamidation, oxidation, lipid oxidation, and glycation are factors that could affect the stability of IAPP.Citation2
IAPP sequence
Genetic mutations of IAPP
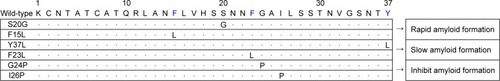
According to a previous study, a naturally occurring missense mutation (S20G) of the human IAPP gene is associated with early onset or more severe types of T2DM.Citation17 The S20G mutation results in increased hydrophobicity and amyloidogenic characteristics of IAPP, which could increase its fibrillogenic potential.Citation18 In addition, S20G IAPP showed a nearly twofold increased rate of the formation of amyloid fibrils, resulting in more than threefold greater aggregation and consequently higher cytotoxicity than the wild-type protein.Citation18 F15, an aromatic residue in IAPP that conserves its hydrophobicity, has been suggested to play a significant role in the amyloid biosynthesis pathway.Citation19 In an in silico study, an F15L mutation generated from a single-point mutation, which altered the α-helix and β-sheet propensities of the protein, resulted in rapid amyloid formation.Citation20 In another in silico study, the Y37L and F23L IAPP mutations resulted in decreased rates of amyloid fibril formation.Citation20 The replacement of tyrosine (Y) with leucine (L) resulted in greater flexibility of the C-terminus, with loss of the steric zipper interactions, and the F23L mutation, in which phenylalanine (F) was replaced with leucine (L), showed slower amyloid formation.Citation21 Over-all, the rate of aggregate formation was reduced significantly in these two mutations compared to other mutants and the wild-type protein.Citation21 Interestingly, the single point mutants G24P and I26P also showed potential for inhibiting amyloid aggregation.Citation22–Citation24 A list of the genetic mutations of IAPP is shown in . Mutations by displacement of one amino acid residue could greatly affect the rate and property of amyloid formation. These aforementioned studies described suggest a close correlation for balancing the reversal or the rate of fibril formation in reducing or increasing cytotoxicity.
Figure 2 List of genetic mutations of IAPP determined from in silico studies, and naturally occurring (eg, S20G) mutations.
Abbreviation: IAPP, islet amyloid polypeptide.
Comparison of IAPP sequences among species and establishment of transgenic rodent models
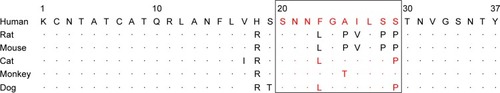
IAPP residues at the N-terminus and C-terminus are conserved in mammals, whereas the amyloidogenic core region is species specific (). Sequence homology has been found between primate and human IAPPs, and these peptides form islet amyloids that lead to the development of T2DM. In contrast, the presence of three proline residues in rat and mouse IAPPs renders the protein water soluble, thereby giving it nonamyloidogenic properties by providing a water-soluble environment; as a consequence, T2DM does not typically occur in rodents.Citation25,Citation26 Since the cytotoxicity of IAPP seems to be dependent on its propensity for oligomer formation, the prevention of fibril formation by the proline residues of IAPP 20–29 in the rat and mouse is very likely to be the cause of their reduced IAPP cytotoxicity, which is hardly detected in these species.Citation2
Figure 3 Alignment of IAPP amino acid sequences from different species.
Abbreviation: IAPP, islet amyloid polypeptide.
Since pancreatic β-cell apoptosis and the induction of diabetes have been confirmed in h-IAPP transgenic rat and mouse models, it is a viable hypothesis that amyloids are associated with the induction and progression of diabetes.Citation27 Indeed, an h-IAPP transgenic model formed toxic IAPP oligomers that eventually generated endoplasmic reticulum stress-induced apoptosis and T2DM characteristics such as hyperglycemia, impaired insulin secretion, insulin resistance, and hyperglucagonemia.Citation28,Citation29
IAPP processing
IAPP is encoded by an 89-residue coding sequence that produces the 67-amino acid pro-IAPP residue. Mature IAPP is then generated by the following process. First, pro-IAPP is formed after cleavage of the 22-amino acid signal peptide before it undergoes proteolysis and posttranslational modification. Sixteen amino acids at the C-terminus are removed by proprotein convertase 1/3 (PC1/3), followed by truncation of eleven amino acids at the N-terminus by proprotein convertase 2 (PC2).Citation30 Then, the C-terminus carboxypeptidase E removes the terminal lysine and arginine residues,Citation31 and peptidylglycine α-amidating monooxygenase (PAM) adds an amine group to the C-terminus. Finally, a disulfide bond is generated between the cysteine 2 and cysteine 7 residues to produce the mature IAPP protein made up of 37 amino acids (IAPP sequence: KCNTATCATQRLANFLVHSSNNFGAILSSTNVGSNTY).Citation32 Both an intact intramolecular disulfide bond and C-terminal amidation are required for IAPP to have normal biological activity. Therefore, the factors that prevent the formation of the disulfide bond and/or cause deamidation, as well as the absence of PC2, contribute to the aggregation of IAPP.
Absence of PC2
Impaired processing of pro-IAPP can affect amyloid formation in T2DM. The first enzyme in the pro-IAPP processing pathway is PC1/3, which plays a crucial role in removing the amino acids of the C-terminus. In the absence of PC1/3, PC2 takes over at both the N-terminal and C-terminal cleavage sites as part of normal posttranslational modification.Citation33 In contrast, because PC2 usually only functions after PC1/3 in the pro-IAPP processing pathway, its absence creates a block at the level of the pro-IAPP convertase intermediate (NH2-terminally extended pro-IAPP). This intermediate acts as a critical initiating factor to induce intracellular and extracellular amyloid formation, and finally induces β-cell death.Citation34,Citation35
Deamidation
Deamidation is an important factor in amyloid formation and affects protein structure, folding, stability, and aggregation.Citation36,Citation37 Deamidation of the side chain at Asn and Gln is a spontaneous reaction and a nonenzymatic posttranslational modification.Citation38 There are six Asn residues and one Gln residue in the IAPP sequence that can be subject to deamidation effects. In addition, for IAPP to properly perform its biological function, it should have some form of C-terminal amidation. Thus, once the C-terminal amide group is exposed to deamidation, the protein structure will be affected. Deamidation may influence the protein structure and stability by adding a negative charge, and thus change the amyloid formation kinetics such that unmodified IAPP (deamidated form) accelerates amyloid formation, creating the seeds for aggregation.Citation39 In other words, if an impurity is produced by deamidation, it induces amyloid formation, thereby affecting the purified peptide’s secondary structure and aggregation behavior.Citation40
Reduction of disulfide bond formation
In one study, the intramolecular disulfide bond of C2–C7 was shown to be the critical site for the formation of IAPP fibrils; however, the role of the intrinsic disulfide bond in the IAPP monomer structure as well as the specific mechanism of aggregation remain unclear.Citation41 The N-loop that is generated by the disulfide bond (C2–C7) of IAPP is highly conserved in the rigid structure.Citation42,Citation43 Although the N-loop is not involved in direct molecular interactions, it nevertheless affects the IAPP aggregation kinetics indirectly, since N-loop removal was found to change the mass/length distribution and kinetics of the h-IAPP fiber.Citation44,Citation45 The absence of the disulfide bond decreases the extent of the helix at the N-terminal region, but favors random coiling and β-sheet formation, which can affect the protein kinetics such as reducing the rate of IAPP aggregation by secondary nucleation.Citation41 In fact, early oligomerization is connected to helix formation. The S–S bridge, which stabilizes the N-terminal helix of h-IAPP, collapses the protein into an amorphous aggregate with less β-sheets. These factors were shown to facilitate the initialization of h-IAPP aggregation, encoded at the monomeric level.Citation41,Citation46,Citation47 Likewise, the disulfide bond determines the morphology of the fibril (eg, stabilizing the amyloid fibril in the folded state), and also plays a role in limitation by topologically restraining the polypeptide during amyloid fibril arrangement.Citation48 Therefore, the disulfide bond may ultimately reduce the toxicity of the amyloid fibril.Citation49
Copper(II) ion effect
Copper ions have been associated with many diseases.Citation50 Furthermore, calcium and zinc ions were shown to be involved in maintaining the native structure of IAPP, by inhibiting amyloid aggregation.Citation51,Citation52 Although the precise impact of copper ions on IAPP is still a matter of debate,Citation53 copper metabolism is thought to be associated with the pathological mechanism of amyloidosis in diabetes.Citation54 In one previous study, copper inhibited IAPP fibrillization,Citation52 whereas in another study, copper contributed to h-IAPP aggregation and cytotoxic oligomer formation.Citation55 Copper can possibly interact with the C2, C7, H18, and Y37 regions in the IAPP sequence.Citation55 These copper–IAPP complexes have metallopeptide complex structures with low aggregation potentialCitation56 and may instead produce granular oligomers, which are the main cause of increased copper-mediated h-IAPP cytotoxicity. Granular oligomers are expected to show the characteristics of toxic oligomers, inducing membrane destabilization and, ultimately, cell apoptosis.Citation55,Citation57,Citation58 In addition, copper-promoted reactive oxygen species (ROS) generation, such as H2O2, and mitochondrial disruption affect the degeneration of islet cells, leading to the production of caspase-3 and poly (ADP-ribose) polymerase (PARP), which in turn promote apoptosis.Citation59–Citation61 Furthermore, noncomplexed copper ions (ie, free copper ions) also show toxicity and can easily turn into reactive copper ions that generate cell-damaging ROS.Citation56 Therefore, treatment with a copper-chelating agent has a beneficial effect against the pathogenesis of T2DM and diabetes complications, by lowering the copper levels, and hence the ROS levels.Citation62
Heparin
The heparin-binding property of amyloid β-protein is dependent on the state of aggregation of this amyloidogenic protein since heparin does not interact with nonfibrillar forms.Citation63 A similar heparin study was conducted with IAPP. Heparin enhanced IAPP fibrillization, and the binding property was found to be dependent on the length of the heparin polysaccharide fragment and the aggregation state. The negatively charged heparin helix essentially binds with the positively charged N-terminal cross β-sheet of the IAPP fibril.Citation64 Short heparin fragments of 2–8 saccharides showed a better protective effect against cell cytotoxicity than fragments of 20 saccharide or longer, which exhibited no protective function.Citation65
Conclusion
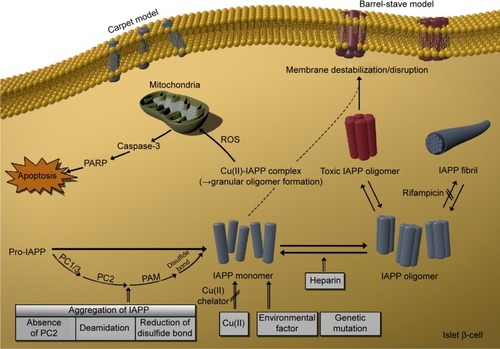
A wide range of diabetes-specific biomarkers has been identified. However, it appears that it is the unusual protein properties induced by such factors and the effect of protein aggregation on β-cell apoptosis that ultimately induce the development of diabetes. In particular, IAPP oligomers generated from IAPP monomers lead to β-cell-specific toxicity, ultimately causing diabetes. This review presented the known factors that appear to trigger IAPP aggregation. demonstrates how pro-IAPP decomposes into the IAPP monomer through the posttranslational process, and how these IAPP monomers can create and organize into toxic IAPP oligomers in β-cells. As illustrated in , the mechanistic factors that influence IAPP aggregation include the absence of PC2 enzyme, deamidation, reduction of disulfide bonds, environmental factors in the cell, genetic mutations, copper metal ions, and heparin. Other factors can also affect amyloid aggregation. Further research on these factors could ultimately aid in the inhibition of amyloid aggregation, which would be a breakthrough for the treatment of diabetes. To date, lack of knowledge about the toxic IAPP oligomer, the ultimate causative factors for reduction of β-cell mass, and the cause of cell death has hindered the ability in treating diabetes and limiting its progression, even in the prediabetic stage, and for selecting or developing novel therapeutic agents. Therefore, determining the mechanism by which the formation of toxic IAPP oligomers leads to T2DM should be an important research goal with the aim of elucidating the pathogenesis of this disorder and establishing new treatments.
Figure 4 Causative factors of toxic oligomer formation.
Abbreviations: IAPP, islet amyloid polypeptide; PARP, poly (ADP-ribose) polymerase; ROS, reactive oxygen species; PC1/3, proprotein convertase 1/3; PC2, proprotein convertase 2; PAM, peptidylglycine α-amidating monooxygenase; Cu(II), copper (II) ion.
Acknowledgments
This research was supported by the Industrial Core Technology Development Program (grant number 10049051, Development of bench-top automatic immunoassay system with intelligent quality control features for screening cancer or chronic diseases in local clinical setting) funded by the Ministry of Trade, Industry and Energy (MI, South Korea) and and the Korea Health Technology R&D Project (HI14C3331) through the Korea Health Industry Development Institute (KHIDI), Korea Ministry of Health & Welfare.
Disclosure
The authors report no conflicts of interest in this work.
References
- KlöppelGLöhrMHabichKOberholzerMHeitzPIslet pathology and the pathogenesis of type 1 and type 2 diabetes mellitus revisitedPathol Immunopathol Res198542110125
- ClarkANilssonMIslet amyloid: a complication of islet dysfunction or an aetiological factor in Type 2 diabetes?Diabetologia200447215716914722650
- NguyenPTAndrakaNDe CarufelCABourgaultSMechanistic contributions of biological cofactors in islet amyloid polypeptide amyloidogenesisJ Diabetes Res2015
- YoungAGedulinBVineWPercyARinkTGastric emptying is accelerated in diabetic BB rats and is slowed by subcutaneous injections of amylinDiabetologia19953866426487672483
- WookeyPJLutzTAAndrikopoulosSAmylin in the periphery II: an updated mini-reviewSci World J2006616421655
- GunasekaranUGannonMType 2 diabetes and the aging pancreatic beta cellAging (Albany NY)20113656521765202
- StroudJCLiuCTengPKEisenbergDToxic fibrillar oligomers of amyloid-β have cross-β structureProc Natl Acad Sci2012109207717772222547798
- WiltziusJJSieversSASawayaMRAtomic structure of the cross-β spine of islet amyloid polypeptide (amylin)Protein Sci20081791467147418556473
- MeierJJKayedRLinC-YInhibition of human IAPP fibril formation does not prevent β-cell death: evidence for distinct actions of oligomers and fibrils of human IAPPAm J Physiol Endocrinol Metab20062916E1317E132416849627
- PounyYRapaportDMorANicolasPShaiYInteraction of antimicrobial dermaseptin and its fluorescently labeled analogs with phospholipid membranesBiochemistry1992314912416124231463728
- OjciusDMYoungJDECytolytic pore-forming proteins and peptides: is there a common structural motif?Trends Biochem Sci1991162252291654003
- JayasingheSALangenRLipid membranes modulate the structure of islet amyloid polypeptideBiochemistry20054436121131211916142909
- DomanovYAKinnunenPKIslet amyloid polypeptide forms rigid lipid–protein amyloid fibrils on supported phospholipid bilayersJ Mol Biol20083761425418155730
- AhmadEAhmadASinghSArshadMKhanAHKhanRHA mechanistic approach for islet amyloid polypeptide aggregation to develop anti-amyloidogenic agents for type-2 diabetesBiochimie201193579380521215287
- AbediniARaleighDPThe role of His-18 in amyloid formation by human islet amyloid polypeptideBiochemistry20054449162841629116331989
- JhaSSnellJMShefticSRpH dependence of amylin fibrillizationBiochemistry201453230031024377660
- SakagashiraSSankeTHanabusaTMissense mutation of amylin gene (S20G) in Japanese NIDDM patientsDiabetes1996459127912818772735
- SakagashiraSHiddingaHJTateishiKS20G mutant amylin exhibits increased in vitro amyloidogenicity and increased intracellular cytotoxicity compared to wild-type amylinAm J Pathol200015762101210911106582
- MarekPAbediniASongBAromatic interactions are not required for amyloid fibril formation by islet amyloid polypeptide but do influence the rate of fibril formation and fibril morphologyBiochemistry200746113255326117311418
- TuL-HRaleighDPRole of aromatic interactions in amyloid formation by islet amyloid polypeptideBiochemistry201352233334223256729
- BernhardtNABerhanuWMHansmannUHMutations and seeding of amylin fibril-like oligomersJ Phys Chem B201311750160761608524294935
- AbediniAMengFRaleighDPA single-point mutation converts the highly amyloidogenic human islet amyloid polypeptide into a potent fibrillization inhibitorJ Am Chem Soc200712937113001130117722920
- MengFRaleighDPAbediniACombination of kinetically selected inhibitors in trans leads to highly effective inhibition of amyloid formationJ Am Chem Soc201013241143401434220873820
- MillerCZerzeGlHMittalJMolecular simulations indicate marked differences in the structure of amylin mutants, correlated with known aggregation propensityJ Phys Chem B201311750160661607524245879
- HaatajaLGurloTHuangCJButlerPCIslet amyloid in type 2 diabetes, and the toxic oligomer hypothesisEndocr Rev200829330331618314421
- WestermarkPEngströmUJohnsonKHWestermarkGTBetsholtzCIslet amyloid polypeptide: pinpointing amino acid residues linked to amyloid fibril formationProc Natl Acad Sci19908713503650402195544
- ButlerAEJangJGurloTCartyMDSoellerWCButlerPCDiabetes due to a progressive defect in β-cell mass in rats transgenic for human islet amyloid polypeptide (HIP rat) a new model for type 2 diabetesDiabetes20045361509151615161755
- MatveyenkoAVButlerPCβ-Cell deficit due to increased apoptosis in the human islet amyloid polypeptide transgenic (HIP) rat recapitulates the metabolic defects present in type 2 diabetesDiabetes20065572106211416804082
- HuangC-JLinC-YHaatajaLHigh expression rates of human islet amyloid polypeptide induce endoplasmic reticulum stress–mediated β-cell apoptosis, a characteristic of humans with type 2 but not type 1 diabetesDiabetes20075682016202717475933
- SankeTBellGSampleCRubensteinASteinerDAn islet amyloid peptide is derived from an 89-amino acid precursor by proteolytic processingJ Biol Chem19882633317243172463053705
- MarzbanLSoukhatchevaGVerchereCBRole of carboxypeptidase E in processing of pro-islet amyloid polypeptide in β-cellsEndocrinology200514641808181715618358
- RobertsALeightonBToddJMolecular and functional characterization of amylin, a peptide associated with type 2 diabetes mellitusProc Natl Acad Sci19898624966296662690069
- MarzbanLTrigo-GonzalezGZhuXRole of β-cell prohormone convertase (PC) 1/3 in processing of pro-islet amyloid polypeptideDiabetes200453114114814693708
- MarzbanLRhodesCJSteinerDFHaatajaLHalbanPAVerchereCBImpaired NH2-terminal processing of human proislet amyloid polypeptide by the prohormone convertase PC2 leads to amyloid formation and cell deathDiabetes20065582192220116873681
- WangJXuJFinnertyJFurutaMSteinerDFVerchereCBThe prohormone convertase enzyme 2 (PC2) is essential for processing pro-islet amyloid polypeptide at the NH2-terminal cleavage siteDiabetes200150353453911246872
- TakataTOxfordJTDemelerBLampiKJDeamidation destabilizes and triggers aggregation of a lens protein, βA3-crystallinProtein Sci20081791565157518567786
- NilssonMRDobsonCMChemical modification of insulin in amyloid fibrilsProtein Sci200312112637264114573875
- BischoffRKolbeHVDeamidation of asparagine and glutamine residues in proteins and peptides: structural determinants and analytical methodologyJ Chromatogr B Biomed Sci Appl19946622261278
- DunkelbergerEBBuchananLEMarekPCaoPRaleighDPZanniMTDeamidation accelerates amyloid formation and alters amylin fiber structureJ Am Chem Soc201213430126581266722734583
- NilssonMRDriscollMRaleighDPLow levels of asparagine deamidation can have a dramatic effect on aggregation of amyloidogenic peptides: implications for the study of amyloid formationProtein Sci200211234234911790844
- LaghaeiRMousseauNWeiGEffect of the disulfide bond on the monomeric structure of human amylin studied by combined Hamiltonian and temperature replica exchange molecular dynamics simulationsJ Phys Chem B2010114207071707720429571
- VaianaSMBestRBYauW-MEatonWAHofrichterJEvidence for a partially structured state of the amylin monomerBiophys J200997112948295719948124
- CopeSMShindeSBestRBGiovannaGVaianaSMCyclic N terminal fragment of amylin forms non amyloid fibers: implications for intra-and inter-molecular interactions in amylinBiophys J20131042389a390a
- GoldsburyCGoldieKPellaudJAmyloid fibril formation from full-length and fragments of amylinJ Struct Biol2000130235236210940238
- YonemotoITKroonGJDysonHJBalchWEKellyJWAmylin proprotein processing generates progressively more amyloidogenic peptides that initially sample the helical stateBiochemistry200847379900991018710262
- KooBWMirankerADContribution of the intrinsic disulfide to the assembly mechanism of islet amyloidProtein Sci200514123123915576552
- AbediniARaleighDPA role for helical intermediates in amyloid formation by natively unfolded polypeptides?Phys Biol20096101500519208933
- MossutoMFBolognesiBGuixerBDisulfide bonds reduce the toxicity of the amyloid fibrils formed by an extracellular proteinAngew Chem Int Ed2011503170487051
- MonsellierERamazzottiMTaddeiNChitiFAggregation propensity of the human proteomePLoS Comput Biol2008410e100019918927604
- Uriu-AdamsJYKeenCLCopper, oxidative stress, and human healthMol Aspects Med2005264–526829816112185
- BrenderJRHartmanKNangaRPRRole of zinc in human islet amyloid polypeptide aggregationJ Am Chem Soc2010132268973898320536124
- WardBWalkerKExleyCCopper (II) inhibits the formation of amylin amyloid in vitroJ Inorg Biochem2008102237137518022240
- SinopoliAMagrìAMilardiDThe role of copper (II) in the aggregation of human amylinMetallomics20146101841185225080969
- DrewSCMastersCLBarnhamKJAlanine-2 carbonyl is an oxygen ligand in Cu2+ coordination of Alzheimer’s disease amyloid-β peptide – relevance to N-terminally truncated formsJ Am Chem Soc2009131258760876119496610
- YuY-PLeiPHuJWuW-HZhaoY-FLiY-MCopper-induced cytotoxicity: reactive oxygen species or islet amyloid polypeptide oligomer formationChem Commun2010463769096911
- LeeECHaESinghSCopper (II)–human amylin complex protects pancreatic cells from amylin toxicityPhys Chem Chem Phys20131530125581257123793354
- MirzabekovTALinM-CKaganBLPore formation by the cytotoxic islet amyloid peptide amylinJ Biol Chem19962714198819928567648
- JansonJAshleyRHHarrisonDMcIntyreSButlerPCThe mechanism of islet amyloid polypeptide toxicity is membrane disruption by intermediate-sized toxic amyloid particlesDiabetes199948349149810078548
- MaLLiXWangYZhengWChenTCu (II) inhibits hIAPP fibrillation and promotes hIAPP-induced beta cell apoptosis through induction of ROS-mediated mitochondrial dysfunctionJ Inorg Biochem201414014315225108186
- ZorovDBJuhaszovaMSollottSJMitochondrial ROS-induced ROS release: an update and reviewBiochim Biophys Acta20061757550951716829228
- MasadAHayesLTabnerBJCopper-mediated formation of hydrogen peroxide from the amylin peptide: a novel mechanism for degeneration of islet cells in type-2 diabetes mellitus?FEBS Lett2007581183489349317617411
- TanakaAKanetoHMiyatsukaTRole of copper ion in the pathogenesis of type 2 diabetesEndocr J200956569970619461160
- WatsonDJLanderADSelkoeDJHeparin-binding properties of the amyloidogenic peptides Aβ and amylin dependence on aggregation state and inhibition by congo redJ Biol Chem19972725031617316249395501
- JhaSPatilSMGibsonJNelsonCEAlderNNAlexandrescuATMechanism of amylin fibrillization enhancement by heparinJ Biol Chem201128626228942290421555785
- AlexandrescuATJhaSPatilSMGibsonJAlderNNNelsonCEEffects of heparin on amylin fibrillizationBiophys J20121023243a
