Abstract
Introduction
Laryngeal cancer is the most common head and neck cancer worldwide. It is urgent to identify the mechanisms underlying laryngeal cancer pathogenesis. In the present study, we investigated the biological functions of Peripherin 2 (PRPH2) in laryngeal cancer and uncovered the molecular mechanism underlying this disease.
Methods
Laryngeal cancer tissues were used to analyze the expression of PRPH2. In vitro transwell matrigel invasion assay and annexin V anoikis assay in laryngeal cancer cells were conducted to investigate PRPH2 related biological functions. Quantitative real-time PCR and Western blotting were performed to investigate the expression and mechanism of PRPH2 in laryngeal cancer.
Results
We found that the expression of PRPH2 was significantly downregulated in laryngeal cancer tissues. Overexpression of PRPH2 suppressed the invasion and anoikis inhibition of laryngeal cancer cells. Furthermore, PRPH2 overexpression increased the phosphorylation of YAP and LATS1 and decreased the activities of Rho GTPases, while PRPH2 knockdown had opposite effects. Inhibitors of the Hippo pathway abrogated PRPH2 knockdown-induced laryngeal cancer cell invasion and anoikis inhibition.
Discussion
These results suggested that PRPH2 suppresses laryngeal cancer cell invasion and anoikis inhibition by activating Hippo signalling. PRPH2 may serve as a potential therapeutic target for laryngeal cancer in the future.
Introduction
Laryngeal cancer is the most common head and neck cancer worldwide. The increased incidence of laryngeal cancer has been reported in recent years.Citation1,Citation2 Until recently, conservative surgery and radiotherapy alone or in combination have been advised for the treatment of laryngeal cancer. Thus, there is an urgent need to identify the mechanisms underlying laryngeal cancer pathogenesis. Because invasion and metastasis are the main causes of mortality in patients with solid tumours, these factors have received much attention in recent studies.Citation3–Citation5 However, the current knowledge of the molecular mechanisms underlying invasion and metastasis in laryngeal cancer remains scarce.Citation6–Citation8
The Hippo signalling pathway plays an important role in regulating the invasion and metastasis of cancer cells.Citation9–Citation11 Hippo signalling includes the following kinase cascade. Macrophage Stimulating 1/2 (MST1/2) in coordination with the regulatory protein SAV1 activates Large Tumour Suppressor Kinase 1/2 (LATS1/2), which phosphorylates and inactivates Yes-Associated Protein (YAP)/Tafazzin (TAZ). Then, YAP/TAZ are restrained in the cytoplasm and lose their ability to transcriptionally activate related genes. Many biological factors such as contact inhibition, cell polarity/adhesion molecules, and cellular metabolic status can activate Hippo signalling.Citation12,Citation13
Peripherin 2 (PRPH2), also known as RDS, was initially identified as a cause of natural retinal degeneration in rats.Citation14 Retinal outer segment membrane protein 1 (ROM1) and PRPH2 form complexes through both covalent and non-covalent interactions that are important to the formation and maintenance of photoreceptor outer segments.Citation15–Citation18 PRPH2 is a transmembrane glycoprotein that is intrinsic to the curvature formation of each disc and flattened surface morphology. Deficiency of this protein results in cellular disorganization and cellular apoptosis activation via unknown mechanisms.Citation15,Citation19 Nevertheless, the link between PRPH2 and Hippo signalling has not been reported.
In the present study, we found that PRPH2 expression was significantly downregulated in laryngeal cancer tissues. The overexpression of PRPH2 could significantly suppress invasion and anoikis inhibition in laryngeal cancer cells. Furthermore, the effects of PRPH2 on the biological behaviours of laryngeal cancer cells were found to be dependent on Hippo signalling activation.
Methods and Materials
Cell Culture
Human laryngeal cancer cell lines, including Hep-2, TU212, TU686, M2e, M4e and AMC-HN-8, were purchased from the Cell Bank of the Chinese Academy of Sciences. Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% (v/v) foetal calf serum (FCS) and 1% antibiotics was used here. The cells were incubated at 37 °C in a humidified incubator under 5% CO2 conditions.
Clinical Samples
Human laryngeal cancer (16 cases) and corresponding normal tissues (12 cases), in which 12 cases were paired, were obtained from the Department of Ear-Nose-Throat, The First Hospital of Hebei Medical University. The human tissue microarray, containing 48 cases of laryngeal cancer samples, was purchased from Alenabio. All the patients were provided with written informed consent before enrollment and in compliance with the Declaration of Helsinki. The study was approved by the by the ethical review committee of the First Hospital of Hebei Medical University (directed by the World Health Organization Collaborating Centre for Research in Human Production).
Quantitative Real-Time PCR
Total RNA of cells or tissues was extracted by TRIzol (Takara) and reverse transcribed by the PrimeScript RT-PCR kit (Perfect Real Time). Quantitative real-time PCR analyses were performed with SYBR Premix Ex Taq (Takara) on a 7500 real-time PCR system (Applied Biosystems) at the recommended thermal cycling settings: 1 cycle at 95 °C for 30 seconds, followed by 40 cycles of 5 seconds at 95 °C and 31 seconds at 60 °C. The primer sequences used were: PRPH2, forward 5′-CAGAAGAAGCGGGTCAAGTTG-3′ and reverse 5′-GCTCCTCTTTCGGAGTTCAATC-3′; CTGF, forward 5′-TGGAGATTTTGGGAGTACGG-3′ and reverse 5′-CAGGCTAGAGAAGCAGAGCC-3′; ANKRD1, forward 5′-GTGTAGCACCAGATCCATCG-3′ and reverse 5′- CGGTGAGACTGAACCGCTAT-3′; CYR61, forward 5′-CCCGTTTTGGTAGATTCTGG-3′ and reverse 5′-GCTGGAATGCAACTTCGG-3′; and β-actin, forward 5′-CTCCATCCTGGCCTCGCTGT-3′ and reverse 5′-GCTGTCACCTTCACCGTTCC-3′.
Western Blotting and GTPase Pull-Down Assays
The cells were lysed in lysis buffer, and the proteins were separated by SDS-PAGE under reducing conditions. The membrane was blocked in phosphate-buffered saline/Tween-20 containing 5% BSA. Then, antibodies against PRPH2 (Abcam), phospho-YAP (Cell Signalling), YAP (Cell Signalling), phospho-LATS1 (Cell Signalling), LATS1/2 (Cell Signalling), GAPDH (Sigma) and species-specific secondary antibodies were used to incubate the membrane. The secondary antibodies were detected by the Odyssey imaging system (LI-COR). GTPase pull-down assays were performed according to standard procedures as previously described.Citation20
Lentivirus Production and Cell Transduction
The 293T cells were cotransfected with the pEZ-lv105 vector (GeneCopoeia) by using Lipofectamine 2000 (Invitrogen) for virus packaging. The viruses were harvested at 24, 48 and 72 hrs after transfection. After detecting the virus titres, 1×105 cells were infected with 1×106 recombinant lentivirus-transducing units by using 5 μg/mL of polybrene (Sigma).
siRNA Transfection
Small interfering RNAs duplexes targeting PRPH2 were produced by Genepharma. The transfection steps were performed according to the manufacturer’s protocols.
Hippo Inhibitors
Inhibitors of Hippo (Verteporfin and Peptide 17) were purchased from Selleck. Verteporfin (chemical formula: C41H42N4O8) and Peptide17 (XMU-MP-1, chemical formula: C93H144ClN23O21S2) are small molecular compounds that can inhibit the interaction of YAP-TEAD.Citation21,Citation22 To inhibit the interaction of YAP-TEAD, Verteporfin and Peptide 17 were added into M2e or AMC-HN-8 cells infected with siRNA of PRPH2 or NC. All cells were incubated at 37 °C.
Invasion Assay
Laryngeal cancer cells were detached and resuspended in serum-free DMEM medium. The cells were plated at 2×104 cells in 0.1 mL onto Matrigel (BD)-coated inserts (Millipore) seated on a 24-well plate. Then, 5% FBS DMEM medium was added to the bottom chamber. The cells were incubated at 37 °C. After 48 hrs, the filters were fixed and stained with 0.1% (w/v) Crystal Violet. Non-invading cells were removed, and invading cells were counted under a microscope at 400× magnification. Three grids per field were counted, and the experiments were repeated twice.
Anoikis Assays
For these assays, 5×105 Laryngeal cancer cells were cultured on poly-HEMA-treated 12-well plates for 48 hrs at 37 °C in a 5% CO2 atmosphere. After incubation, adherent cells were detached with 0.25% trypsin/0.01% EDTA in 1× PBS. Detached and suspended cells were harvested in complete DMEM medium and centrifuged at 1000 rpm/5 mins. The cells were washed with 1× PBS and stained with 100 μl binding buffer containing 1.75 μg/mL Annexin V and 1.75 μg/mL propidium iodide (PI). The cells were incubated at room temperature for 15 mins and analysed by flow cytometry (BD).
Statistical Analysis
Data were presented as the means ± standard error of the mean (SEM). Student’s t-test and one-way ANOVA were used for comparisons between groups. Values of P<0.05 were considered statistically significant.
Results
PRPH2 Is Significantly Downregulated in Laryngeal Cancer Tissues, and PRPH2 Expression Is Suppressed by Epigenetic Methylation
To investigate the expression level of PRPH2 in laryngeal cancer tissues, we collected tissues from 16 laryngeal cancer cases and 12 normal cases. Quantitative real-time PCR revealed that the expression of PRPH2 was significantly downregulated in laryngeal cancer tissues (). Among 12 paired laryngeal cancer and normal tissues, PRPH2 expression was also significantly downregulated in laryngeal cancer tissues (). Human laryngeal cancer tissue microarray (n=48) showed that the expression of PRPH2 was downregulated in 67.80% of laryngeal cancer tissues compared to normal tissues ().
Figure 1 The expression of PRPH2 is downregulated in laryngeal cancer tissues, and DNA methylation suppresses PRPH2 expression. (A) The mRNA expression of PRPH2 in 16 cases of laryngeal cancer (n=16) and 12 cases of normal tissues (n=12). **P<0.01, Laryngeal cancer vs. normal tissues. (B) The mRNA expression of PRPH2 in 12 paired laryngeal cancer and normal tissues. **P<0.01, Laryngeal cancer vs. normal tissues (n=12 in each group). (C) The expression of PRPH2 was downregulated in 67.80% laryngeal cancer tissues (n=48). (D–F) Relative mRNA expression of PRPH2 in TU686 (D), M4e (E) and Hep-2 (F) cells after treatment with DAC and TSA (repeat 3 times for each group). *P<0.05, TSA 600nM vs. vehicle in TU686 cells; **P<0.01, DAC vs. vehicle, DAC+TSA vs. vehicle in TU686, M4E and TU686 cells.
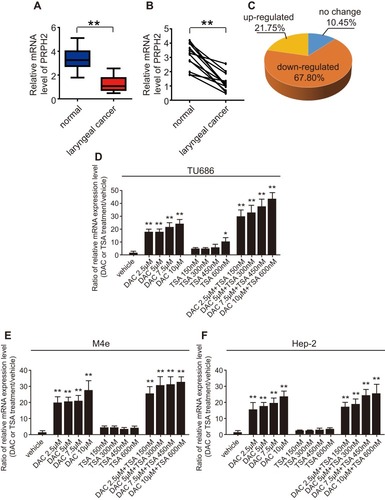
Furthermore, we investigated the reason for PRPH2 downregulation in laryngeal cancer. Notably, after treatment with 5-aza-20-deoxycytidine (DAC), a specific methyltransferase inhibitor, and trichostatin A (TSA), a histone deacetylase inhibitor, methylation was observed in all 3 tested laryngeal cancer cell lines (TU686, M4E and TU686 cells), and histone acetylation was observed in 1 cell line. These results suggested that epigenetic methylation is the main cause of the suppression of PRPH2 expression in laryngeal cancer (–).
Overexpression of PRPH2 Suppresses Invasion and Anoikis Inhibition in Laryngeal Cancer Cells
To further investigate the biological functions of PRPH2 in laryngeal cancer, we detected the expression level of PRPH2 in 6 laryngeal cancer cell lines. As shown in , the expression of PRPH2 in TU686 and M4e cells was obviously lower than that in other laryngeal cancer cell lines. We further established stable PRPH2-overexpressing TU686 and M4e cell lines by the transduction of lentivirus carrying the PRPH2 gene named Lenti-PRPH2. Western blot analyses revealed that PRPH2 was successfully overexpressed in both TU686 () and M4e () cells.
Figure 2 Overexpression of PRPH2 suppresses the invasion and anoikis inhibition of laryngeal cancer cells. (A) Expression of PRPH2 in laryngeal cancer cell lines, including Hep-2, TU212, TU686, M2e, M4e and AMC-HN-8 cells (repeat 3 times for each group). **P<0.01, TU686 or M4E vs. M2e or AMC-HN-8. (B and C) The protein expression levels of PRPH2 in TU686 (B) and M4E (C) cells, infected with Lenti-PRPH2 or Lenti-vector. Statistical analyses of PRPH2 expression in the two groups are shown below (repeat 3 times for each group). **P<0.01, Lenti-vector vs. Lenti-PRPH2. (D) Representative photos of invaded TU686 cells infected with Lenti-PRPH2 or Lenti-vector. Statistical analysis of invaded TU686 cells in the two groups is shown right (repeat 3 times for each group). **P<0.01, Lenti-vector vs. Lenti-PRPH2. (E) Flow cytometry analysis of anoikis of TU686 cells infected with Lenti-PRPH2 or Lenti-vector. Flow cytometry statistical analysis of apoptotic TU686 cells is shown right (repeat 3 times for each group). **P<0.01, Lenti-vector vs. Lenti-PRPH2. (F) Representative photos of invaded M4e cells infected with Lenti-PRPH2 or Lenti-vector. Statistical analysis of invaded M4e cells in the two groups is shown right (repeat 3 times for each group). **P<0.01, Lenti-vector vs. Lenti-PRPH2. (G) Flow cytometry analysis of anoikis of M4e cells infected with Lenti-PRPH2 or Lenti-vector. Flow cytometry statistical analysis of apoptotic M4e cells is shown right (repeat 3 times for each group). **P<0.01, Lenti-vector vs. Lenti-PRPH2.
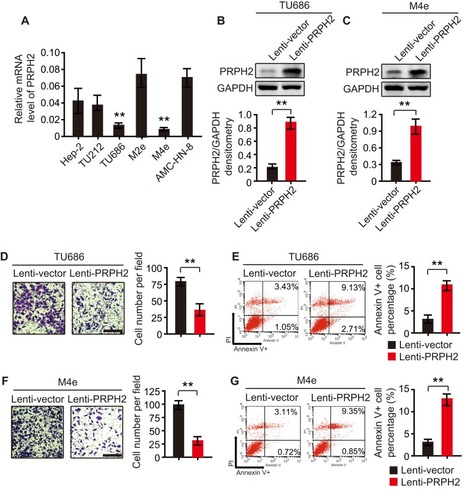
We first investigated the role of PRPH2 in the invasion of laryngeal cancer cells. By Transwell Matrigel invasion assay, we found that the overexpression of PRPH2 suppressed the invasion of TU686 and M4e cells ( and ) after 48 hrs. Furthermore, as shown by annexin V anoikis assay, we found that the overexpression of PRPH2 promoted anoikis in TU686 and M4e cells ( and ) after 48 hrs.
PRPH2 Overexpression Increases the Phosphorylation of YAP/LATS1 Signalling and Decreases Rho GTPase Activities
To investigate the underlying mechanism of the association of PRPH2 with laryngeal cancer, we performed GESA analysis and found that PRPH2 was closely related to the Hippo signalling pathway (). Western blot analyses revealed that the Hippo signalling pathway plays important roles in laryngeal cancer invasion and metastasis. Interestingly, the overexpression of PRPH2 in TU686 cells significantly increased the phosphorylation of YAP and LATS1 (). Then, PRPH2-overexpressing and control TU686 cells were serum starved for 24 h. As shown by pull-down assays, the activities of RhoA and Cdc42 were found to be significantly suppressed by PRPH2 overexpression ().
Figure 3 PRPH2 overexpression increases the phosphorylation of YAP/LATS1 and decreases Rho GTPases activities. (A) GESA analysis showed that PRPH2 was closely related with Hippo signalling pathway. (B) Western blot analysis of phospho-YAP, YAP, phospho-LATS1 and LATS1 in PRPH2 overexpressed and control TU686 cells (repeat 3 times for each group). Phospho-YAP/YAP and phospho-LATS1/LATS1 densitometry are shown on the right. (C) TU686 cells were serum starved for 24 h and the RhoA and Cdc42 activities were measured by pull-down assays in PRPH2 overexpressed and control cells (repeat 3 times for each group). Active-RhoA/total-RhoA and active-Cdc42/total-Cdc42 densitometry are shown on the right. (D) The mRNA levels of CTGF, ANKRD1 and CYR61 in PRPH2 overexpressed and control TU686 cells (repeat 3 times for each group). **P<0.01, Lenti-vector vs. Lenti-PRPH2.
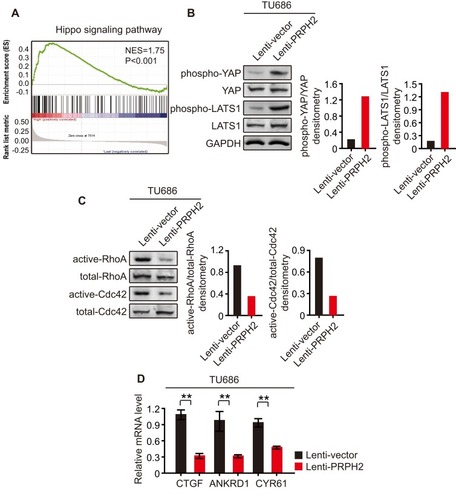
We also detected the mRNA levels of the following canonical target genes of YAP: Connective Tissue Growth Factor (CTGF), Ankyrin Repeat Domain 1 (ANKRD1), and Cysteine Rich Angiogenic Inducer 61 (CYR61). Additionally, CTGF, ANKRD1 and CYR61 mRNA levels were significantly suppressed in PRPH2-overexpressing TU686 cells (). These results demonstrated that the Hippo pathway was activated in PRPH2-overexpressing laryngeal cancer cells, which suppressed laryngeal cancer cell invasion and anoikis inhibition.
Knockdown of PRPH2 Promotes Invasion and Anoikis Inhibition in Laryngeal Cancer Cells, and These Effects Can Be Abrogated by Hippo Pathway Inhibitors
Furthermore, we knocked down PRPH2 in M2e and AMC-HN-8 cells, which express high levels of PRPH2, by using siRNAs (called si-PRPH2-1 and si-PRPH2-2). Western blot analyses revealed that PRPH2 was silenced in M2e () or AMC-HN-8 () cells. Transwell Matrigel invasion and annexin V anoikis assays showed that the knockdown of PRPH2 could promote invasion and anoikis inhibition in laryngeal cancer cells (–).
Figure 4 PRPH2 knockdown promotes the invasion and anoikis inhibition of laryngeal cancer cells, which can be abrogated by Hippo pathway inhibitors. (A and B) The protein expression level of PRPH2 in M2e (A) and AMC-HN-8 (B) cells, infected with siRNA of PRPH2 or negative control (NC). Statistical analyses of PRPH2 expression are shown below (repeat 3 times for each group). **P<0.01, NC vs. si-PRPH2-1 or si-PRPH2-2. (C and E) M2e and AMC-HN-8 cells were infected with siRNA of PRPH2 or NC, then treated with 50 nM Verteporfin (YAP-TEAD inhibitor) or 50 nM Peptide 17 (YAP-TEAD inhibitor), respectively. The invaded M2e (C) and AMC-HN-8 (E) cells were analysed after 48 hrs (repeat 3 times for each group). **P<0.01, NC vs. si-PRPH2-1 or si-PRPH2-2, si-PRPH2-1 vs. si-PRPH2-1+Veterporfin or si-PRPH2-1+Peptide17, si-PRPH2-2 vs. si-PRPH2-2+Veterporfin or si-PRPH2-2+Peptide17. (D and F) M2e and AMC-HN-8 cells were infected with siRNA of PRPH2 or NC, then treated with 50 nM Verteporfin or 50 nM Peptide 17, respectively. The apoptotic M2e (D) and AMC-HN-8 (F) cells were analysed after 48 hrs (repeat 3 times for each group). **P<0.01, NC vs. si-PRPH2-1 or si-PRPH2-2, si-PRPH2-1 vs. si-PRPH2-1+Veterporfin or si-PRPH2-1+Peptide17, si-PRPH2-2 vs. si-PRPH2-2+Veterporfin or si-PRPH2-2+Peptide17. (G) PRPH2 decreases Rho GTPases activities, and subsequently increases the phosphorylation of YAP/LATS1 and activates Hippo signalling, thereby suppressing laryngeal cancer cell invasion and anoikis inhibition.
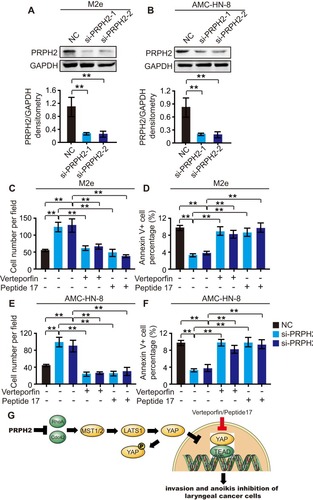
By the administration of verteporfin (an inhibitor of YAP-TEAD) and peptide 17 (YAP-TEAD inhibitor I, an inhibitor of YAP-TEAD), we further investigated the correlation between PRPH2 and Hippo signalling. Verteporfin and peptide 17 were added to PRPH2-silenced and control M2e and AMC-HN-8 cells. Verteporfin and peptide 17 abrogated PRPH2 knockdown-induced M2e and AMC-HN-8 cell invasion ( and ). Moreover, anoikis inhibition in M2e and AMC-HN-8 cells induced by PRPH2 knockdown was also abrogated by these inhibitors ( and ).
These results indicated that PRPH2 suppresses laryngeal cancer cell invasion and anoikis inhibition by activating Hippo signalling ().
Discussion
Few studies on PRPH2 in cancers, particularly those on the detailed biological functions and related mechanism of PRPH2 in laryngeal cancer, have been reported in recent years. In the present study, the precise roles of PRPH2 in laryngeal cancer were investigated for the first time. The downregulated expression of PRPH2 may lead to the development and progression of laryngeal cancer. The biological function experiments revealed that PRPH2 overexpression could suppress the invasion and anoikis inhibition of laryngeal cancer cells, which indicated that PRPH2 is involved in the invasion and anoikis inhibition of laryngeal cancer.
Invasion and metastasis are major concerns during the prognosis and progression of cancer. Hippo signalling is very important in the invasion and metastasis of cancer cells.Citation9–Citation11 YAP contributes to metastasis via multiple mechanisms. YAP interacts with TEAD and FOS in the nucleus to reprogram gene expression to induce epithelial-mesenchymal transition (EMT). YAP also antagonizes E-cadherin junction assembly by regulating actin cytoskeleton organization and contributes to EMT. Furthermore, YAP activation promotes stiffening of the extracellular matrix of cancer-associated fibroblasts (CAFs) to enhance YAP nuclear localization in cancer cells. Such interplay between cancer cells and CAFs might amplify the effects of YAP during tumorigenesis.Citation23–Citation26
In the present study, the overexpression of PRPH2 increased the phosphorylation of YAP and LATS1, restraining these proteins in the cytoplasm and inhibiting their transcriptional activation. Furthermore, these results were confirmed by the detection of Rho GTPase activities and canonical YAP target gene expression. Meanwhile, knockdown of PRPH2 lead to an opposite effect. By using inhibitors of the Hippo pathway, it was found that PRPH2 knockdown induced laryngeal cancer cell invasion, and anoikis inhibition could be abrogated by these inhibitors, indicating that PRPH2 suppressed laryngeal cancer cell invasion and anoikis inhibition by activating Hippo signalling pathway.
In conclusion, the results of the present study showed that PRPH2 plays an important role in laryngeal cancer cell invasion and anoikis inhibition. The overexpression of PRPH2 in laryngeal cancer cells suppresses Rho GTPase activities, activates Hippo signalling, and suppresses invasion and anoikis inhibition in laryngeal cancer cells. The use of PRPH2 may be a future therapeutic strategy for laryngeal cancer.
Disclosure
The authors report no conflicts of interest in this work.
Acknowledgements
This study was funded by Hebei Province Natural Science Foundation.
References
- Chu EA, Kim YJ. Laryngeal cancer: diagnosis and preoperative work-up. Otolaryngol Clin North Am. 2008;41:673–695. doi:10.1016/j4tc.2008.01.01618570953
- Fu Q, Liu P, Sun X, et al. Ribonucleic acid interference knockdown of IL-6 enhances the efficacy of cisplatin in laryngeal cancer stem cells by down-regulating the IL-6/STAT3/HIF1 pathway. Cancer Cell Int. 2017;17:79. doi:10.1186/s12935-017-0448-028878571
- Gupta GP, Massagué J. Cancer metastasis: building a framework. Cell. 2006;127:679–695. doi:10.1016/j.cell.2006.11.00117110329
- Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3:453–458. doi:10.1038/nrc109812778135
- Wang J, Ge W, Wang Y, et al. MiR-613 suppressed the laryngeal squamous cell carcinoma progression through regulating PDK1. J Cell Biochem. 2017. doi:10.1002/jcb.26468
- Rothman KJ, Cann CI, Flanders D, et al. Epidemiology of laryngeal cancer. Epidemiol Rev. 1980;2:195–209. doi:10.1093/oxfordjournals.epirev.a0362237000536
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. doi:10.3322/caac.v63.123335087
- Tamaki A, Miles BA, Lango M, et al. AHNS series: do you know your guidelines? Review of current knowledge on laryngeal cancer. Head Neck. 2017. doi:10.1002/hed.24862
- Han Q, Lin X, Zhang X, et al. WWC3 regulates the Wnt and hippo pathways via dishevelled proteins and large tumour suppressor 1, to suppress lung cancer invasion and metastasis. J Pathol. 2017;242:435–447. doi:10.1002/path.2017.242.issue-428543074
- Tong R, Yang B, Xiao H, et al. KCTD11 inhibits growth and metastasis of hepatocellular carcinoma through activating hippo signaling. Oncotarget. 2017;8:37717–37729. doi:10.18632/oncotarget.v8i2328465479
- Varzavand A, Hacker W, Ma D, et al. α3β1 integrin suppresses prostate cancer metastasis via regulation of the hippo pathway. Cancer Res. 2016;76:6577–6587. doi:10.1158/0008-5472.CAN-16-148327680681
- van Rensburg HJJ, Yang X. The roles of the hippo pathway in cancer metastasis. Cell Signal. 2016;28:1761–1772. doi:10.1016/j.cellsig.2016.08.00427519476
- Juan WC, Hong W. Targeting the hippo signaling pathway for tissue regeneration and cancer therapy. Genes. 2016;7:55. doi:10.3390/genes7090055
- Démant P, Iványi D, van Nie R. The map position of the rds gene on the 17th chromosome of the mouse. Tissue Antigens. 1979;13:53–55. doi:10.1111/j.1399-0039.1979.tb01136.x419532
- Stuck MW, Conley SM, Naash MI. Retinal degeneration slow (RDS) glycosylation plays a role in cone function and in the regulation of RDS·ROM-1 protein complex formation. J Biol Chem. 2015;290:27901–27913. doi:10.1074/jbc.M115.68369826420485
- Zulliger R, Conley SM, Mwoyosvi ML, et al. Oligomerization of Prph2 and Rom1 is essential for photoreceptor outer segment formation. Hum Mol Genet. 2018;27:3507–3518. doi:10.1093/hmg/ddy24029961824
- Conley SM, Stuck MW, Watson JN, et al. Prph2 initiates outer segment morphogenesis but maturation requires Prph2/Rom1 oligomerization. Hum Mol Genet. 2019;28:459–475. doi:10.1093/hmg/ddy35930307502
- Stuck MW, Conley SM, Naash MI. PRPH2/RDS and ROM-1: historical context, current views and future considerations. Prog Retin Eye Res. 2016;52:47–63. doi:10.1016/j.preteyeres.2015.12.00226773759
- Chakraborty D, Conley SM, Al-Ubaidi MR, et al. Initiation of rod outer segment disc formation requires RDS. PLoS One. 2014;9:e98939. doi:10.1371/journal.pone.009893924897172
- Zhang Z, Chometon G, Wen T, et al. Migration of epithelial cells on laminins: RhoA antagonizes directionally persistent migration. Eur J Cell Biol. 2011;90:1–12. doi:10.1016/j.ejcb.2010.09.00520971525
- Schmidt-Erfurth U, Hasan T. Mechanisms of action of photodynamic therapy with verteporfin for the treatment of age-related macular degeneration. Surv Ophthalmol. 2000;45:195–214. doi:10.1016/S0039-6257(00)00158-211094244
- Zhang Z, Lin Z, Zhou Z, et al. Structure-based design and synthesis of potent cyclic peptides inhibiting the YAP-TEAD protein-protein interaction. ACS Med Chem Lett. 2014;5:993–998. doi:10.1021/ml500160m25221655
- Salcedo Allende MT, Zeron-Medina J, Hernandez J, et al. Overexpression of yes associated protein 1, an independent prognostic marker in patients with pancreatic ductal adenocarcinoma, correlated with liver metastasis and poor prognosis. Pancreas. 2017;46:913–920. doi:10.1097/MPA.000000000000086728697132
- Qiao Y, Chen J, Lim YB, et al. YAP regulates actin dynamics through ARHGAP29 and promotes metastasis. Cell Rep. 2017;19:1495–1502. doi:10.1016/j.celrep.2017.04.07528538170
- Li C, Wang S, Xing Z, et al. A ROR1-HER3-lncRNA signalling axis modulates the hippo-YAP pathway to regulate bone metastasis. Nat Cell Biol. 2017;19:106–119. doi:10.1038/ncb346428114269
- Nokin MJ, Durieux F, Peixoto P, et al. Methylglyoxal, a glycolysis side-product, induces Hsp90 glycation and YAP-mediated tumor growth and metastasis. Elife. 2016;5:e19375. doi:10.7554/eLife.1937527759563
