Abstract
Background
Pentraxin 3 (PTX3) is an acute phase protein, involved in antibacterial resistance. Recent studies have shown PTX3 levels to be elevated in the presence of a bacterial infection and in a murine sepsis model.
Objective
We aim to investigate if sputum PTX3 can be used as a biomarker for bacterial infection in subjects with COPD.
Materials and methods
Sputum samples from 142 COPD patients (102 men) with a mean (range) age of 69 years (45–85) and mean (SD) post-bronchodilator percentage predicted forced expiratory volume in 1 second (FEV1) of 50% (19) were analyzed for PTX3, using a commercial assay at stable state and during an exacerbation. Association with bacteria, from culture, quantitative real-time polymerase chain reaction (qPCR) and colony-forming units (CFU) was investigated.
Results
The geometric mean (95% CI) PTX3 level at stable state was 50.5 ng/mL (41.4–61.7). PTX3 levels correlated with absolute neutrophil count in sputum (r=0.37; P<0.01), but not FEV1 or health status. There was a weak correlation between PTX3 and bacterial load (CFU: r=0.29, P<0.01; 16S qPCR: r=0.18, P=0.05). PTX3 was a poor predictor of bacterial colonization (defined as >105 CFU/mL at stable state) with a receiver-operating characteristic (ROC) area under the curve (AUC) of 0.59 and 95% confidence interval (CI) 0.43–0.76 (P=0.21). During an exacerbation, there was a modest increase in PTX3 (fold difference 0.15, 95% of difference 0.02–0.29; P=0.02), and PTX3 fared better at identifying a bacteria-associated exacerbation (ROC AUC 0.65, 95% CI 0.52–0.78, P=0.03).
Conclusion
PTX3 is associated with bacterial infection in patients with COPD, but its utility as a biomarker for identifying a bacteria-associated exacerbation warrants further studies.
Introduction
COPD is characterized by persistent airflow limitation, which is progressive and associated with an enhanced inflammatory response to noxious agents.Citation1 Exacerbations of COPD have a significant effect on the health of patientsCitation2 and are associated with microbialCitation3–Citation7 and airway inflammation.Citation8 Chronic bacterial infection, defined as >105 colony-forming units (CFU),Citation9 occurs in 30% of patients at stable stateCitation10 and increases up to 50% during an exacerbation.Citation3 Guidelines for treatment of an exacerbation advocate the use of systemic corticosteroidsCitation11 and antibiotics;Citation12 however, this is usually without evidence of a bacterial infection as standard culture techniques are not rapid.Citation13 The occurrence of resistant bacteria is a growing concernCitation14 and deemed an emerging medical catastrophe.Citation15 The rapid identification of a bacterial exacerbation is urgently needed to aid clinical treatment decisions. One such possible marker is pentraxin 3 (PTX3). PTX3 is a soluble pattern recognition receptor,Citation16 recognizing pathogen-associated molecular patterns expressed by microorganisms.Citation17 PTX3 is induced in response to proinflammatory stimuliCitation16 and toll-like receptor (TLR) interactionsCitation17 and contributes to innate resistance to pathogens.Citation16 PTX3 levels have been shown to be elevated in a murine bacterial infection model,Citation18 in sepsis,Citation19–Citation21 and in inflammatory rheumatic disease.Citation22 In this study, we aim to determine whether sputum PTX3 is a sensitive biomarker for bacterial colonization at stable state and for a bacterial exacerbation in patients with COPD.
Materials and methods
Subjects and sampling
COPD subjects entering a longitudinal study looking at biomarkers in COPD were analyzed, where subject inclusion and exclusion criteria, study design, and measurements are as previously described.Citation3 In brief, subjects attended a stable state visit every 3 months over a 12-month period and also during exacerbations and 2 weeks post-exacerbation. An exacerbation event was defined according to Anthonisen criteriaCitation23 and NICE guidance,Citation24 and a bacteria-associated exacerbation was defined as >107 CFU/mL as previously described.Citation3 At each visit, participants underwent pre- and postbronchodilator spirometry; blood was collected by standard venipuncture and sputum induction for sputum collection. If participants were unable to perform sputum induction, spontaneous sputum was collected. Health status and symptoms were measured using the St George’s respiratory questionnaire (SGRQ),Citation25 MRC dyspnea scale,Citation26 the chronic respiratory disease questionnaire (CRQ),Citation27 and the visual analog score (VAS).Citation28 Chronic bacterial infection (colonization) was defined as >105 CFU/mL at stable stateCitation9 and in this cohort was associated with a high positive predictive value of a respiratory pathogenic microorganism (). All subjects gave written informed consent, and the study was approved by the Leicestershire, Northamptonshire and Rutland ethics committee (reference number: 07/H0406/157).
Table 1 Proportion of subjects below and above the cutoff of 105 CFU/mL that had a negative or positive microbiology results at stable state
Sputum processing
Sputum processing involved plug selection, followed by a dispersion step with Dulbecco phosphate-buffered saline and a mucolytic step with dithiothreitol as previously described.Citation3 A filtration step to remove debris for cytospin preparation and quantification of cell differential count was then performed.Citation29 Samples with a cell viability of <40% and a squamous contamination >20% were excluded. CFUs were prepared for semi-quantitative analysis using standard techniques.Citation30 A further 500 μL of the filtrate was processed by SYBR green (Applied Biosystems®; Life Technologies Corp., Carlsbad, CA, USA) quantitative polymerase chain reaction (qPCR) for Haemophilus influenzae and Staphylococcus aureus bacterial DNA, and a Taqman qPCR assay for the quantification of Streptococcus pneumoniae and Moraxella catarrhalis as described previously.Citation3 Viral RNA was extracted, using in-house assays, from an additional sputum plug for PCR analysis, as previously described.Citation3 Only samples with available sputum microbiology were analyzed. A sputum eosinophilia was defined as a sputum eosinophil count of >3%.Citation31
Pentraxin 3 Quantikine ELISA
A Human Pentraxin 3/TSG-14 Quantikine ELISA Kit (R&D Systems, Oxfordshire, UK) was used to measure PTX3 levels; the assay was carried out as per manufacturer’s protocol and read using an EnVision plate reader (Perkin-Elmer, MA, USA). The lower limit of detection for PTX3 is 0.31 ng/mL. Readings taken at 560 nm wavelengths were subtracted from the readings taken at 450 nm to correct for any optical imperfections within the plate. The average optical density from the negative control was subtracted from each well to remove any background noise present. All standards, negatives, and samples were run in duplicate on each plate. The standards were plotted against the optical density value. The R2 value was >0.97.
Statistical analysis
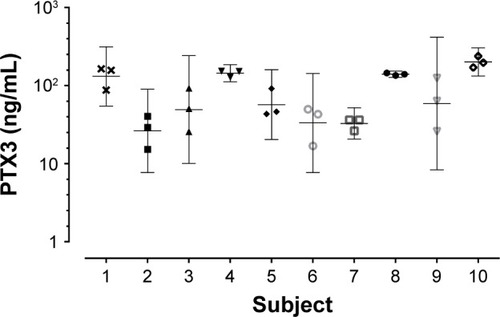
GraphPad Prism version 6 (GraphPad Software, Inc., La Jolla, CA, USA) and Statistical Package for the Social Sciences (SPSS) Statistics version 22 (SPSS, Inc., Chicago, IL, USA) were used for statistical analysis. The Kolmogorov–Smirnov test was applied for normality. PTX3 levels were log-normal transformed. All parametric data were displayed as mean and standard deviation (SD), all non-parametric data were displayed as median (interquartile range [IQR]), and log-transformed data were presented as geometric mean and 95% confidence interval. Paired and unpaired t-test and one-way analysis of variance (ANOVA) tests were used to compare two, three, or more groups, respectively. Receiver-operating characteristic (ROC) curves were utilized to measure the sensitivity and specificity of PTX3 as a biomarker. Repeatability analysis was conducted in 10 subjects over three stable visits and the coefficient of variation was used to test repeatability over time. For multiple exacerbation visits, only the first exacerbation was analyzed. A probability of P<0.05 was considered to be statistically significant.
Results
PTX3 was measured at stable state in 148 individual subjects, 6 of these were unreadable and so excluded from analysis; thus, a total of 142 subject samples were analyzed. A paired exacerbation result was available in 95 subjects. The baseline characteristics of the COPD subjects are presented in .
Table 2 Baseline characteristics of the 142 COPD subjects included in this analysis of pentraxin 3 levels
Stable state
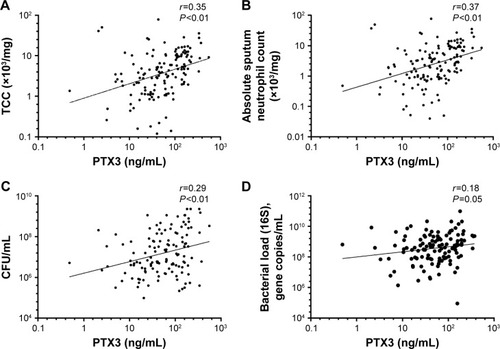
The geometric mean (95% CI) of PTX3 was 50.5 ng/mL (41.4–61.7). Ex-smokers compared to current smokers had higher PTX3 levels (geometric mean [95% CI]: ex-smokers 61.3 ng/mL [48.5–77.4] vs current smokers 40.6 [28.3–58.3], P=0.05). PTX3 was not associated with severity of COPD, defined by Global Initiative for Chronic Obstructive Lung Disease (GOLD) (2007)Citation32 or treatment with or without inhaled corticosteroids (ICS). There were no correlations seen between PTX3 and the post-bronchodilator forced expiratory volume in 1 second (FEV1) (r=−0.13; P=0.15), subject health (MRC: r=0.04, P=0.66; CRQ: r=−0.07, P=0.40; SGRQ: r=0.05, P=0.54; and VAS: r=−0.01, P=0.91), CRP (r=0.13, P=0.14), or absolute blood neutrophils (r=0.09, P=0.27). PTX3 was associated with sputum total cell count (r=0.35, P<0.01; ) and neutrophilic airway inflammation (r=0.37, P<0.01; ). PTX3 was associated with bacterial load, measured by CFU/mL (r=0.29, P<0.01; ) and total 16S qPCR (r=0.18, P=0.05; ). Non-eosinophilic subjects had significantly higher PTX3 levels than eosinophilic subjects (fold difference [95% CI] 0.30 [0.10–0.50]; P<0.01). For individual pathogens, pathogen-specific qPCR demonstrated a weak but non-significant correlation of PTX3 with S. pneumoniae (r=0.33; P=0.38) but not M. catarrhalis (r=0.01; P=0.98) or H. influenzae (r=−0.04; P=0.84). There were too few single S. aureus qPCR isolates to analyze. PTX3 was not a predictor of bacterial colonization at stable state (ROC area under the curve [AUC] 0.59, 95% CI 0.43–0.76, P=0.21) and was stable over time with a coefficient of variation 10% ().
At exacerbation
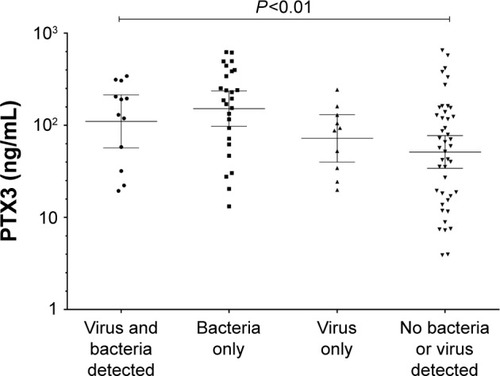
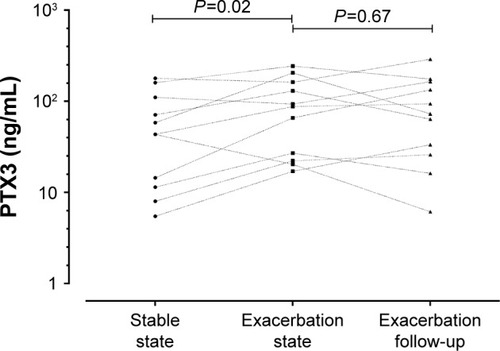
During an exacerbation, there was a significant increase in PTX3 levels from a stable to exacerbation state (fold difference 0.15, 95% of difference 0.02–0.29; P=0.02). Subjects with an increase in PTX3 at the exacerbation were also more likely to culture a pathogen (). PTX3 at exacerbation correlated with CFU/mL (P<0.01, r=0.30) but not total 16S (P=0.91, r=−0.01). At exacerbation, there was a moderate but non-significant correlation of PTX3 with H. influenzae single isolate pathogen-specific qPCR (r=0.41, P=0.18) but not M. catarrhalis (r=−0.12, P=0.82). There were too few isolates of S. pneumoniae and S. aureus to analyze. A PTX3 level of 118.0 ng/mL had a sensitivity and specificity of 60%, respectively (ROC AUC 0.65, 95% CI 0.52–0.78, P=0.03) to determine a bacterial exacerbation. The presence or absence of viruses at the onset of an exacerbation did not affect PTX3 levels (fold difference [95% CI] of 0.03 [−0.25 to 0.32]; P=0.81). The combination of bacteria and virus detection at exacerbation demonstrated similar PTX3 levels to bacteria alone (). In a small subset of subjects (n=11) with sputum PTX3 measured at a stable, exacerbation and exacerbation follow-up visit, a trend to increase in PTX3 from stable to exacerbation state was seen in the 11 paired subjects and a trend to reduction at follow-up ().
Table 3 Difference in culture growth between subjects that saw an increase in PTX3 levels compared with those that saw a decrease from stable to exacerbation state
Discussion
In this study, we aimed to determine if PTX3 was a sensitive biomarker for bacterial colonization at stable state and for a bacterial exacerbation in subjects with COPD, by measuring PTX3 concentrations in sputum. We have demonstrated for the first time that PTX3 in subjects with COPD correlates with bacterial burden, both at stable state and exacerbations and increases during an exacerbation event. PTX3 did not correlate with symptoms or lung function and was a reasonable predictor of a bacteria-associated exacerbation.
Previous studies in non-COPD cohorts have shown PTX3 to rapidly increase due to an infection.Citation19,Citation33 PTX3 has been shown to be elevated in COPD subjects compared with healthy controls in plasmaCitation34 and sputum.Citation35 PTX3 levels have been correlated with the number of neutrophils present in sputum and have been shown to be elevated in current and ex-smokers compared with non-smokers.Citation36 Our results support these statements. To our knowledge, no study has looked into PTX3 as a viable biomarker for a bacterial colonization or bacterial exacerbation in COPD subjects, though this has been suggested.Citation37
PTX3 levels have previously been linked to inflammatory diseases;Citation22 so it was not surprising to see a correlation between PTX3 levels and neutrophils in sputum, a pro-inflammatory cell,Citation38 and also a cell involved in PTX3 storage.Citation17 PTX3 has also been implemented in bacterial infection,Citation18–Citation21 and our study supports this as we found a correlation between bacterial load (CFU/mL) in sputum at stable and exacerbation states implying that PTX3 expression in the lungs is driven by bacterial infection. This was further highlighted when PTX3 expression was relatively unchanged with the presence of a virus. Although non-significant, we did identify a moderate correlation of H. influenzae individual pathogen-specific qPCR with PTX3 during an exacerbation but not at stable state. This is unsurprising as H. influenzae is a commonly isolated pathogen in COPD patientsCitation39 and is the main driver of inflammation.Citation40 Further work is needed to fully investigate the role of individual pathogens in PTX3 expression.
Limitations
This study has a few limitations. First, we did not have data on PTX3 in healthy controls, which would support whether levels detected in the COPD population are elevated; an additional infection control, such as bronchiectasis, would provide added value. However, this is the first study to look at infection (chronic and thus colonization) versus acute (bacteria-associated) exacerbations demonstrating modest utility of PTX3. Finally, advances in qPCR mean it is now possible to measure bacteria more accurately using plasmidsCitation41 and to exclude dead bacteria.Citation42 As this was not available at the time of this work, we cannot confirm if the modest associations seen are a consequence of the inclusion of live and dead bacteria, and both these advancements in the field would greatly enhance this study.
Conclusion
To conclude, this study found that PTX3 correlates with bacterial load and that PTX3 is not sensitive or specific enough to be used as a biomarker for bacterial colonization but could be a potential biomarker for a bacteria-associated exacerbation in patients with COPD. However, further work is warranted to fully evaluate this.
Author contributions
MB is the guarantor of the content of the manuscript, including the data and analysis. All authors participated sufficiently in the work, the data analysis, and/or the preparation of the manuscript, and have reviewed and approved the manuscript for submission. MB and CEB contributed to the design of the study. MD, SJT, MRB, VM, KH, CEB, and MB contributed to data analysis, and all authors contributed to data interpretation and to the writing of the manuscript.
Acknowledgments
The authors thank Mrs Beverley Hargadon, Mrs Susan McKenna, Ms Amisha Singapuri, and Mrs Maria Shelley for their assistance in patient characterization. They thank Mrs Carlene White, Mrs Debbie Parker, and Mr Mitesh Pancholi for their assistance in sputum processing. They also thank Professor Sebastian Johnston and Dr Tatiana Kabadze for their assistance in viral PCR processing.
This work was funded by the Medical Research Council, the Wellcome Trust & Academy of Medical Sciences, and the Oxfordshire Health Services Research Committee. This paper presents independent research funded by the National Institute of Health Research, and the views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health.
Disclosure
All authors declare no financial support from, or relationship with any organization that may have an interest, for the submitted work. MB is funded by a National Institute of Health Research (NIHR) Post-doctoral Fellowship (PDF-2013-06-052) and Oxford Health Sciences Research Committee. MB has received travel support from AstraZeneca, Boehringer Ingelheim, and GlaxoSmithKline and consultancy honoraria outside the submitted work from AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, and Pfizer. CEB was funded by a Wellcome Trust Senior Fellowship. CEB has received grant support and consultancy honoraria outside the submitted work from AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Medimmune, Novartis, and Roche. The authors report no other conflicts of interest in this work.
References
- VestboJHurdSSAgustíAGGlobal strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2013187434736522878278
- GarveyCRecent updates in chronic obstructive pulmonary diseasePostgrad Med2016128223123826560514
- BafadhelMMcKennaSTerrySAcute exacerbations of chronic obstructive pulmonary disease: identification of biologic clusters and their biomarkersAm J Respir Crit Care Med2011184666267121680942
- MonsóERuizJRosellABacterial infection in chronic obstructive pulmonary disease. A study of stable and exacerbated outpatients using the protected specimen brushAm J Respir Crit Care Med19951524131613207551388
- BanerjeeDKhairOAHoneybourneDImpact of sputum bacteria on airway inflammation and health status in clinical stable COPDEur Respir J200423568569115176680
- SethiSMurphyTFInfection in the pathogenesis and course of chronic obstructive pulmonary diseaseN Engl J Med2008359222355236519038881
- PapiABellettatoCBraccioniFInfections and airway inflammation in chronic obstructive pulmonary disease severe exacerbationsAm J Respir Crit Care Med2006173101114112116484677
- BhowmikASeemungalTSapsfordRWedzichaJRelation of sputum inflammatory markers to symptoms and lung function changes in COPD exacerbationsThorax200055211412010639527
- MiravitllesMMarínAMonsóEColour of sputum is a marker for bacterial colonisation in chronic obstructive pulmonary diseaseRespir Res2010111585820470372
- RosellAMonsóESolerNMicrobiologic determinants of exacerbation in chronic obstructive pulmonary diseaseArch Int Med2005165889189715851640
- National Clinical Guideline Centre2010Chronic Obstructive Pulmonary Disease: Management of Chronic Obstructive Pulmonary Disease in Adults in Primary and Secondary careLondonNational Clinical Guideline Centre Available from: http://guidance.nice.org.uk/CG101/Guidance/pdf/EnglishAccessed September 1, 2015
- Global Strategy for diagnosis m, and prevention of COPD Available from: www.goldcopd.orgAccessed March 14, 2016
- Recommendations of the Clinical Subcommittee of the Medical/Scientific Advisory Committee of the Canadian Cystic Fibrosis FoundationMicrobiological processing of respiratory specimens from patients with cystic fibrosisCan J Infect Dis19934316616922346442
- ErkanLUzunOFindikSKatarDSanicAAticiAGRole of bacteria in acute exacerbations of chronic obstructive pulmonary diseaseInt J Chron Obstruct Pulmon Dis20083346346718990975
- BerkowitzFEAntibiotic resistance in bacteriaSouth Med J19958887978047631202
- BalharaJKoussihLZhangJGounniASPentraxin 3: an immuno-regulator in the lungsFront Immunol2013412723755050
- KunesPHolubcovaZKolackovaMKrejsekJPentraxin 3(PTX 3): an endogenous modulator of the inflammatory responseMediators Inflamm2012201292051722577258
- SoaresACSouzaDGPinhoVDual function of the long pentraxin PTX3 in resistance against pulmonary infection with Klebsiella pneumoniae in transgenic miceMicrobes Infect2006851321132916697676
- VänskäMKoivulaIHämäläinenSHigh pentraxin 3 level predicts septic shock and bacteremia at the onset of febrile neutropenia after intensive chemotherapy of hematologic patientsHaematologica20119691385138921880642
- Bastrup-BirkSSkjoedtMOMunthe-FogLStromJJMaYJGarredPPentraxin-3 serum levels are associated with disease severity and mortality in patients with systemic inflammatory response syndromePLoS One201389e7311924039869
- HuttunenRHurmeMAittoniemiJHigh plasma level of long pentraxin 3 (PTX3) is associated with fatal disease in bacteremic patients: a prospective cohort studyPLoS One201163e1765321423699
- HollanIBottazziBCuccovilloIIncreased levels of serum pentraxin 3, a novel cardiovascular biomarker, in patients with inflammatory rheumatic diseaseArthritis Care Res (Hoboken)201062337838520391484
- AnthonisenNRManfredaJWarrenCPWHershfieldESHardingGKMNelsonNAAntibiotic therapy in exacerbations of chronic obstructive pulmonary diseaseAnn Int Med198710621962043492164
- Chronic obstructive pulmonary disease2016 Available from: https://www.nice.org.uk/guidance/Accessed November 1, 2015
- WeatherallMMarshSShirtcliffePWilliamsMTraversJBeasleyRQuality of life measured by the St George’s respiratory questionnaire and spirometryEur Respir J20093351025103019164350
- MRCMedical Research Council Dyspnoea scale/Breathlessness scale [webpage on the Internet] Available from: https://www.mrc.ac.uk/research/facilities-and-resources-for-researchers/mrc-scales/mrc-dyspnoea-scale-mrc-breathlessness-scale/Accessed November 1, 2015
- ChauvinARupleyLMeyersKJohnsonKEasonJOutcomes in cardiopulmonary physical therapy: chronic respiratory disease questionnaire (CRQ)Cardiopulm Phys Ther J2008192616720467499
- BrightlingCEMonterioWGreenRHInduced sputum and other outcome measures in chronic obstructive pulmonary disease: safety and repeatabilityRespir Med20019512999100211778799
- PizzichiniEPizzichiniMMEfthimiadisAHargreaveFEDolovichJMeasurement of inflammatory indices in induced sputum: effects of selection of sputum to minimize salivary contaminationEur Respir J199696117411808804934
- PyeAStockleyRAHillSLSimple method for quantifying viable bacterial numbers in sputumJ Clin Pathol19954887197247560197
- WagenerAHde NijsSBLutterRExternal validation of blood eosinophils, FENO and serum periostin as surrogates for sputum eosinophils in asthmaThorax201570211512025422384
- RabeKFHurdSAnzuetoAGlobal strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2007176653255517507545
- BottazziBDoniAGarlandaCMantovaniAAn integrated view of humoral innate immunity: pentraxins as a paradigmAnn Rev Immunol201028115718319968561
- KurtOKTosunMKurtEBTalayFPentraxin 3 as a novel biomarker of inflammation in chronic obstructive pulmonary diseaseInflammation2015381899325303877
- SchwingelFLPizzichiniEKlevestonTPentraxin 3 sputum levels differ in patients with chronic obstructive pulmonary disease vs asthmaAnn Allergy Asthma Immunol2015115648548926508705
- Van PottelbergeGRBrackeKRPauwelsNSVermassenFEJoosGFBrusselleGGCOPD is associated with reduced pulmonary interstitial expression of pentraxin-3Eur Respir J201239483083821920889
- LiuSQuXLiuFWangCPentraxin 3 as a prognostic biomarker in patients with systemic inflammation or infectionMediators Inflamm2014201442142925530683
- MantovaniACassatellaMACostantiniCJaillonSNeutrophils in the activation and regulation of innate and adaptive immunityNat Rev Immunol201111851953121785456
- BafadhelMHaldarKBarkerBAirway bacteria measured by quantitative polymerase chain reaction and culture in patients with stable COPD: relationship with neutrophilic airway inflammation, exacerbation frequency, and lung functionInt J Chron Obstruct Pulmon Dis2015101075108326089657
- BarkerBLHaldarKPatelHAssociation between pathogens detected using quantitative polymerase chain reaction with airway inflammation in COPD at stable state and exacerbationsChest201414714655
- KleinDQuantification using real-time PCR technology: applications and limitationsTrends Mol Med20028625726012067606
- NockerASossa-FernandezPBurrMDCamperAKUse of propidium monoazide for live/dead distinction in microbial ecologyAppl Environl Microbiol2007731651115117